Abstract
Objective
The benefit of a nasal corticosteroid in the treatment of persistent post-infectious smell disorders is not as clear in previous studies as is assumed for olfactory training. This study would therefore like to describe the treatment strategies using the example of a persistent olfactory dysfunction as a result of a proven infection with SARS-CoViD-2-virus.
Methods
Twenty patients (average age of 33.9 ± 11.9 years) with hyposmia were included in this study from December 2020 to July 2021. Every second patient received additionally a nasal corticosteroid. The two resulting randomized groups of equal size were screened with the TDI test, a 20-item taste powder test for the assessment of retronasal olfaction and otorhinolaryngological examination. The patients were asked to train twice daily using a standardized odor training kit and followed up after 2 months and 3 months, respectively.
Results
We documented a significant overall improvement in olfactory ability over the investigation period in both groups. While the TDI score steadily increased on average under the combination therapy, the rise under olfactory training alone was initially steeper. This short-term interaction effect over mean two months was not statistically significant. According to Cohen, however, a moderate effect (eta 2 = 0.055, Cohen`s d = 0.5) can still be assumed. This effect could be explained by a possibly higher compliance at the beginning of the sole olfactory training due to the lack of further drug treatment offers. When the training intensity decreases, the recovery of the sense of smell stagnates. Adjunctive therapy ultimately outweighs this short-term benefit.
Conclusions
The results reinforce the recommendation of early and consistent olfactory training on patients with dysosmia due to COVID-19. For continuous improvement of the sense of smell, an accompanying topical treatment seems at least to be worth consideration. The results should be optimized with larger cohorts and using new objective olfactometric methods.
Introduction
Even if the pandemic currently seems to have less of an impact on daily life, the long-term effects of coronavirus disease 2019 (COVID-19) remain a challenge in medical practice and therefore a topic of scientific interest. 1 Impairments of the human sense of smell as a result of a corona virus infection were reported from the beginning of the pandemic.2-5 Though many patients seem to recover their sense of smell after a few weeks, a certain proportion of them develop a long-lasting olfactory disorder post-COVID-19.4,6-8 In addition to hyposmia and anosmia, persistent parosmia is reported with striking frequency.9,10 Less surprising is that olfactory dysfunction is commonly associated with gustatory impairments.11-13
Even vaccinations cannot completely protect against an infection and thus against possible disorders of the olfactory function. 14 As of October 2022, there is disagreement about the therapeutic options for post-viral olfactory disorders such as those resulting from Coronavirus Disease 2019. 15 In our opinion, we are still a long way from a rational therapy recommendation that can claim to be a standard. 16 The commonly used therapy with a topical nasal corticosteroid (TNC) has an ambiguous grade of recommendation.17,18 A positive effect of olfactory training (OT) on the recovery from post-infectious olfactory disorders has already been shown in studies.19-21 This study aims to compare these two therapeutic approaches to the treatment of persistent olfactory dysfunction. Most data on loss of smell rely on the evaluation of orthonasal olfactory performance. An extended assessment also seems advisable for the treatment concept. Therefore, we also aimed to assess retronasal olfactory function over the study period. Based on the current state of the literature, we considered a parallel further control group of patients without any therapy recommendation to be ethically unacceptable.
Material and Methods
Subjects
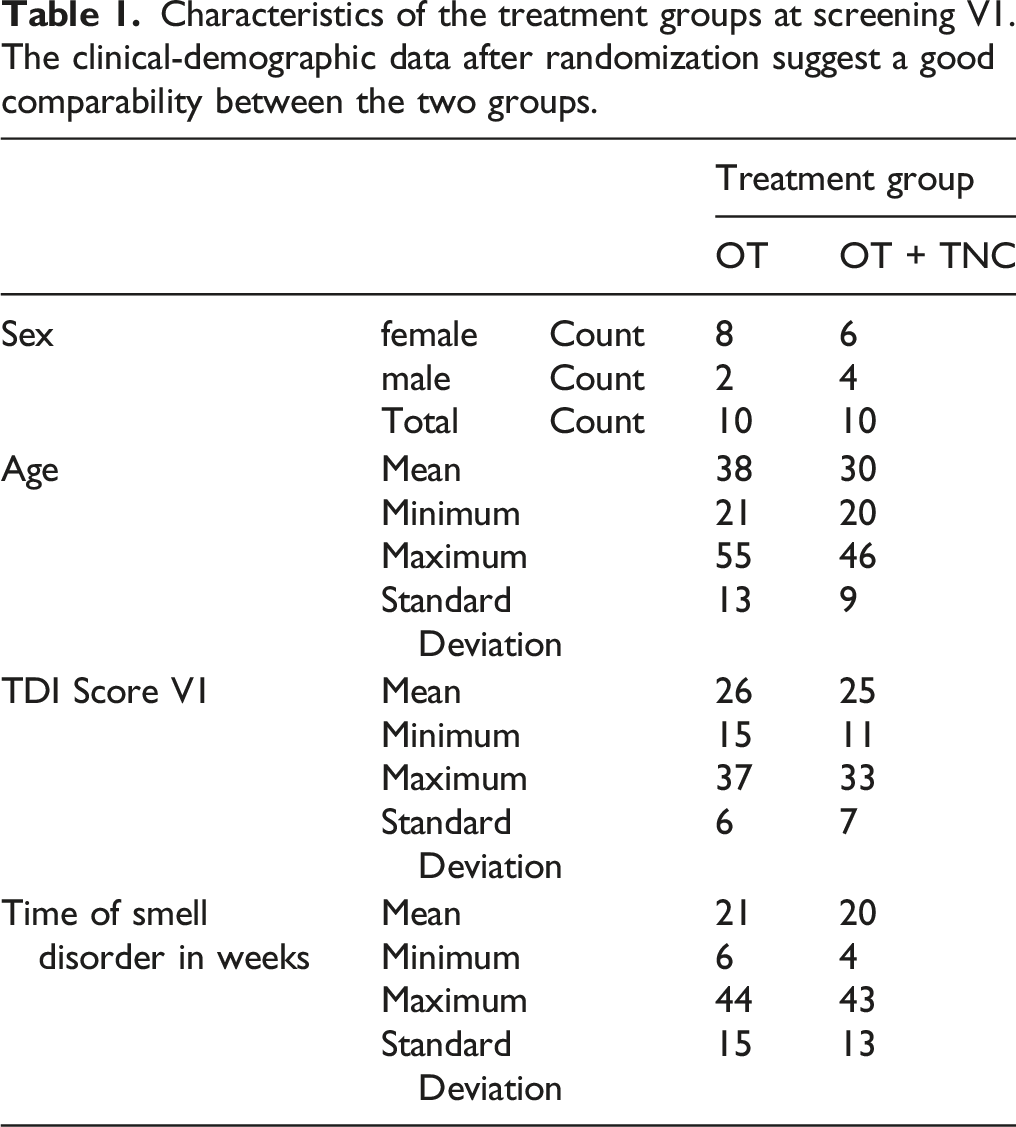
Twenty non-hospitalized patients (age between 21 and 54 years) with a sudden loss of smell after a confirmed COVID-19 diagnosis were recruited for this study. The acute infection was on average 5 months (4–40 weeks) before screening. No patient showed persistent symptoms of COVID infection except for olfactory loss. Between December 2020 and July 2021, 14 female and 6 male participants were included in the study. All selected patients were SARS-CoV-2 negative at screening. Predefined exclusion criteria were: Age below 18 or over 80, pregnancy, olfactory disorder with neurodegenerative or congenital etiology. Two patients had anosmia, 9 hyposmia, 5 parosmia together with hyposmia and 4 had predominantly parosmia. A follow-up visit was scheduled at 2 months and a final visit 3 months after screening. At the baseline and follow-up visits, all patients underwent olfactory testing and an otorhinolaryngological examination. They were randomized by alternating assignment to the treatment pathways. This resulted in two therapy groups, one with olfactory training alone (OT) and the other with additional topical nasal corticoid (OT plus TNC). Four patients terminated their participation in the study early at their own request. Three of them reported the success of the therapy without this being able to be confirmed in a final visit. One patient experienced a severe deterioration in her sense of smell despite the training. Because of the beginning of Lent, she waived the offered therapy escalation. For ethical reasons a further control group with corresponding symptoms and without any therapeutic recommendation was deliberately renounced.
Olfactory testing
Orthonasal olfactory function was evaluated by the “Sniffin’Sticks” (Burghart Messtechnik GmbH, Germany). The battery test comprises three olfactory tasks: threshold (T), discrimination (D) and identification (I). The sum of the scores from the three subtests makes up the “threshold discrimination identification score” (TDI score). A TDI value of less than 16 points was rated as anosmia, up to 30.5 points as hyposmia and a value above 31 points as normosmia.22,23 The change in the scores between the sessions is correspondingly denoted here by way of example with ΔTDI_V1 = TDI_V2−TDI_V1. This applies to the components of TDI as well as to the score of an extended psychophysical test to determine the retronasal olfactory function. 24 This 20-point test was evaluated using flavored powders lacking distinctive tastes administered to the oral cavity.
Treatment
Olfactory training (OT) was explained to each patient. All patients were given a commercially available Odor Training Set at the screening visit (“Smell Training Kit” Dos Medical-BV, Heteren, Netherlands) containing the four internationally recognized standard fragrances (rose, lime, eucalyptus and cloves). Patients were asked to perform sniffing sessions twice a day for at least 5 minutes minimum. For each session of smell, training patients were asked to mindfully sniff four odors for approximately 10 seconds each. The comparison group received a concomitant therapy using a steroid containing nasal spray (TNC). After checking for potential contraindications to TNC, the patients received the standard preparation NASONEX® (mometasone furoate monohydrate aqueous nasal spray, 50 micrograms/actuation) to be used twice a day in each nostril.
Statistical analysis
All analyses were performed using the Statistical Package for the Social Sciences (SPSS Statistics for Windows, Vers. 28, IBM, Armonk, NY, USA). Data are presented as mean ± standard deviation (SD) or median and quartile. A probability value of less than 0.05 was considered statistically significant. Mixed-Design ANOVA was used for a combination of ordinary, variance and covariance analysis with measurement repetitions. To explore the evolution of quantitative variables (T, D, I, TDI and R score) before, under and after the OT, we employed several tests. As confirmed by Lilliefors test and Shapiro–Wilk test (p > 0.05), we can largely assume a normal distribution with a few exceptions. For groups of exactly the same size, we consider the slight deviations from the normal distribution for the ANOVA to be acceptable. 25 Sphericity can be assumed according to the Mauchly test (p = 0.062). There was homogeneity of error variances as assessed by the Levene test (p > 0.05) as well as a homogeneity of the covariances as assessed by the Box test (p = 0.153).
The influence of other factors (age, gender, duration of the olfactory disorder, duration of treatment, baseline value of olfaction) was modeled additionally using a regression analysis among dependent variables. All patients performed the first follow-up and 16 of 20 the second follow-up after baseline. In the three cases (2 in the OT group and one in OT + TNC group), it has been justified to carry forward the last observation (LOCF). Identical values were added for the last visit, assuming that the already significantly improved olfactory ability remained at least the same as the previous visit (Figure 1). Evolution of olfactory disorder due to COVID-19: Evolution of olfactory disorder due to COVID-19 in dysosmic patients between December 2020 and July 2021 before, under and after olfactory training with (OT plus TNC, n = 10) or without (OT, n = 10) a concomitant topical nasal corticosteroid therapy (TNC). TDI score = Threshold–Discrimination–Identification score; Colored parts cover anosmic (red), hyposmic (yellow) or normosmic (green) patients according to TDI normative; V1 = Baseline; V2 = Follow-up after 2 months; V3 = final visit after 3 months.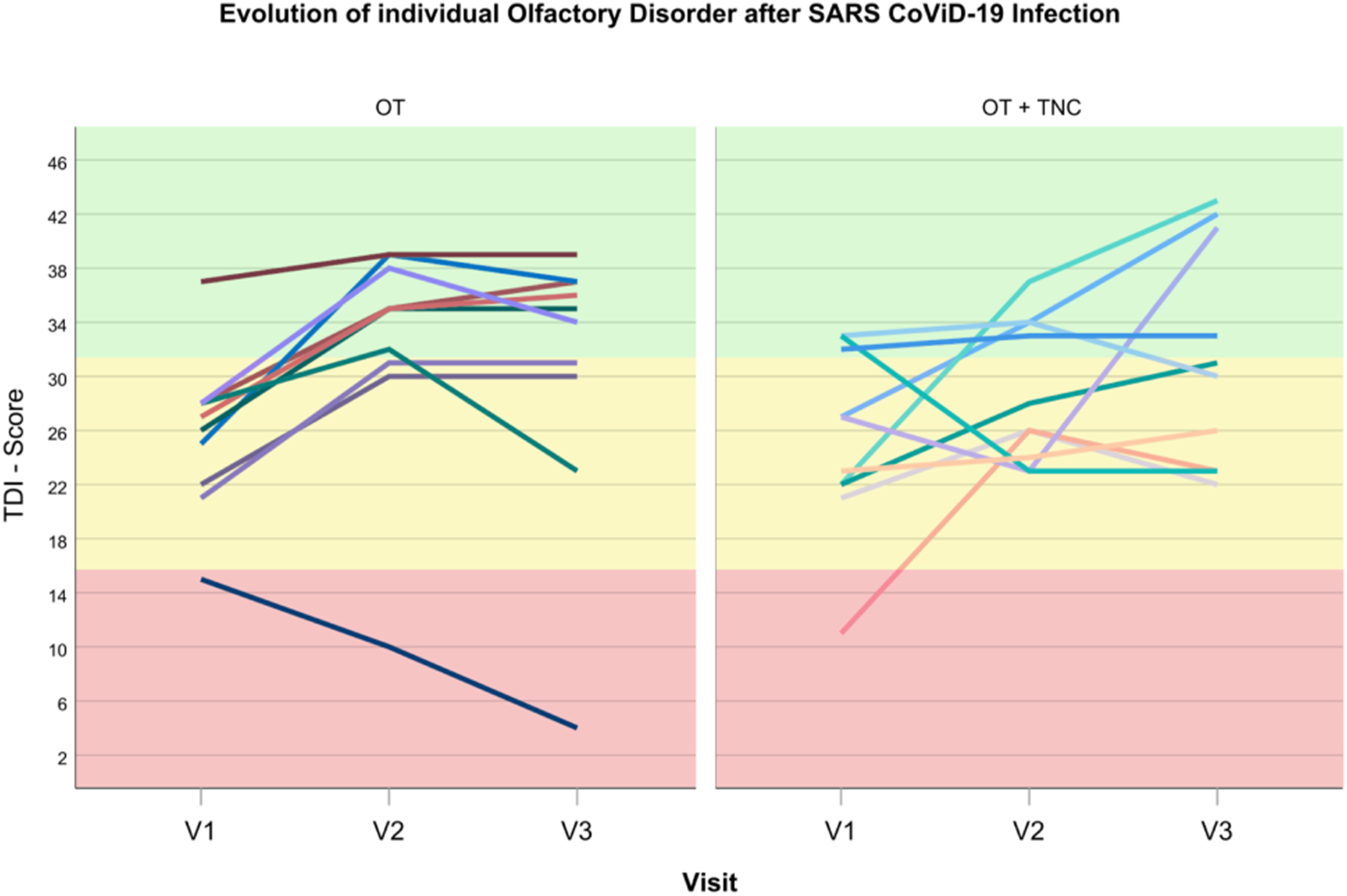
Results
Characteristics of the treatment groups at screening V1. The clinical-demographic data after randomization suggest a good comparability between the two groups.
There was no statistically significant interaction between time and group, F(2) = 1.042, p = 0.363, partial η2 = 0.055. Effect sizes are also considered by the use of eta-square after Cohen (1988). A value of more than d = 0.2 represents small effects, more than d = 0.5 average effects and more than d = 0.8 large effects. According to Cohen, a moderately interaction effect (eta
2
= 0.055, Cohen`s d = 0.5) was found between time and group. While the TDI score steadily increased on average under the combination therapy, the rise under olfactory training alone was initially steeper (Figure 2). After all, a significant main effect for time F(2, 36) = 8.05, p < 0.001, partial η2 = 0.309 can still be reported. The undoubtedly effective factor of spontaneous improvement should statistically benefit both arms of therapy equally and is therefore neglected. Age, gender, the duration of the post-viral olfactory disorder and the total duration of therapy did not show any influence on the change in odor (ΔTDI, ΔT, ΔD, ΔI, ΔR). The subjective improvement in retronasal olfactory ability also did not differ significantly according to the treatment arm. We recognize that despite robust statistical analysis, their performance is not sufficient to rule out these factors entirely. These results should therefore not be overestimated, but should be examined more closely with a larger number of cases. However, the better the initial baseline value of olfaction at screening visit (TDI score), the clearer the improvement in smell up to the first follow-up. Side effects of the therapy were not reported in any case, neither due to olfactory training nor as a result of topical steroid application. Development of mean TDI Scores: Development of mean TDI Scores over mean 3 months in dysosmic patients due to CoViD-19 under olfactory training (OT) with versus without a concomitant topical nasal corticosteroid therapy (TNC).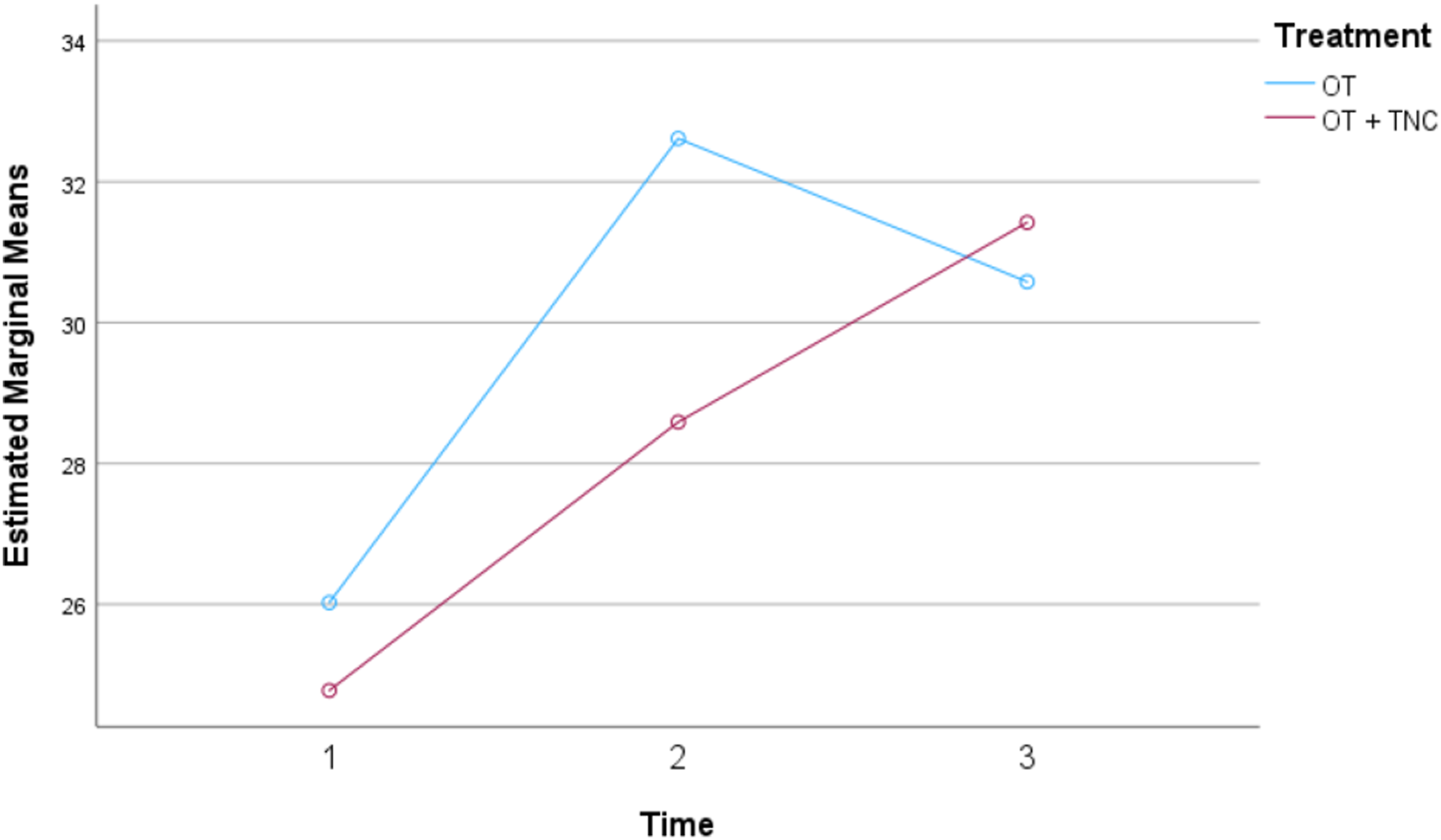
Discussion
The treatment of olfactory disorders after a viral infection such as SARS-CoViD 2 is a challenge and a scientifically relevant topic. Although the majority of patients recover completely after a few weeks, a small proportion displays persistent disturbing symptoms such as hyposmia and parosmia.4,6,7,9,10,13,27 On the global scale of a pandemic, however, even a small proportion corresponds to a large number of people affected, each of whom is significantly impaired in their quality of life.4,28 There is no denying that the therapeutic options are not yet satisfactory. 29 The knowledge gained recently could be groundbreaking and serve as a precedent for many other post-viral olfactory disorders that are far less in focus. The crucial thing is that the cause and the temporal connection of a smell and taste disorder can rarely be assigned as precisely as after a proven corona infection. Based on the time interval of enrollment in this study, it is highly probable that the infections were exclusively caused by a non-omicron virus variant. This is certainly one of the strengths of this study. During the infections, recruitment, screening and follow-up examinations of all subjects, the delta variant of the coronavirus SARS-CoV-2 dominated. 30 Although the sense of smell improved significantly in the majority of cases in this comparative study, we expected the effects of the two therapeutic approaches to be more markedly different. The initial advantage with sole olfactory training could be explained by a possibly higher compliance at the beginning of the sole training due to the lack of further drug treatment offers. When the training intensity decreases, the recovery of the sense of smell stagnates. This follow-up observation was reflected in all models, which we consider crucial for our study. Adjunctive therapy ultimately outweighs this short-term benefit. It is also known that compliance is already insufficient with the regular use of pharmaceuticals, and it is even lower with non-drug therapy recommendations such as smell training. Finally, the insufficient distinction in diagnosis between anosmias and parosmias needs to improve.31,32 The undoubtedly different olfactory disorders should be assessed by extended test methods.
Conclusion
In any case, this study reinforces the recommendation of consistent olfactory training starting as early as possible for patients with dysosmia due to COVID-19. This has proven to be a therapeutically and economically favorable treatment with no side effects for any type of post-infectious olfactory disorder. A therapeutic advantage of a topical nasal corticosteroid was not shown, but cannot be ruled out either. In future investigations with larger cohorts, treatment efficacy could be augmented by established inhalative device-based applicators, which would also improve assessment and compliance. Likewise, smell training could be intensified, monitored and thus standardized using suitable aids. After all, it is long overdue to find a way to better differentiate between hyposmias and parosmias diagnostically. For this purpose, an extended procedure with new subjective and objective olfactometric and imaging tools is being developed.
Footnotes
Author contributions
Conceptualization: Carla Azar, Friedemann Schmidt, Önder Göktas
Formal analysis: Friedemann Schmidt, Daniel Schulze
Funding acquisition: Carla Azar, Önder Göktas
Investigation: Carla Azar, Friedemann Schmidt, Önder Göktas
Writing ± original draft: Friedemann Schmidt
Writing ± review & editing: Friedemann Schmidt, Carla Azar, Önder Göktas
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting anonymized individual cases or case series, especially in the context of this comparative observational study. Further medical examinations required in individual cases outside of our institution are in accordance with the ethics vote no. EA1/112/120 (Ethic Committee Charité Berlin, June 04, 2020)
Statement of human and animal rights
This observational study was performed in accordance with the Declaration of Helsinki on Biomedical Studies Involving Human Subjects.
Statement of informed consent
Verbal informed consent was obtained from a legally authorized representatives for anonymized patient information to be published in this article.
