Abstract
Congenital hypothyroidism rarely causes a clinically significant neck mass in newborns. We present the case of a newborn with congenital hypothyroidism and significantly enlarged goiter and discuss imaging considerations and medical and surgical management. This infant was prenatally discovered to have a midline neck mass on 28 week ultrasound measuring 6.0 cm × 3.4 cm × 5.8 cm. Diagnostic cordocentesis demonstrated elevated thyroid-stimulating hormone (TSH, 361 µIU/mL). Maternal evaluation for thyroid disease and antithyroid antibodies was negative. A Cesarean section at 38 weeks gestation was recommended due to hyperextension of the fetal neck. The infant was intubated for respiratory distress. Postnatal magnetic resonance imaging revealed a 5.5 cm × 4.4 cm × 7.6 cm goiter and laboratory studies confirmed the diagnosis of primary hypothyroidism (TSH 16.7 µIU/mL). Treatment was initiated with intravenous levothyroxine and transitioned to oral supplementation. Serial ultrasounds showed decreased goiter volume over several weeks, with recent volume per lobe being 22% and 44% of original volume. This case demonstrates the importance of prompt diagnosis and initiation of thyroid hormone replacement, allowing for significant goiter regression without surgical intervention and ensuring normal growth and neurodevelopmental outcome. Surgical management should be considered for those with persistent compressive symptoms despite optimal medical management.
Keywords
Introduction
Congenital hypothyroidism occurs in one out of 2000-4000 newborns, with over 95% having no clinical manifestations at birth. Finding of fetal or neonatal goiter is exceedingly rare in these cases (0.1-0.3:10,000 births). 1 Congenital hypothyroidism can be caused by conditions that are intrinsic (primary or central hypothyroidism) or extrinsic to the infant (maternal or environmental factors). Primary hypothyroidism encompasses conditions such as dysgenesis of the thyroid gland and inborn errors of thyroid hormone biosynthesis, otherwise known as dyshormonogenesis.2 -5 Thyroid dysgenesis makes up 75% to 80% of congenital hypothyroidism and dyshormonogenesis makes up 15% to 20%. 5 External causes, including transient maternal effects of autoantibodies, medications, or iodine deficiency can also contribute to congenital hypothyroidism.
Symptoms of congenital hypothyroidism are often subtle, but not recognizing or forgoing treatment can lead to devastating growth and poor neurodevelopmental maturation. Pediatric endocrinologists hasten treatment of congenital hypothyroidism using levothyroxine and monitor response by closely following serum levels of thyroid-stimulating hormone (TSH) and free T4 (fT4), or thyroxine. Maternal TSH does not cross the placenta and fT4 does in small quantities. Experts recommend starting treatment within the first 2 weeks of life as placental transferred fT4 dwindles. Therefore, if fetal hypothyroidism is diagnosed prenatally, intra-amniotic instillation of L-thyroxine is an option for fetal thyroid hormone replacement.1,2,6
Congenital goiters are a rare clinical condition characterized by enlargement of the thyroid gland, which has the potential to compress on the aerodigestive tract. 7 Congenital goiters are most commonly caused by the transfer of maternal antibodies from mothers with Grave’s disease through the placenta. Dyshormonogenesis is another common reason to have a congenital goiter accompanying hypothyroidism.1,2,7 In the literature regarding prenatally diagnosed goiters, intrauterine L-thyroxine has been given if there is concern for complications such as tracheal occlusion, polyhydramnios, or premature delivery and has been shown to stabilize or shrink goiters on ultrasound or magnetic resonance imaging (MRI).1,2,6 There is no detailed report of reduction in goiter size with serial imaging and laboratory work in the postnatal period with levothyroxine treatment. In this article, we present a case report of a newborn with congenital hypothyroidism and a significantly enlarged thyroid goiter, highlighting the imaging considerations, medical management, and the potential need for surgical intervention in such cases.
Case Report
This infant was prenatally identified to have a midline neck mass measuring 4.7 cm × 4.0 cm × 3.2 cm at 26-week growth assessment after prior anatomical ultrasound survey exams were notable for fetal growth restriction. At maternal-fetal medicine evaluation at 35 weeks gestation, the midline neck mass had grown to 6.0 cm × 3.4 cm × 5.8 cm and diagnostic cordocentesis demonstrated elevated TSH (361 µIU/mL). Maternal evaluation for thyroid disease and antithyroid antibodies was negative, and the mother was not using any medications and did not have iodine deficiency. Given the size of the neck mass with elevated TSH, 2 doses of intra-amniotic levothyroxine at 28 and 34 weeks gestation was administered. On the ultrasound, fetal neck extension as well as growth restriction were observed without associated polyhydramnios or tracheoesophageal collapse. Our pediatric otolaryngology, neonatology, and maternal-fetal medicine teams met with the family to discuss an airway plan for delivery and postnatal treatment options. Due to these findings on ultrasound, a Cesarean section was planned and performed at 38 weeks gestation. As part of the delivery plan, the pediatric otolaryngology and neonatology teams discussed airway management, and at delivery, the infant was able to be successfully intubated on the first attempt with a 2.5-inch endotracheal tube by the neonatal team.
On the first day of life, the infant’s TSH was elevated at 16.7 µIU/mL in addition to a normal for age fT4 (0.67 ng/dL). Antibody evaluation was negative, including thyroid-stimulating immunoglobulin and autoantibodies against thyroglobulin thyroid peroxidase (TPO) and TSH receptor. Genetic testing for congenital hypothyroidism genes and thyroid hormone resistance panel (genes TG, TPO, SLC5A5) revealed homozygosity for mutations in the TPO genes associated with autosomal recessive congenital hypothyroidism. Day of life 2 MRI demonstrated a 4.4 cm × 7.6 cm × 5.5 cm heterogenous T1 and T2 hyperintense mass consistent with thyroid goiter, seen in Figure 1. Bedside ultrasonography was used for the subsequent trending of goiter size.
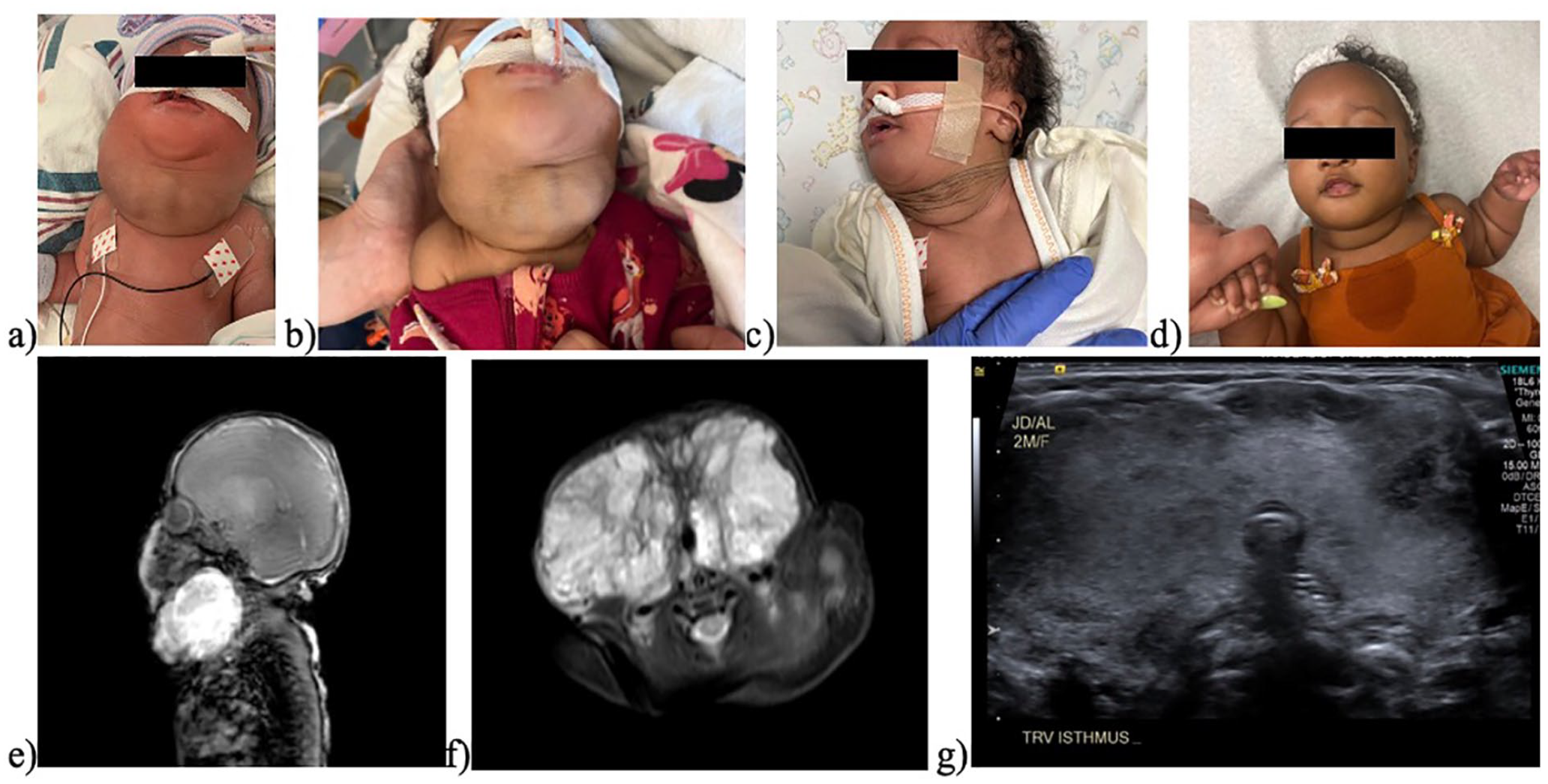
From right to left: (a) Day of life 1, (b) Day of life 13, (c) Day of life 23, (d) 4 months of age (e) Sagittal MRI on day of life 2 showing large mass compressing trachea, (f) Axial MRI on day of life 2 showing compression on the aerodigestive tract and around the trachea, and (g) Thyroid ultrasound axial view at 2 months of age showing less compression on the aerodigestive tract.
The medical team started intravenous levothyroxine at 37.5 mcg daily, which was tapered to 12.5 mcg daily over the first 2 months of life. Serial ultrasonography and labs were performed at 2-week intervals to track change in thyroid size and thyroid hormone levels seen in Table 1. Initial ultrasonography measurements at day of life 1 showed a right lobe at 5.9 cm × 3.7 cm × 5.1 cm (57.6 cc) and left lobe at 5.1 cm × 3.3 cm × 4.0 cm (35.4 cc). After immediate initiation of levothyroxine treatment, at 2 weeks of age, the lobes measured at 4.3 cm × 5.2 cm × 3.2 cm (36.3 cc) on the right and 3.2 cm × 5 cm × 2.6 cm (21.8 cc) on the left with a decrease in volume from original of 63% and 62%, per respective lobe. The thyroid goiter volume continued to decrease over time, with the most recent measurement at 10 weeks of age of 3.0 cm × 3.8 cm× 2.4 cm (12.7 cc) for the right lobe and 2.5 cm× 4.0 cm× 3.1 cm (15.9 cc) for the left lobe. This volume decrease from baseline resulted in a right lobe volume of 22% and left lobe volume of 44% of the original volume while on maintenance therapy of 12.5 mcg of levothyroxine. Clinical examination and MRI and ultrasound imaging demonstrated rapid reduction of the goiter’s size with stabilization in size and gross appearance at 2 to 3 months of age (Figure 1).
Treatment of Hypothyroidism With Resultant Laboratory Values and Volumes Measured by Ultrasound.
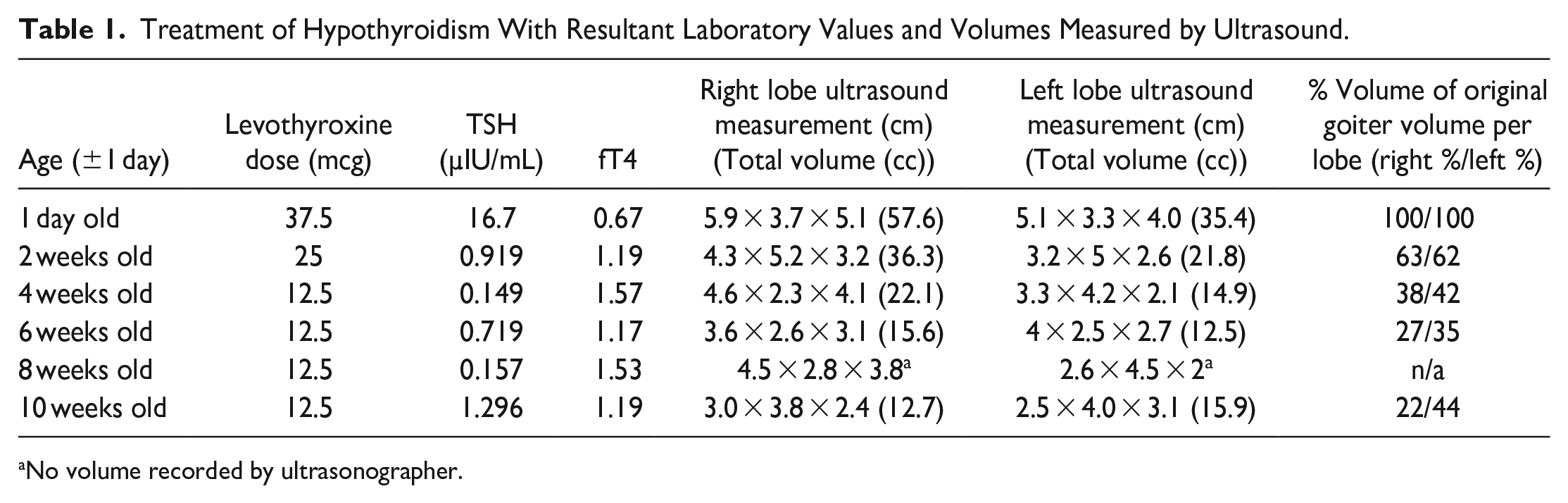
No volume recorded by ultrasonographer.
Regarding compressive effects from the goiter, the patient was intubated without difficulty after birth for respiratory distress refractory to positive pressure therapy. An attempt at extubation at approximately 3 weeks of life was not successful and reintubation was performed. Successful extubation occurred at 1 month of age when the thyroid right lobe measured 4.6 cm × 2.3 cm × 4.1 cm (22.1 cc) and left lobe measured 3.3 cm × 4.2 cm × 2.1 cm (14.9 cc), which correlated to the right lobe volume of 38% and the left lobe volume of 42% of original thyroid volume, well over a 50% decrease in total size. The patient was subsequently weaned to nasal cannula and to room air by 3 months of age. Oral intake was accomplished by 2 months of age, and the infant was discharged home on daily levothyroxine 12.5 mcg at this time. At 3 months of age, the patient successfully completed a swallow study without aspiration.
Discussion
There is limited literature on the management of congenital hypothyroidism with goiter, particularly regarding the changes in goiter size with medical management. We report a case that demonstrates significant goiter size reduction with medical management of congenital hypothyroidism with dyshormonogenetic goiter. Current literature is limited on this topic to a single case report that describes reduction of gland size with 2 different ultrasound measurements, and there is lack of literature documenting the concurrent changes in goiter volume with hormone treatment and levels. 7
Expedient neonatal treatment of congenital hypothyroidism is paramount to ensure appropriate neurodevelopmental growth. Specific therapy depends on the underlying etiology. In infants with a congenital goiter, assessment of maternal causes, such as hypothyroidism and iodine deficiency, as well as intrinsic causes, by thyroid function tests and thyroid antibody tests, should be performed. Infants with thyroid dyshormonogenesis typically present with high TSH, low fT4, and negative thyroid antibodies. They require supplementation with levothyroxine, starting with initially high doses to rapidly correct the TSH levels, followed by gradual dosage reduction once TSH stabilized. 7
In infants with congenital hypothyroidism from primary causes, thyroid hormone deficiency causes high TSH, which stimulates any present thyroid follicular cells to compensate for defective function and increases the thyroglobulin levels, often creating a goiter. 5 In our case of thyroid dyshormonogenesis, we found that medical treatment of hypothyroidism with levothyroxine accomplished rapid correction of laboratory values and over time and significantly reduced goiter volume measured on serial ultrasonography. This approach allowed us to avoid surgical treatment by aggressive medical therapy in conjunction with appropriate airway management to allow time for the goiter to diminish in size and reduce compression on the aerodigestive tract.
For imaging, an initial MRI was performed to examine the parenchyma and confirm the diagnosis of goiter. A repeat MRI was not medically necessary to compare the intrinsic properties of the thyroid parenchyma, but color Doppler ultrasonography was a readily available and useful tool for monitoring and trending the reduction and stabilization of gland size. The pediatric endocrinology team coordinated laboratory value draws on the same day as bimonthly ultrasounds to help guide levothyroxine treatment and normalize TSH levels, as recommended in the literature. 6 Frequent monitoring of TSH and fT4 is necessary as infant weight changes quickly and hormone dose titration is needed to maintain appropriate levels. 6 Previous research suggests administration of levothyroxine at a dose of 10 to 15 µg/kg/day in infants yields the most optimal IQ outcomes. Additionally, initiating treatment earlier (day of life 12-30 vs later than 30 days) has been associated with improved average IQ. 6
To date, no reports have linked early postnatal treatment to improvements in goiter size, airway maintenance, or swallowing function. There are limited published descriptions of intra-amniotic administration of L-thyronine to treat fetal hypothyroidism and reduce or stabilize goiter size, but knowledge of its effect on goiter size and neonatal outcomes is sparse.8,9 Figueiredo et al followed a similar patient with perinatal goiter who was treated with intra-amniotic levothyroxine injections and started on immediate treatment postnatally, but measurements of goiter were not tracked. 10 Case reports note that older children with large goiters despite appropriate treatment for hypothyroidism, as well as worsening goiter size upon stopping levothyroxine supplementation.2,3,5,10
Surgical treatment of congenital goiter is rare and is utilized for extreme cases of aerodigestive compression that are unable to be conservatively managed. Thyroidectomy is also reserved for cases with concerning features of malignancy of the thyroid on imaging and is used in exceptional circumstances. Another surgical option includes tracheostomy for severe airway compression, failure to extubate safely, or severe tracheomalacia refractory to positive pressure. These options are discussed thoroughly with the entire care team before proceeding and fortunately, did not need to be explored in this patient.
We found that allowing time for the goiter to decrease in volume with thoughtful medical management alone allowed for safe extubation once there was reduced compression on the trachea and aerodigestive tract and a deferred need for surgical intervention. Over time, the goiter continued to shrink to a more cosmetically appealing and functional size with meticulous correction of hormone deficiency with levothyroxine supplementation as appreciated on serial ultrasonography. Our case supports the approach of expedient and aggressive medical management of congenital hypothyroidism with goiter to allow for significant volume reduction and reduction of compression on the aerodigestive tract to lessen the need for surgical intervention.
Footnotes
Acknowledgements
The author(s) have nothing to acknowledge or disclose.
Authors’ Note
Presented at: American Academy of Otolaryngology—Head and Neck Surgery Annual Meeting, Nashville, TN 2023.
Data Availability
All data were accessed through our secure electronic medical record and stored on a secure password-protected server.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution policy does not require ethical approval and is exempt for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
