Abstract
Keywords
Introduction
The frontal sinus drainage pathway is a complex system responsible for draining mucus produced in the frontal sinus into the nasal cavity. 1 Numerous anatomic variations in frontal sinus drainage make it one of the most complex anatomical regions in head and neck anatomy. 2 The frontal sinus anatomy is so intricate and unique that these variations have even been researched as a way to identify the deceased through forensics and differentiate monozygotic twins.3,4 Even with advancements in technology to delineate the frontal sinus labyrinth, such as computed tomography (CT) and intraoperative stereotactic image guidance systems for functional endoscopic sinus surgery (FESS), the safety and success of an operation still depends greatly on the surgeons’ understanding of these anatomical variants. 2
In order to provide clear communication between surgeons and in surgical planning, a few classification systems throughout history have been utilized to organize the frontal sinus drainage pathway. One of the original classification schemes was described by Bent and Kuhn et al in 1994 which organized sinus cells into frontal cells (types 1–4) and ethmoid cells. 5 In 2014, the European Position Paper on the Anatomical Terminology of the Internal Nose and Paranasal Sinuses discussed the cells in terms of anterior versus posterior and medial versus lateral to the frontal sinus drainage pathway. 6 Finally in 2016, the International Frontal Sinus Anatomy Classification (IFAC) system was published as a collaborative effort between many specialists in the field. 7 This new system took aspects of the Kuhn et al and Lund et al descriptions and focused on anatomical relationships to surrounding structures including the frontal sinus drainage pathway. The IFAC system was created to be more surgically relevant and remove confusion from prior classifications. 7 Figure 1 presents CT imaging examples of each cell type in the IFAC system.
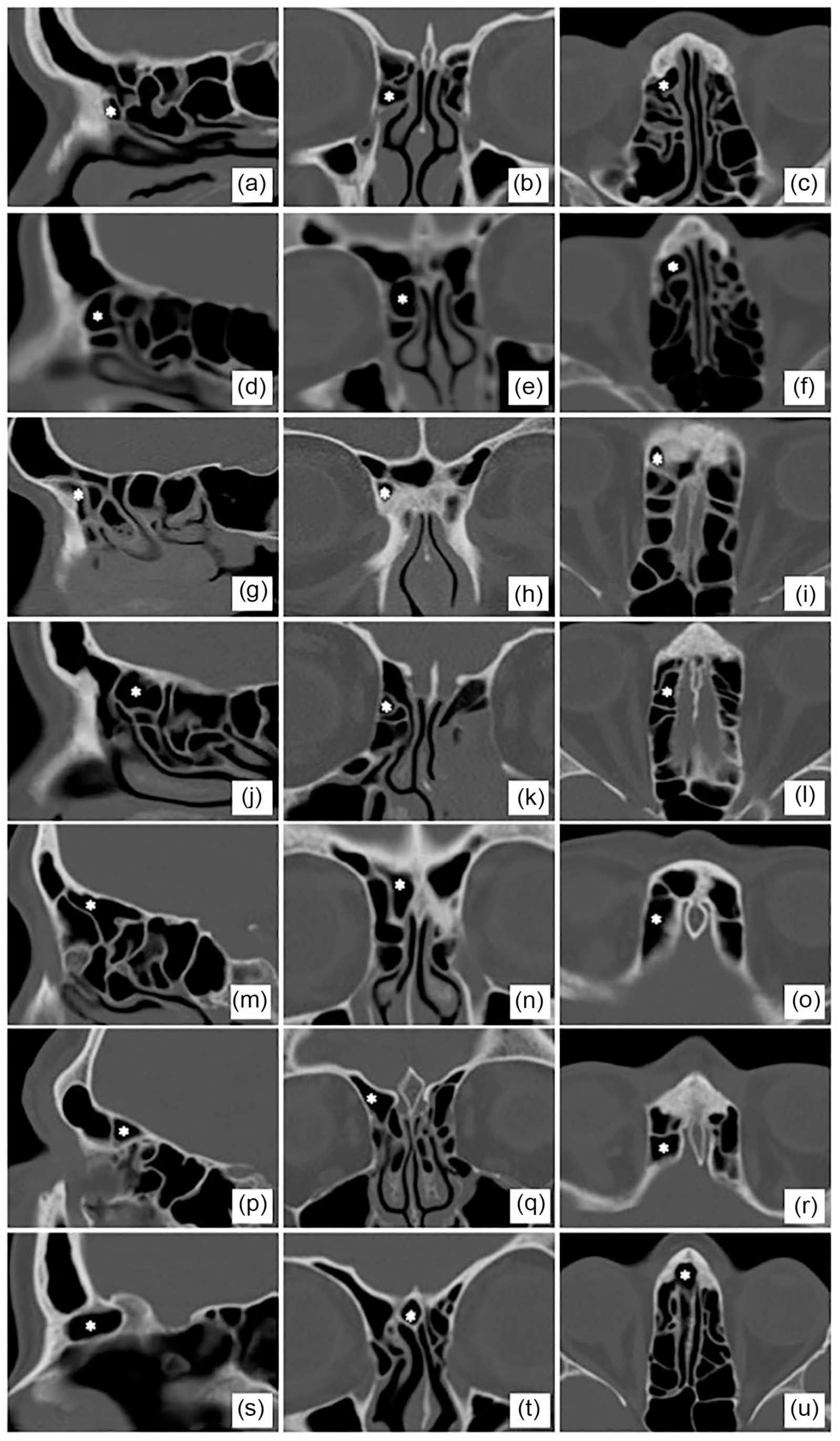
IFAC cell types in sagittal, coronal, and axial CT images. (a-c) agger nasi, (d-f) supra agger, (g-i) supra agger frontal, (j-l) supra bulla, (m-o) supra bulla frontal, (p-r) supraorbital ethmoid, and (s-u) frontal septal.
Previous studies have compared frontal sinus anatomy using the Kuhn system or solely described the distribution of cells found in a population using the IFAC system.8,9 As sinus anatomy has marked variation between individuals, we hypothesized there may be significant differences in frontal sinus anatomy between different races, sex, and those with frontal sinus disease. We therefore designed this study to expand upon current literature and used the IFAC system to classify and compare the frontal sinus anatomy between these patient groups. Improved understanding of these anatomical variants in different populations may have implications in performing safe frontal sinus surgery, particularly in settings without image guidance, as well as gain insight into pathoetiology of frontal sinus disease.
Methods
After obtaining Institutional Review Board exemption (#2010088325) from Indiana University, a single-center, retrospective review of consecutive adult patients with non-contrasted CT sinus or maxillofacial imaging (Current Procedural Terminology [CPT] codes 70486-70488) from 2015 to 2020 was performed. CT scans had 1.0 mm slice thickness in order to detect anatomical discrepancies with sufficient sensitivity. Patients classified into the chronic rhinosinusitis with frontal sinus involvement (CRFS) group were identified from rhinology clinic records who had a diagnosis of chronic rhinosinusitis (CRS). CRS was determined as defined by the current clinical practice guidelines from the American Academy of Otolaryngology—Head & Neck Surgery (AAO—HNS) for chronic sinusitis. 10 This included the presence of frontal sinus mucosal thickening on CT imaging and ≥12 weeks of at least 2 sinonasal symptoms including facial pain-pressure-fullness, anterior and/or posterior mucopurulent drainage, nasal congestion, and/or hyposmia/anosmia. Patients who met the diagnostic CRS criteria and demonstrated persistent CRS symptoms with frontal sinus mucosal disease significant enough to warrant frontal sinus surgery (CPT codes 31276, 31296) despite ≥3 months of intranasal saline irrigations and intranasal steroids were selected for inclusion as the CRFS group. This group included patients with sinonasal polyps, which is also a diagnostic criterion for CRS in the AAO—HNS guidelines. Patients classified as the non-CRFS group were trauma patients who underwent CT imaging as part of standard institutional emergency services protocol and had no evidence of frontal sinus mucosal thickening. Exclusion criteria for both groups included a history of previous sinus surgery, alternate sinusitis diagnosis or etiology (eg, allergic fungal sinusitis, acute recurrent sinusitis, cystic fibrosis, etc), lesions obscuring normal anatomy (eg, tumors), hypoplastic frontal sinuses, fractures involving the frontal sinus or frontal drainage pathway, or sinus disease that inhibits delineation of frontal sinus anatomy (eg, severe sinus opacification).
Patient age, sex, and presence of CRFS were recorded for each patient. CT images were examined in the coronal, axial, and sagittal planes in standard bone windows. Reviewers (LAH, AJJ) were blinded to patient sex, race, and age. Cells were classified according to the IFAC system and grouped as anterior (agger nasi, supra agger cell, supra agger frontal cell), posterior (supra bulla cell, supra bulla frontal cell, supraorbital ethmoid cell), and medial (frontal septal cell). 7 Any discrepancy in classification was disputed among authors.
Statistical analyses were performed using the SPSS v27.0 (IBM Corp., Armonk, NY, USA). Two-sided one-way Welch’s analysis of variance (ANOVA) with Tukey post-hoc testing and Pearson’s χ2 were used for the racial discrepancy analyses. Two-sided Welch’s t-test or Fisher’s exact test were used for both the sex and presence of CRFS analyses. Effect sizes were calculated as Cohen’s d (2 groups) or ω2 (≥3 groups) for continuous variables, odds ratio (OR) for binary categorical variables, and Cramer’s v for categorical variables with ≥3 groups. Corresponding 95% confidence intervals (95% CIs) were calculated. Statistical significance was determined at P < 0.05.
Results
Overview
A summary of the study population is listed in Table 1. A total of 184 patients (368 scans) were included in the analysis, 90 (48.9%) of which had CRFS. The cohort consisted of 96 (52.2%) females with mean age 43.3 ± 17.2 years and racial distribution of 50 white (27.2%), 50 black (27.2%), 45 Hispanic/Latino (24.5%), and 39 Asian (21.2%) patients.
Cohort Overview and Summary.
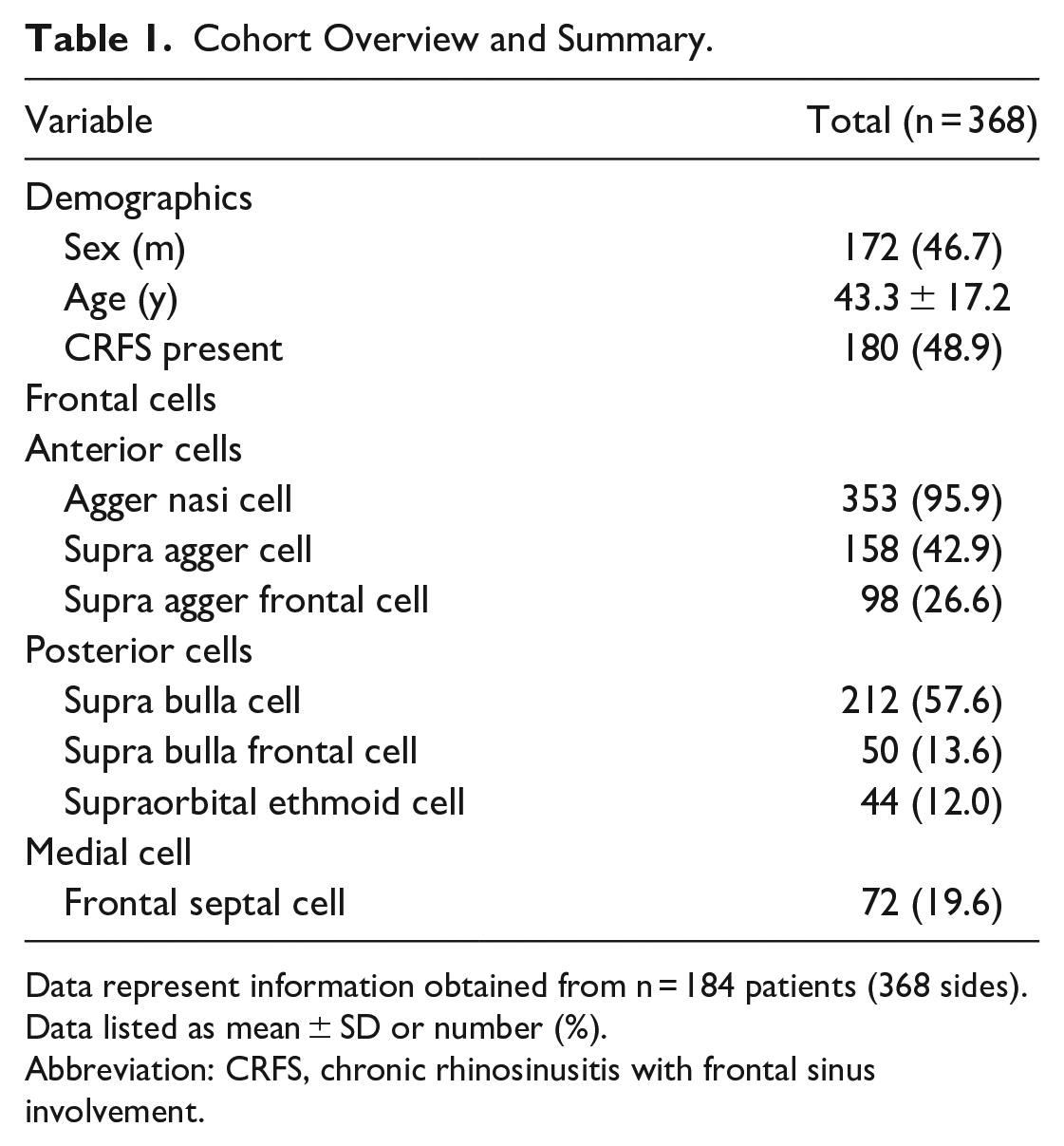
Data represent information obtained from n = 184 patients (368 sides). Data listed as mean ± SD or number (%).
Abbreviation: CRFS, chronic rhinosinusitis with frontal sinus involvement.
Anteriorly, the agger nasi cell was present in 95.9% of the population, with a supra agger and supra agger frontal cell present in 42.9% and 26.6% of patients, respectively. For the posterior cells, the supra bulla cell was found in 57.6% of patients, the supra bulla frontal cell was found in 13.6%, and the supraorbital ethmoid cell in 12% of patients. Medially, the frontal septal cell was identified in 19.6% of patients.
Variations in Race
As shown in Table 2, the cohort was analyzed by race with regard to frontal sinus cell anatomy. No difference in sex distribution was identified, but CRFS was more prevalent in the Asian population compared to white, black, and Hispanic/Latino groups (64.1% vs 40.0-48.9%, P = 0.013). Asian patients were also younger on average than white patients (40.9 ± 13.2 vs 46.6 ± 15.5 years, P = 0.018). The supra agger cell was more common in the white population (57%, P = 0.009), and the supraorbital ethmoid cell was more prevalent in Asians (21.8% vs 7.8%-12.0%, P = 0.017).
Racial Variations in Frontal Sinus Anatomy.
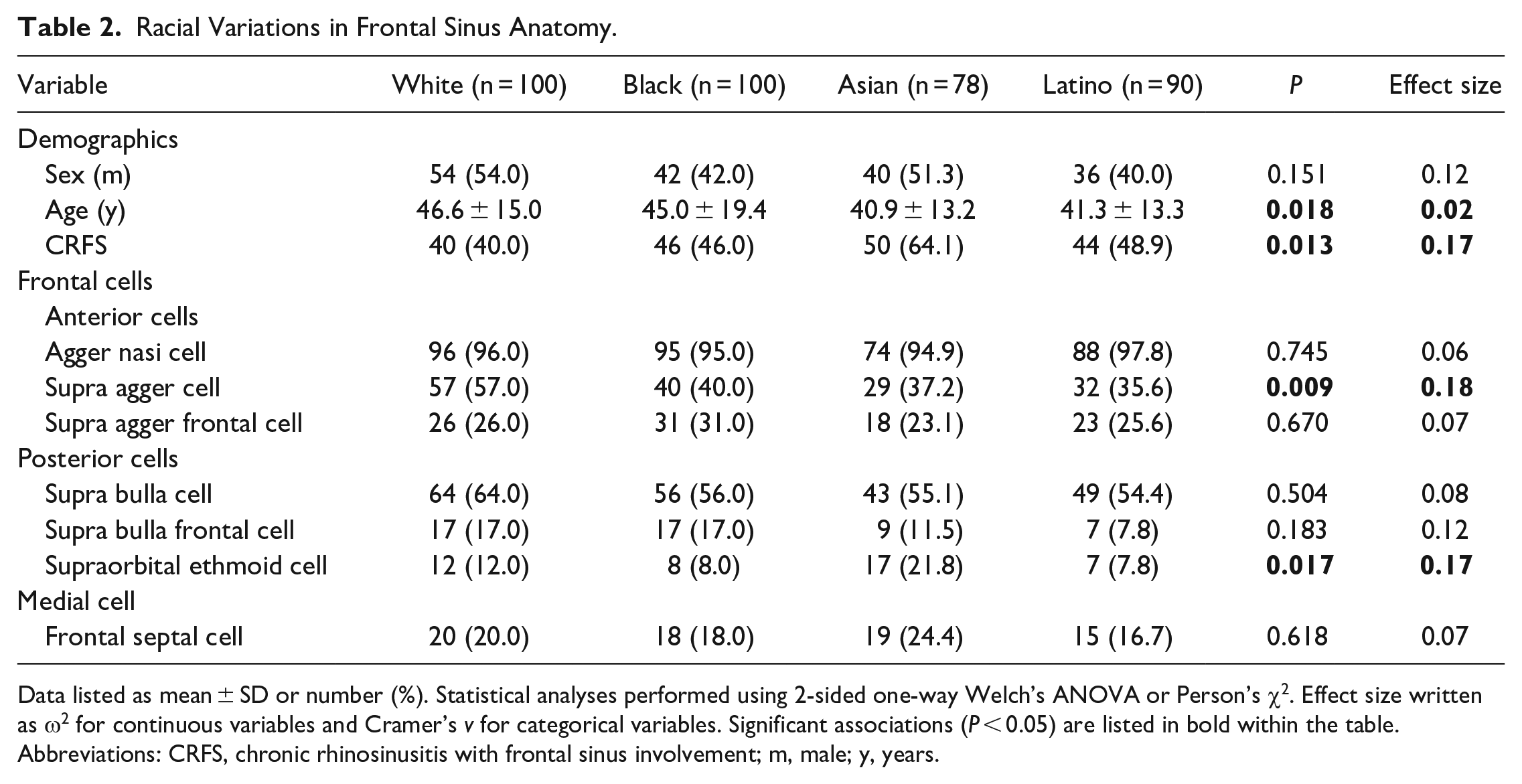
Data listed as mean ± SD or number (%). Statistical analyses performed using 2-sided one-way Welch’s ANOVA or Person’s χ2. Effect size written as ω2 for continuous variables and Cramer’s v for categorical variables. Significant associations (P < 0.05) are listed in bold within the table.
Abbreviations: CRFS, chronic rhinosinusitis with frontal sinus involvement; m, male; y, years.
Variations in Sex
Table 3 shows the results when the cohort was analyzed according to sex. There were no statistical differences in age or racial distributions between sexes. The supra agger frontal cell was more prevalent in males (32.0% vs 21.9%, P = 0.034). The supraorbital ethmoid cell was similarly more prevalent in males (18.6 % vs 6.1%, P < 0.001).
Sex Variations in Frontal Sinus Anatomy.
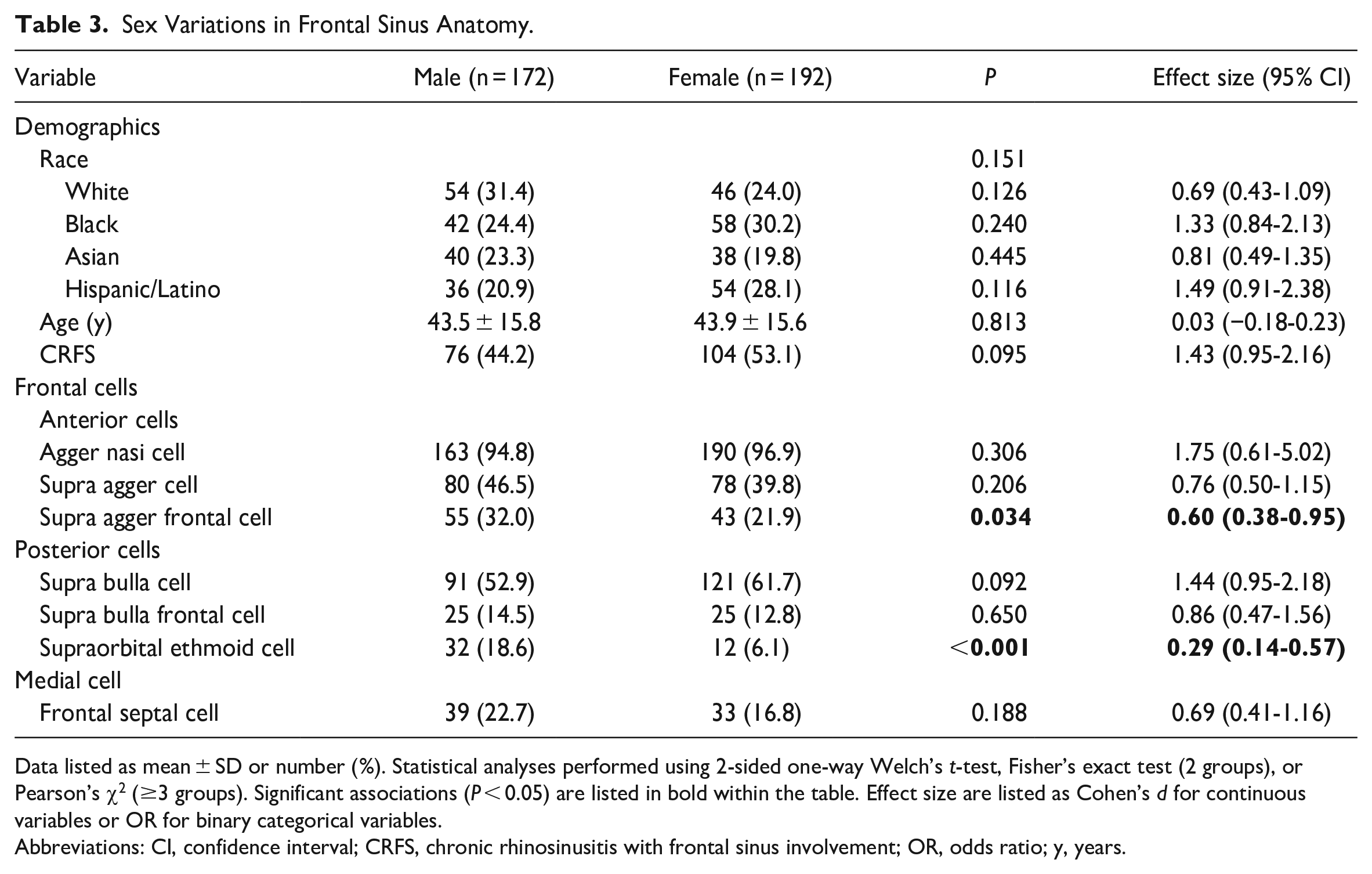
Data listed as mean ± SD or number (%). Statistical analyses performed using 2-sided one-way Welch’s t-test, Fisher’s exact test (2 groups), or Pearson’s χ2 (≥3 groups). Significant associations (P < 0.05) are listed in bold within the table. Effect size are listed as Cohen’s d for continuous variables or OR for binary categorical variables.
Abbreviations: CI, confidence interval; CRFS, chronic rhinosinusitis with frontal sinus involvement; OR, odds ratio; y, years.
Variations in CRFS
As mentioned, a control group of non-sinusitis trauma patients was compared to a cohort of patients diagnosed with CRS involving the frontal sinus (Table 4). There were no differences in age or sex distributions. Caucasians comprised a higher percentage of the control population (31.9% vs 22.2%, P = 0.046), while Asian patients had a higher rate of CRFS (27.8% vs 14.9%, P = 0.003). Both supra agger cells (50.0% vs 35.6%, P = 0.006) and supra bulla cells (65.4% vs 49.4%, P = 0.002) were identified more frequently in control patients. In contrast, supraorbital ethmoid cells had greater prevalence in patients with CRFS (16.1% vs 8.0%, P = 0.024).
Variations in Frontal Sinus Anatomy in CRFS.
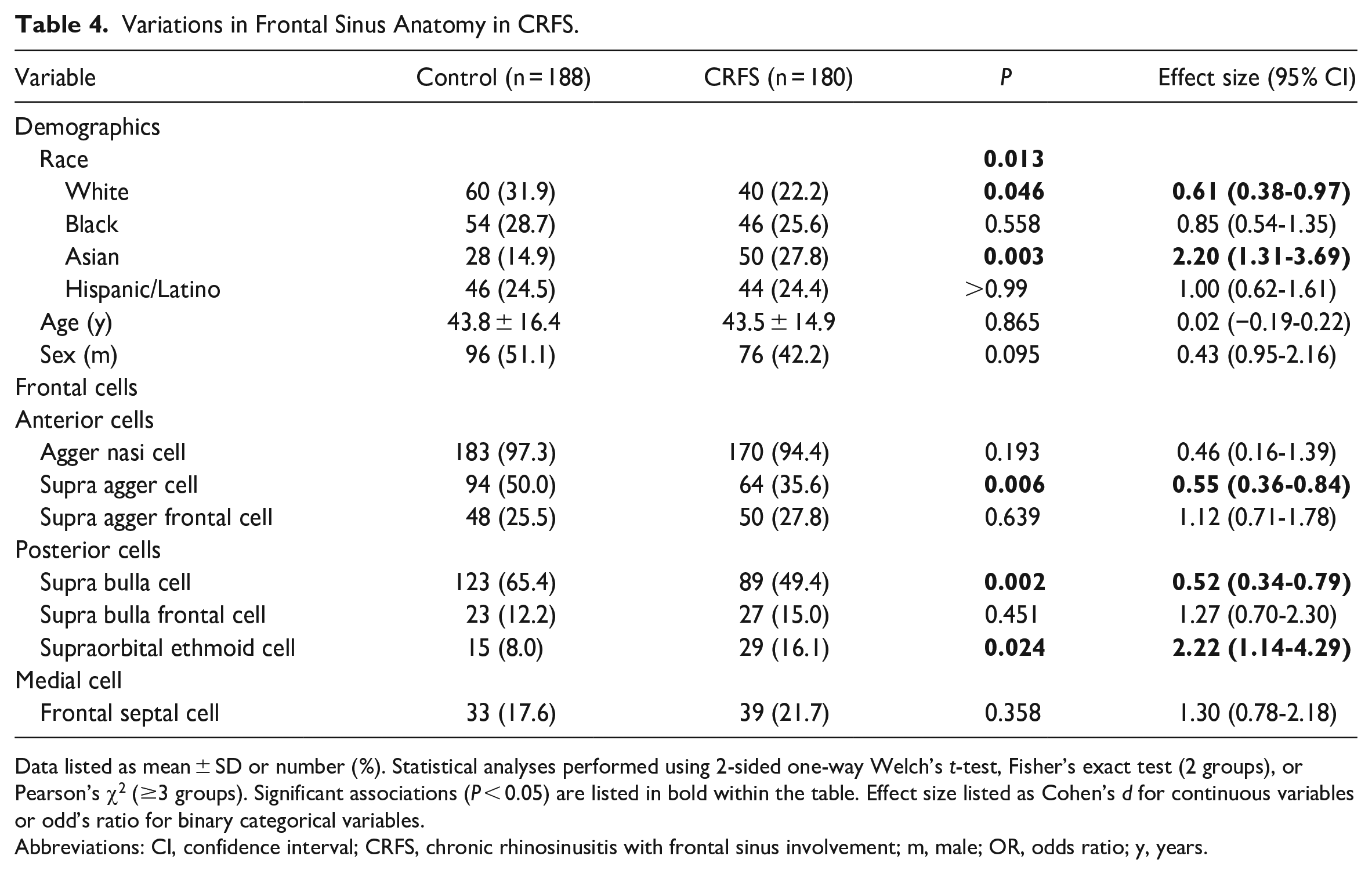
Data listed as mean ± SD or number (%). Statistical analyses performed using 2-sided one-way Welch’s t-test, Fisher’s exact test (2 groups), or Pearson’s χ2 (≥3 groups). Significant associations (P < 0.05) are listed in bold within the table. Effect size listed as Cohen’s d for continuous variables or odd’s ratio for binary categorical variables.
Abbreviations: CI, confidence interval; CRFS, chronic rhinosinusitis with frontal sinus involvement; m, male; OR, odds ratio; y, years.
Discussion
Although the traditional Kuhn frontal cell classification is still utilized clinically and taught in otolaryngology training, it is likely to be replaced by international consensus of the IFAC system. It is therefore important for the frontal sinus surgeon to understand its nomenclature and relevant anatomical differences between patient demographics and disease states within this system. Herein, we provide one of the largest radiographic analyses of the frontal sinus anatomy and the first report of findings between sexes and several races utilizing the IFAC system and nomenclature.
The Kuhn and IFAC systems have some overlapping definitions of cells, including the agger nasi cell and supraorbital ethmoid cell. 5 Other cells identified in the Kuhn system have counterparts that are named slightly different in the IFAC system. The suprabullar cell is now called the supra bulla cell, the frontal bulla is called supra bulla frontal, and the inter-frontal sinus septal cell is now called the frontal septal cell. The type 1–4 frontal cells in the Kuhn system were consolidated and renamed. Type 1 is now the supra agger cell, type 2 would be multiple supra agger cells, and types 3 and 4 are supra agger frontal cells. The definitions of these in the IFAC system were meant to be clearer and utilize surrounding structures such as the anterior ethmoidal artery in defining cells.
Five previous investigations have evaluated the prevalence of each cell type using the IFAC system.8,11–14 Compared to these studies, our results fall within the ranges previously reported in literature. Prior investigations have similarly identified the nearly ubiquitous agger nasi cell in 95.5% to 97% of patients, further proving its utility as an excellent radiographic and surgical landmark. However, we noted moderate variation in the presence of other frontal sinus cells. The supra agger cell and supra agger frontal cells had prevalence ranging 16.3% to 50% and 11% to 36% of patients, respectively. Large variation was also demonstrated in the posterior cells with the presence of supra bulla cell at 46.2% to 89%, supra bulla frontal cell at 4.3% to 53%, and supraorbital ethmoid cell at 5.5% to 42%. Medially, the frontal septal cell was more consistent across literature with a range of 8.3% to 28%. These differences are likely due to either a difference in populations studied (race, age, sex) or expected variation from sampling. Of the aforementioned reports, none reported racial composition except Fawzi et al, which analyzed only Malaysian patients. 13 Choby et al and Tran et al excluded patients with sinus disease while Fawzi et al, Sommer et al, and Nofal & El-Anwar specifically studied sinusitis patients.8,11–14 Our study included several racial groups and patients with CRFS, which overall represents a balanced sample of patients presenting to clinic.
Fawzi et al also compared cell types to presence of sinusitis using the IFAC system. 13 They identified a significant association between the supraorbital ethmoid cell and frontal septal cell with the development of frontal sinusitis. We similarly determined the supraorbital ethmoid cell was associated with CRFS. The difference in our exact cell ratios and the contrast in frontal septal cell significance may be related to differences in the populations studied. The Fawzi et al investigation was completed in Malaysia with only an Asian population represented in analysis and included only patients with known sinus disease. 13 Two other studies, looking at German and Indian groups, found no significant association between any IFAC cell and sinusitis.12,15 Similarly, Nofal et al examined patients with a diagnosis of CRS undergoing FESS and did not identify any difference in the distribution of infected frontal sinus cells between patients with and without frontal sinusitis. 14
Regarding differences between sex, no previous study has reported this analysis to compare findings. However, sex hormones have demonstrated an effect on the development of craniofacial structure which could provide an explanation for our findings. 16 Regarding race, other investigators have shown that supraorbital ethmoid cells were associated with a Caucasian population and supra bulla cells were more common in the East Asian population.17,18 Both of these studies were completed prior to the IFAC system being released and utilized an alternative classification system, which could explain why our results do not show the same patterns. There are many studies examining the relationship between race and craniofacial development, one of which showed differences in obstructive sleep apnea risk based on facial phenotype across different ethnicities. 19 This theory gives merit to our interest in sinonasal anatomic differences between ethnic groups, as genetic differences impact the development of the craniofacial bone structure and, subsequently, the frontal sinus development.
Presence of an agger nasi cell or incomplete removal of an agger nasi has been correlated with frontal sinusitis and persistent disease.20,21 Given the agger nasi is a variable-sized cell, patients with a large agger nasi cell obstructing the nasofrontal duct were found to be twice as likely to require surgical intervention. 20 We found that a supra agger cell was actually more prevalent in patients without frontal sinus mucosal thickening. This could indicate that the presence of a supra agger cell diminishes the size of the agger nasi, acting more as a septation and division of the agger nasi, or that the supra agger cell displaces the agger nasi in such a fashion to actually widen the infundibulum. The etiology for the supraorbital ethmoid cell contributing to frontal sinus mucosal disease in our population could stem from a superior cell encroaching on the infundibulum, causing a blockage upstream in the sinus drainage pathway. Further analyses are required to delineate anatomical factors and development that predispose to frontal sinus obstruction and disease.
Understanding anatomical patterns and variation may improve surgeon preparedness prior to intervention and awareness of at-risk groups for frontal sinus disease. Ensuring appropriate opening of the obstructing frontal sinus cells in patients with CRFS can improve postoperative outcomes. This could be useful in circumstances where image CT imaging is unavailable, particularly in resource-limited settings. However, these sex and racial differences should not replace meticulous examination of preoperative CT sinus imaging when it is available, as this is the most important factor in surgical preparedness.
Our investigation is limited in its retrospective bias and utilizing a regional sample. We excluded distorted imaging that could not be accurately classified using the IFAC system, which excluded patients with severe frontal sinus disease from the study. As previously investigated, IFAC classification accuracy is limited with extensive CRS and most useful in patients with lower Lund–Mackay scores. 22 Therefore, our findings are biased toward mild to moderate as opposed to severe frontal sinus disease due to the exclusion criteria. Because of our clinical practice setting and regional sampling, we were unable to include the American Indian, Alaskan native, native Hawaiian, Pacific islander, or other populations in our racial analysis, which is a commonly noted drawback in many study populations. Future studies are needed to determine surgical implications of frontal sinus anatomy on patient outcomes.
Conclusion
The IFAC system provides a more surgically relevant and comprehensive system by which to classify the frontal sinus anatomy and drainage pathway. Key differences in frontal sinus anatomy exist between different races and those with CRFS. Our investigation identified chronic frontal sinus disease to be associated with the presence of a supraorbital ethmoid cell and Asian patients. Knowledge of these anatomical differences and relationships can improve patient care and surgical planning.
Footnotes
Author’s Note
This manuscript was presented as a poster at the 125th Annual American Academy of Otolaryngology—Head & Neck Surgery Meeting in Los Angeles, CA, on October 3–6, 2021.
Author Contributions
Lauren A. Howser: data collection, data analysis, manuscript drafting and editing; Alexander J. Jones: idea conception, data collection, data analysis, manuscript drafting and editing; Satyan B. Sreenath: idea conception, manuscript drafting and editing; Jonathan Y. Ting: idea conception, manuscript editing; Elisa A. Illing: idea conception, manuscript drafting and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Indiana University Kuali Coeus Institutional Review Board (#2010088325).
Statement of Informed Consent
Informed consent for patient information to be published in this article was not obtained because of its retrospective study design.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Indiana University Kuali Coeus Institutional Review Board-approved protocols.
