Abstract
Introduction
Adult laryngeal hemangiomas (ALHs) are rare and usually involve the supraglottic or glottic region.1,2 The causative factors are thought to be vocal abuse, cigarette smoking, and laryngeal trauma (i.e., intubation).1,2 The treatment of ALHs has not been studied systematically and the optimal surgical approach for treating these hemangiomas remains controversial. Some mild asymptomatic cases do not require any medical intervention, while most scholars suggested early intervention for symptomatic cases, those with a risk of tumor rupture, or psychological stress.3,4 Leaving ALHs untreated in some cases can lead to life-threatening hemoptysis or airway obstruction.
The challenge with adult hemangiomas is that unlike in children, they are not responsive to medications/propranolol. Traditionally, corticosteroids were considered the first-line medical treatment for hemangiomas, as they induce tumor shrinkage, relieve symptoms, and have high response rates, with particularly good outcomes seen in pediatric patients with infantile hemangiomas.3-5 However, tumor rebound or relapse occurs in approximately 30% of patients after corticosteroid treatment.3-5 Reported treatments include surgical removal, sclerotherapy,6,7 cryosurgery, 8 laser surgery,9-11 radiation therapy, 12 and transoral robotic surgery. 4 However, sclerotherapy requires multiple intralesional applications and causes adverse pulmonary fibrosis. 6 Laser surgery is relatively effective and less invasive than surgical removal,9-11 but laser facilities are expensive and most hospitals lack them. Several authors do not recommend CO2 laser excision for adult cavernous hemangiomas with large-diameter vessels. 13
Low-temperature plasma radiofrequency (LPRF) coblation is an evolving technology that can cut soft tissue and provide hemostasis at lower temperatures (40-70 °C) 14 ; it has achieved good results in the treatment of head and neck hemangiomas. 15 With the recent rapid development of LPRF, hemostasis can be achieved by blocking the blood vessels while ablating the hemangioma, which is very suitable for treating hemorrhagic diseases. 16 However, there are no reports on the use of LPRF ablation strategies for the treatment of ALH. Here, we present a series of patients with ALHs that were excised successfully by LPRF coblation without complications.
Materials and Methods
Ethical Considerations
The study protocol was reviewed and approved by the Institutional Ethics Review Board of Yiwu Central Hospital in Yiwu, Zhejiang, China. Informed consent was obtained from all participants.
Materials
This retrospective case series reviewed patients treated from January 2016 to March 2022. The inclusion criteria were over 18 years old, in good health, diagnosed with laryngeal hemangioma by clinical history and videostroboscopic examination, and no previous surgery or medical treatment for the hemangioma. All patients underwent preoperative computed tomography (CT) of the neck with contrast enhancement. The hemangioma subtypes were diagnosed from the pathology or clinical appearance and radiographic findings. Body mass index (BMI) was classified as normal (<25 kg/m2), overweight (25-29.9 kg/m2), or obese (≥30 kg/m2).
The patients underwent preoperative and postoperative voice and swallowing assessments. The voice assessment consisted of videolaryngoscopy, self-assessment using the Voice Handicap Index (VHI), and calculation of the maximum phonation time (MPT). Videostroboscopic images were evaluated in terms of glottal closure, mucosal wave, and symmetry. Swallowing was evaluated according to the classification proposed by Bazaz et al 17 Using this system, the patient’s swallowing symptoms are classified as none (no difficulty swallowing liquids or solids), mild (no difficulty swallowing liquids; rare difficulty swallowing solids), moderate (no or rare difficulty swallowing liquids; occasional, food-specific, difficulty swallowing solids), or severe (no or rare difficulty swallowing liquids; frequent difficulty swallowing solids) dysphagia.
Surgical Procedure
All the surgical interventions were performed under general anesthesia using a suitable endotracheal tube (internal diameter = 5.5-6.5 mm), with the hemangioma fully exposed under suspension laryngoscopy. Video-assisted laryngoscopy was performed using a 0° rigid endoscope connected to a high-resolution endoscopic video camera. The operative field was amplified for clarity.
The RF coblation system was from China Micro Medical Limited Shanghai Micro Medical Devices (Shanghai, China); the Evac 70 Plasma wand was preferred for the treatment of hemangiomas in the larynx. Throughout the process, the Evac 70 wand was set at an ablation power of 7 and a coagulation power of 3.
The coagulation function of the plasma wand was first used on the tumor, which was gradually coagulated from 1 to 2 mm around the tumor margins to the center. The dark red tumor became gray and decreased in size, revealing the tumor base. Then, the ablation function of the plasma wand was used: the tumor base and surrounding mucosa were removed for capillary hemangiomas (Figure 1), and the swelling tumor body was reduced quickly and finally flattened for cavernous hemangiomas.
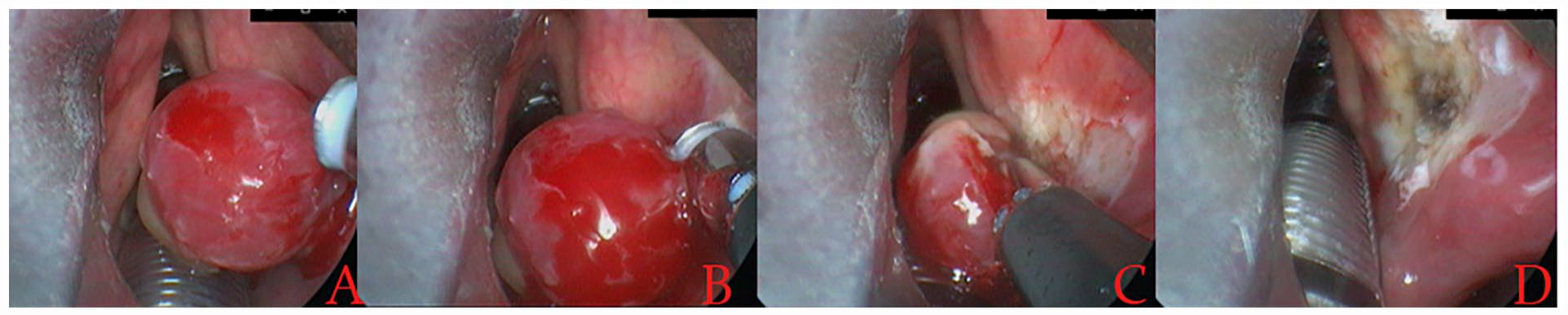
A 76-year-old man with laryngeal capillary fibroangioma. Preoperative (A), LPRF coblation procedure (B and C), and postoperative (D). LPRF, low-temperature plasma radiofrequency.
Postoperative Treatment and Outcome Measures
Oral antibiotics and atomization inhalation were given for 1 week postoperatively and the patients were instructed to remain on strict voice rest for 7 days. Postoperative follow-up was done 1, 3, and 12 months after surgery. The patients were examined for complications, including severe hemorrhage, secondary surgery, dyspnea, and recurrence. In addition, the voice and swallowing were assessed 1 and 3 months after surgery.
Results
Patient Clinical Characteristics
The 23 patients comprised 21 males and 2 females with an average age of 47.8 ± 3.2 years. Table 1 summarizes their age, sex, smoking status, BMI, and clinical diagnosis. The ALHs were diagnosed clinically as six cavernous hemangiomas and 17 capillary fibroangiomas. The 6 patients with cavernous hemangiomas were diagnosed based on the medical history and laryngoscopic and radiographic findings, while the 17 patients with capillary fibroangiomas were diagnosed based on the laryngoscopic examination and postoperative pathology (Figure 2). Preoperative CT found no bone destruction in any patient. The ALH was confirmed via suspension microlaryngoscopic examination under general anesthesia. There were no histopathological specimens, since the hemangiomas were ablated completely.
Clinical Characteristics and Surgical Management.
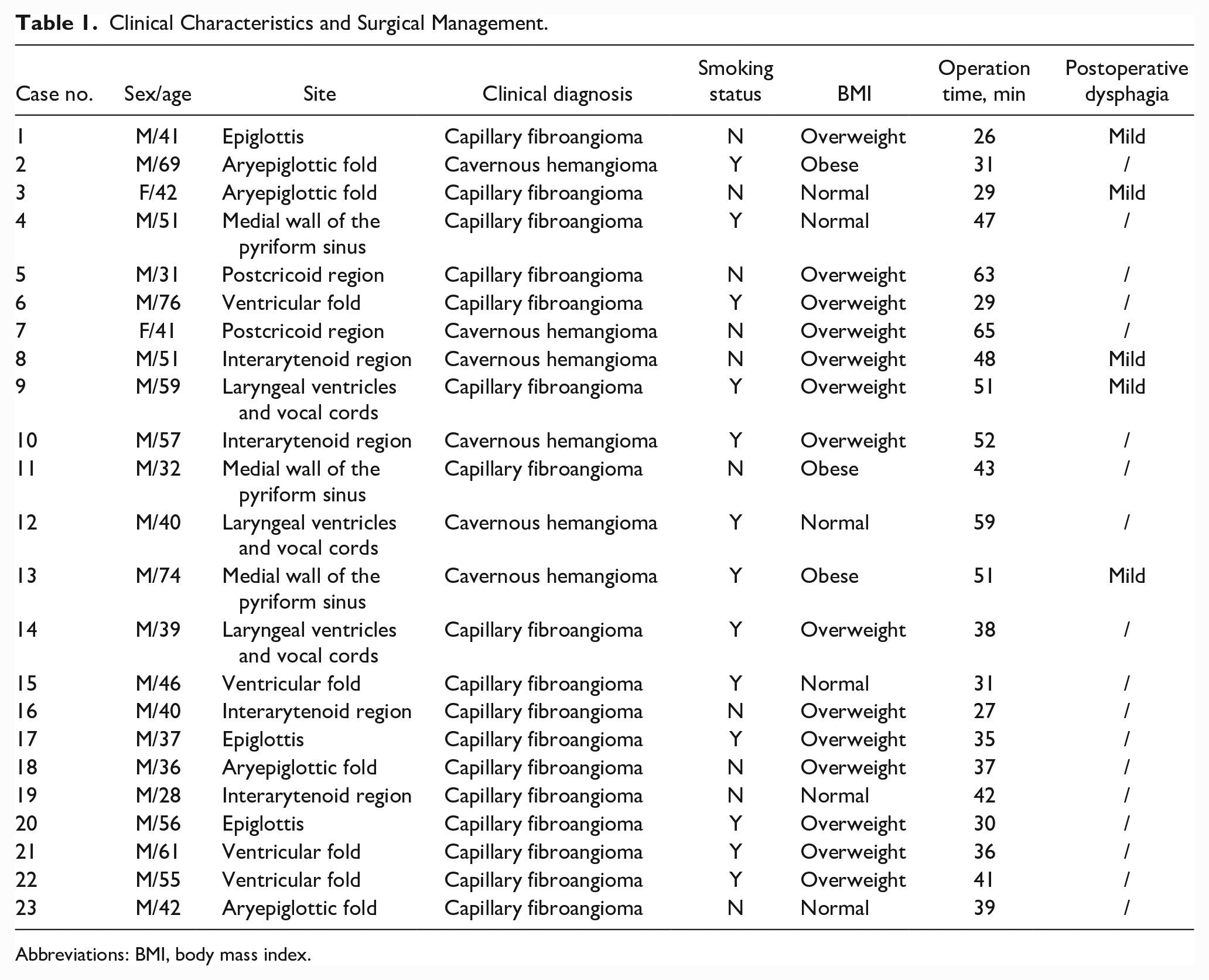
Abbreviations: BMI, body mass index.
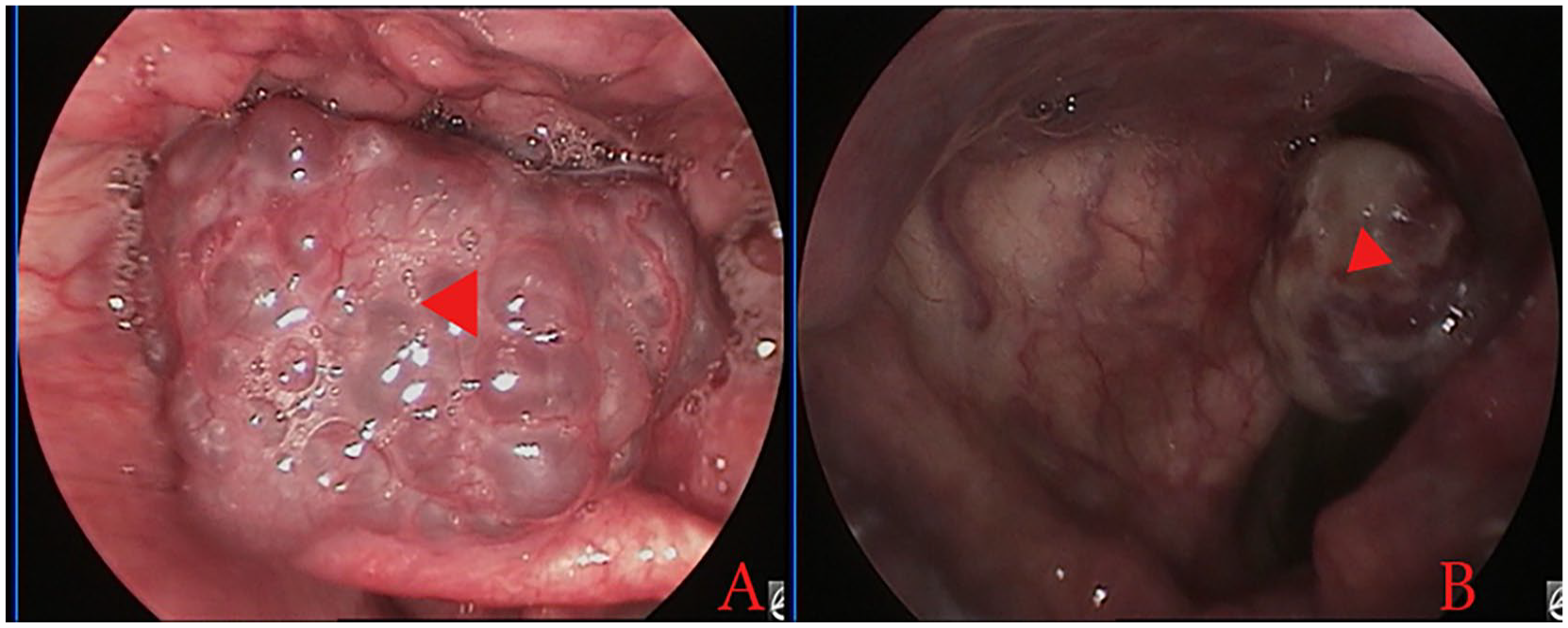
Cavernous hemangioma (A) and capillary fibroangioma (B). The red triangle indicates the tumor.
Major symptoms included foreign-body sensation in the larynx in 14 patients (tumor incidentally discovered on routine laryngoscopic examination, thereby inducing anxiety to seek surgical treatment), hemoptysis or bloody sputum in 7 patients, hoarseness in 3 patients, dysphagia in 2 patients, and difficulty breathing in 1 patient. The sites of the hemangiomas included the aryepiglottic fold, ventricular fold, and interarytenoid region (all n = 4), the medial wall, pyriform sinus, epiglottis, and laryngeal ventricles and vocal cords (all n = 3), and the postcricoid region (n = 2). Intraoperative exposure of the lesion was complete in all patients. The mean operating time was 41.3 ± 9.4 minutes.
Overall Outcome
All 23 laryngopharyngeal hemangiomas were subject to ablation, with a mean blood loss of 3.17 ± 1.12 mL. All 23 cases achieved success after one operation, with no postoperative severe bleeding, dyspnea, dysphagia, dysphonia, or other complications. Six cases had minimal bleeding that did not require surgical intervention. None required postoperative tracheotomy. The patients were followed for 1 year without recurrence.
Before the surgical intervention, 2 (8.7%) of the 23 patients had mild (n = 1) or moderate (n = 1) dysphagia. Postoperative dysphagia was assessed at 1 and 3 months. At 1 month postoperatively, 5 (21.7%) of the 23 patients had mild dysphagia, including 3 new cases. However, at 3 months postoperatively, no patients had any dysphagia.
The mean VHI was 11.2 ± 3.7 preoperatively, 7.1 ± 2.8 at 1 month postoperatively, and 4.8 ± 3.1 at 3 months postoperatively; the preoperative VHI differed significantly from the VHI at 1 and 3 months, postoperatively. In comparison, the mean MPT was 10.8 ± 3.7 seconds preoperatively, 12.6 ± 1.8 seconds at 1 month postoperatively, and 14.1 ± 3.9 seconds at 3 months postoperatively. The preoperative and postoperative mean MPT differed significantly only at 3 months postoperatively.
Preoperative stroboscopic video recording showed that the mucosal wave was decreased in 2 (8.7%) patients and normal in 21 (91.3%) patients. Glottal closure was incomplete in 1 (4.3%) patient and complete in 22 (95.7%) patients. However, at 3 months postoperatively, mucosal wave resolution was normal and glottal closure was complete in all patients. Figure 3 shows the postoperative laryngoscopic images at different follow-up times in a 76-year-old man with capillary fibroangioma.
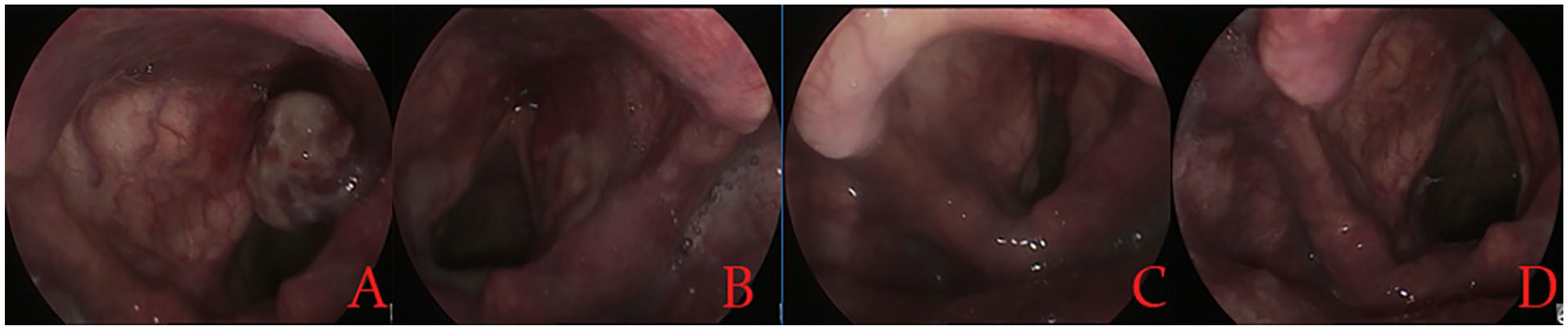
Preoperative (A) and at 2 weeks (B), 4 weeks (C), and 12 months (D) postoperatively.
Discussion
Currently, there is no consensus regarding the treatment of ALH due to its rarity. The treatment of laryngeal hemangioma is challenging because of the complexity of the anatomic structures involved and the potential for severe postoperative complications, including life-threatening hemorrhage and airway obstruction.2-6 The aim of treatment is not only complete resection, but also the preservation of anatomic structures and function.11,18 Therefore, some clinicians believe that it is necessary to intervene early when the lesions are circumscribed to the most superficial submucosa to perform radical excision without injuring the organs of phonation. 19 Sclerotherapy has long been an alternative to surgical resection to treat vascular anomalies. Although it obtains good results, it requires repeated injections and a long treatment cycle for ALH.6,7 Moreover, some sclerosing agents are associated with fibrosis and may be unsuitable for application to the adjacent true vocal fold, ventricle, and interarytenoid area. 6
Laser surgery is effective and less invasive than surgical removal. Lesion excision with a CO2 laser is generally accepted.8-11 Lucioni et al reported no recurrences in 5 of 6 adult supraglottic hemangiomas treated with CO2 laser surgery. 9 Alshaya et al excised an ALH with a CO2 laser and there was no recurrence at 2 years. 10 Shim and Kim reported no recurrence during 18 months of follow-up in a patient treated using a CO2 laser combined with KTP laser photocoagulation. 11 Mesolella et al performed microlaryngeal surgery with a CO2 laser in 2 patients with no recurrence during a 3-year follow-up. 19 Lomeo et al reported 4 cases involving only the glottic area and there were no negative consequences of CO2 laser treatment. 20 Comparing the curative effect of chemotherapy, KTP lasers, and CO2 lasers combined with chemotherapy in the treatment of ALH, Wu et al found no significant difference in efficacy among groups. 3
Although laser therapy has many advantages, it is expensive and only some patients can afford it. The spectrum of a laser beam is usually 577 to 585 nm, and the laser energy concentrates on tissues ranging from 0.75 to 1.2 mm deep, which limits the depth of penetration and the effectiveness on deeper hemangioma tissue. 13 Most scholars suggest that laser therapy is suitable for limited or pedunculated supraglottic superficial hemangiomas, but not advisable for extended lesions or hemangiomas with large vessels.4,9 In addition, the use of a CO2 laser increases the risk of damage to the adjacent mucosa and of developing subglottic stenosis postoperatively. 4
In recent years, LPRF coblation has been used increasingly to treat granulomas, 21 laryngeal carcinomas, 22 and oropharyngeal tumors. 14 A recent article reported the use of LPRF in the treatment of tongue hemangiomas. 23 Here, we used LPRF coblation to remove ALH in 23 patients, with a mean blood loss of 3.17 ± 1.12 mL. All cases achieved success after a single operation without recurrence during the 1-year follow-up. Compared to lasers and electrocoagulation, plasma radiofrequency works at relatively low temperatures and causes less damage to surrounding tissues; furthermore, there are fewer postoperative scars. 14 Several studies have reported the use of LPRF coblation in the treatment of hereditary hemorrhagic telangiectasia and nasopharyngeal fibroangiomas and achieved good results, with the advantages of less trauma, less bleeding, and quick recovery.24,25 In addition, LPRF coblation simultaneously performs resection, hemostasis, irrigation, and suction, which ensures a bloodless operative field and avoids the use of different instruments, thereby shortening the operation. The plasma knife of LPRF coblation can bend over a larger range, enabling adjustment of the angle during the operation with unfavorable laryngoscopic exposure.
In our patients, the plasma wand was placed close to the tumor body for gradual coagulation from the edges around the tumor toward the center; during the coagulation process, the tumor became pale and shrank gradually, blocking its blood supply and reducing its size. Then, the tumor was resected using the ablation function of the plasma wand. We believe that benefits of LPRF coblation include radical excision of the pathology with reduced trauma, better hemostatic control, excellent postoperative functionality, short hospitalization, and low financial costs.
This study assessed the voice and dysphagia preoperatively and postoperatively. At 1 month postoperatively, 21.7% of the patients had mild dysphagia, including 3 new cases. However, no patient had any level of dysphagia at 3 months postoperatively. This may be because the mucosa in the ablation zone did not achieve complete epithelization at 1 month, resulting in odynophagia, while complete epithelization was obtained at 3 months. In addition, the subjective and objective voice assessments showed better voice recovery at 3 months postoperatively. No LPRF coblation-related complications or recurrence were encountered.
The limitations of this study were the small sample size, lack of a control population, and short follow-up.
Conclusion
LPRF coblation is an effective minimally invasive method for treating ALHs with better voice and swallowing recovery. Edge coagulation before ablation resection may reduce the intraoperative bleeding.
Footnotes
Author’s Contributions
Availability of Data and Material
All data generated or analyzed during this study are included in the published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Technology Agency of Yiwu, China (Grant #2021-3-55). The funding supported the study design; collection, analysis, and interpretation of the data; and writing of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
