Abstract
Keywords
Introduction
Laryngotracheal stenosis (LTS) is characterized by an abnormal decrease in the diameter of the upper airway between the larynx and carina. It can have idiopathic or acquired causes, such as endotracheal intubation, tracheostomy, trauma, inflammation, autoimmunity, and tumors. 1 LTS is diagnosed based on the patient’s clinical history and endoscopic examination, with radiographic imaging and pulmonary function tests (PFTs) being adjunctive diagnostic tools. Specifically, spirometry reflects the functional aspects rather than the anatomic severity of LTS.1–4
LTS can be managed through the endoscopic or open surgical approach. The endoscopic approach may involve balloon dilation, cold or laser incision, adjunctive medication with wound-healing modifying agents such as mitomycin C (MMC), or intralesional steroid injections (ILSIs). 1 Endoscopic treatment is considered favorable for LTS with ongoing inflammation, lack of cartilage involvement, or idiopathic type. Incisions into the stenotic soft tissue scar are made through the whole thickness of the scar, saving the normal mucosal lumen. It is a less invasive procedure with lower morbidity compared to the open approach. 5 Steroid use reduces scar proliferation, as well as collagen and glycosaminoglycan synthesis, and inhibits fibroblast growth, which allows scar softening.6–8 Additionally, office-based and intraoperative administration of ILSIs has been found to improve the peak expiratory flow (PEF) by 23.1% (95% CI 6.4%, 39.8%) and 25.1% (95% CI 9.59%, 40.69%), respectively, in patients with idiopathic subglottic stenosis (SGS). 9
The aforementioned management strategies seek to establish a patent airway without dyspnea. However, numerous patients develop restenosis requiring further interventions. A recent systematic review of endoscopic interventions for idiopathic SGS, including cold knife, laser, or dilation (balloon or rigid), in various combinations, with or without wound-healing modifying agents 10 reported that the stenosis recurrence rate ranged from 40 to 100%, with no endoscopic intervention showing superiority. CO2 laser incision monotherapy had the highest recurrence rate (100%) and shortest between-intervention interval (2 months). Contrastingly, ILSIs combined with rigid dilation showed the lowest recurrence rate (40%). Although combined therapy involving balloon dilation, MMC, and ILSIs resulted in a high recurrence rate of 80%, it showed the longest between-intervention interval (21.2 months). However, this was attributed to the prolonged follow-up duration for this intervention compared with other interventions. The existing literature shows the heterogeneity of different endoscopic interventions used in various combinations; these combinations have different recurrence rates with no intervention being obviously superior over the others.
Therefore, we aimed to compare the improvement in PFT parameters following endoscopic balloon dilation in patients with acquired LTS between those who did and those who did not receive ILSIs. Additionally, we aimed to compare the recurrence times and rates between the groups.
Patients and Methods
This retrospective study was conducted at King Saud University Medical City in Riyadh, Saudi Arabia, and approved by the Institutional Review Board of its College of Medicine (Project No.
Statistical Analyses
Statistical analyses were performed using the SPSS program 26 (IBM Corp., Armonk, NY, USA). Between-group comparisons (with ILSIs vs without ILSIs) of continuous and categorical variables were performed using an independent-sample t-test and Fisher’s exact test, respectively. Paired-sample t-tests were used for within-group comparisons of pre- and postoperative measurements. Logistic regression analysis was used for predictors of improvement in PFT. The level of significance was α = .05 for all tests. The normality of continuous data was assessed using a Shapiro–Wilk test. Categorical variables were analyzed using the chi-squared test to determine statistical significance in cross-tabulations. In cases of continuous variables, an unpaired Student’s t-test was employed for normally distributed variables, while the Mann–Whitney U test was used for non-normally distributed variables.
Results
Table 1 presents the demographic characteristics, including the number of cases, sex, smoking status, lung condition, presence or absence of comorbidities, etiology of stenosis, symptom recurrence, type of anesthesia, dilation time, and Cotton–Meyer classification.
Demographic and Clinical Characteristics.
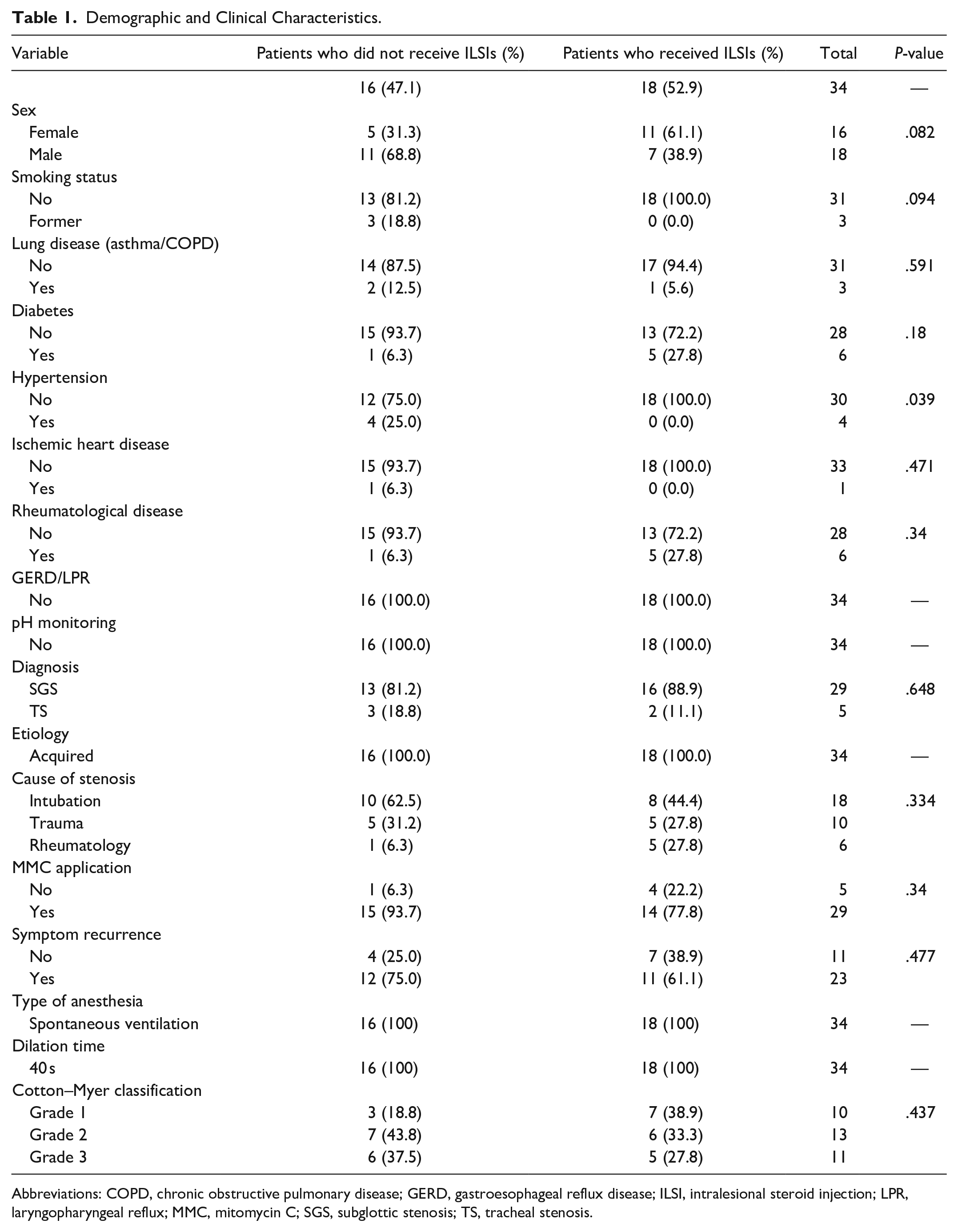
Abbreviations: COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; ILSI, intralesional steroid injection; LPR, laryngopharyngeal reflux; MMC, mitomycin C; SGS, subglottic stenosis; TS, tracheal stenosis.
The group of patients who received ILSIs consisted of 18 individuals, 11 (61.1%) females and 7 (38.9%) males. In contrast, among the 16 patients who did not receive ILSIs, 5 (31.3%) were female and 11 (68.8%) were male. However, the difference in the proportion of males and females between these 2 groups was not statistically significant.
SGS and tracheal stenosis (TS) were diagnosed in 29 (85.3%) and 5 (14.7%) patients, respectively. The main cause of stenosis was intubation (52.9%), followed by trauma (29.4%) and rheumatological causes (17.7%).
ILSIs and MMC were administered to 52.9 and 85.3% of patients, respectively. Among the 18 patients who received ILSIs, symptom recurrence was reported in 11 cases (61.1%). In the group of 16 patients who did not receive ILSIs, symptom recurrence was reported in 12 cases (75.0%). The overall recurrence rate was 67.6%. However, there was no significant difference in the proportions of symptom recurrence between the 2 groups. The calculated between-group difference in the proportion of symptom recurrence was −0.1389 [95% confidence interval (CI): −0.4483, 0.1705].
All patients (n = 34) received spontaneous ventilation, among which 18 received ILSIs and 16 did not receive ILSIs. Similarly, the dilation time of all patients was 40 seconds. According to Cotton–Myer classification, 10 patients were in grade 1, 13 in grade 2, and 11 in grade 3 categories. No significant differences were observed between those who received ILSIs and those who did not receive ILSIs (P = .437).
Patients who did not receive ILSIs were significantly taller than those who received ILSIs (165.22 cm ± 7.42 vs 158.94 cm ± 9.46; P = .041). No significant between-group differences in age, weight, percentage of stenosis, stenosis length, or distance of stenosis from the vocal cords were observed. The mean (standard deviation) time to first reintervention was 8.62 (8.00) and 7.38 (3.20) months for patients who did and those who did not receive ILSIs, respectively (P =.614, 95% CI −6.30, 3.84; Table 2).
Summary Statistics of Demographic and Clinical Characteristics.
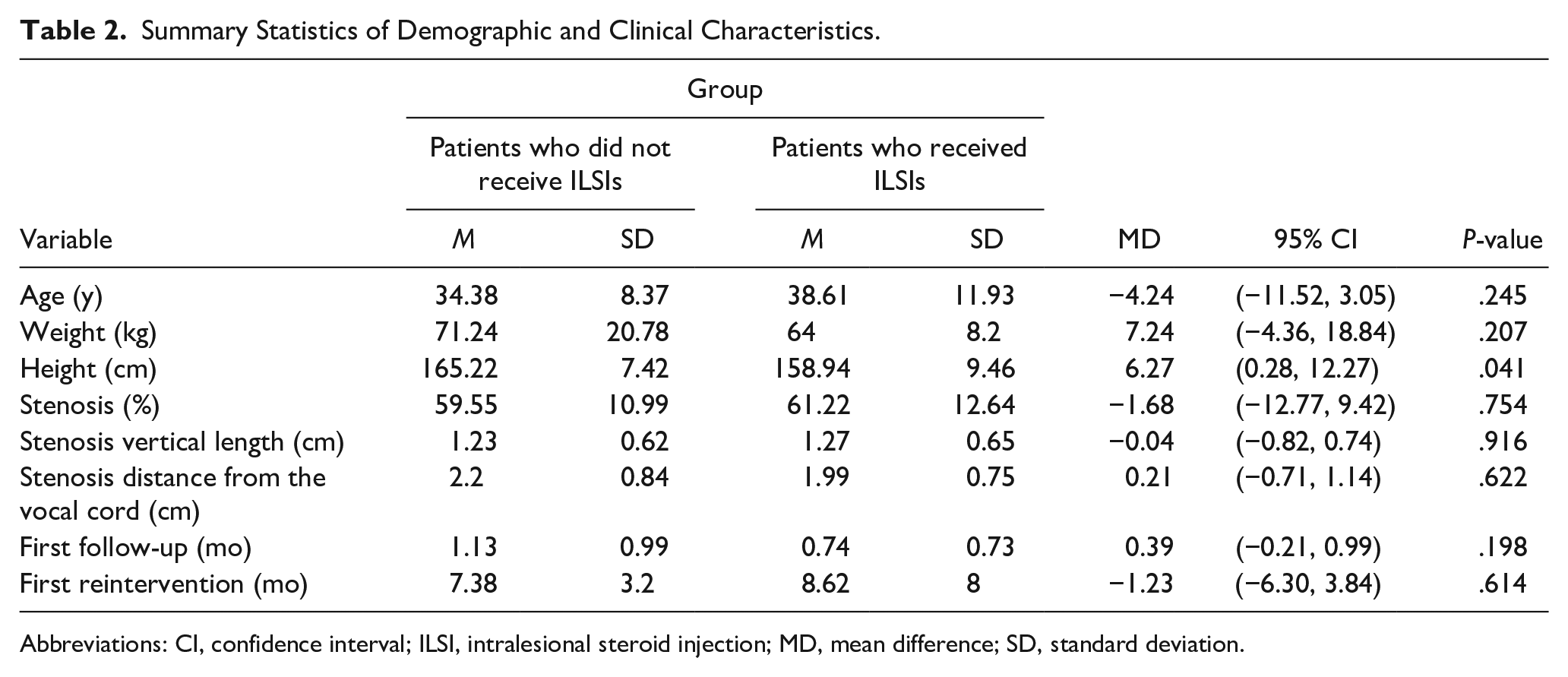
Abbreviations: CI, confidence interval; ILSI, intralesional steroid injection; MD, mean difference; SD, standard deviation.
Tables 3 and 4 show the pre- and postoperative PFT parameters, respectively. Patients who did not receive ILSIs had significantly higher preoperative FEV1 than those who received ILSIs (2.09 ± 0.58 vs 1.59 ± 0.72, mean difference 0.50, P = .035, 95% CI 0.04, 0.96). There were no significant between-group differences in the preoperative PEF; FEV1/PEF; 25%, 50%, and 75% FEF; MMEF; and FVC. Moreover, there were no significant between-group differences in all postoperative measurements (P > .05).
Between-Group Comparisons of Preoperative PFT Parameters.
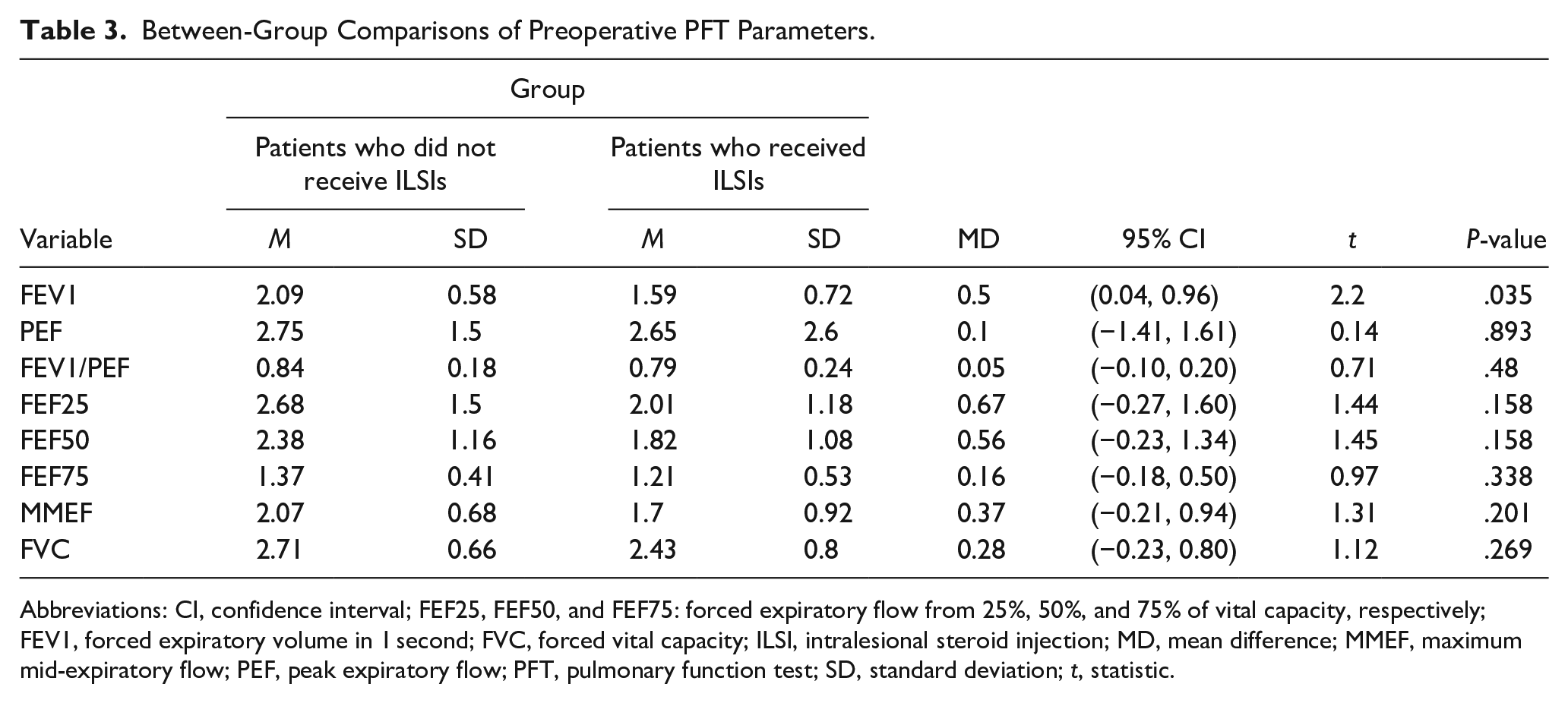
Abbreviations: CI, confidence interval; FEF25, FEF50, and FEF75: forced expiratory flow from 25%, 50%, and 75% of vital capacity, respectively; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ILSI, intralesional steroid injection; MD, mean difference; MMEF, maximum mid-expiratory flow; PEF, peak expiratory flow; PFT, pulmonary function test; SD, standard deviation; t, statistic.
Between-Group Comparisons of Postoperative PFT Parameters.
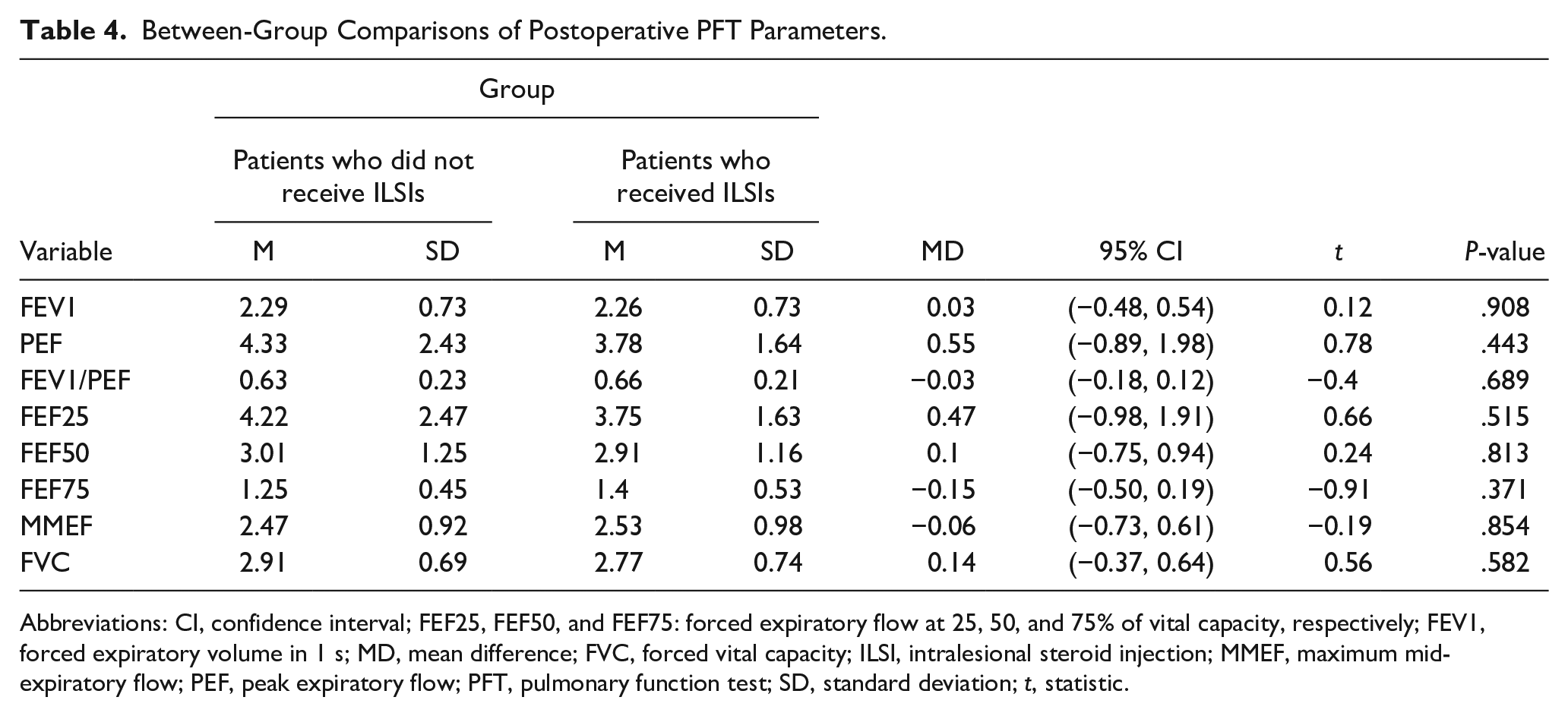
Abbreviations: CI, confidence interval; FEF25, FEF50, and FEF75: forced expiratory flow at 25, 50, and 75% of vital capacity, respectively; FEV1, forced expiratory volume in 1 s; MD, mean difference; FVC, forced vital capacity; ILSI, intralesional steroid injection; MMEF, maximum mid-expiratory flow; PEF, peak expiratory flow; PFT, pulmonary function test; SD, standard deviation; t, statistic.
Among patients who did not receive ILSIs, compared with the preoperative values, the postoperative PEF (Mpre = 2.75 ± 1.50 vs Mpost = 4.33 ± 2.43) and FEF25 (Mpre = 2.68 ± 1.50 vs Mpost = 4.22 ± 2.47) were significantly higher (P = .005 and .008, respectively), while the postoperative FEV1/PEF values (Mpre = 0.84 ± 0.18 vs Mpost = 0.63 ± 0.23) were significantly lower (P = .008). There were no significant within-group differences in the FEV1, FEF50, FEF75, MMEF, or FVC (P = .139, 0.068, .379, .052 and .209, respectively; Table 5).
Within-Group Comparisons (Pre- and Post-Surgery Measurements).
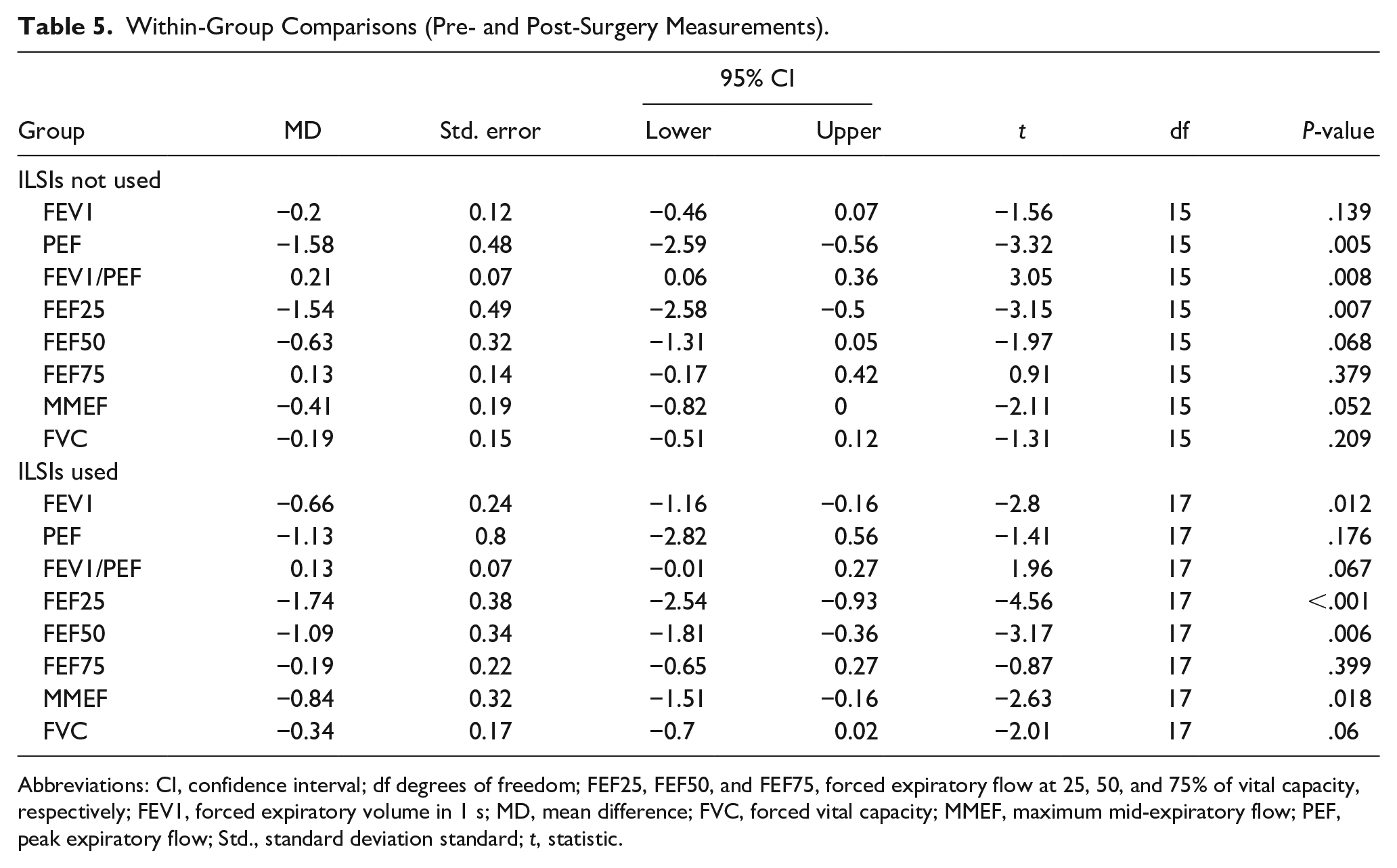
Abbreviations: CI, confidence interval; df degrees of freedom; FEF25, FEF50, and FEF75, forced expiratory flow at 25, 50, and 75% of vital capacity, respectively; FEV1, forced expiratory volume in 1 s; MD, mean difference; FVC, forced vital capacity; MMEF, maximum mid-expiratory flow; PEF, peak expiratory flow; Std., standard deviation standard; t, statistic.
Table 5 shows the results of within-group comparisons of pre- and postoperative measurements.
Among patients who received ILSIs, compared with the preoperative values, the postoperative FEV1 (Mpre = 1.59 ± 0.72 vs Mpost = 2.26 ± 0.73), FEF25 (Mpre = 2.01 ± 1.18 vs Mpost = 3.75 ± 1.63), FEF50 (Mpre = 1.82 ± 1.08 vs Mpost = 2.91 ± 1.16), and MMEF (Mpre = 1.70 ± 0.92 vs Mpost = 2.53 ± 0.98) were significantly higher (P = .012, .001, .006, and .018, respectively).
The pre-post difference in PFT parameters was calculated by subtracting the pre-PFT values from the post-PFT values (Table 6).
Comparison of the Pre-Post Differences in PFT Between Groups.
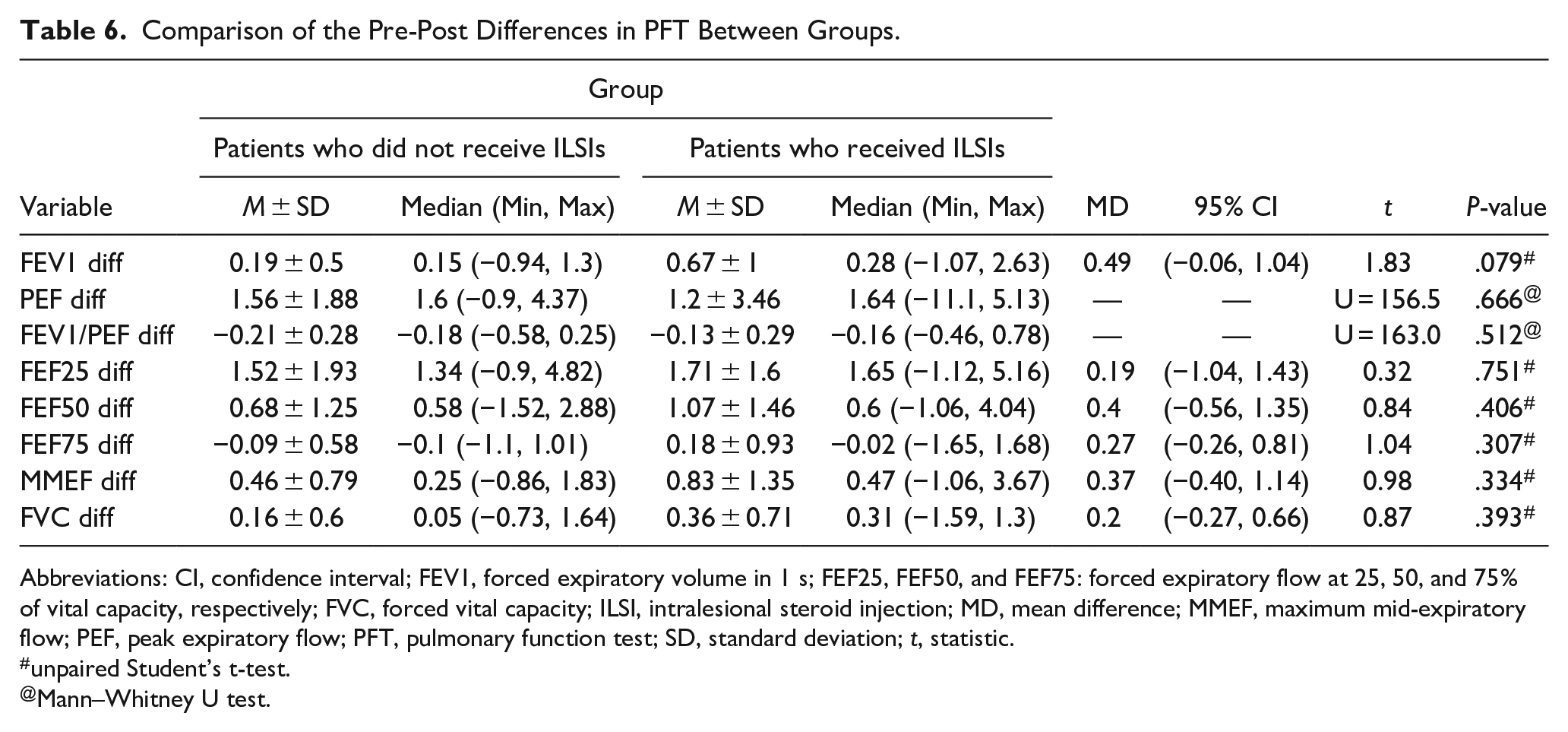
Abbreviations: CI, confidence interval; FEV1, forced expiratory volume in 1 s; FEF25, FEF50, and FEF75: forced expiratory flow at 25, 50, and 75% of vital capacity, respectively; FVC, forced vital capacity; ILSI, intralesional steroid injection; MD, mean difference; MMEF, maximum mid-expiratory flow; PEF, peak expiratory flow; PFT, pulmonary function test; SD, standard deviation; t, statistic.
unpaired Student’s t-test.
Mann–Whitney U test.
Except for the PEF diff variable, all other PFT parameters showed a higher mean difference in patients who received ILSIs than in those who did not. However, this difference was not statistically significant.
Logistic regression analysis was conducted to determine predictors for PFT improvement, which was defined as the difference between the pre- and postoperative FEV1 values. The median difference in FEV1 values was calculated, and values equal to or above the median value were considered to show improvement in PFT. None of the predictive variables included in the regression model showed statistical significance (Table 7).
Logistic Regression Analysis for Predictors of Improvement in PFT.
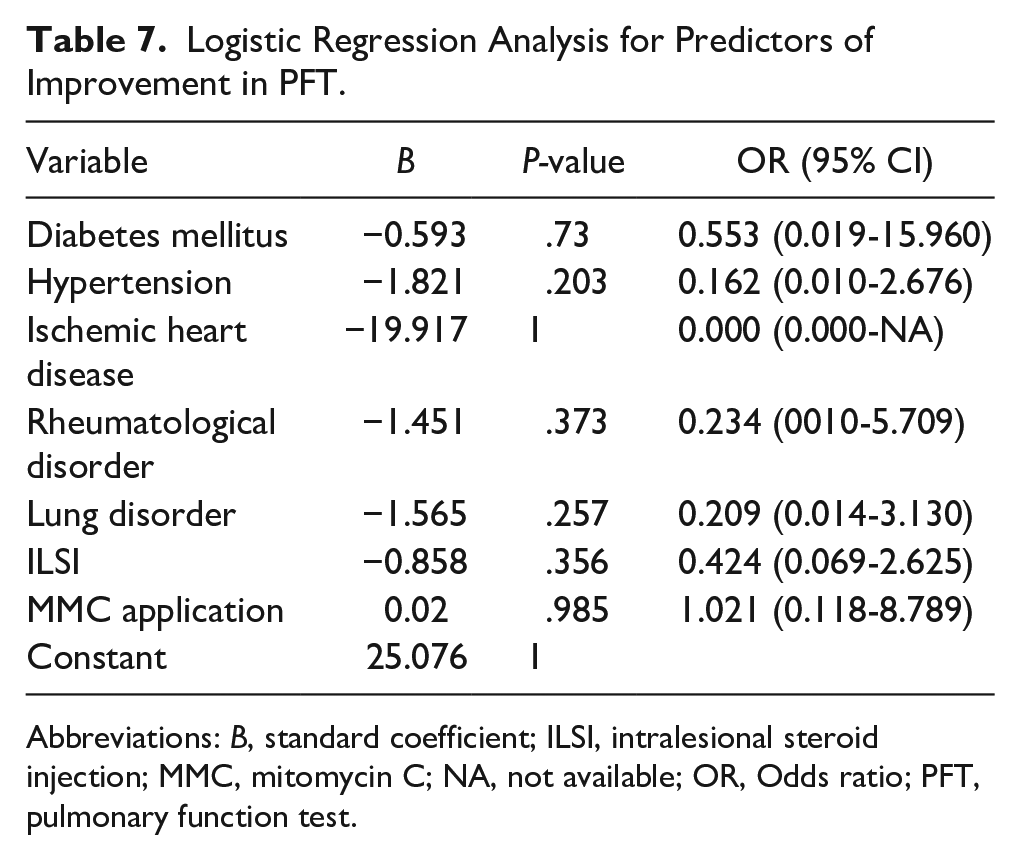
Abbreviations: B, standard coefficient; ILSI, intralesional steroid injection; MMC, mitomycin C; NA, not available; OR, Odds ratio; PFT, pulmonary function test.
Discussion
We observed postoperative improvements after endoscopic balloon dilation in several PFT parameters, including the FEV1, PEF, FEV1/PEF, FEF25, FEF50, and MMEF, consistent with previous related reports.3,11,12 Additionally, the FEV1 was significantly higher under ILSI administration, which is consistent with that observed in the aforementioned reports.
In patients with acquired SGS, including those with an idiopathic etiology, endoscopic dilation improves PFT outcomes, such as the FEV1, PEF, FEV1/PEF, FEF25, FEF50, and MMEF.13–15 Hseu et al. reported that PEF, total peak flow, and FEV1/PEF were the most sensitive and specific parameters for monitoring the effectiveness of an intervention. 16 Similarly, our findings showed improvement in FEV1, FEF25, FEF50, and MMEF among patients who received ILSI, but without statistical significance. The significant improvement observed in PEF and FEF25 in patients who did not receive ILSI might be due to the selection of patients with less severe SGS.
Moreover, the present study showed that in terms of the pre- and postoperative PFT difference, all PFT parameters except PEF showed improvements in mean difference in patients who received ILSIs compared to those who did not. However, this still did not suggest a favorable intervention (all P > .05). Other studies have evaluated the efficacy of PEF as a reliable predictor of disease worsening and the need for an intervention. However, PEF was shown to be less useful than other parameters for the initial diagnosis of SGS.14,17,18
The lack of improvement in PFT using balloon dilation with concurrent ILSI compared to simple dilation could be due to disease etiology or patients’ comorbidity. 19 Our results did not show any factor that predicts PFT improvement. In our study, the mean duration until the first postoperative follow-up visit was 0.74 to 1.13 months. Moreover, no significant between-group difference in the mean interval to the first intervention due to symptom recurrence was observed.
Consistent with our study, Feinstein et al. reported no significant difference in the mean between-intervention interval according to applied adjuvant measures among patients with SGS of various etiologies. 20 Additionally, Hseu et al. reported no significant difference in the mean between-intervention interval. 16 Moreover, office-based and intraoperative administration of ILSIs has been observed to extend the surgery-free post-intervention interval.21–23 Another study showed that among patients with autoimmune, idiopathic, and traumatic SGS, 68.9% showed improved endoscopic dilation following ILSI administration; however, there were no significant differences in the number of reinterventions required to achieve such an outcome. 24
There was no significant difference in the recurrence rate between patients who did or did not receive ILSIs (61.1% vs 75%, P = .477). The overall recurrence rate in our study was 67.6% (95% CI: −0.4483, 0.1705), with previously reported values ranging from 40% to 80%. 10 A randomized controlled trial reported that steroid inhalation improved PFT parameters; however, the recurrence rate was comparable between the inhaled steroid and control groups. 25
Limitations and Recommendations
This study had some limitations. First, this was a small-scale retrospective study. Second, the wide range of involved anatomical areas in patients with SGS and TS makes the data broad and vague. Finally, an additional objective measure other than PFT is required to assess surgical outcomes. A large-scale randomized controlled study is warranted to validate our findings.
Conclusion
PFT parameters improved following endoscopic balloon dilation with FEV1 being significantly high with concurrent ILSI. There was no between-method difference in the treatment effectiveness, although almost all PFT parameters showed a higher mean difference in patients who received ILSIs than in those who did not, with statistically nonsignificant results. Additionally, the restenosis recurrence rate was consistent with that reported in the literature, indicating that the disease is progressive and inflammatory, with no standardized treatment established yet.
Although patients with LTS show improvement and symptom-free intervals after endoscopic management, otolaryngologists still face difficulties in choosing the optimal intervention for these patients.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
