Abstract
Plain Language Summary
The Sino-Nasal Outcome Test-22 (SNOT-22) has shown that the quality-of-life results of sinus surgery after endoscopic sinus surgery (ESS) improve significantly. The amount of change seems to vary a lot from one study to the next, and this difference seems to be caused by things like the pre-op SNOT-22 score, the average age of the subjects, and the length of the tracking period. The results of this study give both a single number value and a range of changes that are likely to happen after surgery. These results can be used to guide projects that aim to improve the quality of care. Also, giving the Sino-Nasal Outcome Test-22 (SNOT-22) to people with chronic rhinosinusitis (CRS) before they have surgery may help them understand what effects they can expect, although this is up to each person to decide. Recent preliminary research shows that using SNOT-22 scores and tissue histopathology together could be a new way to predict how well treatment will work for people with CRS. The accuracy and precision of future analyses are likely to get better as efforts are made to get unbiased data and patient-level metrics from a wide range of patients and doctors.
Introduction
Rhinosinusitis is a commonly occurring medical condition that has a significant impact on the quality of life (QOL) of individuals globally. 1 Chronic rhinosinusitis (CRS), commonly abbreviated as CRS, is a prevalent and complex medical condition that imposes significant societal and financial ramifications. CRS is prevalent in 15% of the populace in developed countries, rendering it the second most frequently occurring persistent ailment. 2 The pathophysiology and etiology of the condition have been thoroughly established in the literature. However, the treatment approach is subject to frequent modifications due to the rapidly evolving nature of the causative organism and antimicrobial resistance, posing a challenge to healthcare providers. This is supported by existing research. 3
CRS has been associated with a range of upper and lower airway complications, including but not limited to nasal polyposis, asthma, and cystic fibrosis. 4 Although not posing a threat to life, rhinosinusitis has a significant impact on daily functioning and QOL. 5 The initial treatment for CRS comprises of nasal saline irrigation, systemic steroids, and anti-inflammatory steroids that effectively drain the paranasal sinuses. 6 Nevertheless, a considerable number of individuals exhibit resistance toward these therapies and necessitate the implementation of functional endoscopic sinus surgery (FESS) to ameliorate their symptoms and enhance their standard of living, given that endoscopic sinus surgery (ESS) possesses the capability to reinstate the typical mucociliary flow. 7 Due to the significance and gravity of this pathological condition, it is imperative to exercise caution when selecting patients for surgical intervention and to comprehend the determinants that forecast surgical outcomes, with the aim of optimizing success and minimizing risk. 8
The evaluation of the QOL of individuals with CRS can be conducted through the utilization of generic questionnaires such as the SF-36. However, it is more suitable to employ disease-specific questionnaires like the SNOT-22 for this purpose. Individuals diagnosed with CRS who have not experienced improvement from pharmacological interventions are suitable candidates for FESS. Additionally, the Sino-Nasal Outcome Test-22 (SNOT-22) is a valuable tool for forecasting the QOL in patients with CRS. 9
The SNOT-22 questionnaire comprises questions that are evaluated on a scale of 0 to 5, resulting in a total score range of 0 to 110. Higher scores on this scale are indicative of lower QOL. According to the referenced source, 10 the typical value for minimal clinically important difference (MCID) is 8.9 points. According to several studies, the utilization of ESS has been found to enhance SNOT-22 scores in adults with CRS.11-15 To incorporate SNOT-22 scores as an outcome measure in quality improvement initiatives, it is imperative to possess knowledge regarding the anticipated spectrum of alteration. Familiarity with the customary amelioration subsequent to ESS and patient factors that could influence results is necessary for citing this source. Taking all factors into account, a systematic review was conducted on a set of studies16-30 that evaluated the alteration in SNOT-22 score following ESS for adult patients diagnosed with CRS. Additionally, a meta-analysis was conducted to determine the average change in SNOT-22.
Materials and Methods
The current meta-analysis was conducted in accordance with the Preferred Reporting Items of Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 31
Eligibility Criteria
For a study to be considered eligible, certain criteria must be satisfied: (a) Prospective studies examining the alteration in SNOT-22 score among individuals with CRS prior to and following EES and (b) investigations assessing the primary endpoints, including the disparity in average SNOT-22 score before and after the surgical procedure, the duration of the follow-up period, and the average age of the participants. The exclusion criteria comprised of clinical trials that had a follow-up duration of <1 month. This meta-analysis excluded studies that involved healthy participants or individuals with disorders other than CRS who were not undergoing EES.
Information Sources
A comprehensive literature review was performed by searching the electronic databases Cochrane Library, EMBASE, and PubMed up until March 2023. Furthermore, pertinent meta-analyses and bibliographies of studies were also examined.
Search Strategy
A comprehensive literature review was performed utilizing the following predetermined criteria for inclusion: The inclusion criteria for the studies were as follows: (i) the studies had to be either prospective or retrospective in nature and had to report the SNOT-22 score in patients with CRS both before and after undergoing ESS; (ii) the studies had to include patients with CRS who were 18 years of age or older; and (iii) the studies had to provide data on the primary outcome. The study aimed to investigate the pre-op SNOT-22 score, the mean difference in SNOT-22 score before and after surgery, the duration of the follow-up period, and the average age of patients. A systematic review was conducted to identify articles published in the English language that used the keywords “Chronic Rhinosinusitis” or “CRS,” “Endoscopic Sinus Surgery” or “ESS,” “22-test Sino nasal test outcome” or “SNOT-22,” “Quality of Life” or “QOL,” “Meta-analysis,” “prospective cohort studies,” and “systematic review.”
The Medical Subject Headings were conjoined with the text keywords in the search strategy using the Boolean operator “AND.” Initially, the elimination of replicated articles was conducted, followed by a thorough screening of the titles and abstracts of the remaining articles. Subsequently, all studies deemed eligible were obtained and subjected to a comprehensive review for the purpose of determining their inclusion or exclusion, in accordance with the inclusion and exclusion criteria outlined by the PRISMA guidelines.
Selection Process
Two authors conducted the literature search independently. A consensus was achieved through deliberation in the case of a disagreement. The data extraction process was carried out by 2 distinct authors in an independent manner. Subsequently, a comparative analysis was conducted on the results obtained from both authors’ extractions. In situations where there were conflicting viewpoints, a consensus was achieved through deliberation. The inclusion of a third author was contingent upon the specific circumstances.
Data Collection Process
A data extraction form that was computerized was created using Microsoft Excel. This form was then employed to document the essential details of the studies that were chosen for the meta-analysis. The aforementioned details encompassed the identity of the primary author, the year of publication, the study design, the geographical location of the study, the total sample size, the average age of the participants in years, the gender distribution of the participants, the type of surgical intervention, the duration of the post-op follow-up period in months, and the principal outcome of the studies that were incorporated.
Risk of Bias Assessment
The methodological validity of each study included in the meta-analysis was assessed using the Cochrane Risk of Bias tool. 32 In the course of extracting data, chosen articles were evaluated and assigned a rating of “low,” “high,” or “some concern” based on their approach to generating random sequences, allocation concealment, participant and staff blinding, outcome assessment blinding, adequacy of outcome data, selective reporting, and other potential sources of bias. Subsequently, the Review Manager (RevMan) [Computer program]. Version 5.4. The Cochrane Collaboration, 2020 33 was employed to generate a summary and graph for the assessment of the risk of bias in terms of quality.
Reporting Bias Assessment
The utilization of Egger’s test, 34 Begg’s test, 35 and Deek’s funnel plot 36 was conducted through the utilization of MedCalc 64 bit version, flagship product of MedCalc Software Ltd 37 to investigate the presence of publication bias.
Synthesis Methods
A meta-analysis was conducted utilizing Comprehensive Meta-Analysis Version 4 38 to ascertain the average alteration in SNOT-22 across all studies. The mean change following surgery was determined using a random effects model with inverse variance weighting, in light of the anticipated heterogeneity. A forest plot and 95% confidence interval (CI) were also utilized. A mixed-effects meta-regression 39 was utilized to assess the influence of specific attributes, such as pre-op SNOT-22, mean change in SNOT-22 score, and average patient age, on the results of various studies. The statistical analysis involved inputting the mean and standard deviation or frequency of each variable in each study into a model to examine its impact on outcomes across studies. This was done by utilizing study-level data in the meta-regression and creating corresponding scatter plots. 40
Results
Literature Search Results
The PRISMA chart that was utilized for research selection is illustrated in Figure 1. A thorough exploration of digital repositories yielded a total of 1214 research investigations. A total of 734 studies were eliminated from consideration on the basis of duplicity and titles deemed to be irrelevant. Subsequently, a total of 480 studies underwent screening based on their title, abstract, and review. Subsequently, a total of 465 cases were eliminated from the study due to insufficient data for constructing a 2 × 2 table, absence of the outcome of interest, or noncompliance with the inclusion criteria. Fifteen pertinent articles were ultimately incorporated in the current meta-analysis. Table 1 displays the characteristics of all trials that were incorporated, including patient demographics. The studies incorporated in the analysis assessed the pre-op and post-op SNOT-22 test scores among patients with CRS who underwent ESS.
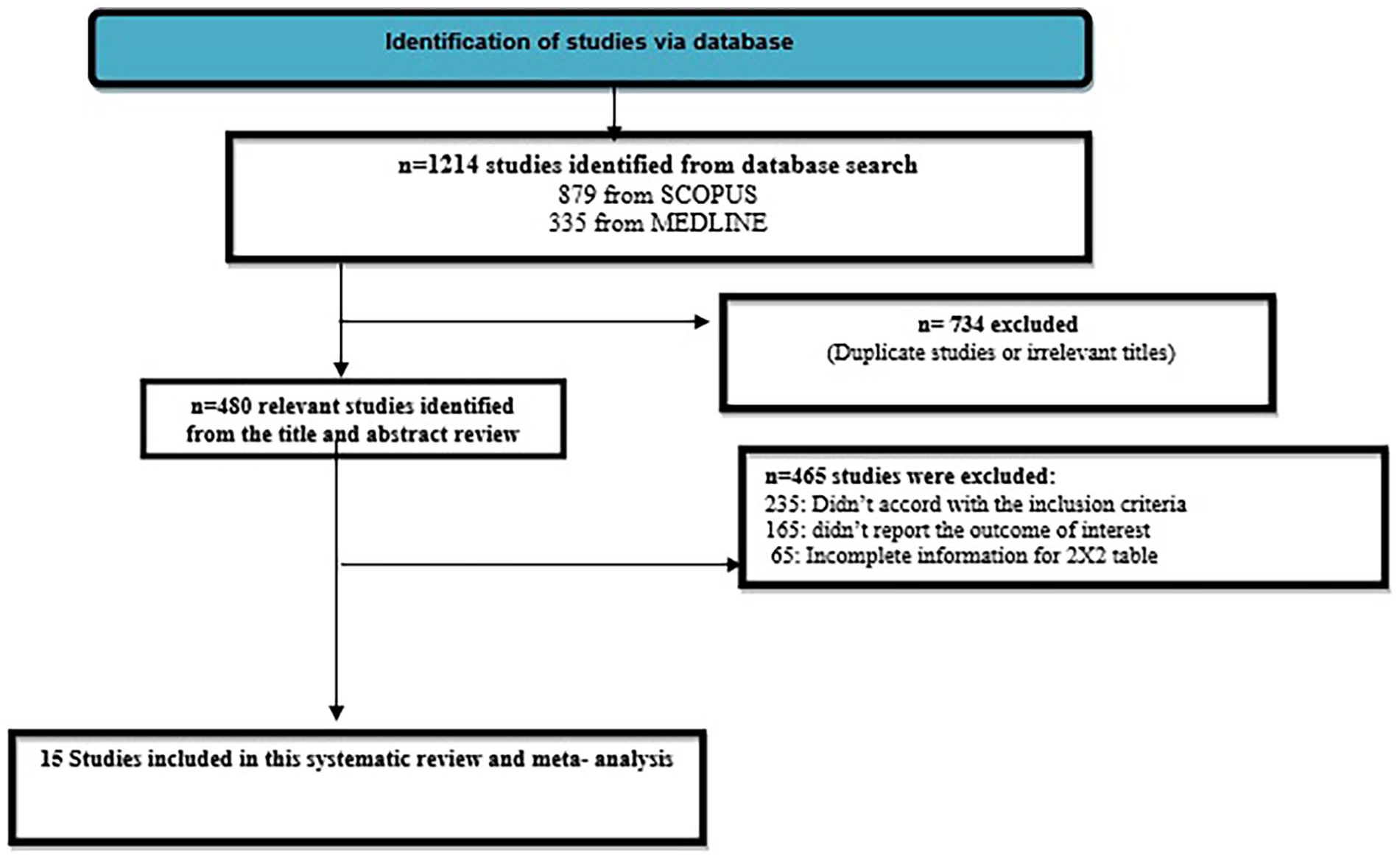
PRISMA flowchart of selection of studies.
Characteristics of the Included Studies.
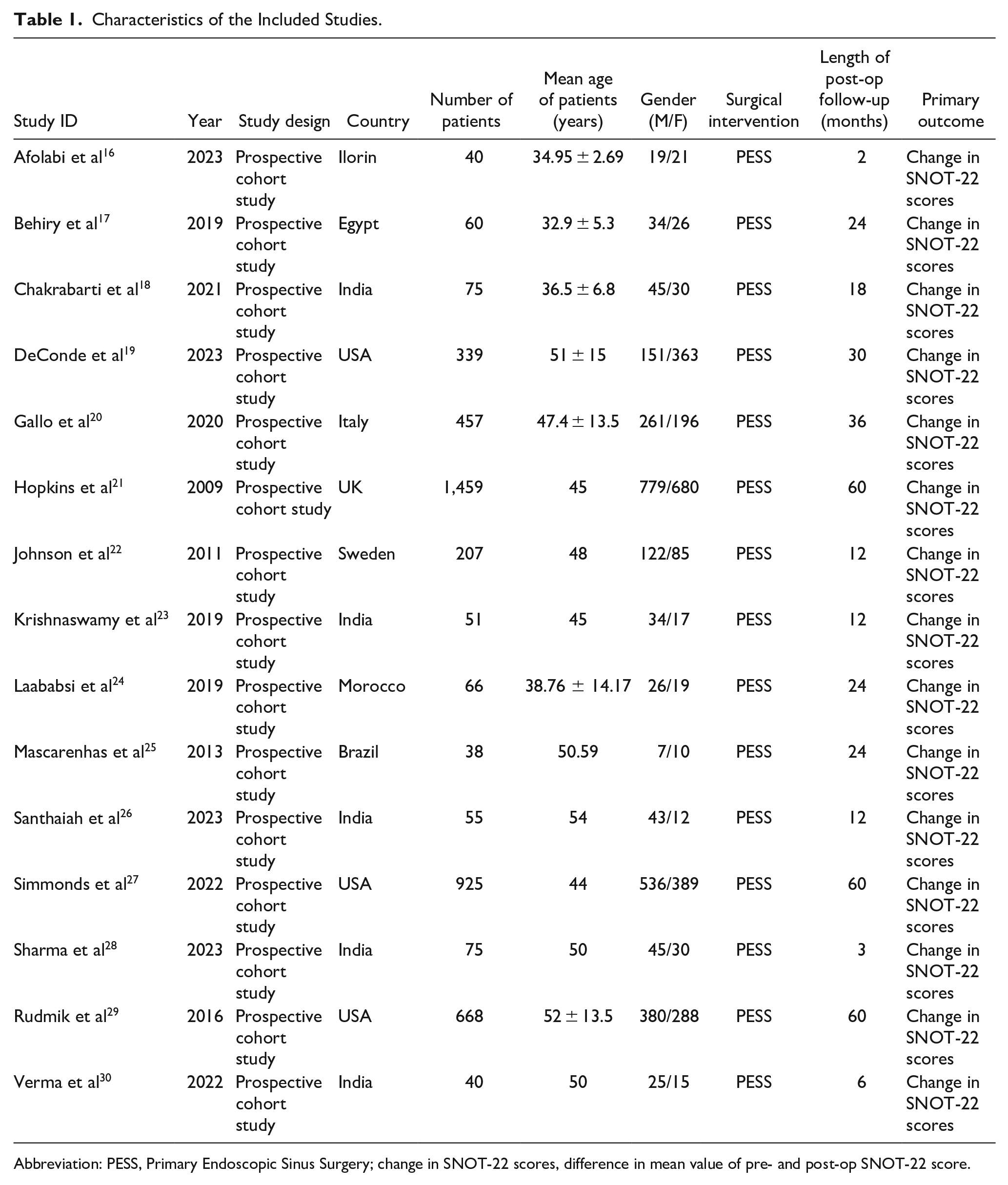
Abbreviation: PESS, Primary Endoscopic Sinus Surgery; change in SNOT-22 scores, difference in mean value of pre- and post-op SNOT-22 score.
Risk of Bias Assessment and Publication Bias
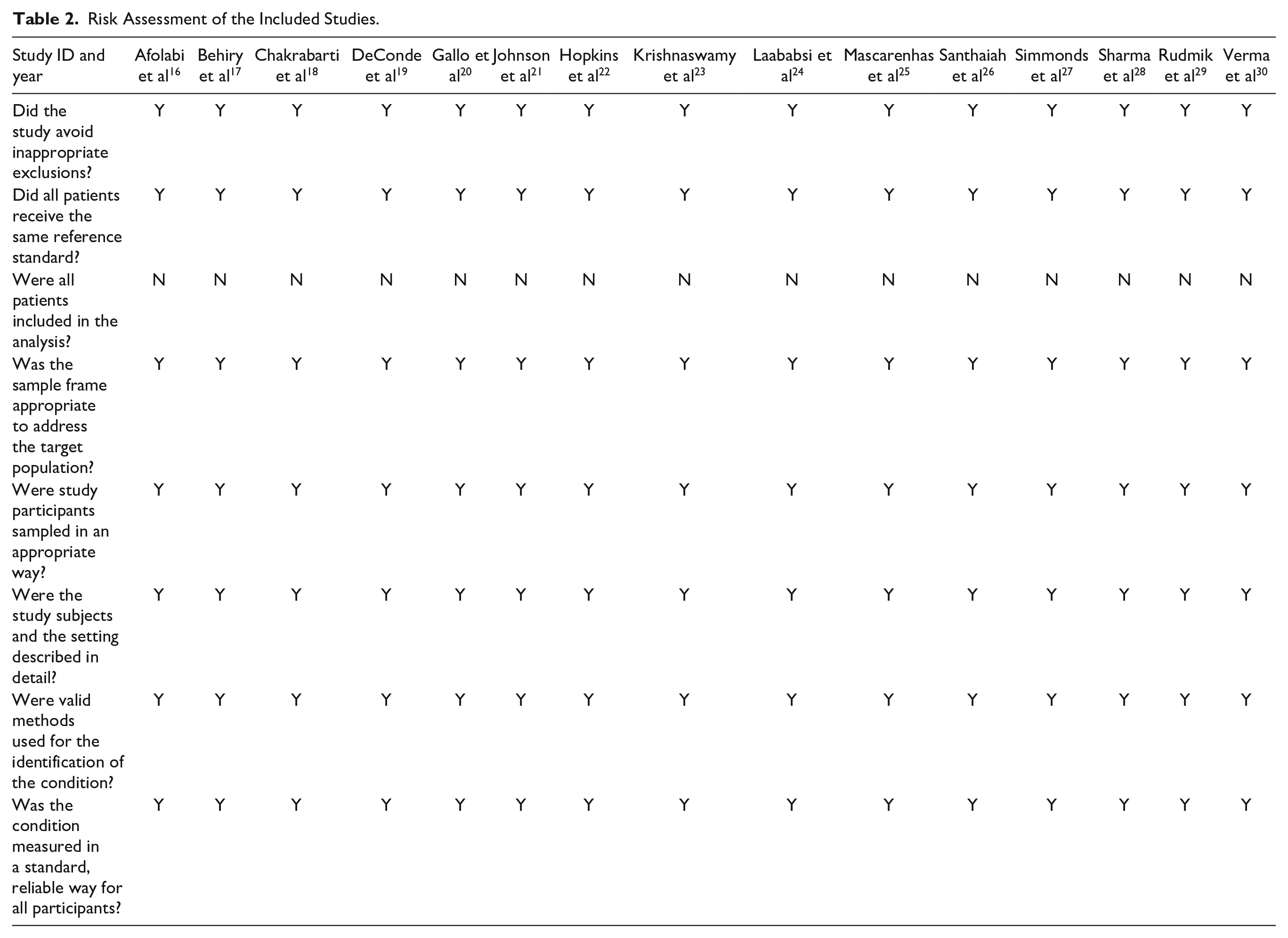
Table 2 displays the assessment of the quality of the studies that were included. Figure 2 provides a concise overview of the potential for bias, while Figure 3 presents a graphical representation of the risk of bias. Out of the 15 studies that were included, 8 were found to have a low risk of bias, while 4 studies were identified to have a moderate risk of bias due to confounding. The 3 studies incorporated in the analysis exhibit a high risk of bias stemming from the measurement of the exposure and potential bias resulting from postexposure interventions. The funnel plot presented in Figure 4 suggests a low likelihood of publication bias, as evidenced by the nonsignificant p values of 0.279 for Begg’s test and 0.467 for Egger’s test.
Risk Assessment of the Included Studies.
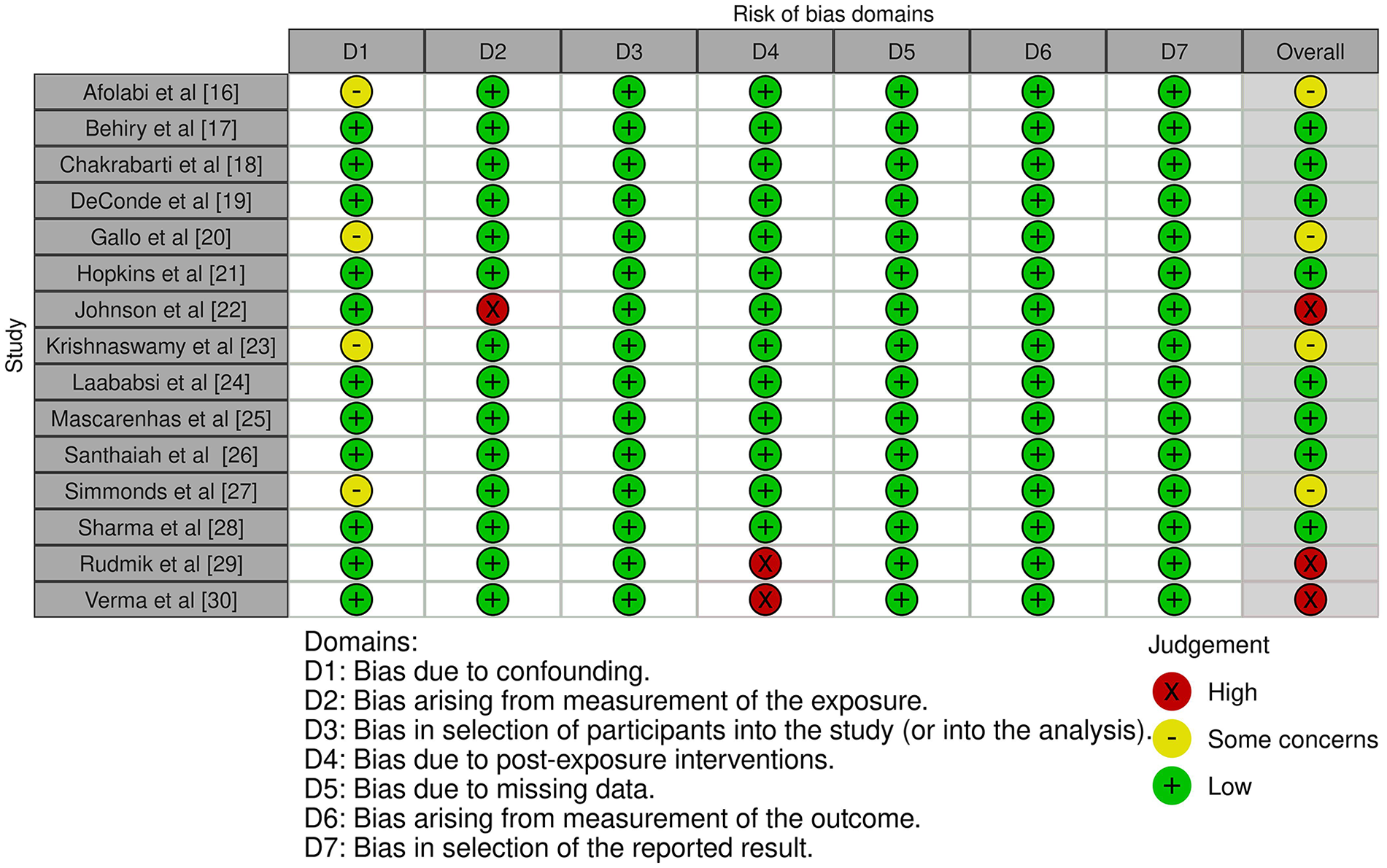
Risk of bias summary.
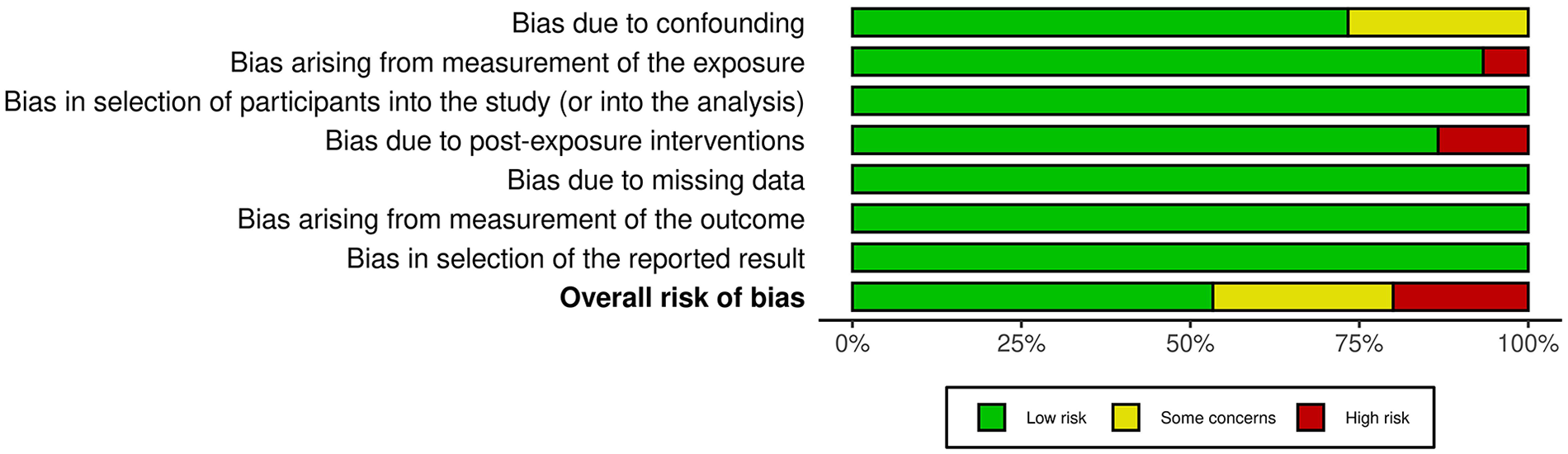
Risk of bias graph.
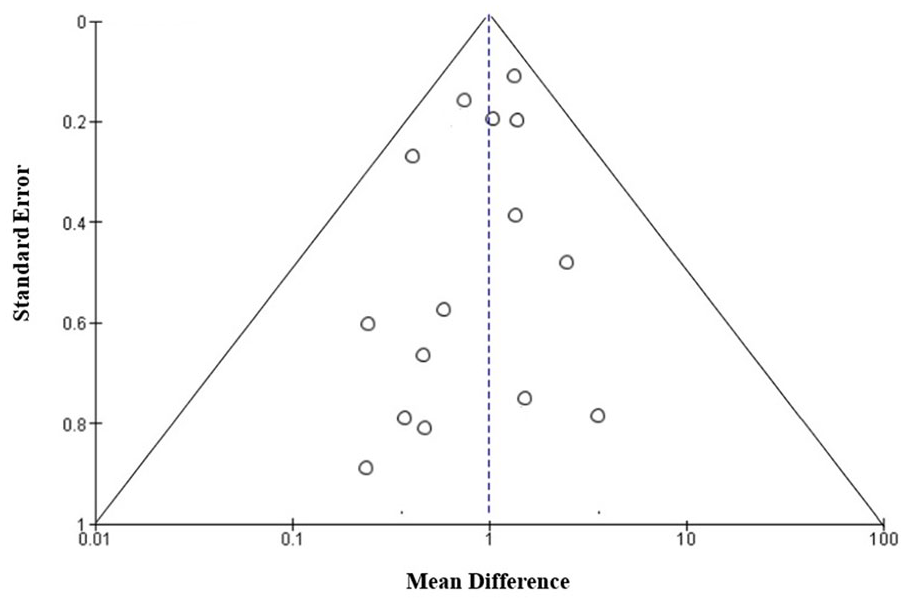
Funnel plot.
Meta-analysis Results
The meta-analysis considered various prospective cohort studies and found that all of them reported a significant statistical difference in the average SNOT-22 scores between the pre-op and post-op periods (P < .05). The magnitude of this difference ranged from 5.1 to 55.4. There was a statistically significant alteration in the average SNOT-22 score across all studies, with a mean difference of 26.02 (95% CI: 12.83-38.60). The findings of individual studies and summary measures are illustrated in Figure 5, as evidenced by the forest plot. The data indicate that 9 studies exhibited a mean change in SNOT-22 and a 95% CI that was greater than or equal to 26.02 (with a range of 12.83-38.60). Conversely, 6 studies demonstrated a mean change and a 95% CI that was less than or equal to the mean of the study. Based on the results of the analysis, it can be inferred that the outcomes of the majority of the studies, specifically 9 out of 15, exhibited a 95% CI that intersected with the mean summary change value of 26.02. As a result, it may be appropriate to classify these outcomes as “good” rather than “fair.” Moreover, there was no notable distinction observed between investigations that utilized the conventional diagnostic criteria for CRS and those that implemented alternative criteria. A random effects model was employed due to the high heterogeneity observed in the study, with an I 2 value of 87.37%.
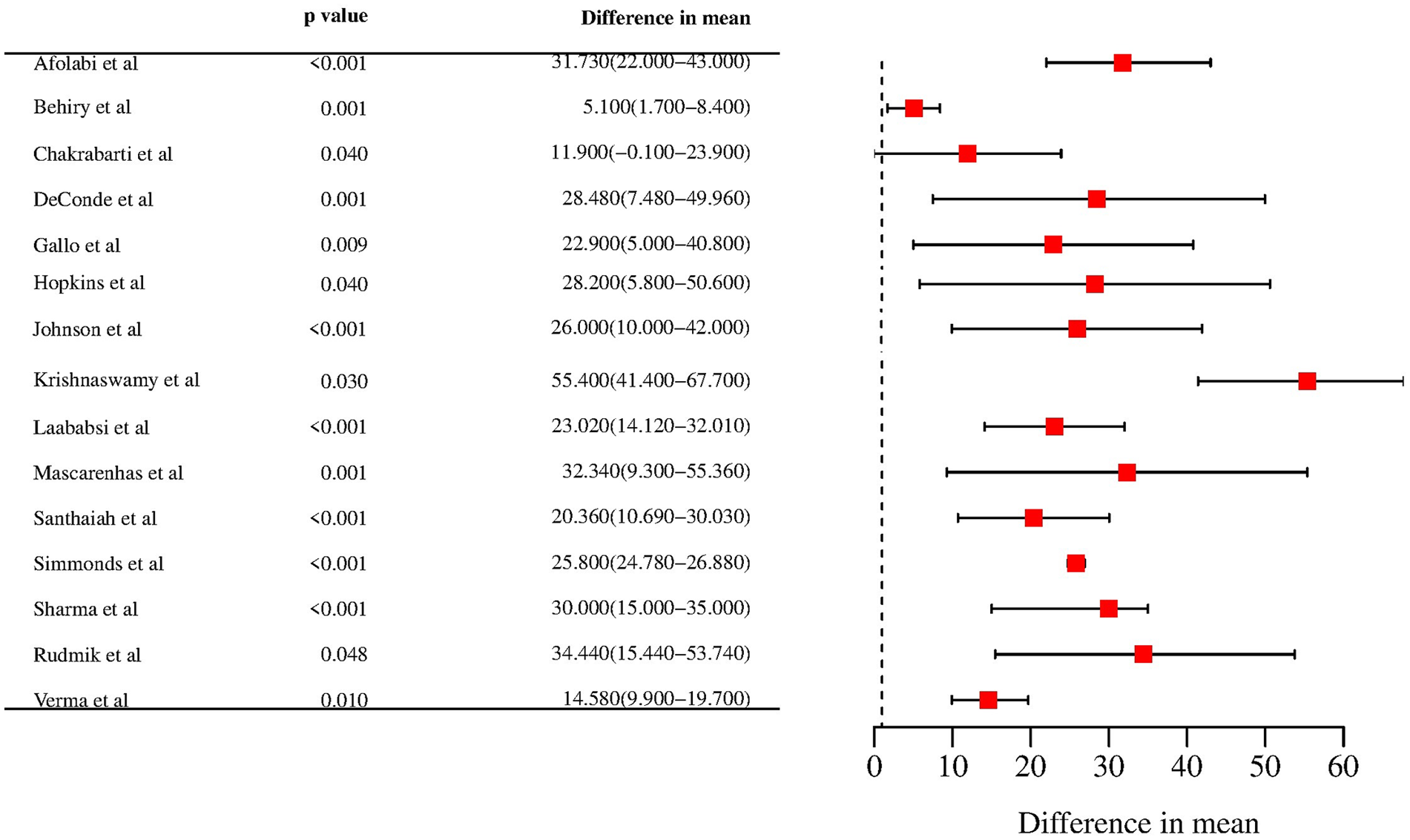
Forest plot for difference in mean value of SNOT-22 score in the included studies.
Across Study Findings (Meta-regression)
The study employed a univariate meta-regression analysis to examine the correlation between patient-specific characteristics and the average alteration in SNOT-22 scores following ESS across the studies that were incorporated. Table 3 displays the primary outcome results, which include the mean change in SNOT-22 score and its corresponding p value. Meanwhile, Figure 6 presents scatter plots that depict the correlation between various study variables, such as the mean change in SNOT-22 score, pre-op SNOT-22 score, mean age of patients, and length of follow-up period. No observable impact was noted on the individual. Based on a stepwise multivariate analysis, it was found that studies with a higher average age and average pre-op SNOT-22 scores exhibited more significant changes in SNOT-22 scores after undergoing ESS. Conversely, studies with a longer average follow-up duration showed less significant changes in SNOT-22 scores. Therefore, the SNOT-22 score prior to surgery, average age of the patients, and duration of the follow-up period remained statistically significant based on the forward, stepwise multivariate modeling.
Statistical Summary of the Primary Outcome: The Difference in Mean Values of Pre-OP and Post-OP SNOT-22 Scores.
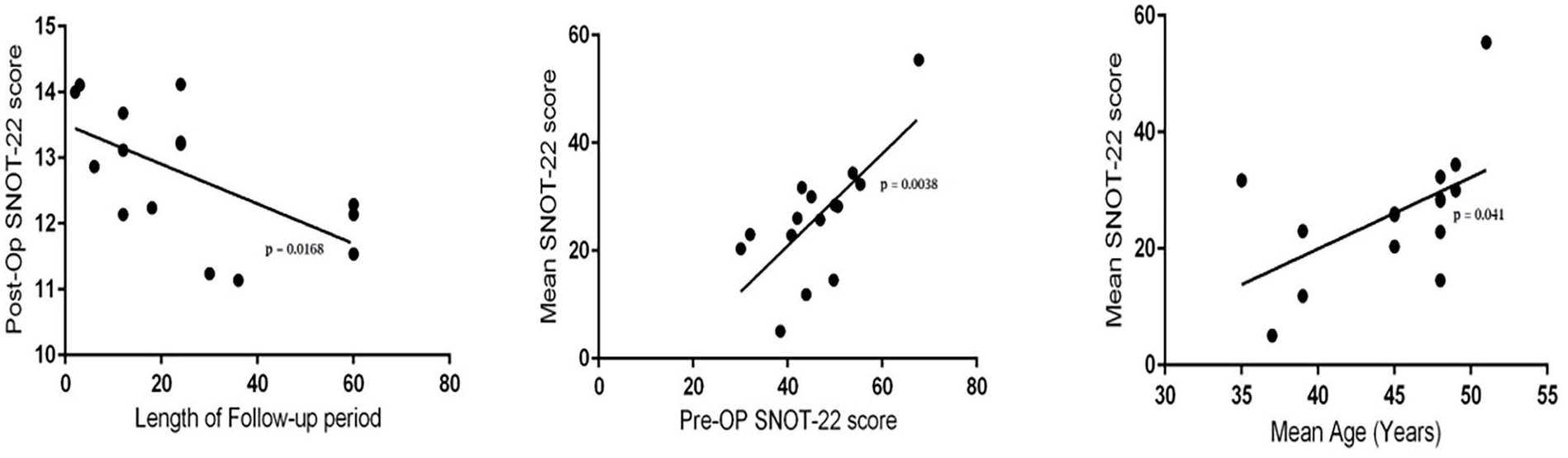
Univariate meta-regression scatter plots for length of follow-up period, Pre-OP SNOT-22 score and mean age.
Discussion
CRS is defined as the persistent inflammation of the paranasal sinuses and nasal channel linings for a duration of 12 weeks or more. This condition encompasses a varied range of illnesses that exhibit similar pathologic and clinical features. 41 Patients diagnosed with CRS are evaluated through the utilization of the SNOT-22, a self-administered assessment tool that has been validated for this purpose. A total of 22 items are assessed on a scale ranging from 0, indicating the absence of any issue, to 5, indicating the most severe symptom imaginable. The SNOT-22 tool is utilized to assess symptoms related to CRS, as well as the impact of the condition on productivity, sleep quality, and social and emotional well-being. This has been documented in previous studies.42,43 SNOT-22 data can be gathered by surgeons both pre- and post-op from patients with CRS. To assess their advancement, a benchmark will be required. 44 The task of identifying appropriate reference points from a multitude of published studies is challenging owing to their heterogeneity. Therefore, a comprehensive synthesis of all the published research on anticipated fluctuations in SNOT-22 is superior to any individual study. The utilization of summary mean and 95% CI would enable healthcare providers to make comparisons between their respective datasets. The attainment of results within the anticipated range installs a sense of assurance, whereas outcomes that fall below this threshold may necessitate further investigation and enhancements in quality. 45
The regression model has the ability to account for baseline characteristics within a surgeon’s practice, including pre-op SNOT-22, mean age of patients, and follow-up time. These factors have been observed to impact the published outcomes across various studies. 46 The systematic review and meta-analysis conducted by several researchers47-49 pertaining to the assessment of SNOT-22 outcomes in patients with CRS who underwent EES have provided summary metrics. These metrics serve as a preliminary reference point based on the latest available data. However, it is important to note that they are not considered to be the most ideal metrics. The statistically significant summary mean (95% CI) of the pre- and post-op changes in the mean SNOT-22 scores of patients with CRS and a P value less than .05 in our study can be utilized as a point of reference for comparative purposes among other studies.
Nonetheless, the data resulting from the integration of all published studies may be subject to various biases. It is plausible that the studies that have been published may have emanated from institutions with more extensive expertise and superior proficiency than initially expected. It is expected that all service providers adhere to certain standards. Research endeavors are prone to emanate from academic institutions, which may result in the studied cohorts not being representative of the societal standards. Alternatively, centers that exhibit substandard outcomes may exhibit a reduced propensity to disseminate their findings, thereby exacerbating the bias in the results. As refinements are made to procedures, criteria are modified, and post-op therapies are enhanced, it is expected that outcomes will evolve over time. The existing body of literature may not necessarily be indicative of this fact.
Limitations
Several constraints must be considered within the scope of this inquiry. The findings are constrained due to the scarcity of studies, amounting to only 15, that exhibit moderate to high degrees of heterogeneity. This is despite the utilization of the recommended level of scientific rigor in the analysis. Secondly, the study utilized aggregated data rather than individual participant data. The inclusion criteria for patients with CRS and the duration of follow-up varied slightly among the studies, potentially leading to some level of internal heterogeneity. This was the situation as the trials exhibited minor variations among them. Thirdly, there was a discernible variation in the precise delineation of primary outcomes among the numerous studies that were included in the analysis. Lastly, it is noteworthy that the search was restricted to publications written in English, which could have potentially introduced some degree of bias in the selection of articles for presentation.
Conclusion
The SNOT-22 has been found to exhibit a significant enhancement in the QOL outcomes of sinus surgery following an ESS. The extent of alteration seems to exhibit significant heterogeneity across the entire body of literature, and this heterogeneity seems to be impacted by several factors, such as the pre-op SNOT-22 score, the average age of the subjects, and the length of the monitoring period. The results of this study offer both a single numerical value and a spectrum of anticipated modifications following surgical intervention. These outcomes can be utilized to advise initiatives aimed at improving the standard of care. Furthermore, administering the SNOT-22 to patients with CRS before undergoing surgical interventions may assist in educating patients regarding their anticipated results, although this is subject to personal interpretation. Recent preliminary research suggests that utilizing a combination of SNOT-22 scores and tissue histopathology may serve as a novel approach for forecasting treatment outcomes among patients with CRS. The precision and accuracy of forthcoming analyses are expected to improve due to endeavors aimed at furnishing unbiased data and patient-level metrics encompassing a broad spectrum of patients and providers.
Footnotes
Acknowledgements
None.
Author Contributions
YF and DL: Conceptualized and designed the study, analyzed data and drafting of the article; WH and ZW: Collected the data and helped in data analysis; YZ: Proofreading and final editing along with guarantor of the article. All authors read and approved the final version of the article.
Consent to participate statement
N/A
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethics
An ethics statement is not applicable because this study is based exclusively on published literature.
Study approval statement
N/A
