Abstract
Congenital bilateral vocal fold paralysis (BVFP) is a rare but significant cause of morbidity in pediatric otolaryngology. The differential diagnosis is expansive, with common etiologies including birth trauma, brainstem neoplasms, and neurologic disorders. There are few known genetic causes of the condition. This report details the first known case of BVFP secondary to a genetic deficiency in MYOD1, a master transcriptional regulator of skeletal muscle cell specification. Genetics consultation and testing may be a useful adjunct in the workup of congenital BVFP and may help guide prognostication, additional workup, counseling, and clinical decision-making.
Introduction
Congenital bilateral vocal fold paralysis (BVFP) impacts less than 1 in 1,000,000 neonates but can carry significant morbidity. 1 Inability to normally abduct the vocal folds during respiration may lead to respiratory failure, feeding difficulty, and failure to thrive. About one-half of cases are idiopathic, with a high rate of spontaneous return of function. 2 Other causes include presumed birth trauma, brainstem neoplasms, neurologic disorders like Arnold–Chiari malformation, and neuromuscular junction defects known as congenital myasthenia syndromes. 3 There are also several named syndromes associated with congenital BVFP including Moebius syndrome, Robinow’s syndrome, 22q deletion syndrome, and Down syndrome. 4 In this report, we describe the first known case of congenital BVFP secondary to congenital myopathy due to mutations in MYOD1, a gene encoding a master regulatory transcription factor of skeletal muscle cell specification.
Case Report
A full-term male infant presented to the hospital in respiratory distress 1 day following an uncomplicated home birth. He was stabilized on continuous positive airway pressure support via face mask. Due to concern for increased work of breathing with advancement of oral feeds, a nasogastric tube was placed, and enteral tube feeds were initiated. Clinical exam was notable for inspiratory stridor, micrognathia, hand and finger anomalies, undescended testes, unilateral hydronephrosis, and hypotonia. Otolaryngology was consulted, and flexible laryngoscopy revealed mild tongue base obstruction and bilateral paramedian vocal cord position with minimal and no movement of the right and left true vocal folds, respectively. Cranial ultrasound and magnetic resonance imaging of the brain demonstrated normal findings. The Genetics service was consulted, and genetic testing, including clinical exome sequencing, was ordered.
Prior to the finalization of genetic testing results, the patient exhibited worsening respiratory failure and inability to wean from noninvasive respiratory support. The driving mechanism was unclear and thought to be multifactorial: vocal fold immobility with reduced glottic airway caliber, micrognathia with possible tongue base obstruction, and/or hypotonia with impaired respiratory effort and restrictive lung physiology. This diagnostic dilemma translated into a treatment dilemma as each etiology requires a different management. The decision was made to proceed with direct laryngoscopy and endoscopy-assisted left true vocal fold suture lateralization to address the glottic airway. Other than the paramedian immobile true vocal folds and mild retrognathia with a grade 2 laryngoscopic view, direct laryngoscopy showed normal airway anatomy with freely mobile cricoarytenoid joints (Figure 1).
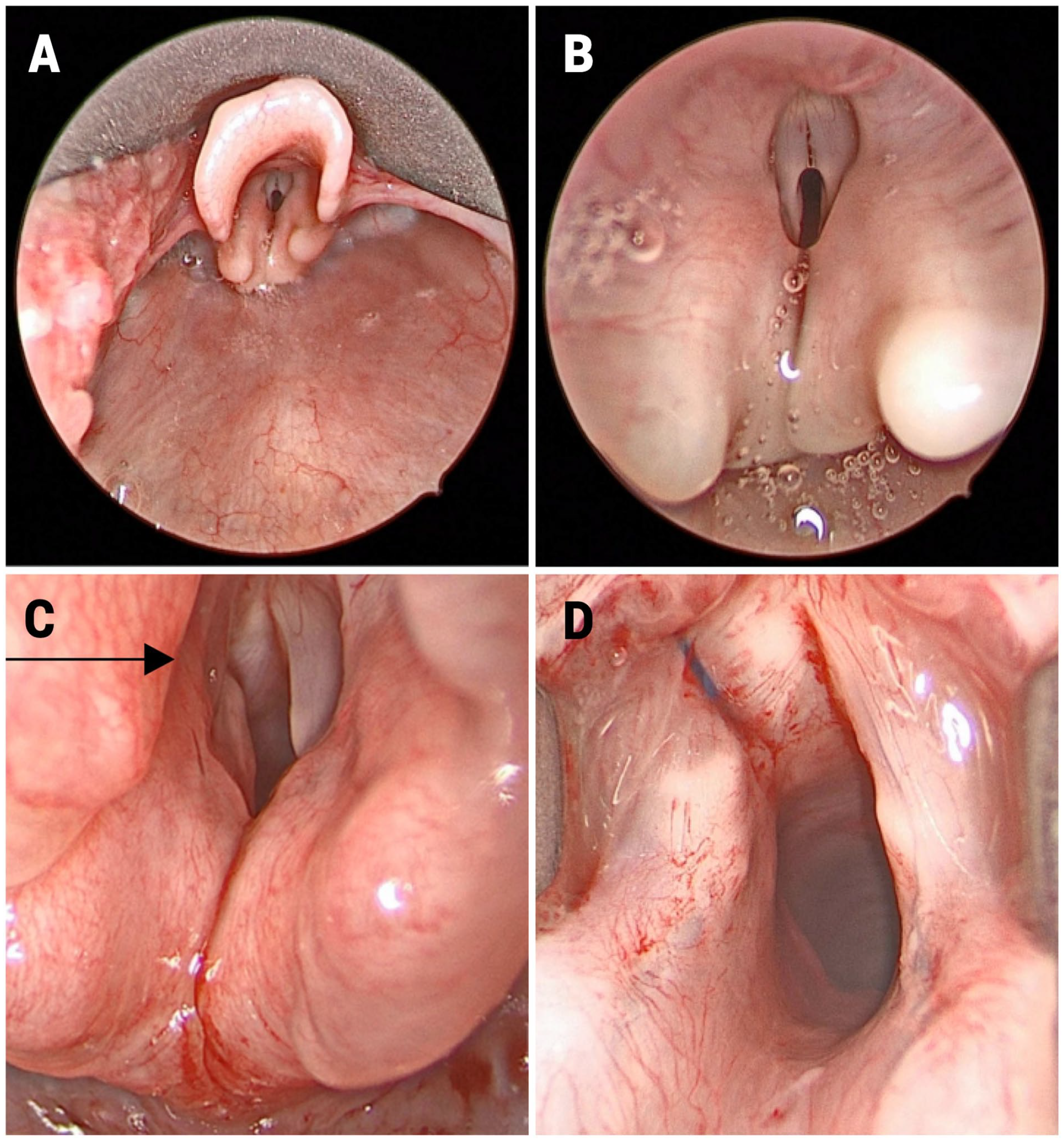
(A) Supraglottic view. Grade 1 laryngoscopic view achieved with anterior cricoid pressure with Parson’s laryngoscope. (B) Paramedian vocal folds. Normal vocal fold anatomy with bilateral paramedian vocal fold position. (C) Left true vocal fold suture lateralization. 4-0 Prolene suture loop lateralization of left true vocal fold. Note resulting increased glottic aperture.
Postoperatively, the patient showed improvement in stridor and work of breathing but continued to struggle with poor weight gain, impaired respiratory effort, and difficulty weaning from noninvasive respiratory support via high-velocity therapy via nasal cannula. A video fluoroscopic swallow study demonstrated moderate oropharyngeal dysphagia characterized by weak and inefficient sucking but without evidence of aspiration. Clinical exome sequencing results revealed pathogenic variants in MYOD1, a gene that encodes a regulatory transcription factor that is a master regulator of skeletal muscle cell specification with roles in differentiation and repair. This led to a diagnosis of MYOD1-related congenital myopathy with diaphragmatic defects, respiratory insufficiency, and dysmorphic facies. Given the genetic findings, the decision was made to proceed with tracheostomy in anticipation of chronic mechanical ventilatory needs. The patient underwent tracheostomy and reversal of the left true vocal fold lateralization suture at 3 months of age. A gastrostomy tube was placed at the time of surgery in anticipation of long-term enteral feeding needs. He did well postoperatively on mechanical ventilation and was discharged at 5 months of age on a home ventilator machine.
Discussion
Congenital BVFP is frequently characterized by stridor and failure to thrive in the neonate. The diagnosis is made with a thorough history and exam including awake flexible laryngoscopy. Airway evaluation under anesthesia is often performed to evaluate for other pathologies and to differentiate vocal fold paralysis from fixation. Additional workup is non-protocolized and varies with patient presentation but often includes imaging to evaluate the central nervous system.
MYOD1 deficiency in humans is rare, and presentation is varied—there have been 3 published cases of lethal fatal akinesia and 2 published cases of milder phenotypes. Reported features include craniofacial anomalies, respiratory insufficiency and diaphragmatic anomalies, hypotonia, arthrogryposis, and renal anomalies.5 -7 Bilateral vocal fold immobility should be considered as a cause of respiratory failure in patients with congenital myopathy, and conversely, congenital myopathies should be considered in the differential diagnosis of BVFP.
To date, there is no established algorithm for management of congenital BVFP. Clinical decision-making typically requires a multi-disciplinary approach and is tailored for the individual patient. The primary aim is to establish a safe airway while minimizing compromise of voice and swallow functions. Traditionally, the gold standard for airway securement was tracheostomy to bypass glottic level airway obstruction. However, the decision to proceed with tracheostomy is not without social and financial consequence for the patient and family and carries high morbidity and mortality risk. A variety of less invasive management options, summarized in Table 1, have come into favor with advancements in medical care and technology.
Watchful waiting on noninvasive respiratory support may be a reasonable initial management option, given the high incidence of spontaneous recovery, particularly in idiopathic cases. However, this patient with multiple congenital anomalies was likely to have an underlying etiology and additional contributing factors. Percutaneous suture lateralization, a reversible endoscopic procedure, was specifically chosen in this case to assess the degree to which glottic narrowing was contributing to the patient’s chronic respiratory failure. 10 Failure of the patient to improve with this intervention helped demonstrate that the patient’s respiratory failure was primarily due to MYOD1 deficiency-related hypotonia and restrictive lung physiology. Given this finding, the decision was made to proceed with tracheostomy to facilitate long-term mechanical ventilation, and the lateralization suture was easily removed at the time of tracheostomy. Additionally, speech–language pathology feeding evaluations consistently demonstrated poor oropharyngeal muscle tone, resulting in inefficient latching, and sucking and premature fatigue with oral feedings. A gastrostomy tube was placed at the time of tracheostomy for tube feed supplementation of the oral feedings and to assist with weight gain.
This is the first reported case of BVFP secondary to a genetically diagnosed congenital myopathy. Long-term prognosis for this patient is unclear, given the rarity of this disease process. However, there has been a notable phenotypic variability in prior published cases of MYOD1 deficiency, with 2 out of 5 previously described patients reportedly doing well in childhood. Genetics consultation and testing may be a useful adjunct in the workup of congenital BVFP in addition to neurologic workup—especially in patients who present with other congenital anomalies such as hypotonia—and may help guide prognostication, additional workup, counseling, and clinical decision-making.
Footnotes
Data Accessibility Statement
Data accessibility statement is not applicable to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent for patient information to be published in this article was not obtained as it has been deemed to not meet criteria for research.

