Abstract
Introduction
Intralabyrinthine schwannoma (ILS) is a benign tumor that occurs in the schwannoma cells of the vestibular nerve or cochlear nerve branches in the labyrinth. ILS is a special type of vestibular schwannoma (VS) [also known as acoustic neuroma (AN)] with a low morbidity rate. 1 Its clinicopathological features and treatment strategies are different from those of general VS. 2,3
ILS can manifest as hearing loss, tinnitus, ear tightness, vertigo, and dizziness and its clinical symptoms are usually related to the location and range of extension of the lesion. Diagnosis of ILS is challenging due to the lack of specific clinical symptoms, signs, and audiological and vestibular function test results, and there is no diagnostic standard at present. In addition, standard computed tomography (CT) and skull magnetic resonance imaging (MRI) have limited utility for identification and diagnosis of ILS, and they can easily miss the lesion thus delaying diagnosis. Thin-layer high-resolution MRI has high sensitivity and specificity for the diagnosis of ILS. It can be used to determine the precise location, scope, size, and anatomical characteristics of the tumor, and can be used for follow-up observation. 4,5
ILS is divided into a number of subtypes according to the location and range of extension of the lesion. 6 VS mostly originates from the vestibular nerve, and the ratio between VS in the vestibular nerve and cochlear nerve is about 3:1. 7 Intracochlear schwannomas (ICS) is the most common subtype of ILS, 8,9 accounting for more than 50%, 6 while intravestibular schwannomas (IVS) is a relatively rare subtype, accounting for only 19% to 31% of cases. 3,6 ILS extending outside the cochlea and vestibule accounts for 31% of cases, and the rate of lesions extending into the internal auditory canal (IAC) is about 15%. 9 The cochlear base turn is often involved in ICS (in 44%-75% of cases). 6,10 The posterior semicircular duct is rarely involved, which suggests that ILS does not typically originate from the inferior vestibular nerve. 3
The treatment of ILS is selected according to the tumor location, size, and growth, and according to the patient’s symptoms, hearing, age, and willingness. 2 Wait-and-scan follow-up observation and surgical excision are the 2 most commonly used treatment methods for ILS. 6,11 In addition, radiotherapy 12 and chemical labyrinthinectomy 13 among other treatment methods, may be selected in some cases. Surgical approaches to VS include microsurgical approaches, exclusive endoscopic approaches, 14 -18 and combined approach, 19,20 while in ILS, TTEA was first reported to be applied in patients with class D hearing and no tumor extension to the CPA in 2018. 21
The surgical approach was selected according to the tumor’s location, size, and extension (translabyrinthine approach, enlarged transcanal labyrinthine approach, transcapsular approach, etc.). 22 Surgeons usually attempt to use the minimally invasive method of otoendoscopy to remove the AN. In addition to traditional microsurgery, endoscopic surgery has also been used in the treatment of ILS (Figure 1). ILS can be removed by endoscopic surgery via the promontory, through the external auditory canal. 21,23 Recently, cochlear implantation after ILS resection has become another novel choice of surgery in several studies, 24 -29 although there is still insufficient evidence due to the small number of cases. Cochlear implantation is mainly used for advanced bilateral sensorineural hearing loss, and has been applied to patients with single-sided deafness recently. Some researchers suggested that residual or recurrent tumor may interfere with the normal use of cochlear implants and requires MRI follow-up, 27 however, more data are required to prove this opinion. In our study, cochlear implantation was not considered in the treatment process because all patients included had profound hearing loss in the affected side while the contralateral ear owns normal hearing.
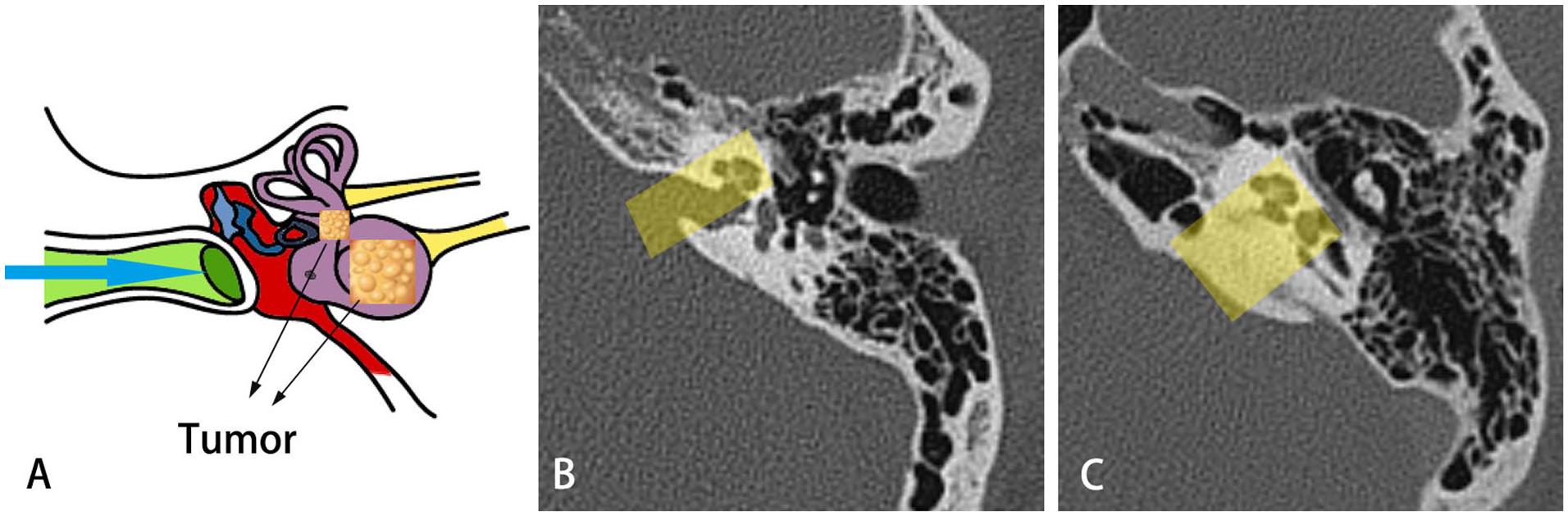
Schematic diagram of the surgery for intralabyrinthine schwannomas. (A) Blue arrow indicates the endoscopic approach to the ear. Tumors are marked by black arrows. (B) A computed tomography scan in coronal view showing the working area (yellow). (C) The introduction of the underwater bone grinding system allows for the expansion of the working area of endoscopic inner ear surgery.
In this article, we introduce our experience with fully endoscope resection of ILS using the transcanal transpromontorial endoscopic approach (TTEA), which removes the tumor through the external auditory canal and reaches the lesion through the promontorial region. 23 We focus on the clinical presentations, surgical aspects, and outcomes.
Material and Method
Three ILS patients (1 man and 2 women, aged 60-62 years), without tumor extension into the IAC and who underwent surgery via the TTEA at the Otolaryngology—Head and Neck Surgery Department of the EENT Hospital of Fudan University in 2020, were included in this study. The inclusion criteria for this study were as follows: ILS with profound hearing loss (PTA > 80 dB, according to the classification of hearing loss by WHO in 2021) or further decrease in residual hearing; rapidly growing tumor or invasion into the IAC, cerebellopontine angle (CPA), or middle ear; refractory vertigo or tinnitus. 10,30 All patients suffered from intractable vertigo (for 4 months to more than 20 years) with profound sensorineural hearing loss (for between 10 and 27 years) (Table 1).
Demographic data and baseline parameters before therapy or when first presenting at our institution.
The study protocol was approved by the Ethics Committee of the Eye, Ear, Nose, and Throat Hospital, Fudan University (approval number: ChiCTR2100042035). Written informed consents were obtained from all 3 patients involved in this study.
Hearing and Facial Function
Hearing function was evaluated preoperatively using the Hearing Classification System according to the American Academy of Otolaryngology—Head and Neck Surgery (AAO-HNS). 31 Facial function was assessed using the House–Brackmann (HB) grading system. 6
All 3 patients showed class D hearing preoperatively and had a history of tinnitus prior to treatment. None of the patients had facial palsy preoperatively.
One of the 3 patients had a history of intermittent poor balance for 20 years, which had been more severe during the past year, and 2 patients had intractable vertigo refractory to medical treatment for 4 to 6 months.
Vestibular Function Examination
All 3 patients’ cervical vestibular evoked myogenic potential (c-VEMP) showed abnormal bilateral vestibular function, and decreased function of the bilateral sacculus inferior vestibular nerve pathway. The ocular vestibular evoked myogenic potential (o-VEMP) showed a bilateral increased response threshold, and decreased function of the bilateral utricle supravestibular nerve pathway. Bithermal caloric tests were performed to evaluate the function of the semicircular canal function. Semicircular canal function was decreased on the lesion side, and vestibular function was characterized by insufficient dynamic and static balance.
Radiologic Assessment
All the patients were diagnosed and evaluated preoperatively by CT scan and gadolinum-enhanced MRI of temporal bone, to plan the surgical approach. All 3 patients underwent enhanced MRI preoperatively. The ILS showed no abnormality on T1-weighted images (T1-WI), heterogeneous high signal intensity filling defect on T2-weighted images (T2-WI), or uneven enhancement (Figure 2). One case showed an abnormal cochlear signal, which was an intracochlear schwannoma; the other 2 cases showed abnormalities in the cochlea and vestibular area, which were schwannomas in the vestibule and cochlea.
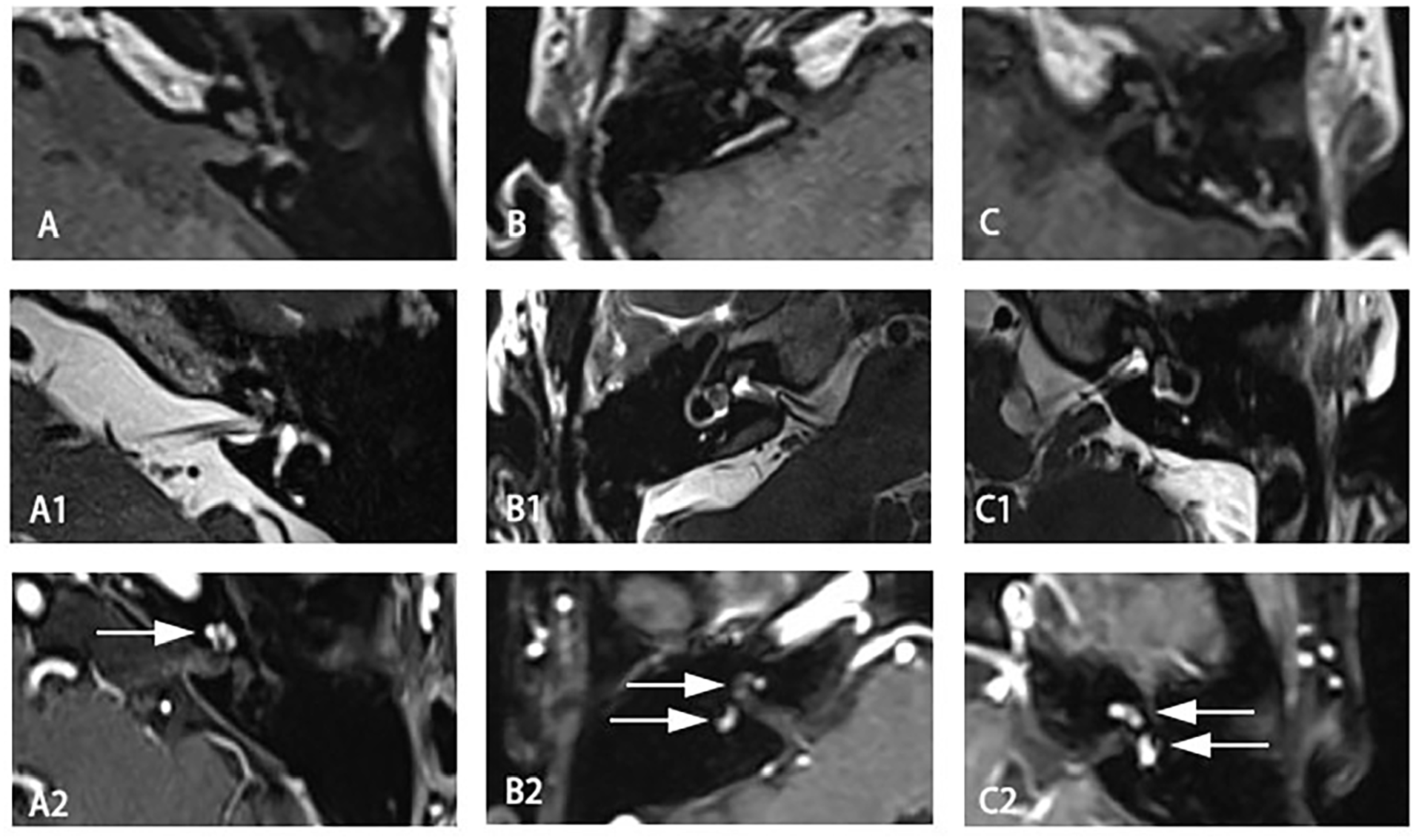
Preoperative enhanced MRI of the temporal bone. (A-C) Intralabyrinthine schwannomas showed no abnormalities on T1-WI. (A1) Tumors confined to the cochlea showed a filling defect on T2-WI. (B1, C1) Tumors in the cochlea and vestibule showed a filling defect on T2-WI. (A2) Typical contrast enhancement of the tumor in the cochlea on enhanced T1-WI (white arrows). (B2, C2) Contrast enhancement of tumors in the cochlea and vestibule on enhanced T1-WI (white arrows).
Postoperative MRI was performed on the day after surgery to check the surgical field and determine whether there was any residual tumor.
Tumor Classification
Tumors were classified according to the Kennedy’s classification. 32 The Kennedy classification was used to assess intralabyrinthine schwannoma’s extension (Table 2). In all 3 patients, the tumors were located in the cochlea and/or vestibular nerve without CPA involvement.
The modified Kennedy Classification System.

Surgical Techniques
The clinical features and tumor characteristics of the patients were evaluated. Cochlear implantation without resection of tumor was not considered since the contralateral ear of all patients preserved normal hearing. All surgical procedures were performed by senior otolaryngologists (DR and WW). Based on the extent of the tumors, all 3 patients were treated using the transcanal transpromontorial endoscopic exclusive approach.
The operation was performed under general anesthesia. The skin around the ear and external auditory canal was sterilized with povidone iodine. After preparation of the skin of the external auditory canal, it was sterilized again and injected with 1% lidocaine with 0.4% adrenaline with endoscopic guidance (0°, 3 mm outer diameter, 140 mm length). The tympanic canal flap was cut in a circular way 1 cm from the tympanic ring. The external auditory canal skin and tympanic membrane were stripped, and the external side wall of the upper eardrum was removed with an electric drill to expose the tympanic structure.
The ossicular chain was removed to expose the medial wall of the tympanic cavity. The stapes was removed, and the vestibular cavity tumor was exposed. The promontory was drilled with a 2-mm electric drill (Visao; Medtronic, Minneapolis, MN, USA) to uncover the basal and medial turns of the cochlea. The tumor was then exposed and removed (Figure 3).
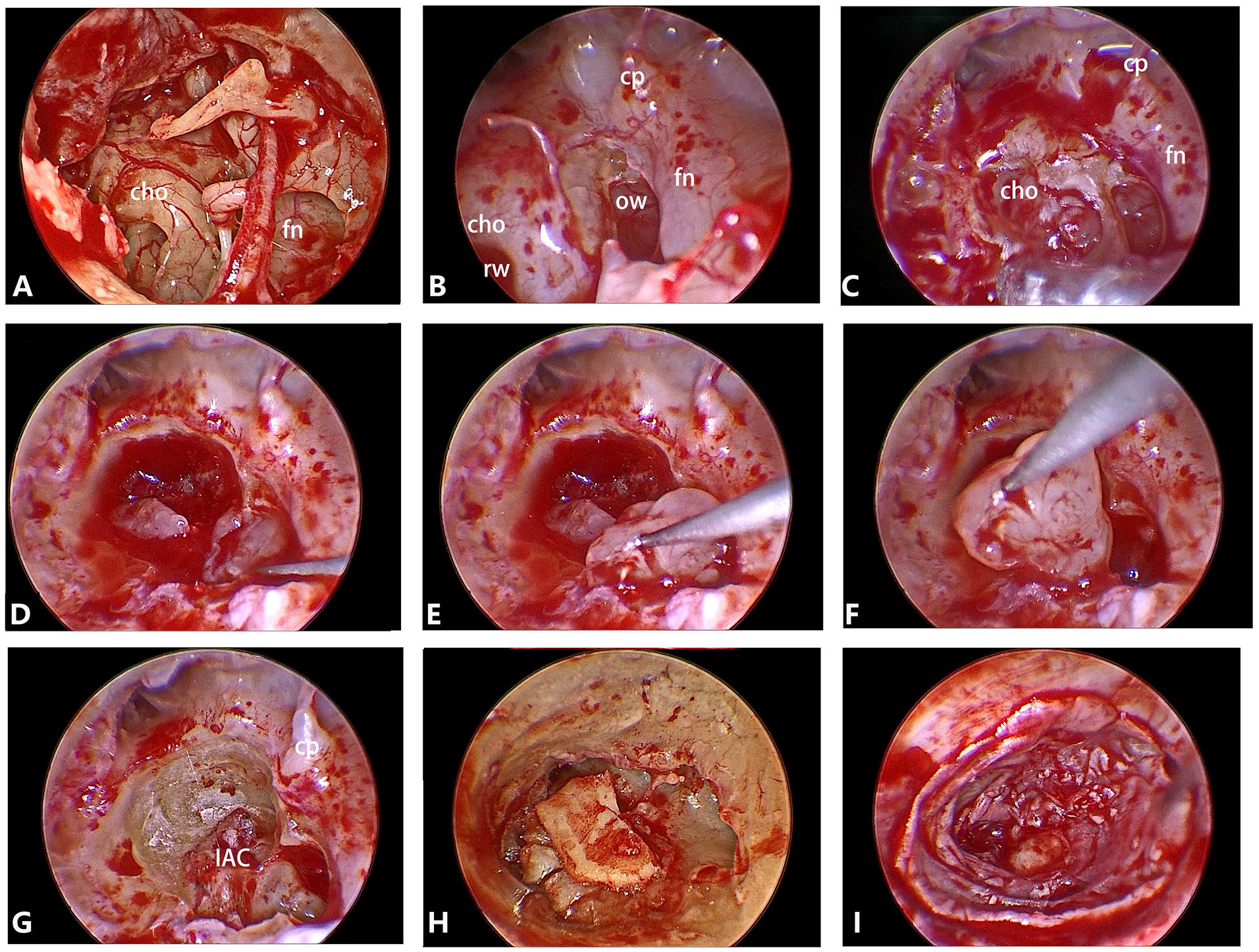
Surgical procedure. (A) The external auditory canal skin and tympanic membrane were stripped, and the external side wall of the upper eardrum was removed with an electric drill to expose the tympanic structure. (B) The stapes was removed to expose the vestibule. (C) The promontory was progressively removed to uncover the tumor in the cochlear turn and vestibule (white arrows). (D-F) Process of removal of intralabyrinthine schwannoma in the vestibule. (G) Close-up view of the IAC after tumor removal. (H) Perichondrium-cartilage was used to prevent cerebrospinal fluid leakage. (I) The flap of the external auditory meatus.
In one case, the stapes was retained without being removed. The bone of the lateral wall of the cochlea was drilled. The tumor was located in the basal and medial turns of the cochlea and was removed.
After tumor removal, the internal auditory meatus was sealed with fat and tragus perichondrium, cartilage, and glue. The Eustachian tube was obliterated with temporal muscle fragments, and the tympanomeatal flap was replaced. Finally, the eardrum and ear canal flaps were restored. Absorbable gelatin sponge was placed in the external auditory canal at the end of the procedure (Figure 3).
Postoperative Care
Observation in the intensive care unit was not necessary, and MRI was performed the next day after surgery. The patient remained in the supine position for 48 hours after surgery, and as no complications occurred, they were discharged from the hospital after 5 days, which is a significantly shorter hospital stay than in translab or middle fossa approach (about 10-14 days in our hospital). The criteria for discharge is as follows: The patient was in good condition, with normal temperature, no abnormal secretion exudation in the external auditory canal, no cerebrospinal fluid leakage, no redness and swelling in the eardrum incision, no severe postoperation vertigo.
Result
The operations were performed successfully in all 3 patients. The average operation time was about 2 hours, ranged from 1 to 2.5 hours. The follow-up period ranged from 10 months to 2 years. No major intraoperative and postoperative complications occurred. There was no facial paralysis, and postoperative facial nerve function was normal (HB grade I). None of the patients had cerebrospinal fluid leakage. The tympanic membrane healed without infection in all cases.
The vertigo, which means the subjective symptoms of dizziness of patients, was relieved in all 3 patients 1 week after the operation. The balance disorder (normal result of patients’ objective vestibular function examination) in all 3 patients had disappeared in ordinary life at 3 months postoperatively. After follow-up for 11 months to 2 years, none of the 3 patients had severe episodes of vertigo, with 1 IC patient complaining of transient episodes of vertigo (1-3 seconds) when climbing or lifting heavy objects. Tinnitus disappeared in 2 patients, while 1 patient had persistent tinnitus after surgery.
Postoperative (on the first day after the operation) MRI was performed in all 3 patients. All patients underwent total resection. MRI at 6 months postoperatively revealed no signs of tumor recurrence or nervous system involvement (Figure 4).
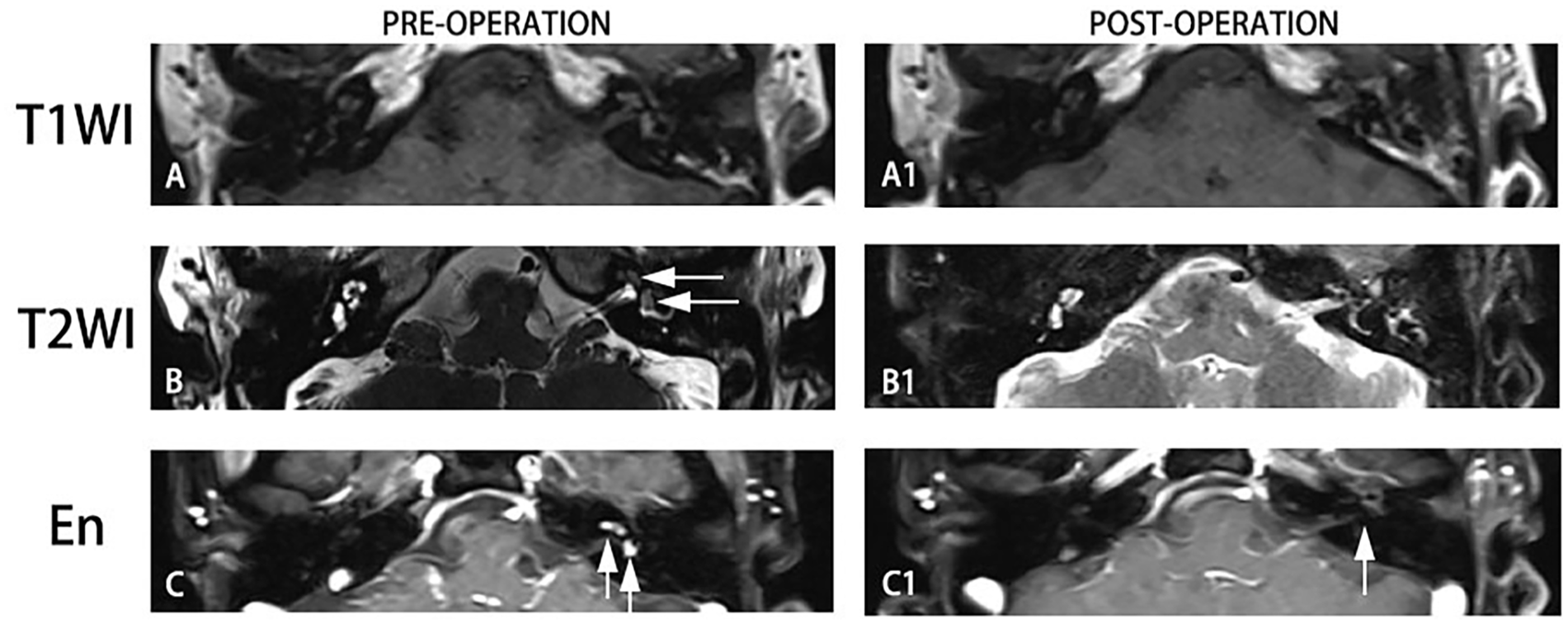
Before operation. (A) Intralabyrinthine schwannomas showed no abnormality on T1-WI. (B) Tumors confined to the cochlea showed a filling defect on T2-WI (white arrows). (C) Contrast enhancement of tumor in cochlea and vestibule on enhanced T1-WI (white arrows). After operation, (A1) no much difference was seen on T1-WI. (B1) The disruption of the labyrinthine structure due to the operation can be seen on T2-WI. (C1) Typical contrast enhancement of tumor disappeared (white arrow).
Discussion
ILS can originate from branches of cochlear or vestibular nerve in the inner ear labyrinth, and can also extend to IAC or even CPA, or laterally to the middle ear and external auditory canal. 22,33 ILS most commonly presents with sensorineural hearing loss, which is typically progressive; some patients show a sudden onset and a few shows fluctuating hearing loss. Speech recognition is abnormal; distortion product otoacoustic emissions may occur in the early stage. Tinnitus is the second most common symptom of ILS, with an incidence of more than 50%. 8 Vertigo symptoms are particularly common in ILS involving the vestibule. c-VEMP and o-VEMP are abnormal. 30 MRI examination showed low signal intensity T1-WI, and heterogeneous high signal intensity T2-WI (filling defect) after enhancement. 30
Our patients first visited a physician due to repeated vertigo, which is similar to Ménière’s disease. The patients were probably initially diagnosed with Ménière’s disease and treated accordingly. The tumors were finally found when the patients underwent gadolinium contrast MRI of the inner ear. MRI revealed the space-occupying lesions in the cochlea and/or vestibule with or without endolymphatic hydrops. Localized enhancement could be seen on gadolinium-enhanced T1-WI, while low-intensity signals (filling defect) were seen in the same part on T2-WI.
The surgical methods used for treatment of ILS include microscopic and endoscopic surgery. Microscopic surgery includes the labyrinthine, expanded labyrinthine, and ear sac approaches, depending on the localization and extension of the tumor. 21 To improve visibility, a large area of bone of the lateral skull base must be ground off when performing traditional surgery. Moreover, due to traction or compression of normal brain tissue during the operation, serious complications such as brain edema and intracranial hemorrhage may occur after the operation.
Endoscopy has been widely adopted in otologic surgery to approach the middle ear and skull base (a minimally invasive approach); surgeons usually apply the endoscopic transcanal approach to reach the IAC and resect VS. 15,21 Unlike traditional microsurgery, transcanal endoscopic AN surgery uses the natural orifice over the middle ear to reach the promontory directly, thus avoiding the removal of excessive amounts of temporal and labyrinthine bone. The endoscope can provide good lighting conditions and enlarged images. Endoscopes with different angles can improve visibility and show areas that are barely visible when using a microscope. Endoscopy allows clear identification of important anatomical structures, and their relationships with the tumor tissue, thus reducing damage and allowing complete removal of the tumor without leaving any residual tumor tissue.
Before the operation, it is necessary to conduct a detailed evaluation according to the imaging findings, determine the extent and condition of the tissue surrounding the lesion, determine the feasibility of completing the operation under total endoscopy, and formulate a careful surgical approach. Taken together, previous reports in the literature and our experience indicate that the TTEA technique was strictly restricted to the growing ILS (Kennedy Classification IV, IC IVC) with class D hearing, and intractable vertigo 34 (patients who underwent medical therapy and lifestyle modifications for at least 6 months without improvement and who had more than 6 vertigo attacks for the last 6 months before the surgery 35 ) or intolerable tinnitus nonresponsive to medical treatment. In addition, the TTEA surgical method is the suitable way to resect ILS tumors of the cochlea and vestibule. Endoscopic surgery increases the accuracy and safety of the ILS operation with minimal invasiveness. Endoscopic surgery reduced operation time and complications, and promoted faster postoperative recovery, good postoperative outcomes, complete tumor resection, shorter hospitalization times, and lower costs in comparison to traditional microscopic surgery. Neurological function was well preserved, and no facial nerve injury or cerebrospinal fluid leakage occurred. The patients recovered quickly after operation. There was no need to remove the sutures after surgery, and the patients could quickly return to their normal lives.
However, blood easily adheres to the endoscope, which must be cleaned continuously to achieve clear vision. When using endoscopy in operation, care is required to avoid intraoperative bleeding, which would obscure the visual field. During the operation, to facilitate exposure of the surgical field, we usually ensure that the tympanic canal flap is completely “sleeved” by the tympanic ring, and completely strip off the skin flap while the malleus handle remains on the tympanic membrane. This is convenient for bone grinding and promotes clear vision. The malleus handle was usually left on the tympanic membrane, which could be used for positioning when restoring the flap. In addition, we grind the bone under high-pressure water flow to wash away blood and bone debris accumulated in the cavity, and thus keep the visual field clear and reduce the operation time.
Our experience indicates that it is necessary to pay close attention at several points during the operation. When removing the tumor, it is necessary to first remove the tumor tissue from the vestibular cavity, and then to remove tumor tissue from the cochlea to delay the occurrence of cerebrospinal fluid leakage during surgery since dissection of tumor from vestibular cavity would not lead to cerebrospinal fluid otorrhea. We preserved the outermost bone shell of the cochlea to prevent internal carotid artery injury. After fully exposing the tumor in the cochlea, it should be removed as completely as possible to avoid cerebrospinal fluid otorrhea, which would affect the visual field and prolong the operation time. When repairing cerebrospinal fluid otorrhea at the bottom of cochlea, care is required to ensure that the muscle or fascia does not compress the facial nerve in the IAC, which may result in facial paralysis. Filling is generally done with the sandwich method using perichondrium, cartilage, or perichondrium and glue. The orifice of the Eustachian tube is closed by muscles.
Our follow-up results were satisfactory. There were no cases of tumor recurrence and no severe vertigo attacks. Only 1 IC subtype patient experienced transient vertigo, and only during certain postures and actions. Long-term follow-up to check the postoperative vestibular function (balance table test) in 3 patients also indicated a return to normal. One patient had a brief vertigo episode after the operation, which may have been because the vestibular cavity was not opened. The vestibular receptor still had some functionality, resulting in vertigo attack in a specific posture. We believe that the stapes could be routinely removed, and the vestibular cavity should be filled and closed for patients with profound hearing loss, even those of the IC type, to avoid postoperative vertigo.
In the process of the invention and trial use of TTEA as a new surgical technique, there were doubts about the higher probability of complications of facial paralysis and cerebrospinal fluid leakage. 36 However, with the accumulation of experience brought by time and the reference of previous precursor studies, we have achieved good surgical results in both IVC and IC patients, and avoided the occurrence of facial paralysis and cerebrospinal fluid leakage after setting strict surgical indications. We hope that it can be used for reference by future otorhinolaryngologists and contribute to the further development of endoscopic surgery in ILS. However, there are some limitations in this study, such as the small case number due to the restrictive criteria for this surgery. Also, long-term follow-up was required to evaluate the outcome of surgery, which is not included in our current study. In addition, for patients with poor hearing in the opposite ear, whether TTEA could be applied deserves careful weighing, considering the possibility of retaining the cochlea structure and a later cochlear implantation.
Conclusion
Here we reported a case series about fully endoscope resection of ILS using the TTEA, which has great advantages in removing tumor completely, minimal complications and short postoperative recovery period, to add to the case data of this rare disease and shares our team’s unique experience. While the preoperative evaluation is important, the TTEA technique was strictly restricted to the growing ILS (Kennedy Classification IV, IC IVC), with class D hearing, and intractable vertigo or intolerable tinnitus that does not respond to medical treatment. According to our experience, when resecting the ILS by TTEA, we should routinely remove the stapes and stuff the vestibular cavity for patients without serviceable hearing, even for the IC type, which might be better for the recovery of vertigo.
Footnotes
Acknowledgements
Thanks to the cooperation of anesthesia and nursing team to complete the operation.
Data Availability Statement
Data openly available in a public repository.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC; grant nos. 81771017 and 81970880 to DR); Natural Science Foundation of Shanghai (grant no. 22ZR1410100 to DR); General project of scientific research fund of Shanghai Health Committee (grant no. 202040286 to JW); the “Zhuo-Xue Plan” of Fudan University to DR; Shanghai 2020 “Science and Technology Innovation Action Plan”—“One Belt One Road” International Cooperation Project (grant no. 20410740600 to YY), and Clinical Research Plan of SHDC (grant no. SHDC2020CR1049B to YY).
