Abstract
Spontaneous intracranial hypotension is an uncommon but increasingly recognized condition characterized by an orthostatic headache secondary to low cerebrospinal fluid pressure. Vestibulocochlear symptoms are common but rarely the only presenting feature and can be challenging to differentiate from Meniere’s disease. We present a case series that highlights the common vestibulocochlear symptoms and a review of the literature to increase awareness amongst otolaryngologists and highlight the path to diagnosis and management of this condition
Keywords
Introduction
The classical neurological manifestation of spontaneous intracranial hypotension (SIH) is orthostatic headache, but the headaches can be non-orthostatic, exertional, valsalva related, or the syndrome may be acephalgic. Interscapular spinal pain, diplopia, visual field changes, cognitive and personality changes, gait unsteadiness, tremor, cervical radicular symptoms, and vestibulocochlear symptoms may occur. SIH clinical presentations to the otolaryngologist may be difficult to identify against the more common presentations of low-frequency sensorineural hearing loss, tinnitus, and vertigo like Meniere’s disease. In cases with appropriate management of SIH, these vestibulocochlear symptoms resolve.
SIH is an uncommon cause of orthostatic headache secondary to low cerebrospinal fluid, affecting approximately 2-5 per 100,000 people. Women are twice as commonly affected than men, with peak incidence occurring at 30-50 years of age. 1 SIH most commonly relates to spinal dural tears or a cerebrospinal fluid (CSF)–venous fistula, frequently within thoracic regions. Leakage of CSF leads to reduced CSF volume and the associated postural headache.1,2 The headache typically intensifies in the upright position and is relieved by lying flat; however, this is not true for all patients. These headaches are often coupled with, or even preceded by vestibulocochlear symptoms including tinnitus, aural fullness, hearing impairment, dizziness, or vertigo. A recent systematic review reported rates of hearing changes in approximately 70% of patients, which is an important consideration for the otolaryngologist. 1
There are only nine case reports in the otolaryngology literature highlighting the vestibulocochlear symptoms that accompany SIH.3-12 Sakano et al 6 reported low-frequency sensorineural hearing loss, vertigo and tinnitus, similar to Meniere’s disease. Additionally, however, there have been several case reports demonstrating low-frequency sensorineural hearing loss in patients with SIH.3,5,7-12 In some of those reports, consecutive audiograms showed an improvement in the recumbent position7,9,12 and resolution following treatment. Furthermore, tinnitus and aural fullness has been reported in several cases.3-6,10,12
Diagnosis of SIH can be difficult. In many but not all cases, the magnetic resonance imaging (MRI) brain (with gadolinium) will demonstrate all or some of the typical features for CSF hypovolemia including meningeal enhancement, sagging of the brain, pituitary enlargement, and subdural fluid collections. 2 If positive, then MRI spine imaging followed by referral to an interventional radiologist for consideration of a Dynamic CT Myelogram is the next step. Opening pressure is measured in almost all cases, and can be useful in the individual’s treatment journey. Identification of a bone spur, leaking enlarged perineural sleeve cyst or diverticulum or CSF–venous fistula allows targeted management. This is initially done by blood/fibrin patching, progressing to transvenous fistula embolization or surgical ligation, ablation or closure depending upon the lesion. 2 Cases with a strongly suggestive clinical history, but negative initial MRI present a greater diagnostic challenge and referral to a clinician with expertise in headache or an interventional radiologist is required.
This article reports a series of cases of SIH presenting with prominent vestibulocochlear symptoms, as would often present to an otolaryngologist. We review the literature in this area and highlight the clinical features that should raise suspicion of the diagnosis of SIH, and the pathway to diagnosis of this very treatable disorder.
Case Reports
Case 1
A 61-year-old female presented to an otolaryngologist with an 8 week history of imbalance, veering toward the right with intermittent right-sided otalgia. The patient denied any sensation of rotation, vertiginous symptoms, nausea, vomiting, or tinnitus. Her clinical examination was relatively unremarkable; however, she demonstrated significant disequilibrium. Her tympanograms were type A on right and negative type C on left. She had a normal cranial nerve examination and vestibulo-ocular reflex, as evident on a head impulse test. A referral to a vestibular physiotherapist and neurologist was made. Her vestibular physiotherapy assessment demonstrated significant imbalance, unable to walk unaided, elicited a right beat nystagmus upon valsalva, but symptoms intermittently resolved with a right sided Epley’s maneuvre.
The neurologist noted postural elements to her symptoms, relieved by lying flat and worsened throughout the day when upright. Further history noted a blunt injury to her mid thoracic region 3 months prior to her first episode of right ear fullness and imbalance. Subsequent episodes occurred a month later following a short haul flight and gradually became more frequent and progressed to include otalgia, aural fullness, and headache.
A cranial and total spinal MRI with contrast was performed demonstrating subtle cranial meningeal contrast enhancement, dural thickening (Figure 1), and pituitary engorgement (Figure 2). In the thoracic region, perineural cysts were noted at T6/7 and T7/8 bilaterally and right T11/12. Subsequent referral was made to an interventional neuroradiologist for a CT myelogram as a more definitive investigation and ongoing management. CT myelography did not demonstrate a clear leak. Initially, all levels were treated with targeted epidural blood patches. Subsequent focal patches were targeted to the T11/12 perineural cyst leading to surgical ligation and resolution of her symptoms. Four months post-surgery, she had recurrence of her symptoms. A repeat CT myelogram suspected a right T12 leak (Figure 3) and or fistula. At surgery she was found to have a prominent CSF–venous fistula, which was ligated. She has since had resolution of her symptoms and no recurrences in the last 18 months.
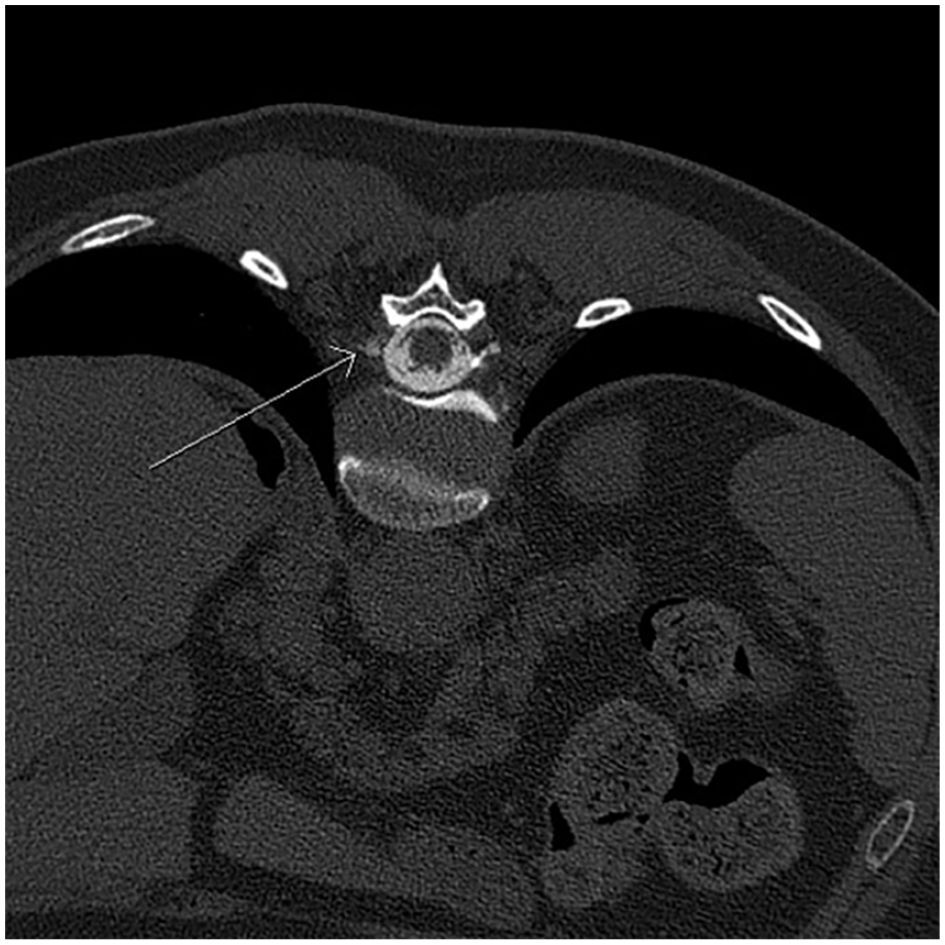
T12 vertebrae cerebrospinal fluid–venous fistula (arrow).
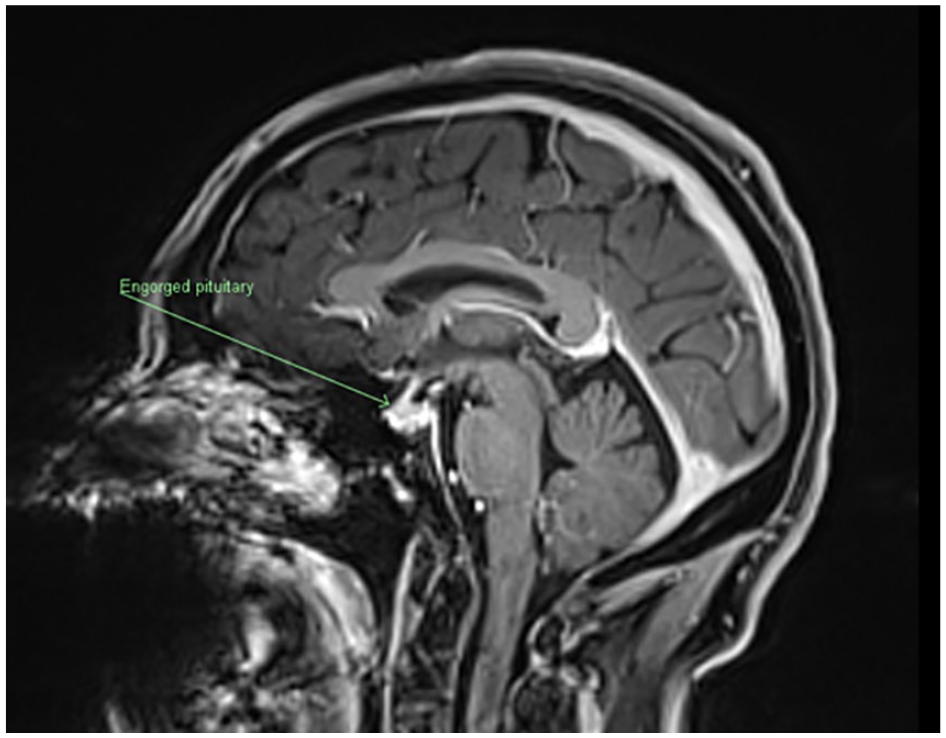
Magnetic resonance imaging brain demonstrating pituitary engorgement (arrow).
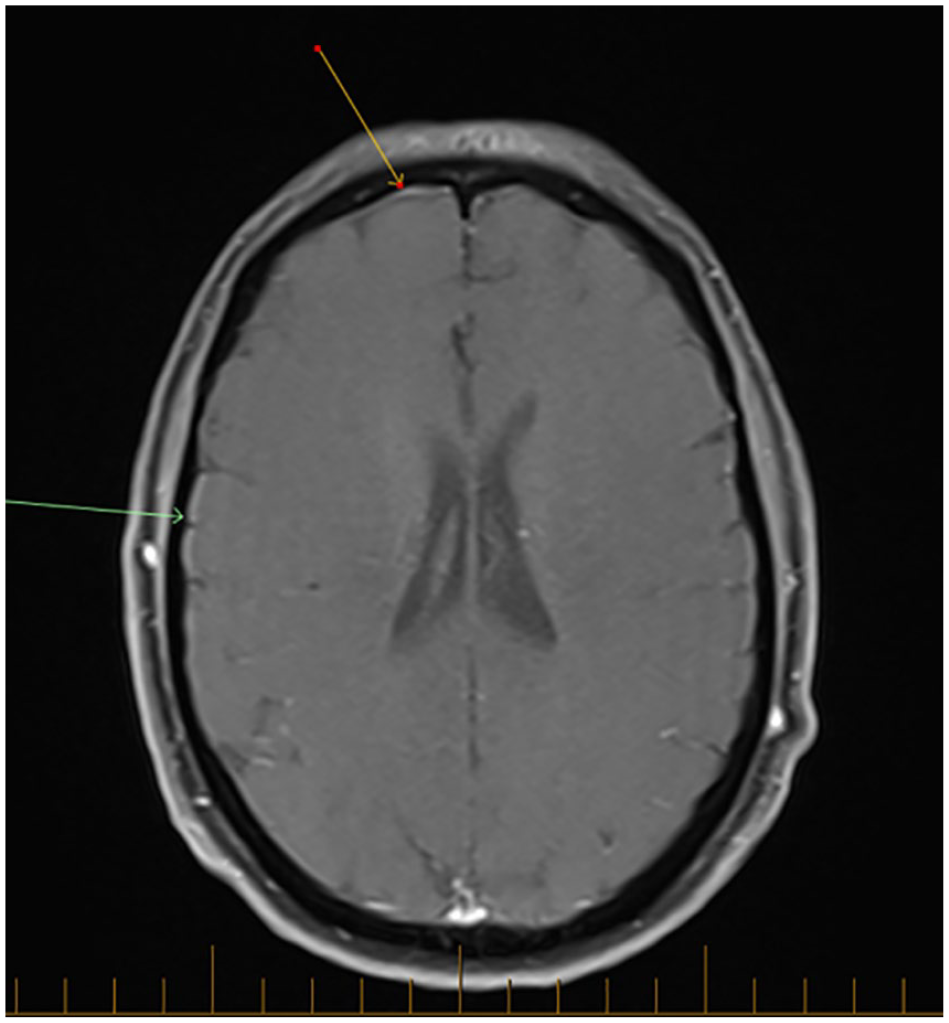
Magnetic resonance imaging brain demonstrating subtle meningeal thickening (arrows).
Case 2
A 76-year-old female presented with a 3-month history of fluctuating bilateral hearing loss, aural fullness and left-sided tinnitus with occasional generalized pressure headache, and left-sided aural fullness progressing over time to bilateral. She reported intermittent vertiginous episodes and pulsatile tinnitus. There was no postural element to her symptoms, even reporting occasionally waking with headache. She had a past medical history of hypertension, hypothyroidism, and gastro-esophageal reflux disease. She sustained a whiplash injury 40 years prior and concussion aged 13. Her otological and neurological examinations were normal, and she was systemically well. An audiogram, demonstrated bilaterally symmetrical sensorineural hearing loss (Figure 4). A cranial MRI was suggestive of intracranial hypotension with dural meningeal enhancement and thickening. Her CT myelogram demonstrated an opening CSF pressure of 11.5 cm H2O (normal pressures 10-18 cm H2O). A fibrin patch to a small T8 diverticulum did not resolve her symptoms. A repeat myelogram demonstrated a CSF–venous fistula at her right T8 nerve root. She was subsequently referred for neurosurgical intervention and had significant improvement in her symptoms.
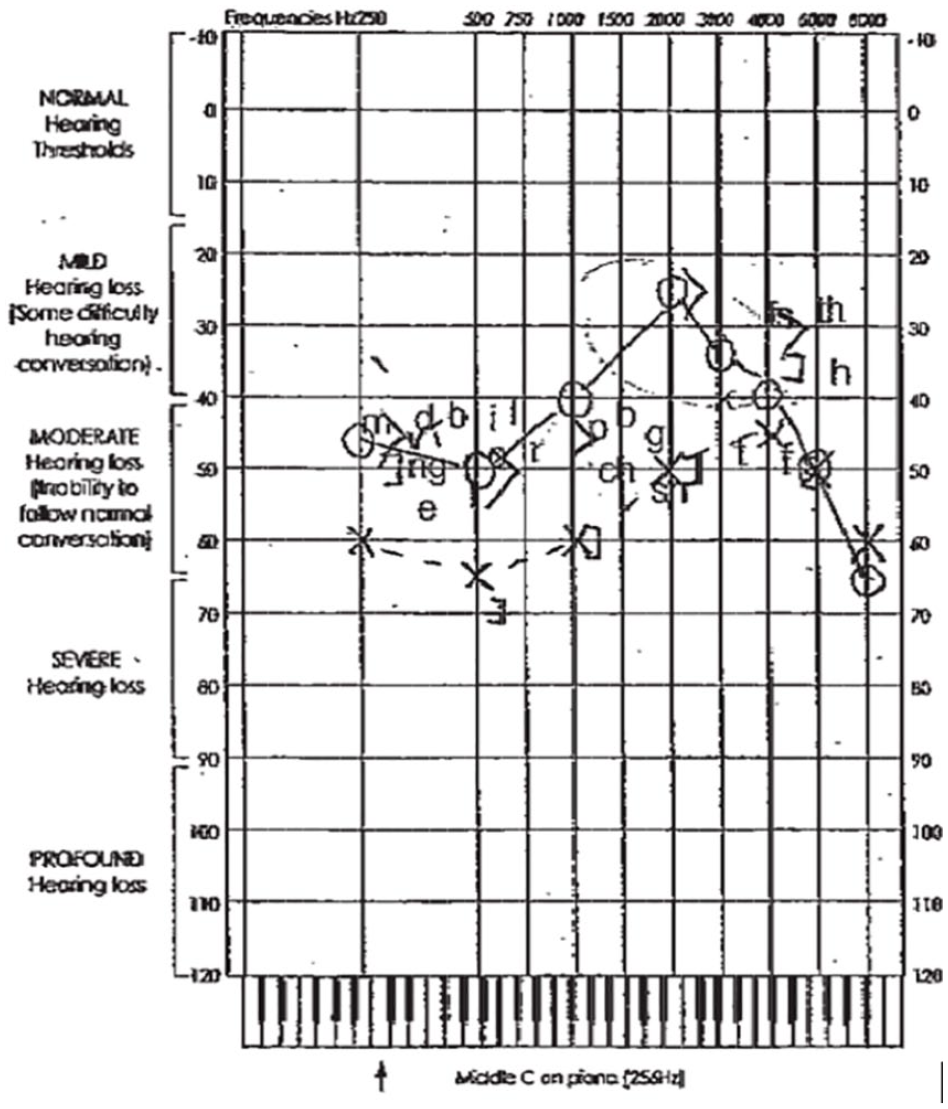
Audiogram demonstrating bilateral sensorineural hearing loss.
Case 3
A 66-year-old female presented to the otolaryngologist with an 8 month history of sudden onset left-sided tinnitus, with intermittent right-sided aural fullness, deafness, and postural instability. Her symptoms did not vary with posture, and she denied any headache. Her clinical examination was unremarkable, and her pure tone audiogram was normal with bilateral type A tympanograms. A cranial MRI demonstrated features of SIH including dural thickening, engorgement of the dural venous sinus, and pituitary gland (Figure 5). Spinal MRI revealed several enlarged perineural sleeve cysts within her thoracic spine, particularly on the right at T5 and T6. Her CSF opening pressure was low at 8.5 cm H2O, and a CSF–venous fistula (Figure 6) was noted on CT myelography. Her symptoms resolved with embolization to close the fistula.
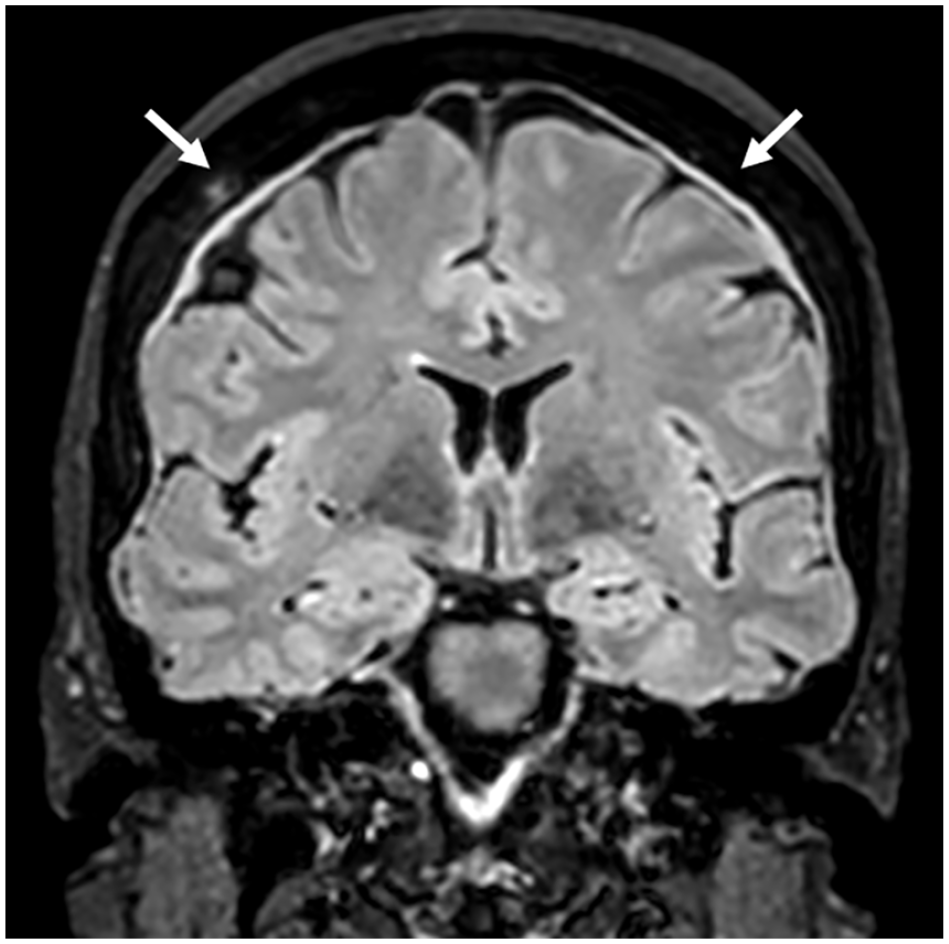
Magnetic resonance imaging demonstrating dural thickening (white arrows).
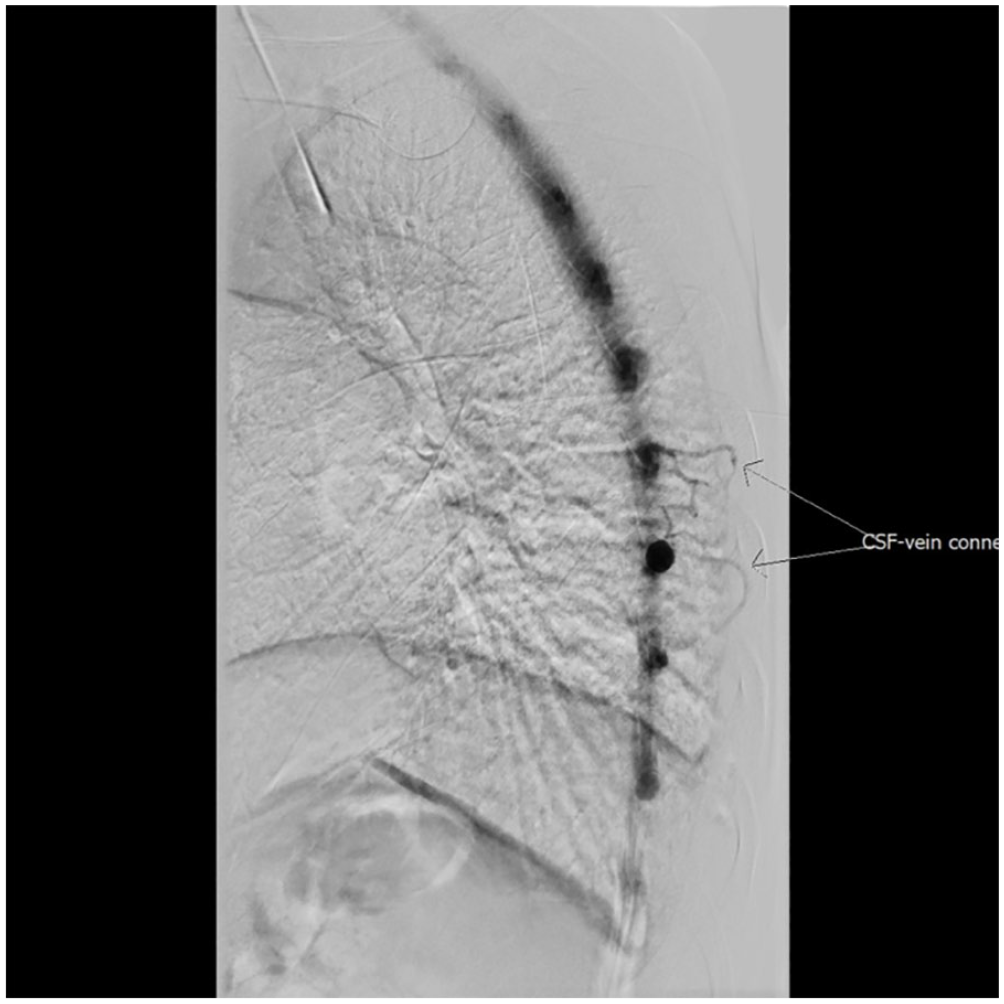
Computerized tomography demonstrating cerebrospinal fluid–venous fistula (arrows).
Case 4
A healthy 58-year-old female presented with several month history of low pitched tinnitus with occipital headache and intermittent vertigo. It was noted that her headaches were worse with bending forward. There was no associated nausea and no reported history of trauma. A cranial MRI demonstrated dural thickening without any obvious spinal CSF leak. At CT myelogram, the CSF opening pressure was borderline low (10 cm H2O), and a CSF–venous fistula was identified at the right T9 nerve root. She was subsequently referred for transvenous embolization of the fistula with resolution of her symptoms.
Case 5
A 66-year-old lady presented to her neurologist with a 2 year history of progressive non-positional headache, brain fog, left-sided hearing loss, dysnomia, and dysguesia. A cranial MRI demonstrated significant brain sag, dural thickening, and venous distension. Spinal MRI did not reveal any epidural CSF collection, but a few lower thoracic perineural sleeve cysts were noted. She underwent CT myelogram, with an opening pressure of 9 cm H2O, demonstrating a CSF–venous fistula on her right T11 nerve root. Following transvenous embolization, she had resolution of her symptoms.
Case 6
A 71-year-old female presented with a 12-month history of low pressure postural headache with associated vertigo, left ear fullness, and visual disturbances. Of note her headache was worse when lying flat. She was investigated for acute confusion, vertigo, and memory loss and noted to have dural thickening and other features suggestive of a CSF leak and SIH (Figure 7). CT myelography revealed opening pressures were low (8 cm H2O), and a CSF–venous fistula was demonstrated at the right T6 nerve root alongside a large diverticulum (Figure 8). She was referred for transvenous embolization but eventually chose surgery (Figure 9) and has since had no further symptoms.
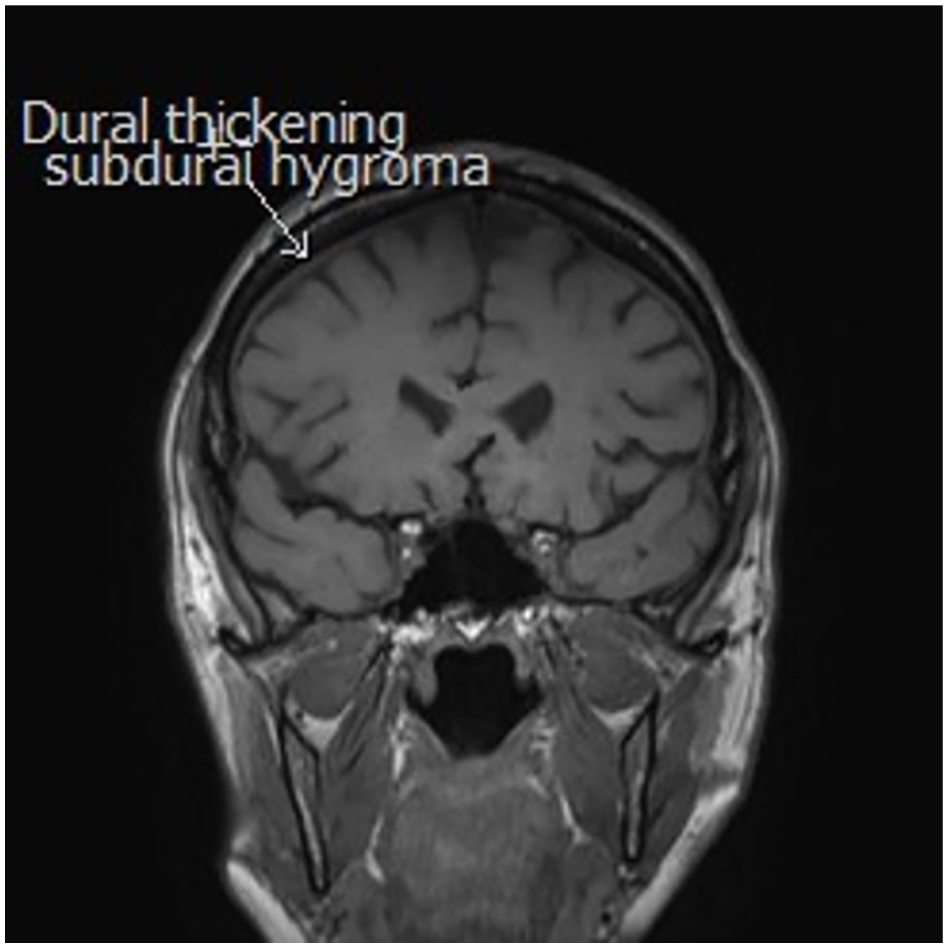
Cranial MRI demonstrating dural thickening and subdural hygroma (white arrow) in SIH.
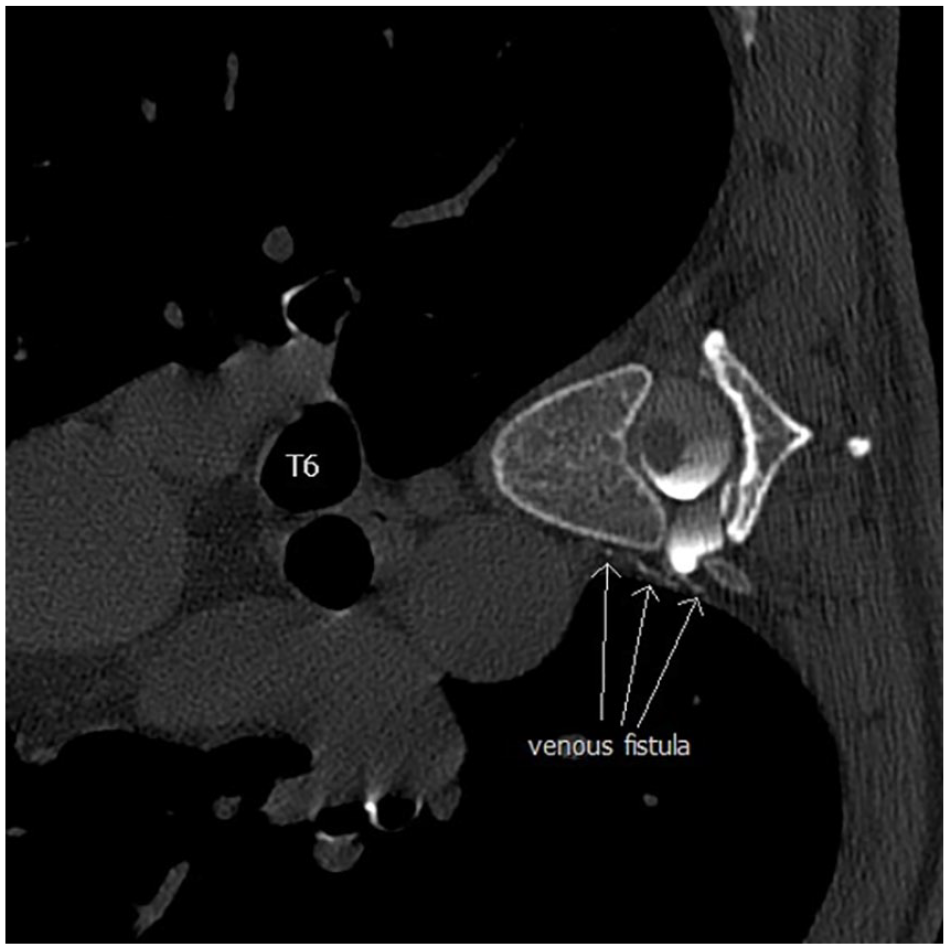
CT image of T6 nerve root cerebrospinal fluid–venous fistula (white arrows).
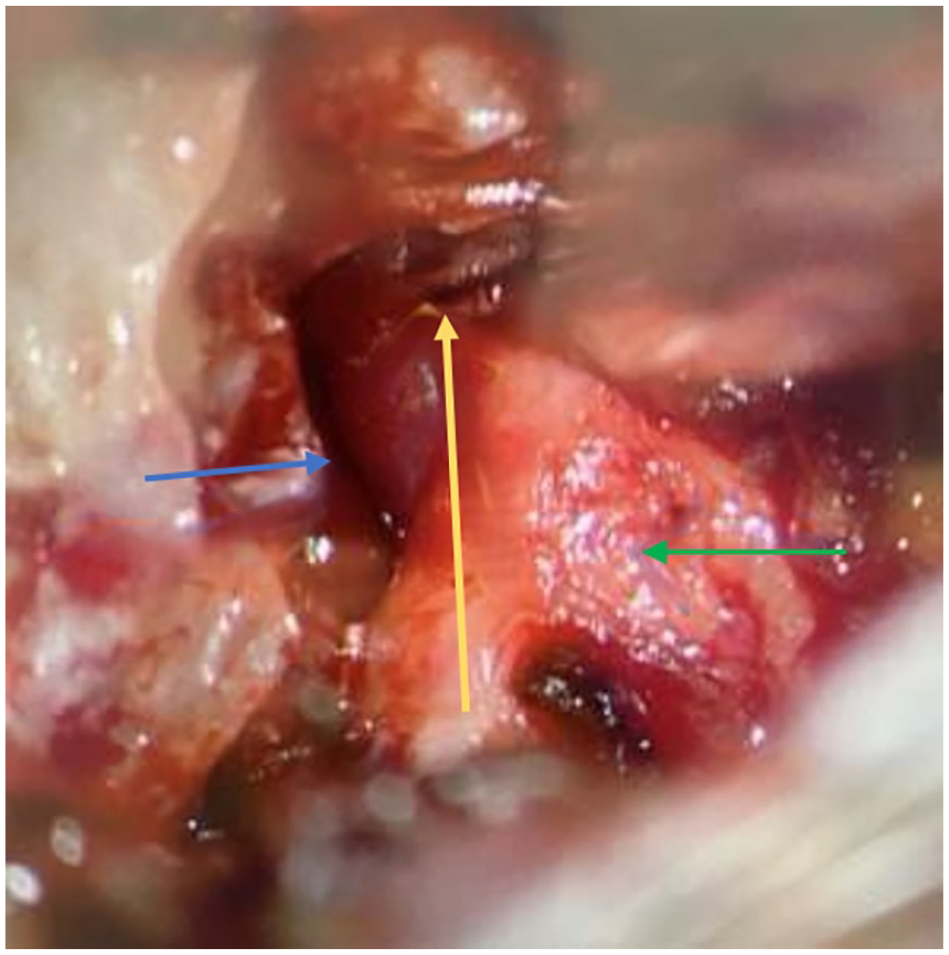
Intraoperative photo showing fistula (yellow arrow) and the adjacent vein (blue arrow) and nerve (green arrow).
Discussion
SIH is secondary to CSF hypovolemia, either from a spinal leak or CSF–venous fistula. CSF fluid leaks occur from tearing of the arachnoid mater as a result of nerve root diverticulum, an outpouching of the arachnoid mater, or by trauma, commonly associated with osteophyte spurs. 2 This leads to an effusion of CSF in the extradural compartment. Conversely, CSF–venous fistulas are a pathological communication between the subarachnoid space and a paraspinal vein, allowing for unregulated drainage of CSF into the circulatory system. 13 Ultimately, the loss of CSF causes traction on pain-sensitive fibers within the dura mater when in the upright position leading to the classical orthostatic headache. 2 The headache is typically relieved by lying flat; however, this is not true for all patients. Given the variability of headache and non-specific nature of other symptoms including vestibulocochlear, the diagnosis of SIH is often delayed before definitive diagnosis and management.
The cochlear aqueduct runs from the subarachnoid space within the posterior cranial fossa through the petrous temporal bone into the inner ear and, thus, a direct connection between CSF and perilymph.14,15 The low CSF pressure that is transmitted through the cochlear aqueduct leads to a reduction in perilymph within the cochlear turns and compensatory expansion of endolymph. 6 As a result, the patient develops varying degrees of vestibulocochlear symptoms, including sensorineural hearing loss, tinnitus, aural fullness, and occasionally vertigo. Previous studies have reported rates of vestibulocochlear symptoms in up to 70% of patients.14,16 The variability of the anatomy of the cochlear aqueduct described in previous studies may account for the lower rates of vestibulocochlear symptoms in patients with SIH. 6 Choi et al 16 also demonstrated 54% of patients had unilateral or bilateral low-frequency sensorineural hearing loss, and a majority of patients’ hearing loss resolved following resolution of their orthostatic headache. A few case reports have demonstrated the positional element to hearing loss, performing better on audiological assessment when lying flat compared with the upright position.12,14 All the cases reported above had vestibulocochlear symptoms, some of which were the only symptom noted or the primary complaint and hence referred initially to an otolaryngologist. Moreover, given the similarity of symptoms between SIH and Meniere’s disease, many patients might be initially misdiagnosed prior to the correct diagnosis being made. 8 The rise in endolymphatic pressure is similar pathophysiologically to the endolymphatic hydrops seen in Meniere’s disease and thus also produces a low-frequency sensorineural hearing loss. 17 Thus, when taking a detailed history for patients with vestibulocochlear symptoms, clarifying whether there is a postural element could help narrow the differential diagnoses.
Diagnostic criteria includes CSF opening pressures <6 cm H2O, radiological findings of spinal CSF leak, or intracranial hypotension and temporal headache in relation to low CSF pressure. 18 SIH, however, cannot be diagnosed in a patient who has had a dural puncture within the last month. Evidence of intracranial hypotension on MRI commonly includes brainstem sag, pachymeningeal enhancement, and dural venous engorgement but can also have pituitary enlargement, posterior fossa superficial siderosis, or subdural collections. 13
In the cases we present, opening pressures were low (8-11.5 cm H2O). All had dural enhancement. No leaks detected on MRI spine; however, all had CSF–venous fistulas.
The decreased CSF volume is believed to be the driving force for the development of SIH symptoms. 13 SIH is still a very variable condition and does not always present as per diagnostic criteria. A 2018 review of the literature outlined 10 common myths regarding SIH, and how no single test is sensitive enough to make diagnosis. Clues to the diagnosis of SIH when patients with vestibulocochlear symptoms present for otolaryngologist review are classically an orthostatic quality to their symptoms, however not absolute. 19 Other clues to the diagnosis include the temporal association of symptoms such as interscapular pain, diplopia, gait unsteadiness, or cognitive symptoms. However, no single test has high enough sensitivity to rule out SIH. 19
If SIH is suspected on cranial MRI (Table 1), patients should undergo full-spinal MRI looking for the site of leak and referred to a center for dynamic CT myelography. A study of 103 patients demonstrated a sensitivity of a full-spinal MRI identifying the leak to be approximately 92%. 20 However, this is felt to be overestimated as MRI is limited in its ability to pick up spinal fistulas. 13 CT myelography is the gold standard in our institution in diagnosing all types of CSF leaks.
Radiological features of cerebrospinal fluid hypovolaemia. 13

After identifying the cause for SIH, there are several management options available. Conversative measures including caffeine, oral hydration, and bed rest, all of which are rarely effective long term, providing only temporary relief. In the management of CSF leaks, epidural blood patching is the most common technique used, involving injecting autologous blood or fibrin into the epidural or perineural (extradural) space to seal (or slow) the dural defect. The efficacy of blood patching varies, with patients often requiring more than 1 patch to obtain resolution of symptoms, but has shown to be effective, depending upon the study, in up to 90% of cases.2,13 CSF–venous fistulae can be treated with targeted patching, transvenous embolization, or surgery, the second being a significantly less invasive option compared with surgery. Surgical ligation of the nerve root and accompanying vein is, however, relatively straight forward through a small incision. 13 A 2020 prospective study demonstrated clinically significant improvement in symptoms in 100% of cases for the management of CSF–venous fistulae. 21 Ultimately, with appropriate management patients with SIH will have positive outcomes and significant reduction in their symptoms.
SIH is an uncommon condition that has become increasingly recognized amongst neurologist and neuroradiologists. The associated vestibulocochlear symptoms may lead to initial presentation to an otolaryngologist. This case series and review of literature highlights the common symptoms the otolaryngologist might be presented with and the importance of differentiating it from other differential diagnoses such as Meniere’s disease.
Footnotes
Acknowledgements
We thank Mr Arul Bala for providing us with the intraoperative photo.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case series was obtained from Ramsay Health Care WA/SA Human Research Ethics Committee.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
