Abstract
Introduction
Airborne fungi are ubiquitous in the environment and are commonly associated with airway inflammatory diseases, and the vast majority of healthy persons and patients suffering from chronic rhinosinusitis can cultivate fungi in their nasal mucus.1,2 Under the protection of the mucosal immune barrier, fungi are not susceptible to disease. However, fungi can become pathogens when they are not cleared by the mechanical action of mucus. Unlike inert and nonviable material that settle on the mucus layer, fungi can multiply and create a ball or concretion of fungal hyphae, commonly known as a fungal ball (FB). Mycoses or FBs of the sinuses were first reported in 1885, and Aspergillus-associated sinusitis was most common. 1 FB rhinosinusitis (FBS) is a concretion or collection of fungal debris usually within a single sinus, and the maxillary and sphenoid sinuses are most commonly affected.
The fungal colonization does not have an invasive component, but it may cause a mixed inflammatory reaction in the involved sinus. This reaction occasionally involves sinuses within the same functional unit, such as the frontal and anterior ethmoid in the case of maxillary sinus FBs. 3 Imaging examination indicates sensitivity and specificity in FBS diagnosis. Computed tomography (CT) reveals a high-density area, sclerosis of the lateral sinus wall, erosion of the inner sinus wall, and irregular material surface, whereas magnetic resonance imaging (MRI) reveals a low-intensity area on T2-weighted MRI.4,5
Most local anatomical variants have potential risk factors for FB formation. Septal deviation, among other variants, including concha bullosa, is implicated along with infundibulum length compared with normal controls.6-9 However, a repeat study failed to confirm this association with the septum and instead suggested an association with the deviation away from the involved side. 10 Differences in the anatomical variation of the bilateral nasal cavities can lead to differences in nasal ventilation, but it remains unclear whether such differences in ventilation can cause FBS. Nevertheless, few studies have reported that differences in bilateral nasal ventilation may affect FBS localization. To investigate this issue, this retrospective study was conducted on anatomical variants and nasal ventilation in patients with FBS.
Nasal resistance (NR) is an objective measure of the ventilation resistance in one or both sides of nasal cavity during calm breathing, which serves as an indicator for assessing physiological status and patency of the nasal cavity. There are 3 methods currently used for rhinomanometry: active anterior rhinomanometry (AAR), passive anterior rhinomanometry, and active posterior rhinomanometry. AAR is the most frequently used method, and is considered simple, fast, and well tolerated. In 2016, the International Standardization Committee on the Objective Assessment of the Nasal Airway suggested that the logarithmic effective resistance measured using rhinomanometry was a parameter with high diagnostic relevance. 11
Acoustic rhinometry (AR) utilizes the principle of acoustic reflection to measure nasal cavity geometry, such as nasal cavity volume (NCV) and nasal cross-sectional area (NCA) under transient breath-holding conditions and is not influenced by subjective factors of the subjects. AAR and AR allow doctors to evaluate nasal airway patency; however, there are also several factors that may affect the measurements, such as sex, age, and physiological changes.12-14 NCA in pregnant women decreases significantly between the first and third trimesters, 12 and both age and sex affect the average minimum cross-sectional area (MCA), with men having a larger average NCV than women. 14 To avoid these interfering factors, this study compared the anatomical variations and differences in ventilation between the affected and unaffected nasal cavities of patients with FBS to investigate the relationship between these differences and FB localization.
Materials and Methods
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The protocols and experimental procedures used in the present study were reviewed by the ethics committee of our hospital, and ethical approval documents were obtained. Informed consent was not obtained because this was a retrospective study in which there was no direct contact with the patients; however, patient-identifiable data were not recorded.
Two senior doctors participating in this study used the medical record information management system to search for discharge diagnoses with “sinusitis” as the keyword in chronological order and identified all types of sinusitis among patients hospitalized at our facility from January 2021 to December 2022. We reviewed the surgical records and pathological staining of patients, and established a rediagnosis based on DeShazo’s initial criteria and description for FB. 15 Study participants were selected according the following inclusion criteria: patients (1) with complete data, including data on previous medical history, and surgical records, and with CT examination, nasal endoscopy, nasal resistance (NR) measurement, and AR performed in our hospital before surgery; (2) without polyps or polypoid mucosa in the nasal cavity through nasal endoscopy; and (3) with CT images showing isolated paranasal sinus lesions or inflammation involving adjacent sinuses showing mucosal thickening. The exclusion criteria were as follows: (1) previous surgery on the nasal cavity or paranasal sinuses; (2) nasal bone or nasal septum fracture due to trauma within 2 years; (3) severe structural abnormalities in the nasal cavity leading to inability to measure NR and perform AR; (4) systemic antihistamine (including cold drugs) and/or corticosteroid use within 1 week before hospitalization; and (5) intranasal antihistamine and/or corticosteroid use within 24 hours before NR measurement and AR. We reviewed CT images, NR and AR results, as well as anatomical variations (ie, septum deviation, concha bullosa, and Haller cells). NR, NCA, and NCV were collected from the affected and unaffected sides for analysis.
NR and AR
The patients’ nasal resistance was measured using a rhinomanometer (NR6, GM Instruments Ltd., Kilwinning, UK), acoustic rhinometry (A1, GM Instruments Ltd., Kilwinning, UK) was performed by an experienced doctor in a quiet room at 20 C to 25 C, and AAR was performed according to the guidelines of the International Committee on Standardization of Rhinomanometry. The subjects were seated and instructed to familiarize themselves with the testing process, and the subjects’ cooperation was obtained before the measurement. The measurement process was completed using a computer. The test was repeated 4 to 5 times for the same subject, the average value was taken, and the results were recorded. AAR is an important parameter measured using rhinomanometers and is equal to the pressure difference between the anterior and posterior nostrils divided by the flow rate of air within the nasal cavity. The most commonly used parameter in AR assessment is the MCA. Moreover, several authors have presented NCA for various nasal cavity sections; for example, at the point of the second narrowing, which corresponds to the head of the middle nasal concha, and we named it the second MCA (sMCA) in this study. Another commonly used parameter is the NCV for a selected section (range, 0-7 cm; See Figure 1). 16
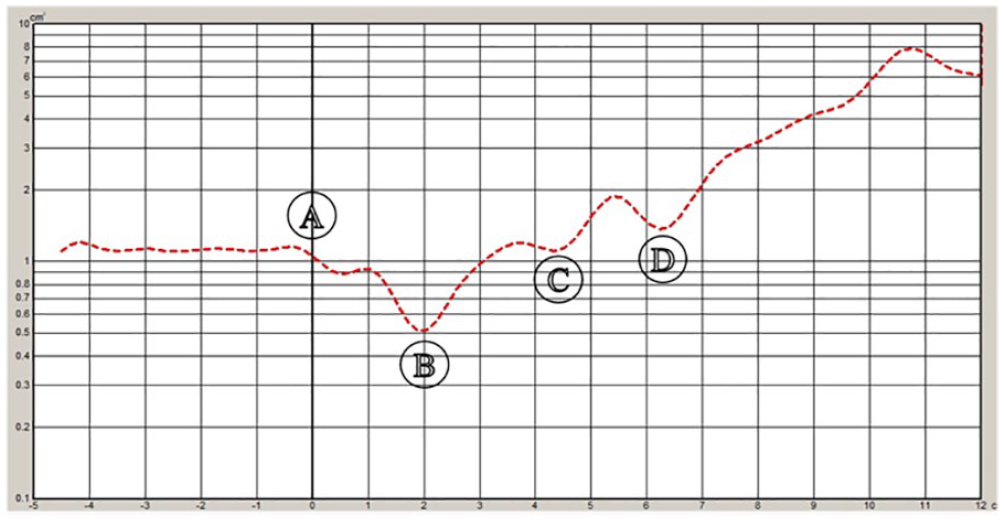
Schematic Diagram of acoustic rhinometry results.
Statistical Analyses
IBM SPSS version 19 for Windows (IBM Corp., Armonk, NY, USA) was used for the analyses. Descriptive statistics were used to present demographic and clinical characteristics, and anatomical abnormalities. Categorical variables are presented as frequencies and percentages, whereas continuous variables were first subjected to the Shapiro–Wilk test. If the data followed a normally distributed distribution, we utilized the mean ± standard deviation and conducted a paired sample t-test to analyze the study variables in both observation and control groups, and then the actual power analysis was performed using G*Pcalculator 3.1.9.7 (http://www.gpower.hhu.de/) based on the differences between the 2 groups. If the data were not normally distributed, the median was used, and Kruskal–Wallis analysis of variance was used. The relationships between FB localization and anatomical risk factors, bilateral NR ratio, NCA ratio, and differences in NCV were analyzed. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using multiple logistic regression analyses. Variable 0 was defined as FB occurring in the left sinus, and variable 1 was defined as FB occurring in the right sinus during data analysis. Additionally, a dummy variable was set equal to 1 if the septal deviation was directed to the affected side or if concha bullosa and Haller cells existed on the affected side, and 0 otherwise. A P value of <.05 was considered statistically significant at the 95% CI.
Results
In total, 152 patients were rediagnosed with fungal rhinosinusitis, and 95 patients with FBS were included in this study. The 33 male (34.7%) and 62 female (65.3%) patients were aged between 25 and 89 (mean 52.8) years. The most common site of FB was the maxillary sinus (83, 87.4%), followed by the sphenoid sinus (9, 9.5%; see Table 1). The analysis of anatomical variations is summarized in Table 2. A total of 52 (54.7%) patients were diagnosed with septal deviation, which was more common on the affected side (33, 63.5%) than in the unaffected side. However, multivariate logistic analysis showed no correlation between septal deviation and FB occurrence (OR = 0.707; 95% CI, 0.312-1.598; P = .404) (see Table 4). Additionally, concha bullosa and Haller cells were more common on the unaffected side than in the affected side; however, due to the small number of cases, statistical analysis was not applicable.
Participants’ Demographic and Clinical Data.
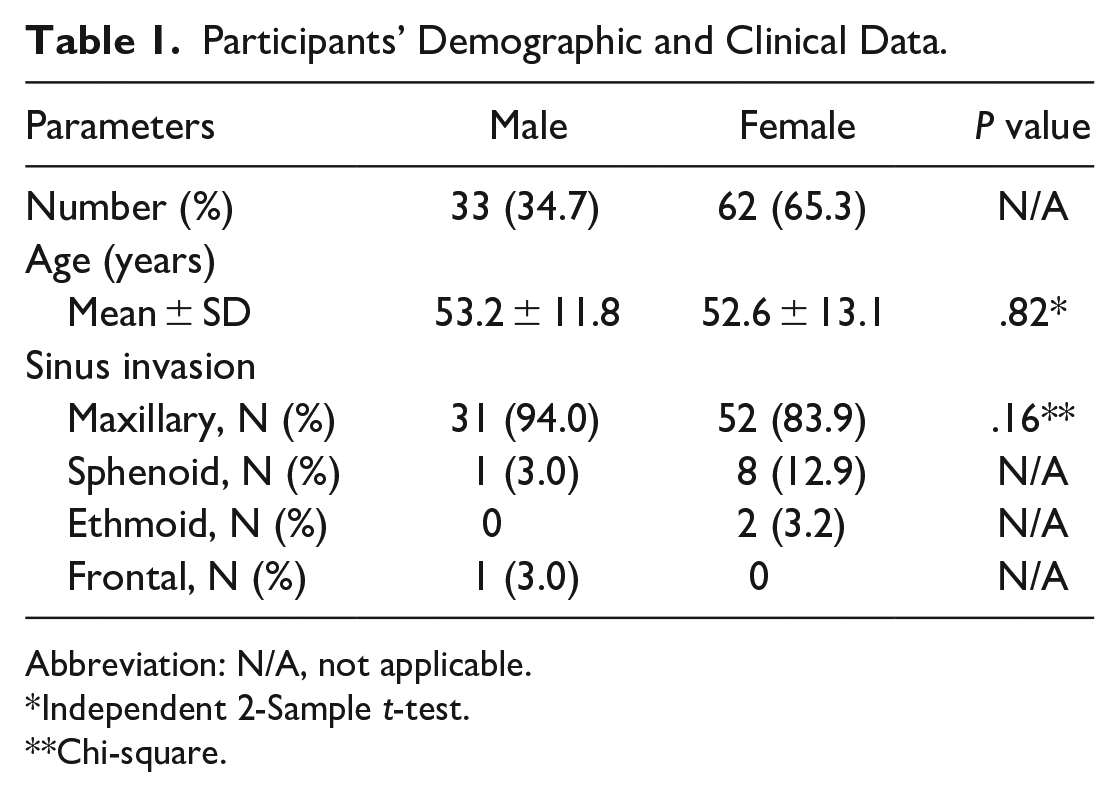
Abbreviation: N/A, not applicable.
Independent 2-Sample t-test.
Chi-square.
Prevalence of Anatomical Variations in Affected and Unaffected Sides.
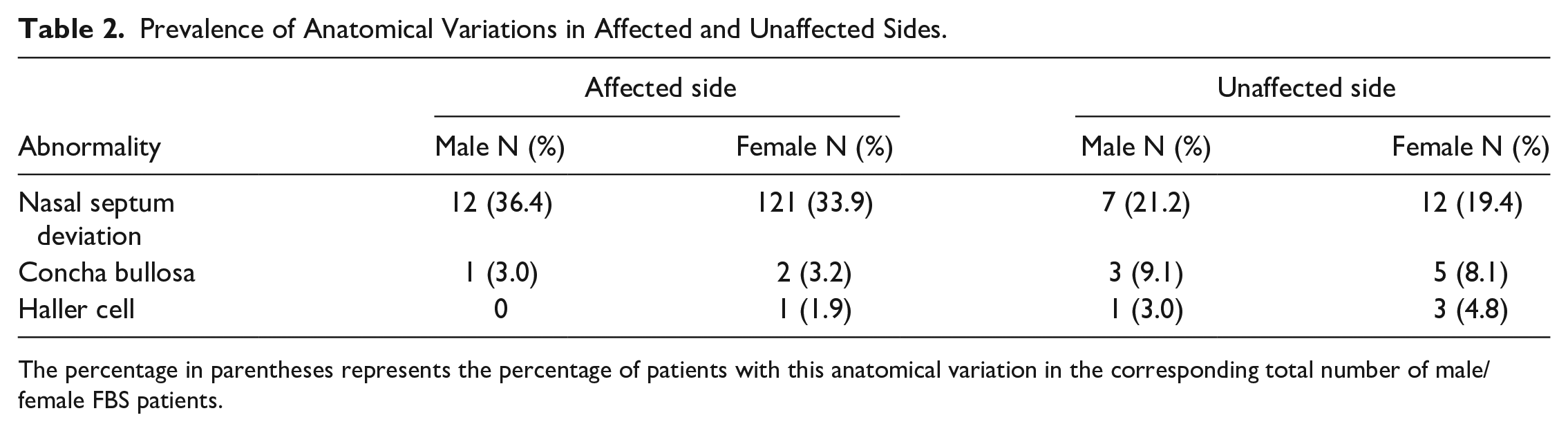
The percentage in parentheses represents the percentage of patients with this anatomical variation in the corresponding total number of male/female FBS patients.
We compared the affected and unaffected sides of the nasal cavity, respectively, as the observation and control groups. Significant differences were found in the NR, MCA, sMCA, and NCV between these 2 groups (P < .05, see Table 3). Moreover, based on the comparison of the average values of NR, sMCA, MCA, and NCV between 2 sets of 95 paired samples, we conducted a power analysis and found that: the actual power of NR, sMCA, and NCV remained above 0.9, but unfortunately, the actual power of MCA was only 0.6 (see Table 3). This low power of MCA implies that the statistical results should be interpreted with caution. To further validate our findings, we conducted a multivariate logistic analysis, which included controlling for sex and age. Specifically, we analyzed the ratios of bilateral NR, MCA, and sMCA, as well as the difference in bilateral NCV. The results showed that a higher left-to-right NR ratio was associated with an increased likelihood of FBs being present in the left sinus (OR = 0.185; 95% CI, 0.061−0.558; P < .01). Additionally, when the left-to-right sMCA ratio was higher, and the FB was more in the right sinus (OR = 3.194; 95% CI, 1.593−6.405; P = .001). Moreover, when the difference between left and right NCV was greater and FB occurred more commonly in the right sinus (OR = 1.435; 95% CI, 1.196-1.721; P < .001). However, there was no correlation found between the left-to-right MCA ratio and FB localization (see Table 4).
Summary of Nasal Resistance and Acoustic Rhinometry.
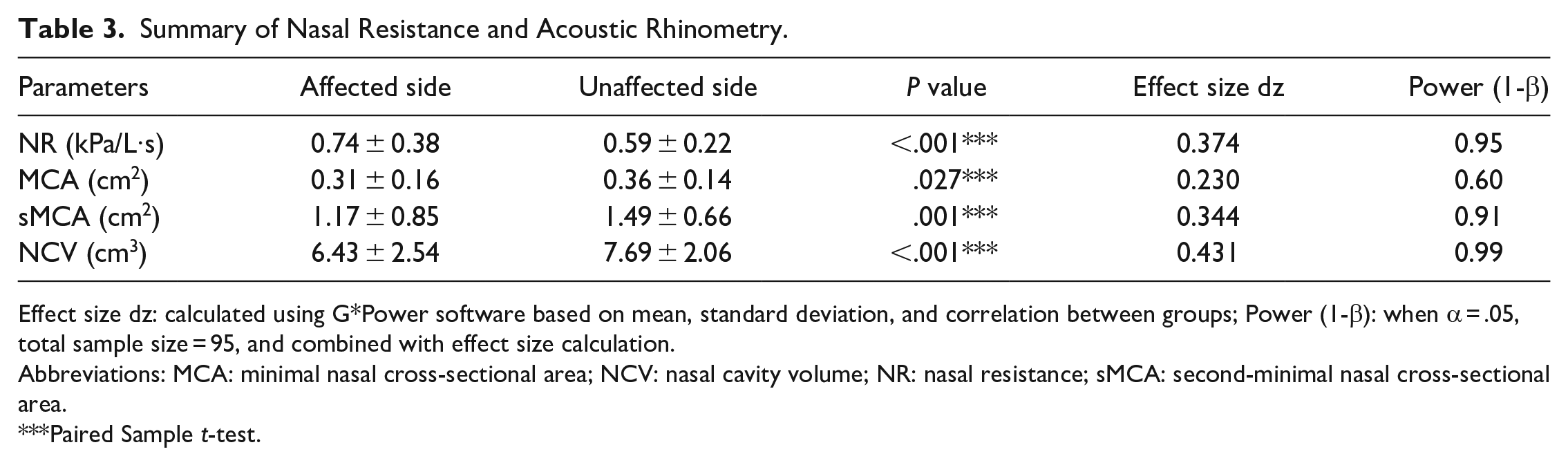
Effect size dz: calculated using G*Power software based on mean, standard deviation, and correlation between groups; Power (1-β): when α = .05, total sample size = 95, and combined with effect size calculation.
Abbreviations: MCA: minimal nasal cross-sectional area; NCV: nasal cavity volume; NR: nasal resistance; sMCA: second-minimal nasal cross-sectional area.
Paired Sample t-test.
Multivariate Analysis of Factors Affecting the Localization of FB.
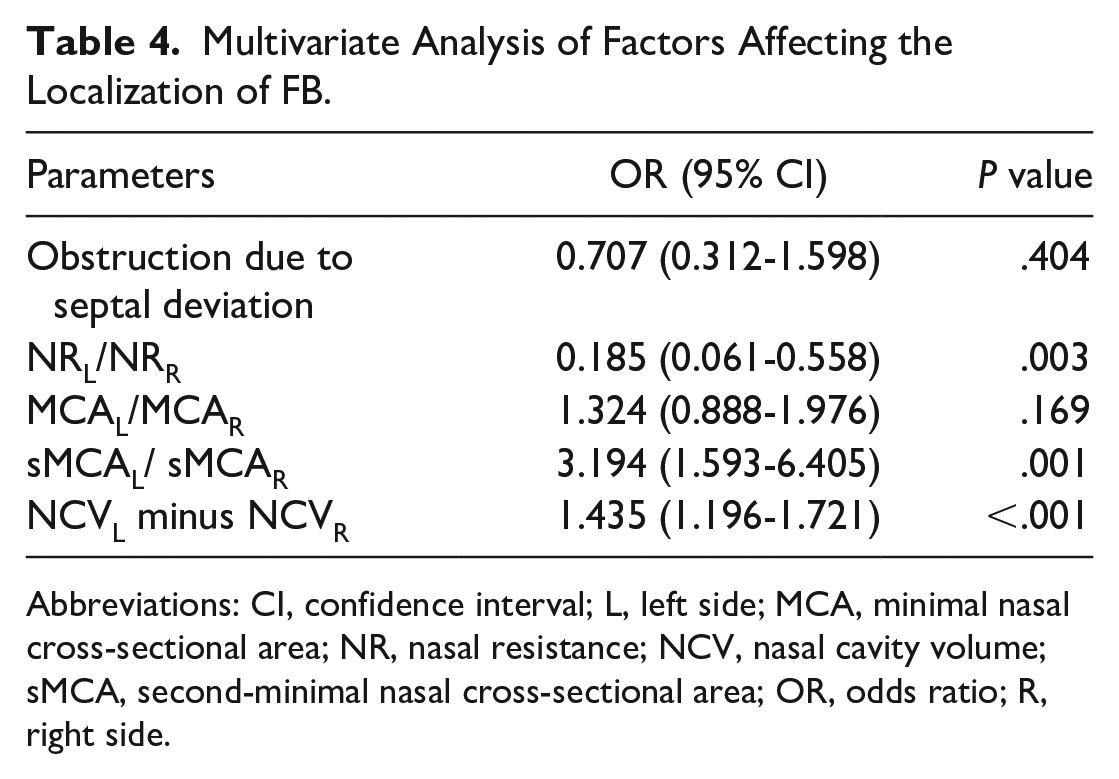
Abbreviations: CI, confidence interval; L, left side; MCA, minimal nasal cross-sectional area; NR, nasal resistance; NCV, nasal cavity volume; sMCA, second-minimal nasal cross-sectional area; OR, odds ratio; R, right side.
Discussion
FBS tends to occur in patients with normal immunity, and there is a higher incidence of FBS in females with a ratio as high as 2:1,5,10 which is consistent with our findings. Environmental and hormonal factors may be involved in FB pathogenesis,17-19 but this remains unclear. FBs are commonly found in a single sinus, with the maxillary sinus being the most frequent location, followed by the sphenoid sinus.10,18,20 Isolated opacification of the sphenoid sinus is observed in <5% of patients with paranasal sinus disease. 21 Primary isolated FBs of the ethmoid and frontal sinuses are rare and infrequently reported to grow in the concha bullosa.22,23 The different incidences above may be due to differences in ventilation of each sinus caused by the anatomical position; however, this is only conjecture.
FBS Pathogenesis
FB pathogenesis remains unclear, but 2 possible routes of entry have been suggested: the so-called “aerogenic” pathway in which high quantities of airborne fungal spores enter the sinus through the natural ostium, and the odontogenic “iatrogenic” pathway in which fungal colonization of the maxillary sinus occurs through iatrogenic oroantral communication secondary to dental extraction.8,9,24,25 The fungal infection in the maxillary sinus may be caused by the destruction of its bottom wall mucosal barrier function due to chemical and physical trauma. However, this theory cannot account for FBs arising in remote sinus cavities. Therefore, other factors may also contribute to FB formation. It is believed that the closure of the ostium creates anaerobic and low-pH environments that promote fungal growth and transform saprophytic colonization into a pathological form. 26
Disputes Over the Correlation Between Anatomical Variation and FBS
Most local anatomical variants are potential risk factors for FB formation.6-9 Septal deviation, along with other variants, including concha bullosa, has been implicated in infundibulum length compared with normal controls. 7 However, a repeat study failed to replicate this association with the septum and instead suggested an association with deviation away from the involved side. 6 Supporters believe that poor sinus ventilation is associated with disease development and that ventilation is influenced by anatomical variation in the paranasal sinuses, such as sinonasal anatomical variations, especially the presence of a concha bullosa, and/or a narrow and long infundibulum. Additionally, the consequences of the traumatic effects caused by the wall shear stress of the high-velocity airflow and the increased chance of inhaling fungal spores are considered the pathogenesis of FBs. 6 However, a large cross-sectional study involving 538 patients with FBS found no correlation among septal deviation, Haller cells, or concha bullosa when using the unaffected side as a control. 10 With the above reports, the European Position Paper on Rhinosinusitis and Nasal Polyps believes that anatomical variants are not a major contributor to FB formation. In this study, we observed that patients with FBS with nasal septal deviation had a higher prevalence of convex surface on the affected side compared to the unaffected side. Whereas, the concha bullosa and Haller cells were more commonly found on the unaffected side than on the affected side. However, our multivariate logistic regression analysis did not reveal any correlation between these anatomical variations and FB localization.
Correlation Between Nasal Ventilation and FBS
Anaerobic and low-pH environments are believed to favor fungal growth, and pathogenesis is enhanced by anaerobic conditions in poorly ventilated sinus cavities.26,27 Examination of NR combined with AR can objectively reflect the patency and geometric shape of the nasal cavity, which allows for detection of obstruction levels. A study found that patients with FBS had significantly different NR, NCV, and MCA parameters compared to normal volunteers: the NR increased while the NCV and MCA decreased; the distance of the MCA from the nostril appeared to move backward compared with normal volunteers, and the above differences may be risk factors for FBs. 28 We have reviewed multiple studies, but only few studies have investigated the correlation between FBS incidence and its risk factors, such as NR and NCV.
During this study, 95 patients with FBS underwent nasal resistance measurement and AR to record the NR, MCA, sMCA, and NCV of both the affected and unaffected sides. The affected and unaffected sides were used as the observation and control groups, respectively, for statistical analyses. We found that the NR in the observation group was significantly greater than that in the control group (P < .001), and the sMCA and NCV in the observation group were significantly lower than those in the control group (P < .01). Simultaneously, we performed a multivariate logistic analysis on the correlation between the above observation indicators, combined with the sex and age of the patients, and FB localization. Our finding indicates that FBs tend to occur on sides with higher NR and lower sMCA or NCV (P < .01). These findings suggest that differences in the bilateral NR and NCA may play a crucial role in FB formation. We believe that there are 3 possible reasons for this conclusion: (1) an increase in NR and a decrease in NCV may lead to reduced nasal ventilation, resulting in a low-ventilation state of nasal cavity that is conducive to anaerobic fungal growth; (2) differences in bilateral NR and NCV may also cause variations in the number of fungi inhaled into the nasal cavity or sinuses; and (3) multivariate analysis revealed no difference in the MCA between the observation and control groups, while a significant difference was observed in the sMCA located in the posterior segment of the nasal cavity. This suggests that the NCA located near the head of the middle nasal concha has a more significant impact. Owing to the presence of a small NCA, a high-velocity airflow may form locally during breathing, leading to traumatic effects caused by wall shear stress and ultimately FBs growth. Our team is developing a computer model to visualize nasal airflow velocity, direction, and flow status at different positions in the nasal cavity. This technology may be useful for measuring and understanding the role of nasal ventilation in paranasal sinus diseases.
Clinical Significance
Assuming that our conjecture is correct, it could have significant value in the treatment of patients with FBS. While functional endoscopic sinus surgery (FESS) remains the primary treatment for FBS and advancements in endoscopy and surgical techniques have significantly reduced recurrence rates, evidence suggests that persistent inflammation and recurrence can still occur. Two studies with longer follow-up have reported a recurrence rate of 3.2% and 4%.5,29 Despite this, many researchers still advocate simple maxillary sinus natural ostium opening for the treatment of maxillary sinus FBs. However, posttreatment inflammation was observed in 50% of patients with FB, while 17.9% experienced mucus deposition and 14% had residual debris after undergoing a simple antrostomy. Although an antrostomy was combined with an inferior meatal window, none had residual debris or inflammation.30,31 Therefore, modified medial maxillectomy or FESS with both middle and inferior meatal antrostomies is an effective surgical option for removing maxillary sinus FBs and should be considered to achieve better outcomes.30-32 In this study, we conclude that in the surgical treatment of FBS, it is crucial to address issues affecting NR and NCV on the affected side. Although anatomical variations do not affect FB localization, correcting nasal septal deviation on the affected side and removing the concha bullosa and Haller cells can reduce NR and increase NCV on the affected side. These measures may be significant in preventing recurrence of FB.
Study Limitations
Being retrospective, several limitations of this study should be acknowledged. The retrospective analysis may have excluded cases with incomplete data, which could potentially impact the actual results. Additionally, due to equipment updates and parameter changes, we have not reviewed a significant number of previous cases. These reasons led to the low effect size and power of paired t-test for bilateral MCA differences in patients of this study, indicating that more data may be necessary to demonstrate this in the future. During this study, we excluded patients with recurrent FBS as they constitute a distinct group. Patients with FBS treated with FESS may experience changes in mucosal ciliary function that can impact FB colonization, which is the next step we need to take. Finally, long-term postoperative follow-up was not investigated in this study, which resulted in our inability to obtain information on postoperative complications in FBS patients and verify our conclusions. Therefore, conducting a prospective study is our next task.
Conclusion
There is no significant correlation between the presence of FBs and anatomical variations such as nasal septal deviation, concha bullosa, or Haller cells. However, increased NR and decreased NCA and volume may play important roles in FB localization. These findings can guide the selection of surgical methods for patients with FBS.
Footnotes
Correction (November 2023):
Article updated to correct Affiliation 1.
Author Contributions
Tao Xu: data curation; statistical analysis; investigation; data curation; and writing original draft; Yin-Feng Wang: conceptualization and funding acquisition; Xiao-Tao Guo: statistical analysis; Ya-lin Wang: nasal resistance and acoustic rhinometry; and Jing Luo: conceptualization; data curation; and review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Natural Science Foundation of Anhui Province (1808085MH294).
Ethical Approval
The protocols and experimental procedures in the present study were reviewed by the Ethics Committee of the First Affiliated Hospital of the University of Science and Technology of China, and ethical approval documents were obtained (2023−RE−086, March 17, 2023).
Data Availability Statements
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
