Abstract
Introduction
Reconstruction of soft tissue defects following oncologic resection in patients with head and neck cancer requires a wide armamentarium of donor tissue options depending on the size and location of the defect as well as vessel access and various patient comorbidities. Despite the successful development of microvascular free tissue transfer, pedicled axial flaps remain relevant. They are the reconstructive option of choice for many smaller defects and for cases in which free tissue transfer has either failed or is not a suitable option. Flap failure for both microvascular free flaps and axial flaps is a rare but major complication that threatens the integrity of the reconstruction and can cause significant morbidity and rarely even mortality. Failure of axial flaps is usually secondary to insufficient perfusion of the rotated tissue, leading to distal necrosis. This is the result of either poor flap design, vascular pedicle compromise, or poor wound healing. 1,2 Free tissue transfers most commonly fail as a result of venous congestion, and less commonly, arterial compromise. 3 -6
Regardless of the type of flap performed, angiogenesis and neovascularization of the transferred tissue are fundamental for flap success. The exact mechanism by which the flap relies on the underlying wound bed versus the adjacent tissue remains unclear. The relative contribution of the pedicle versus angiogenesis from the underlying wound bed and adjacent skin in the distal portion of an axial flap is an important concept to understand for flap design purposes. This is especially true when the distal portion of a pedicled flap crosses into an adjacent angiosome, where the blood supply is random rather than relying on the pedicle. It is therefore important to have a reliable animal model for studies on axial flap survival.
The mouse dorsum has 4 angiosomes supplied by bilateral lateral thoracic arteries (LTAs) and bilateral deep circumflex iliac arteries. There can be a minor contribution to each angiosome from the intercostal perforators. 7 The LTA-based flap can be used to study distal axial flap necrosis by designing its length to cross into another angiosome, resulting in inadequate vascularization of the distal portion of the flap. The majority of studies using the LTA axial flap have placed a silicone barrier over the muscle bed after the flap has been raised to prevent early angiogenesis or growth factor signaling from the underlying wound bed. 8 -10 Despite the frequent use of a silicone barrier, it is unknown what effect this barrier has on the percentage and rate of flap necrosis or how dependent the LTA axial flap is on direct contact with the underlying wound bed early after surgery.
There are no comparisons of viability between flaps that utilize or do not utilize a silicone barrier for wound bed isolation in a mouse model of an axial LTA flap. We believe that understanding the dependence of an axial flap on the recipient wound bed will allow for appropriate flap design to answer specific questions about ischemia–reperfusion injury and flap necrosis. Our goal is to use the LTA axial flap to understand the dependence of long axial flaps on wound bed vasculature by investigating the effect of a silicone barrier under a variety of conditions.
Materials and Methods
Animal Models
All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Alabama at Birmingham. SKH-1 nude, immunocompetent mice (Charles River Laboratories, Hartford, CT, USA), aged 4 to 6 weeks, were used for this study. Mice were housed in a pathogen-free environment, allowed free access to food and water, and were kept on a standard cycle of 12 hours of light and 12 hours of darkness. Mice were divided into 5 groups: no silicone (NS) (n = 7), silicone in the proximal 50% of the wound bed [proximal silicone (PS)] (n = 8), silicone in the distal 50% of the wound bed [distal silicone (DS)] (n = 5), silicone over the full length of the wound bed with pedicle preservation [full silicone, pedicle preserved (FSp)] (n = 5), and silicone over the full length of the wound bed with pedicle sacrifice [full silicone, pedicle sacrificed (FSs)] (n = 5). Mice were excluded if they demonstrated auto-cannibalism (n = 7), which resulted in uneven group numbers.
Flap Harvest
Mice were anesthetized with isoflurane (VetOne®, Boise, Idaho; Distributed by MWI Animal Health® Boise, Idaho), and the dorsum was disinfected with 70% isopropyl alcohol wipes. Mice were positioned on a sterile operating board under a direct heating lamp to ensure body temperature was maintained throughout surgery. A marking pen was used to draw the intended 1 cm × 4 cm axial flap overlying the right side of the dorsum based on the LTA. A length of 4 cm was chosen to ensure the distal end of the flap crossed into the second angiosome supplied by the deep circumflex iliac artery. A width of 1 cm was chosen to surgically divide contributions from the intercostal perforators. These flap dimensions were based on a prior study demonstrating that a LTA flap with dimensions of 1.5 cm × 3.5 cm avoids distal ischemic necrosis. 11 Boundaries of the flap were the midline of the dorsum (medial), right axillary line (lateral), infrascapular line (superior), and the iliac crest (inferior) (Figure 1A). Using sterile technique, flaps were raised in a caudal to cranial direction and consisted of skin, subcutaneous tissue, and a thin layer of panniculus carnosus muscle. Care was taken to preserve the LTA as it entered the skin flap from a superolateral direction (Figure 1B). For the FSs group, the pedicle was located on the undersurface of the flap and was deliberately cut with a knife. Hemostasis was achieved with direct pressure. After the flap was elevated, it was sutured in its original position for the NS group. The flap was sutured with a simple interrupted 5-0 polypropylene suture (©Ethicon US, LLC. Raritan, NJ, USA) for all experimental groups (Figure 1C). For the FSp, FSs, PS, and DS groups, a piece of medical grade silicone sheeting (dimensions 1 × 2-4 cm, thickness 0.5 mm) (Invotec International, Inc.®; Jacksonville, FL, USA) was secured to the underlying wound bed with simple interrupted 5-0 polypropylene suture (Figure 1D-F). After the procedure, mice were given a subcutaneous injection of carprofen/buprenophrine (manufactured by Vericore Limited Dundee, UK and distributed by Pfizer Animal Health, Pfizer, New York, NY, USA) for post-procedural pain control. The duration of each procedure was approximately 45 minutes. Post-procedure, flaps were monitored daily by visual inspection.
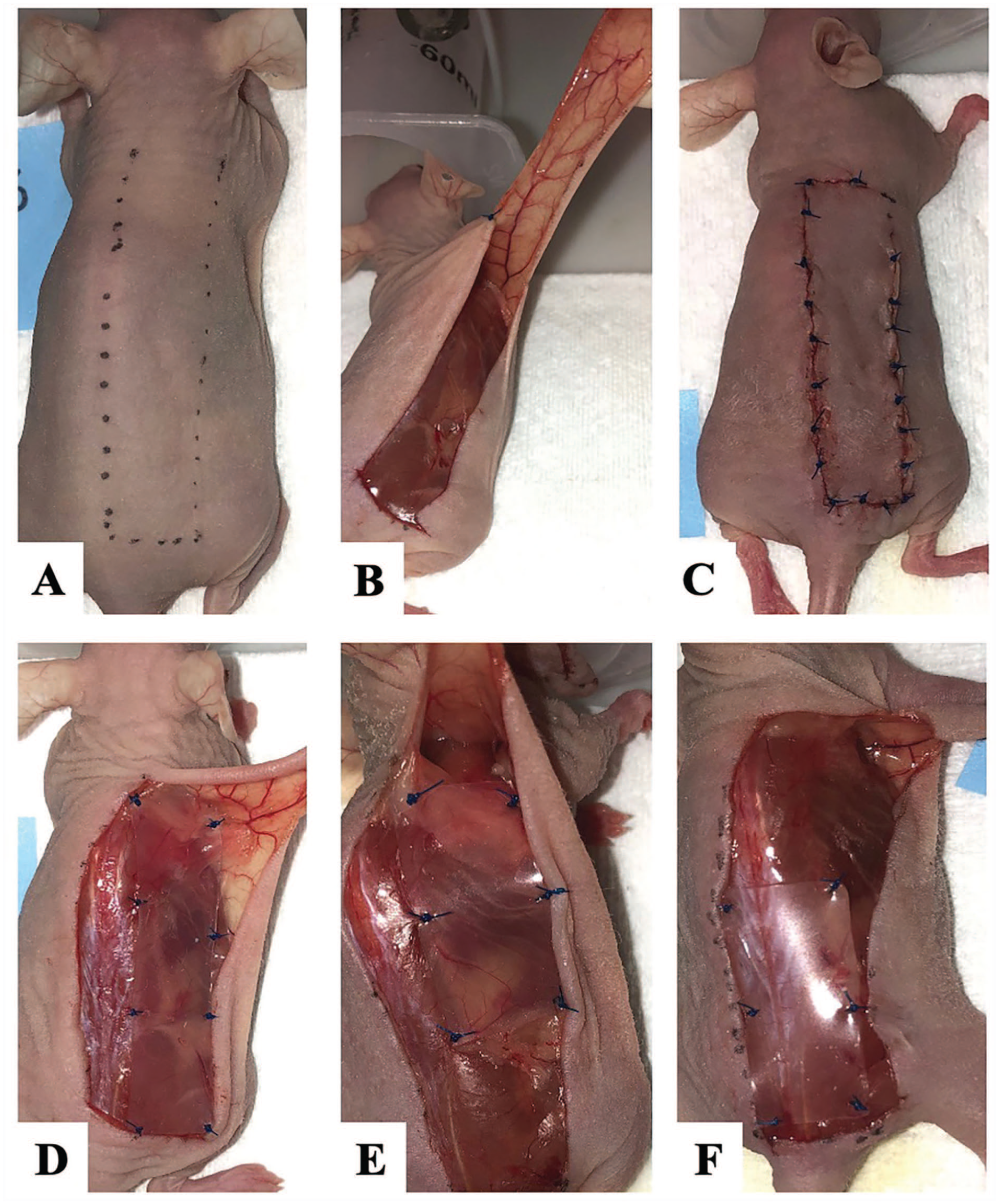
Lateral thoracic artery (LTA) axial flap procedure.
Analysis of Flap Necrosis
High-resolution photographs were taken of each mouse flap daily from postoperative day (POD) 0 (immediately after surgery) through POD 6 for documentation of flap necrosis. Photographs were taken each day using an iPhone 8s at a distance of 20 cm from the mouse. The end point was defined as POD 6 or earlier for mice flagged by the UAB Animal Resources Program veterinary support staff in accordance with IUCUC’s humane treatment of research animals. Photos were uniformly cropped to highlight the flap on the mouse dorsum. A novel, objective imaging analysis protocol was utilized for flap viability assessment using ImageJ©, an opensource Java image processing program from the National Institute of Health. 12 Images were converted to a gray scale type, and 2 reconstructive surgeons (JF, ER) independently selected a density threshold for the image library to capture the baseline level of intensity change indicating vascular compromise. A uniform rectangular selection frame was created to capture each flap in its entirety (Figure 2). A profile plot was then created based on these frames to represent the relative intensity of the frame content. Each intensity peak was analyzed using the automated software to calculate relative area of necrosis, and results were converted to a percentage of the control timepoint without necrosis (POD 0) for each experimental condition. Venous congestion was assessed by a reconstructive surgeon (CT) blinded to type of flap to assess a more qualitative aspect of flap necrosis.
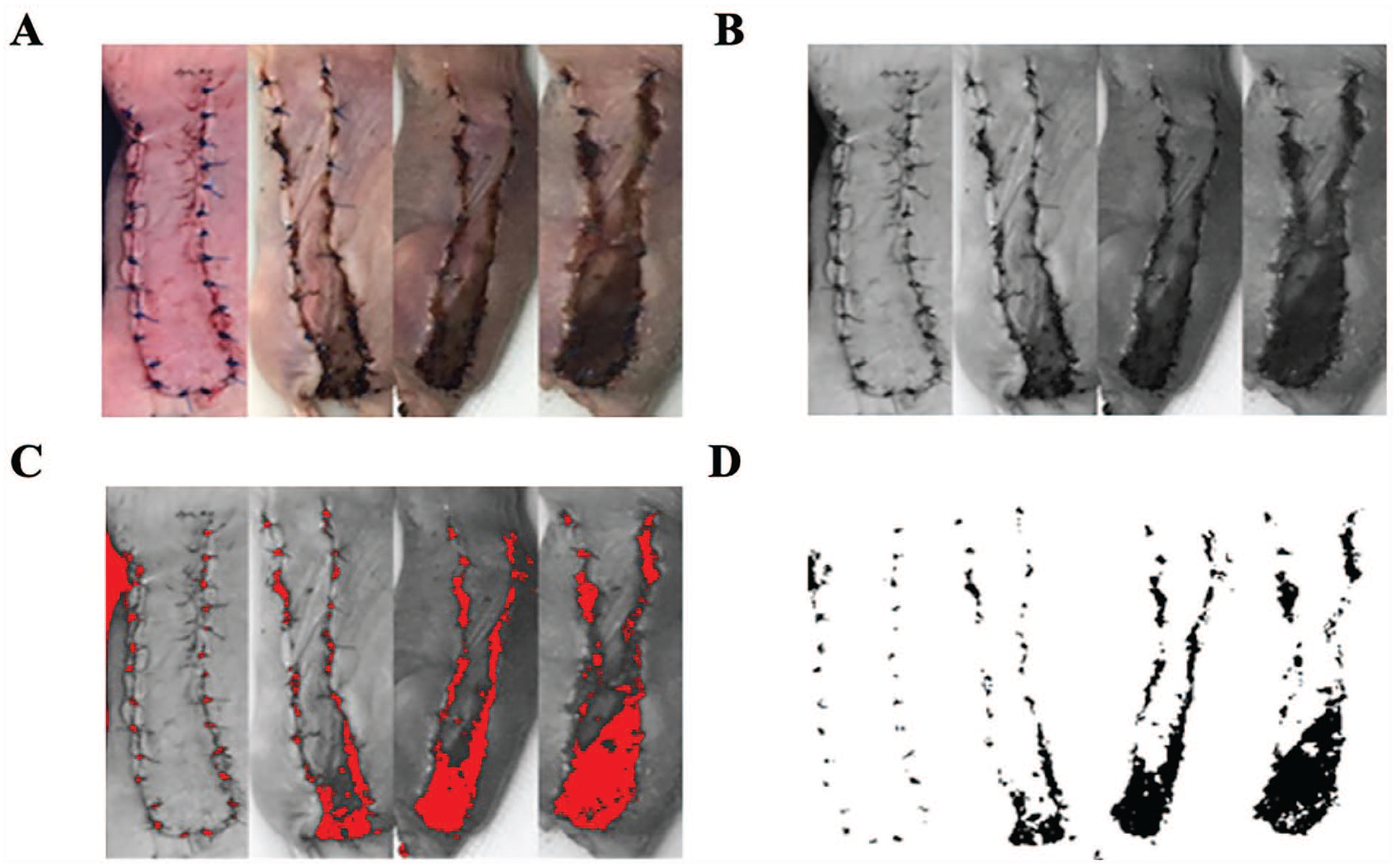
Assessment of flap viability.
Statistical Analysis
Data were analyzed using GraphPad Prism (version 8.0; GraphPad Software, Inc., La Jolla, CA, USA). ANOVA was used to compare the mean percent flap viability of each group, followed by a Dunnett’s multiple comparisons test to compare differences in the mean area of tissue necrosis of each group to the control group. The NS group was used as the control to which the remaining groups were compared. Statistical significance was defined as P < .05.
Results
Average percent flap viability did not significantly differ (P > .05) among the groups with an intact vascular pedicle (NS, FSp, PS, DS), regardless of the presence or position of a silicone barrier. As shown in Figure 3, mean percent flap viability was 97.87%, 98.02%, 95.80%, 94.89%, and 83.66% for the NS, FSp, PS, DS, and FSs groups, respectively. The mean difference in percent necrotic area (with 95% confidence intervals) relative to NS group was −0.15% (−15.09% to 14.09%) for the FSp group, 2.07% (−5.26% to 9.39%) for the PS group, 2.98% (−10.98% to 16.94%) for the DS group, and 14.21% (0.48% to 27.94%) for the FSs group. The only group to demonstrate a significant difference in flap viability compared to the NS group was the FSs group (P = 0.045), in which the pedicle was deliberately sacrificed (Figure 3).
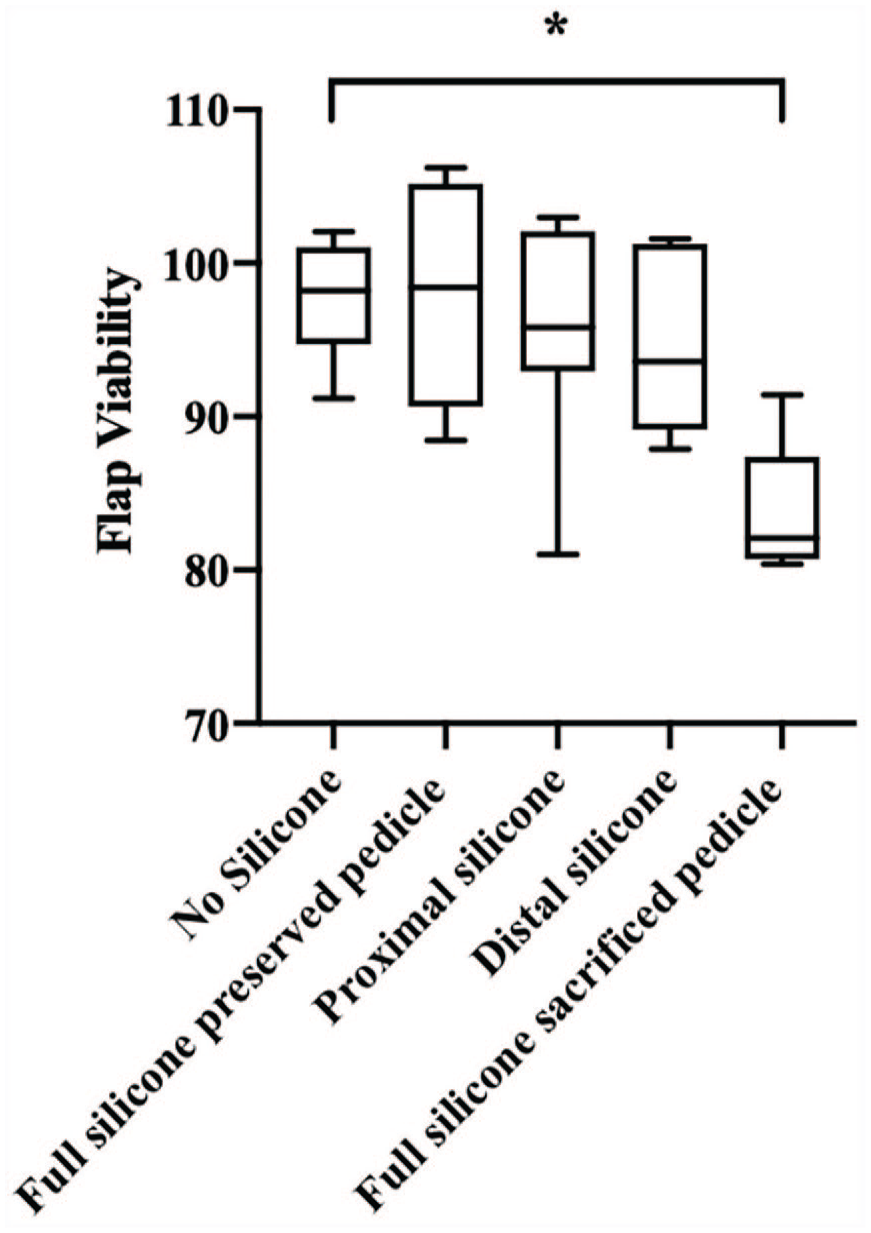
Average percent of flap viability.
All groups had flaps demonstrating some degree of flap necrosis by POD 1. The FSs group was the only group in which all flaps showed obvious necrosis by POD 1. Venous congestion was observed in 11 (44%) flaps among different groups with an intact pedicle including NS (n = 2, 18.2%), DS (n = 4, 36.4%), PS (n = 3, 27.3%), and FSs (n = 2, 18.2%). There is no significant difference (P > .05) in the percentage of flaps with venous congestion between groups. All flaps with venous congestion went on to develop necrosis. None of the FSp flaps demonstrated venous congestion (Figure 4).
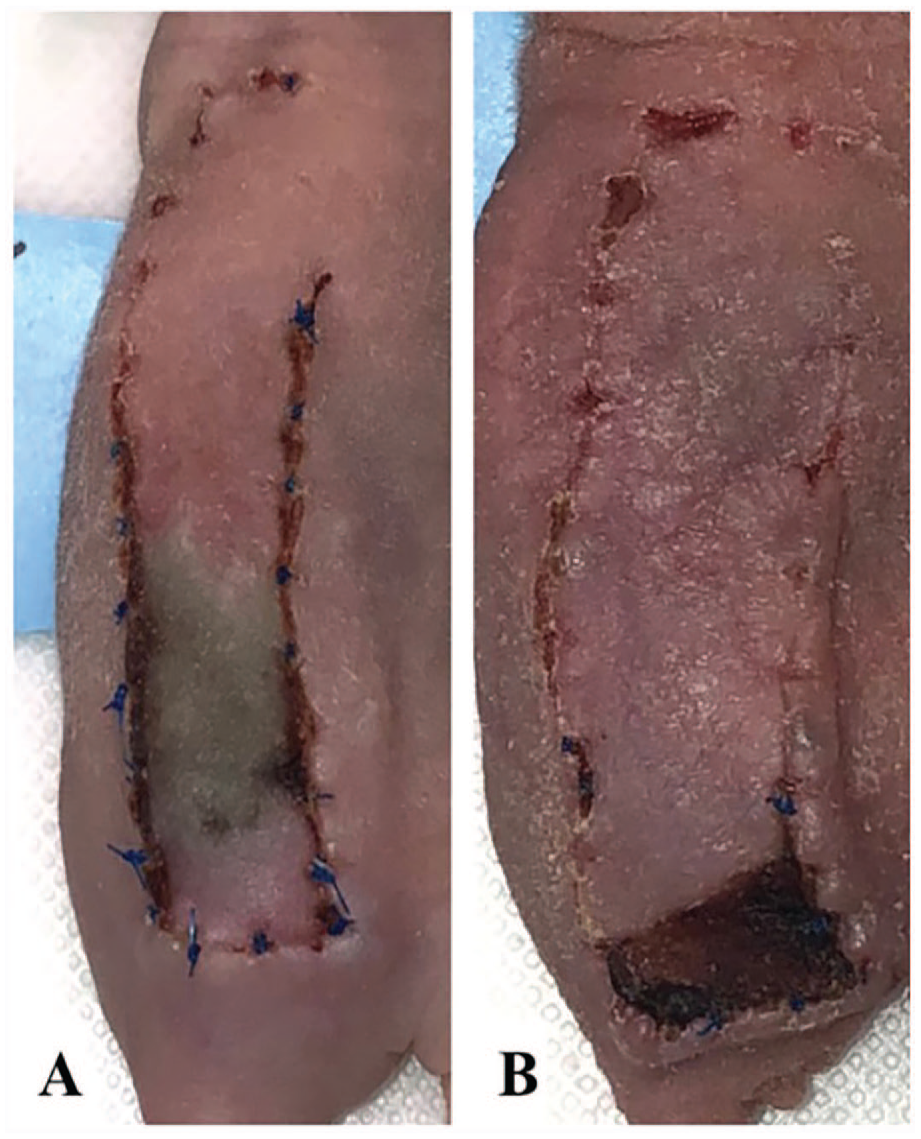
Venous congestions versus necrosis.
Discussion
Here we demonstrate that an axial pattern flap designed to extend beyond the pedicle’s angiosome is not dependent on the wound bed vasculature. The distal aspect of the axial flaps developed necrosis as anticipated based on the design of the flap but isolating the flap from the wound bed with a silicone barrier did not increase the severity of flap necrosis compared to the no silicone control group.
Kaufman et al was the first to report the effects of wound bed isolation on the survival of the distal portion of an axial flap in rats. 13 A polyethylene film was placed proximal, distal, or full length between the flap and the suprafascial areolar layer of the rat dorsum to block angiogenesis from the underlying wound bed to the flap. In their study, rats with a barrier across the distal 55% or the entire length of the flap demonstrated significantly greater necrosis when compared to controls, but not with a barrier in the proximal 45% of the flap. They conclude that contact between the underlying wound bed and the distal aspect of the flap is an absolute necessity to ensure flap survival, as the distal flap portion acts like a skin graft with improved survival when contact was reinforced with a pressure dressing. Our findings contradict those of Kaufman et al in that we did not find any impact on flap necrosis with placement of a silicone barrier along any part of the flap length. This could be due to the difference in angiosomes supplying the rat versus mouse dorsum or the flap design.
Alternative studies in rats and mice have used various materials to create a physical barrier to assess random and axial flap survival in the absence of wound bed angiogenic contributions. 14 -19 All of these studies placed a barrier along the entire length of the flap, and conclusions are mixed as to whether distal flap survival is affected by isolation from the underlying wound bed. For example, Jones et al found that wound bed isolation using a silicone barrier had a negative impact on survival of an axial pattern flap and significantly increased the amount of necrosis in the distal aspect of the flap in rats. 14 Comparable findings in mice were reported by McGuire and Howdieshell. 20 Similar to Kaufman et al, the authors concluded that the barrier prevented processes critical to skin graft survival such as imbibition and angiogenesis. In contrast, the study by Sönmez et al. is the only prior study to describe the LTA axial flap (1.5 cm × 2.5 cm) without use of a silicone barrier, and they found an average percentage distal necrosis of 28.62%. 21 There is significant variability in reported rates of LTA axial flap necrosis in mice ranging from 9.7% to 83% with flap dimensions ranging from 1-1.5 cm × 2.5-4.5 cm. 11,8,9 The reported variability in percent flap necrosis could be due to differences in surgeon technique and variability in flap design with differing flap ratios. The role of the wound bed vasculature in preventing flap necrosis was unclear in the absence of a direct comparison between the presence and absence of a silicone barrier.
It is also important to understand how skin grafts differ from pedicled flaps in their dependence on underlying wound bed vasculature. It is understood that skin grafts survive by imbibition of plasma and ingrowth of new vessels from a muscular bed with an abundant blood supply. 14,22,23 Studies on skin graft healing in murine models have demonstrated signs of angiogenesis from the underlying wound bed at 48 hours after grafting. 24,25 Our goal with the current study was to delineate the role of pedicle contributions from those of the wound bed in the most distal aspect of the flap where the blood supply is random.
Based on these prior studies, we designed a 1 cm × 4 cm flap to ensure the flap crossed into an adjacent angiosome and to achieve some distal necrosis in all groups. Our hypothesis was that the addition of silicone would cause greater distal flap necrosis by preventing inosculation and angiogenesis from the wound bed, and the NS would have the least amount of flap necrosis with no inhibition of angiogenesis. If distal axial flap survival is dependent on angiogenesis from the underlying wound bed, as seen in skin graft survival, our results would have reflected that relationship with increased distal necrosis in any flap with a silicone barrier blocking that process (FSp, DS, and PS) relative to the NS group. However, we found no significant difference in the amount of macroscopic tissue necrosis between the NS, DS, PS, and FSp groups. Our findings demonstrate that the silicone barrier does not increase flap necrosis at the clinical level within the first week of surgery, suggesting that angiogenesis from the wound bed early after surgery is not essential for distal flap survival in an axial model. These findings also argue that the distal portion of an axial flap acts differently than an isolated skin graft, given how contact with the underlying wound bed did not change the amount of necrosis seen. The FSs group, in which the vascular pedicle was deliberately sacrificed, was the only condition in which flap viability was significantly altered. These flaps showed dramatic necrosis early in the postoperative period (POD 1). It is well known that an intact vascular pedicle is critical for survival of an axial flap. These findings suggest that even when the distal portion of an axial flap crosses into an adjacent angiosome, an intact vascular pedicle is the most crucial factor leading to flap survival.
Sönmez et al. analyzed angiogenesis using in vivo bioluminescence imaging to detect Vascular Endothelial Growth Factor Receptor 2 (VEGFR2) activity in murine LTA axial flaps. They demonstrated increased activity of VEGFR2 along the periphery of the flaps not from the underlying wound bed, indicating that the key angiogenesis process is from the periphery. 21 This could explain why placement of a silicone barrier over the wound bed would not affect necrosis patterns. Interestingly, in the Kaufman et al’s article, placement of a barrier between the flap and wound bed plus between the flap and adjacent skin edges had a higher rate of flap necrosis compared to a distal barrier (54.36% ± 2.21% vs 38.47% ± 2.10%, respectively). 13 Though the silicone blocks inosculation and potential angiogenesis from deeper tissues, peripheral cutaneous angiogenesis appears to be crucial in axial flap survival.
There are important limitations to acknowledge in our study. First, our total number of mice for each group was relatively small and not evenly distributed, primarily due to the exclusion of mice that exhibited auto-cannibalism. This is a known risk of performing surgical procedures on a murine model, and precautions were taken to avoid auto-cannibalism, including minimizing the number of mice in a given housing unit and monitoring the flaps for irritated or dehisced areas that could be repaired. A limitation with the flap viability assessment was the way in which the ImageJ© software detected the increased density of the flap sutures on POD 0. This is reflected in the flaps that had viability percentages greater than 100 after POD 0, since the suture color diminished and subsequently dropped below the density threshold. Although cropping the image to exclude suture material was possible, this would have variably diminished the flap area analyzed. Given that the suture material was visible in all murine models, full flap area inclusion ensured consistency across comparative analysis between conditions. Finally, another limitation is the lack of histopathological or immune biochemical studies to quantify necrosis. However, our intention with this study was to rate necrosis based on clinical findings.
Conclusion
In this study, we demonstrate that placement of a silicone barrier does not affect the amount or rate of necrosis seen in the LTA axial flap in a murine model. We show that even when the distal flap crosses into an adjacent angiosome, the main contribution to the survival of the flap is an intact vascular pedicle and does not behave as an isolated skin graft that solely relies on the underlying wound bed. This furthers our understanding of this particular model and advances our ability to create a flap design to answer future hypotheses about ischemia–reperfusion injury, angiogenesis, and neovascularization. We hope that a better understanding of the relationship between the distal aspect of an axial flap and the underlying wound bed in a mouse model will provide translational information for work in human flap design.
Footnotes
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH NCI R R01CA240589, NIH NCI 1R37CA245157-01A1, American Cancer Society RSG-18-006-01-CCE, and the UAB Comprehensive Cancer Center’s Preclinical Imaging Shared Facility P30CA013148.
Ethical Standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guides on the care and use of laboratory animals (University of Alabama at Birmingham IACUC).
