Abstract
Introduction
Vestibular schwannoma (VS) refers to a benign pathological tumor that originates in the lateral internal auditory canal (IAC) near Scarpa’s ganglia, which can extend medially to the cerebellopontine angle (CPA). 1 Treatment of vestibular meningioma includes waiting and observation, surgical treatment, gamma knife treatment, etc. For surgical treatment, the choice of surgical approach is very important for VS resection. The traditional approach to VS resection includes translabyrinthine, middle cranial fossa, and suboccipital retrosigmoid sinus.2-4 The translabyrinthine approach allows the most direct access to the CPA, which has the advantages of fully exposing the facial nerve and drilling extradural. 5 Hearing is sacrificed with the translabyrinthine approach, so it is only suitable for patients who have CPA lesions without considering the preservation of hearing. The retrosigmoid approach has a large operating space and full exposure to the IAC, so it can be used to remove tumors of various sizes in CPA. However, the meningeal incision is farther away from the CPA, and intraoperative pressure and pulling of the cerebellum are required, increasing the risk of postoperative cerebral edema. The middle cranial fossa approach can also preserve hearing, but the temporal lobe is pulled during the operation, the operating space is narrower than that of the retrosigmoid approach, and the exposure to CPA is limited, which is easy to damage the facial nerve, and is more suitable for tumors at the bottom of the ICA.
Due to the special location of the tumor, the otologist and neurosurgeons have been trying to find surgical methods that can resect the tumor with less trauma and complications for patients and preserve hearing and facial nerve functions. The retrolabyrinthine approach was important for hearing preservation, which was first reported by Hitselberger and Pulec 6 in 1972 and has been used to treat cranial nerve pathologies in the temporal bone and CPA for several decades,7-9 such as vestibular nerve section in Meniere disease, trigeminal nerve sensory root amputation in trigeminal neuralgia, facial muscle spasm, facial nerve microvascular decompression, etc. The retrolabyrinthine approach is primarily extradural, which does not need to pull the cerebellum and temporal lobe and can expose the semicircular canal to maintain the integrity of the labyrinth during the approach. However, due to its complex anatomical structure and narrow exposure range, its application scope was limited.
With the development of microscopy, endoscopy, and intraoperative function monitoring of facial nerve and auditory nerve, ear surgeons have recently tried to apply this approach to treat small VS tumors. Hao et al 10 used temporal bone high-resolution computed tomography (HRCT) to predict the size of the retrolabyrinthine space and the degree of exposure to the inner auditory canal Muelleman et al 11 found that, using endoscopic assistance, the retrolabyrinthine approach can expose the entire IAC in specimens. In this article, we want to be innovative in using this surgical approach to remove VS and to study its feasibility and safety through preoperative CT evaluation in patients during operation.
Methods
Ethical Consideration
The study was carried out according to the Declaration of Helsinki. All included patients provided signed informed consent to participate.
Design
Single-center retrospective study.
Study Design and Participants
The study was carried out in the Department of Otolaryngology of the Eye & ENT Hospital, Fudan University, Shanghai, China. Five patients with VS were reviewed who underwent retrolabyrinthine surgical approach between October 2019 and March 2021. The recruited patients were clinically diagnosed with VS through computed tomography (CT) and magnetic resonance imaging (MRI) examinations. The tumor was in ICA-CPA and CT showed enlargement of the IAC. MRI showed isosignal or slightly low signal on T1-weighted images (T1WI), slightly high signal on T2-weighted images (T2WI), and uneven signal and uneven enhancement after enhancement with clinical symptoms, such as hearing loss, tinnitus, and dizziness.
Main Outcome Measures
Pure tone audiometry (PTA), speech discrimination score (SDS), auditory brainstem responses, and auditory steady-state evoked response were used to assess the hearing. The facial function was assessed using the House/Brackmann scale. The sensitive information relevant to patient privacy was removed before analysis and all participants gave their written informed consent to scientific research. Surgery was performed in all patients using a retrolabyrinthine approach.
The 3-Dimensional CT Reconstruction Demonstrated the Retrolabyrinthine Space and Adjacent Structures
A retrolabyrinthine approach refers to performing surgery in the space between the posterior semicircular canal and the anterior wall of the sigmoid sinus. Exposure to the surgical field depends on the distance from the anterior edge of the sigmoid sinus to the posterior semicircular canal and is affected by the position of the jugular bulb. Two-dimensional CT images cannot directly demonstrate the size of the retrolabyrinthine space. To better assess the feasibility of VS resection, the temporal bone of the patients was scanned with a multidetector row CT (Sensation 16; Siemens Medical Systems, Erlangen, Germany) and performed with a scan slice thickness of 0.625 mm, followed by a coronal multiplanar reconstruction. The axial CT scan images were imported into the Mimics 10.0 software (Materialize, Leuven, Belgium) in scan order and used for modeling. Structural boundaries are delineated by an interactive manual and automated division. To better show the adjacency of important structures, we used Mimics 10.0 software to hide soft tissue structures, and combined manual and automatic interactive division to highlight the important bony structures in the temporal bone, including the sigmoid sinus, dura, endolymphatic sac, facial nerve canal, IAC, and the 3 semicircular canals and cochlea (Figure 1). According to the adjacent relationship skeleted by the 3-dimensional reconstruction of the temporal bone, the space between the posterior semicircular canal and the anterior sigmoid sinus can be exposed. And the IAC can be exposed under direct vision, from which the endolymphatic sac can be seen on the superficial side of the IAC. Mastoid skeletonization, maximum exposure of the retrolabyrinthine space, endolymphatic sac treatment, and IAC exposure are the main steps of surgery. Starting with the surgical position, we will introduce the procedure of the retrolabyrinthine approach step by step in detail.
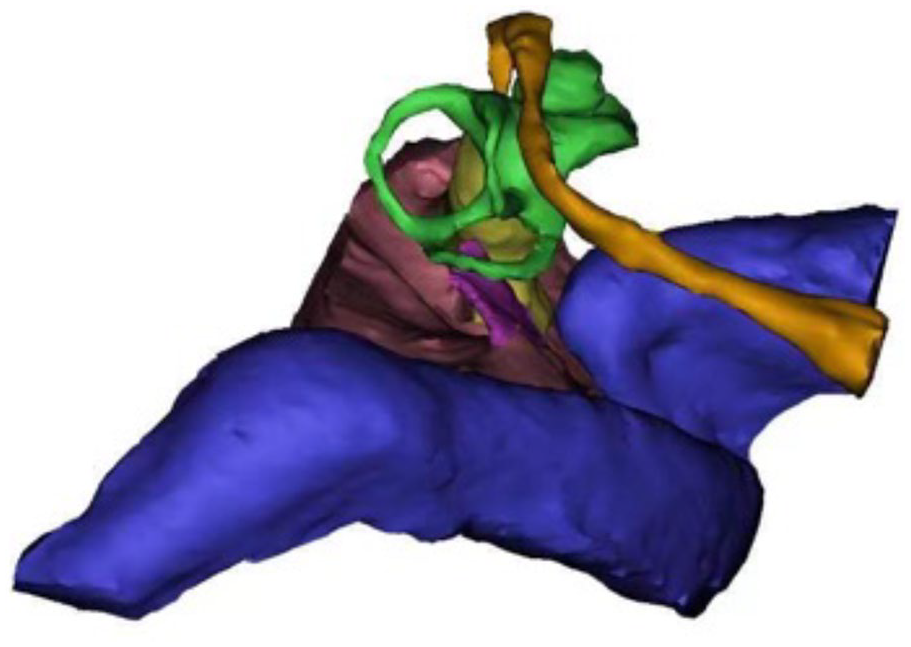
Exposure to the surgical field. The important structures of temporal bone are shown in 3-dimensional proportions, including the sigmoid sinus and jugular vein (dark blue), endolymphatic sac (purple), posterior fossa dura (brown), internal auditory canal (yellow), 3 semicircular canals (green), and facial nerve (orange).
Surgical Technique
Preparation and position
In contrast to most intracranial operations, rigid fixation of the head is not necessary. The patient was placed in the supine position with the head turned laterally to the lesion so that the angle between the shoulder and the occipital bone was open. During the operation, the angle of the operating bed could be rotated axially to adapt to the direct-vision range of the microscope and fully expose the surgical field.
Surgical incision
Parallel to the posterior sulcus, a posterior auricular arc incision was made from the upper edge of the auricle root to the tip of the mastoid process. The middle part of the incision was 1.5 cm away from the posterior sulcus and the lower end was about 0.5 and 1.2 cm away from the root of the auricle, respectively.
Mastoid skeletonization
The exposure to the operative field of the posterior labyrinth approach depends on the distance from the anterior edge of the sigmoid sinus to the posterior semicircular canal and is affected by the position of the jugular bulb. After the mastoidectomy was complete, a large diamond drill was used to remove the bone from the surface of the sigmoid sinus, leaving a thin slice of bone on the surface of the sigmoid sinus. The jugular bulb, horizontal, superior, and posterior semicircular canals are identified and contoured, with the bony labyrinth preserved, and do not blue line. The bone on the surface of the sigmoid sinus, posterior fossa, and jugular bulb can be removed, and the sigmoid sinus and jugular bulb can be elastically pushed and pulled, thus expanding the surgical field.
The anatomical landmarks of the deep surface of the operation cavity included the entrance of the posterior semicircular canal, the jugular bulb, the endolymphatic sac, the dura, the sigmoid sinus, the cone segment of the facial nerve, and the vertical segment of the facial nerve (Figure 2A).
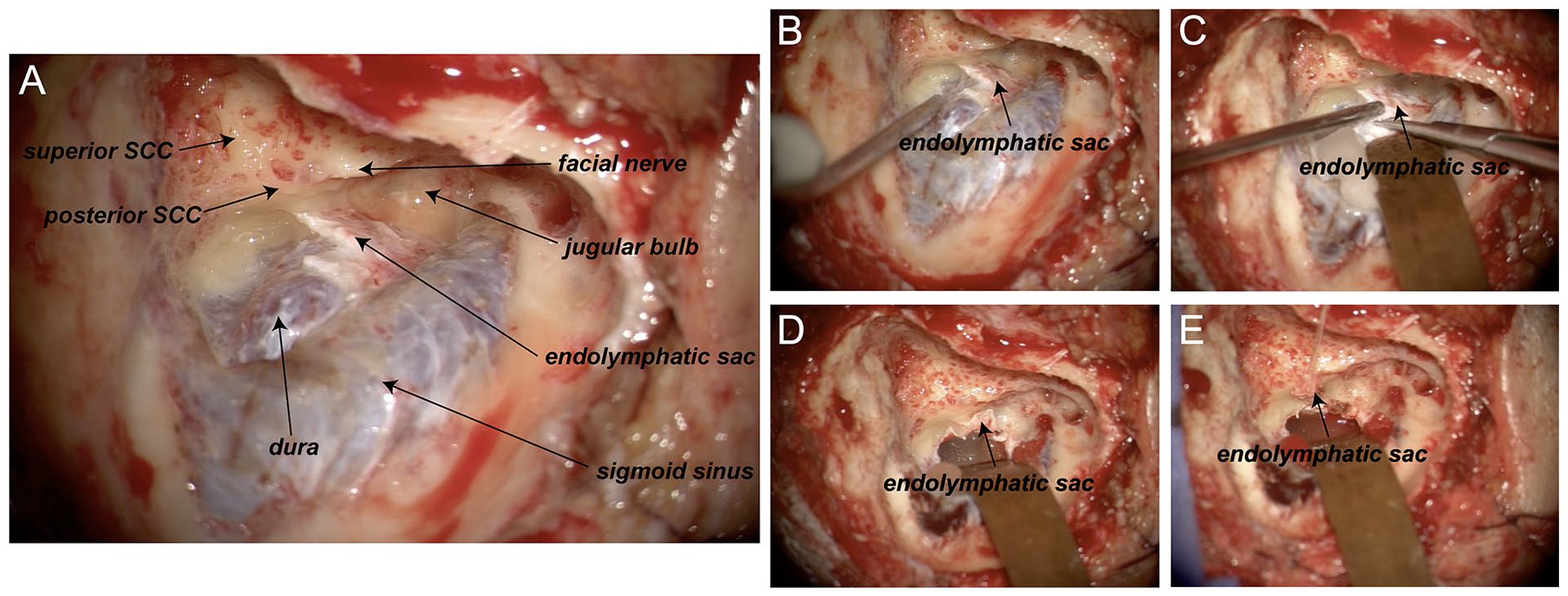
Exposure to the retrolabyrinthine space. (A) Anatomical diagram of the right retrolabyrinthine space fully exposed during surgery. The 3 semicircular canals are covered by a dense bony shell. The endolymphatic sac is located below the posterior semicircular canal, grayish white, and thicker than normal meningeal tissue. The bone on the surface of the sigmoid sinus and dura was completely removed. Handling of endolymphatic sacs. (B-E) The dura was incised around the endolymphatic sac and the endolymphatic sac was turned forward and fixed.
Processing of the endolymphatic sac
The endolymphatic sac is a blind pouch that connects the membranous labyrinth through the endolymphatic vessels, located between the 2 layers of the dura mater in the posterior fossa, below the extension of the external semicircular canal. The proximal end of the endolymphatic vessel and the endolymphatic sac is located in the vestibular aqueduct, which runs medially to the posterior semicircular canal into the vestibule. After adequate exposure of the sigmoid sinus and posterior fossa dura, a septal dissector was used to gently depress and expose the posterior fossa dura to confirm the entry of the endolymphatic sac into the vestibular aqueduct. The dura was incised around the endolymphatic sac, and the endolymphatic sac was turned forward and fixed (Figure 2B -E).
Tumor dissection and reconstruction
Full exposure of the IAC was the key to completely exposing and dissecting the tumor. The 270-degree bone above, below, and behind the IAC was removed (Figure 3A and B). After identifying the tumor, the tumor cyst was cut and the tumor in the cyst was removed. The tumor-carrying nerve was then cut at the bottom of the IAC, and the tumor was pulled medially, thus gradually separated from the facial nerve and the cochlear nerve. Finally, the tumor was completely resected with endoscopic assistance. The intraoperative facial nerve was monitored to protect facial nerve function. The cochlear nerve was anatomically preserved (Figure 3C-F).
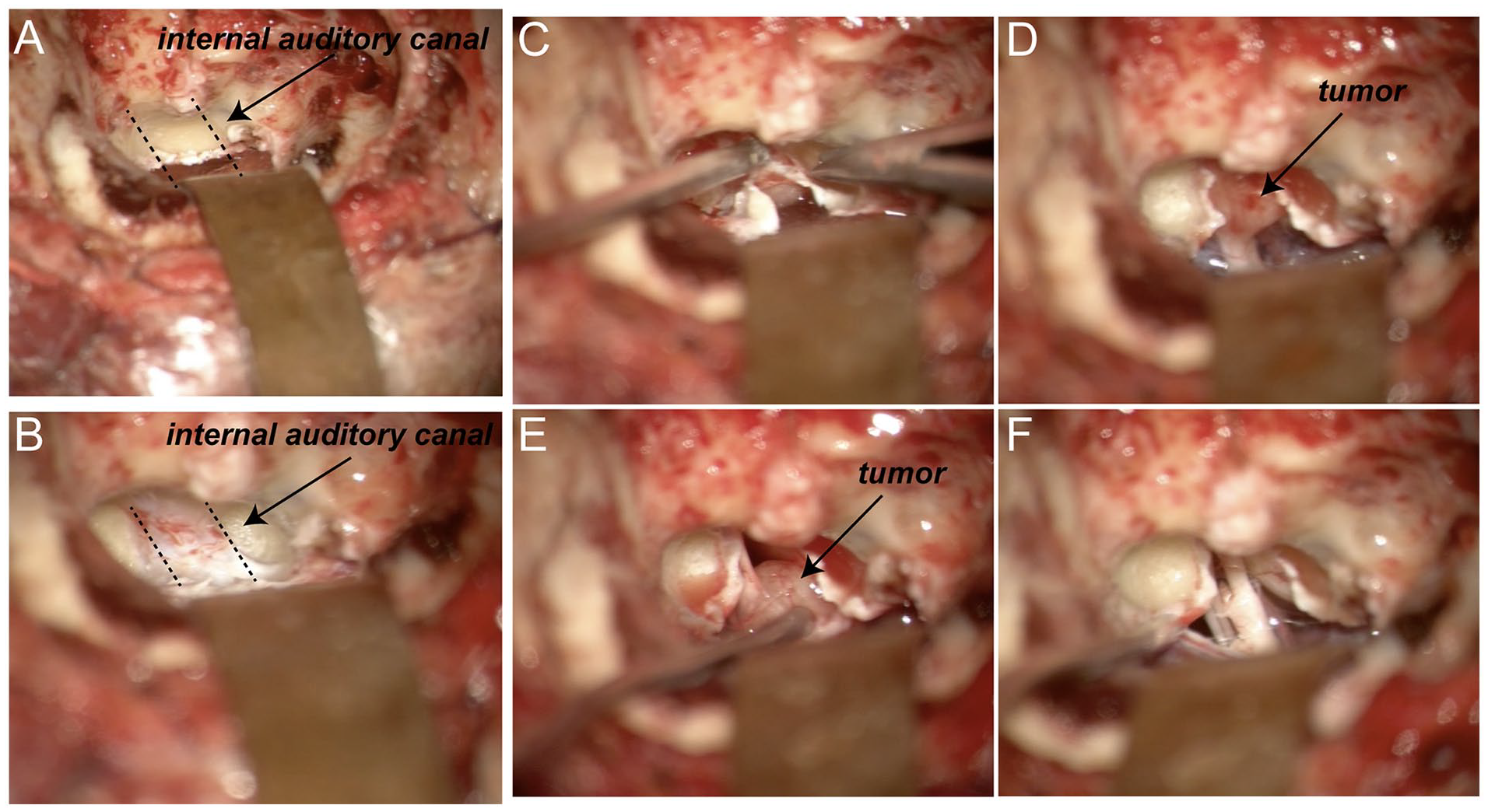
Important surgical process. (A, B) The projection of the internal auditory canal in the retrolabyrinthine space and the surface bone were removed. (C-F) Tumor resection with preserved facial and cochlear nerves.
The operative cavity was washed with antibiotics, artificial meninges were placed, and abdominal fat was taken to fill the operative cavity. Prothrombin was instilled into the surgical cavity. The tympanic sinus entrance was packed with muscle. The incision was sutured layer by layer.
Surgical Case Example
A 61-year-old man was admitted to the hospital with a 3-year history of hearing loss and tinnitus of the left ear. Preoperative PTA suggested moderate and severe hearing loss in the left ear. The hearing classification assessed by the American Academy of Otolaryngology Head and Neck Surgery (AAO-HNS) hearing rating grade was grade C. The facial nerve function assessed using the House/Brackmann scale was grade I.
The CT image showed enlargement of the left IAC and increased soft tissue density shadows from the IAC to the cerebellar pontine angle (Figure 4A). No obvious enlargement of the right IAC was observed. No bone absorption or destruction was observed.
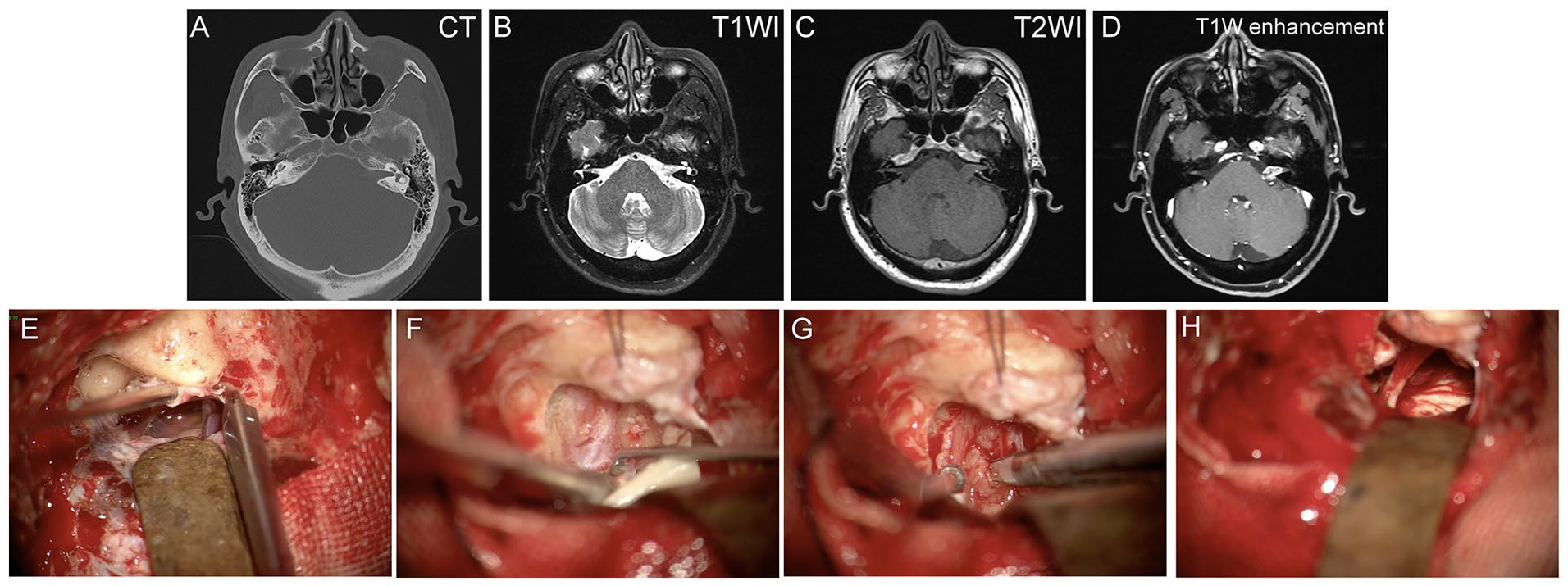
Images and surgical records of a patient with left internal auditory canal–cerebellopontine angle (IAC-CPA) occupying lesion. (A) The left IAC was enlarged and increased soft tissue density shadows were observed in the IAC-CPA area. (B-D) Soft tissue mass from the left IAC to the cerebellar pontine angle with enlargement of the left IAC. It was about 1.0 × 1.5 cm in size with moderate intensity in T1-weighted images (T1WI) and unevenly slightly high intensity in T2-weighted images (T2WI), which was unevenly enhanced after angiography. (E) Endolymphatic sac transposition. (F) Exposed IAC. (G) Tumor dissection. (H) The cranial part of the auditory and facial nerves.
MRI image showed a soft tissue mass in the left IAC to cerebellar pontine angle with enlargement of the left IAC. It was approximately 1.0 × 1.5 cm in size with moderate intensity in T1WI and uneven slightly high intensity in T2WI, which was unevenly enhanced after angiography (Figure 4B-D). The mass was adjacent to the left pedunculus cerebellaris medius, with no obvious pressure on the pedunculus cerebellaris medius and the fourth ventricle.
The decision to undergo resection of VS via the retrolabyrinthine approach was made after the patient was fully informed of the surgical options and associated surgical risks. The surgery was performed by ear surgeons and neurosurgeons together. After mastoid skeletonization, endolymphatic sac transposition, and IAC opening, the tumor was completely removed preserving the the auditory and facial nerves (Figure 4E-H). After the operation, which lasted about 6 hours, the patient was transferred to the postoperative care unit for close observation overnight. Subsequently, the patient was transferred to a general ward and kept under observation for a week until the stitches were removed. During hospitalization, the patient was complicated with vertigo after the operation, and the symptom disappeared after a week without any treatment.
Follow-up
Postoperatively, the patients were followed up in the outpatient clinic by the chief surgeon. Temporal bone MRI, audiometry, SDS, facial nerve function, and neurological examination were performed in patients 6 months after surgery. There were no complications such as intracranial hemorrhage, infection, or facial paralysis in the 5 patients who underwent VS resection by retrolabyrinthine approach. The results of the temporal bone MRI 6 months after surgery indicated that the VS was completely removed in all 5 patients. According to the AAO-HNS hearing rating grade, we compared the auditory function of the patients before and 6 months after surgery. Grades I, II, and III were considered postoperative hearing preservation.
Results
Four of the 5 patients had their hearing preserved after surgery, according to the results of audiometry and SDS 6 months after surgery. Hearing in a patient was not preserved because the tumor adhered to the auditory nerve was damaged during the operation. The function of the facial nerve was judged according to the House/Brackmann (HB) scale. The HB scores of the 5 patients were all grade I preoperative, postoperative, and 6 months postoperative. The demographic data of the patients, tumor sizes, preoperative and postoperative hearing classes, postoperative facial nerve function, and follow-up times are shown in Tables 1 and 2.
Demographic Characteristics of Patients.
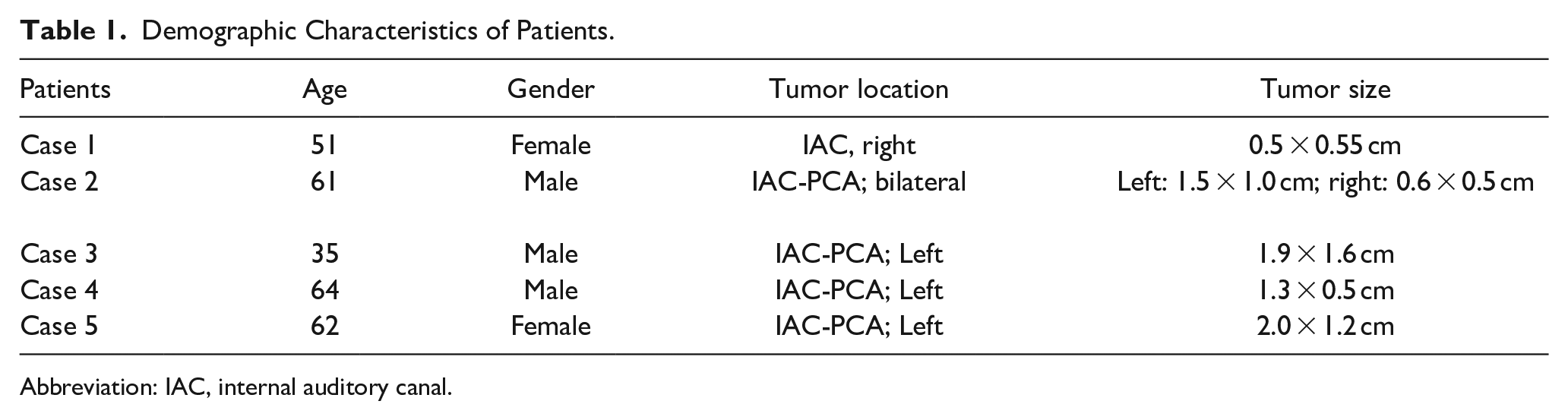
Abbreviation: IAC, internal auditory canal.
Hearing Classification 6 Months After Surgery of Operated Ear According to AAO-HNS Hearing Rating Grade and Facial Function 6 Months After Surgery Assessed by House/Brackmann Scale.
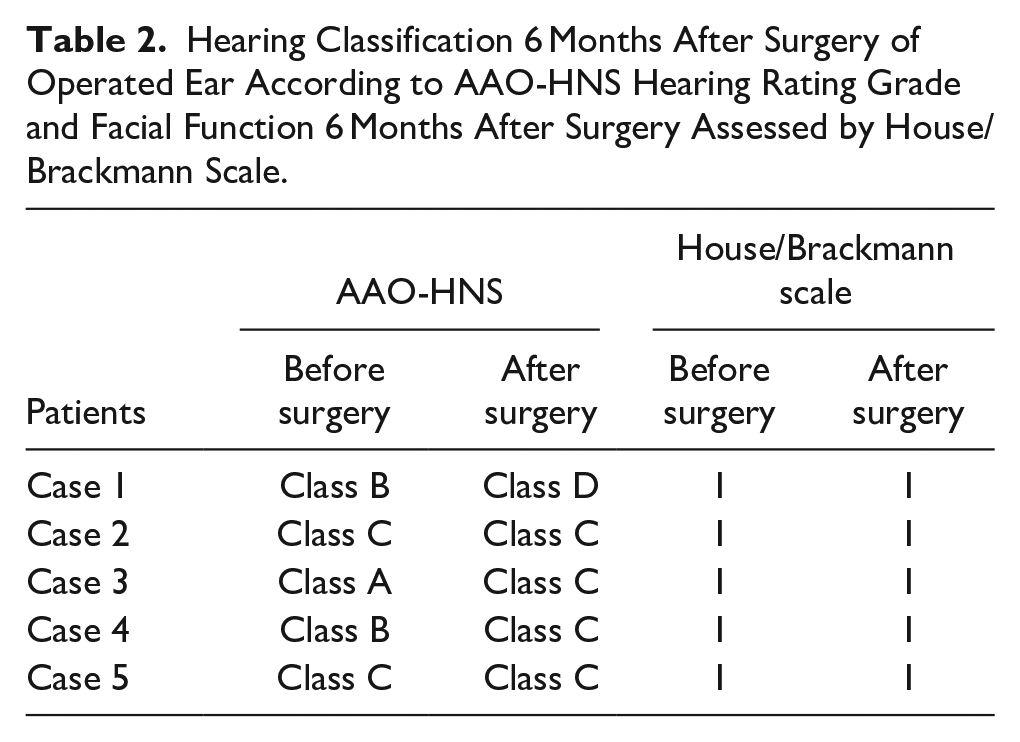
Discussion
VS accounts for about 6% to 8% of intracranial tumors and 80% to 90% of cerebellar pontine angle tumors. The clinical manifestations of VS are related to the size, location, and extent of the tumor. Smaller tumors can be asymptomatic. Tinnitus, sensorineural hearing loss, and vertigo are the most common early symptoms of this disease. As the tumor grows and expands, symptoms such as trigeminal nerve damage, facial paralysis, cerebellar dysfunction, and intracranial hypertension may occur due to increased intracranial pressure and tumor invasion into the posterior fossa. A few tumors that are located in the vestibule, cochlea, or semicircular canals are called intralabyrinthine schwannoma. 12 When invading the jugular foramen area, compression of IX, X, and XI on the cranial nerve can cause the corresponding symptoms of the cranial nerve. The tumor invades the middle cranial fossa, compressing the abducens nerve and oculomotor nerve, causing eye movement disorders, diplopia, etc.
Surgical excision is the accepted treatment of choice. House introduced the operating microscope and microsurgical technology in the surgical treatment of skull base diseases in the 1960s, marking the formal entry of lateral skull base surgery into the era of microsurgery. 13 Due to the rapid development of imaging, microsurgical technology, surgical approach, anesthesiology, and neuromonitoring technology, the diagnostic level and therapeutic effect of VS have made a qualitative leap. At present, cerebellar pontine angle operation, especially VS operation, is gradually turning to low mortality, low complication rate, and high retention rate of neurological function, to improve the postoperative quality of life of patients as much as possible. Currently, the retention rate of facial nerve function after VS surgery is more than 80%. 14 However, the postoperative auditory retention rate has been unsatisfactory, and the auditory retention rate is less than 70% after the operation of VS by the hearing-preserving approach. 15
For VS requiring surgery, in addition to the retrosigmoid approach and the middle cranial fossa approach, the retrolabyrinthine approach is a good option for patients with only a hearing ear or with good hearing. The retrolabyrinthine approach to the CPA is shorter and more direct than the retrosigmoid approach. Furthermore, compared to the middle cranial fossa approach, the larger space of the posterior cranial fossa exposed through the retrolabyrinthine approach reduced the huge risk of hemorrhage causing brainstem compression and the possibility of sequelae caused by temporal lobe compression.
The 3-dimensional CT imaging technique is used to convert 2-dimensional images into intuitive and accurate stereoscopic images, which can not only be observed from different angles but also be used to distinguish the different tissues. Our previous study quantified the surgically relevant anatomic relationships and 3D morphology of the supralabyrinthine space. 16 Hao et al 10 used standard postprocessing images of temporal bone HRCT to predict the size of the retrolabyrinthine space and the degree of exposure to the inner auditory canal. Based on 3-dimensional images, the distance, length, width, and angle between important anatomical marks can be measured to analyze the feasibility of surgery.
In this study, the temporal bone anatomy under the guidance of 3D reconstruction animation of the volunteer’s head spiral CT scan images was used to demonstrate the feasibility of entering the CPA and the IAC through the retrolabyrinthine approach. And VS of different sizes (0.7-2.0 cm) was successfully resected by the retrolabyrinthine approach with preservation of the endolymphatic sac, and facial nerve function was preserved in all 5 patients, the hearing preservation rate is 80%. The results of the 3D reconstruction images showed that the processing of the endolymphatic sac was very important for exposing the area of IAC, and the contour of the posterior semicircular canal was the key to locate IAC and endolymphatic sac. In addition, it was safe to handle the fundus of the IAC in the retrolabyrinthine space by using the semicircular canal as a sign, which effectively avoided damage to the labyrinth. The exposure of the vestibular aqueduct and its relationship to the posterior semicircular canal are also important markers in our surgery. Our practical experience indicated that resection of vestibular neurinoma via retrolabyrinthine approach is feasible, which may be a potential and promising route for hearing preservation.
Footnotes
Author Contributions
YN and WZ designed the studies; XL and HM acquired the data; XL, HM, and YN analyzed the data; and XL and HM wrote the manuscript.
Data Sharing and Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82271169) and the “Clinical research project” (20224Y0319).
