Abstract
Background:
The prevalence of smell dysfunction, along with its risk factors, has been evaluated in previous literature; however, little has been established regarding its relation to other factors such as disease severity, history of coronavirus disease-2019 (COVID-19) vaccinations, and medical treatment for COVID-19 infection. These factors may impact the overall recovery of olfaction in COVID-19 patients.
Objective:
This study investigated the prevalence of early olfactory recovery from COVID-19 infection and its associated factors.
Methods:
This study was a prospective cohort study on 348 COVID-19 patients with a new onset of anosmia or hyposmia. Smell sensation scores on the olfactory self-assessment test were collected at baseline and every week until 6 weeks of follow-up. The primary outcome was the prevalence of early olfactory recovery. The secondary outcomes were analyzing factors associated with early olfactory recovery and the median time to complete recovery of smell loss.
Results:
The prevalence of early olfactory recovery was 63.1% (95% CI: 0.58-0.68), and significant associated factors in multivariate analysis included patients without allergic rhinitis (OR 2.22, 95% CI: 1.12-4.40, P = 0.023) and no alteration of taste perception (OR 1.62, 95% CI: 1.02-2.57, P = 0.042). The median time to complete recovery from smell loss was 2 weeks.
Conclusion:
The present study found that the prevalence of early olfactory recovery within 2 weeks was 63.1%. The median time to olfactory recovery was also 2 weeks. Patients without allergic rhinitis and taste alteration had a significantly better chance of early recovery of olfactory function.
Keywords
Introduction
The coronavirus disease-2019 (COVID-19) primarily causes upper respiratory tract symptoms, with other manifestations potentially affecting the pulmonary, gastrointestinal, and neurological systems. Existing evidence has highlighted a significant association between COVID-19 infection and the development of new smell and taste disorders. 1 These symptoms have become a prominent warning sign for early detection of COVID-19 infection, particularly during the initial stages of the pandemic. Previous studies have reported that the prevalence of olfactory dysfunction in COVID-19 patients ranges from 48% to 74%.2-7 Moreover, olfactory dysfunction appears more prevalent and severe in Delta variant COVID-19 than in other strains. 8 A recent study indicated that around 7% of patients remained anosmic following COVID-19 infection. 9 For individuals whose occupation relies heavily on their sense of smell, such as professional sniffers, olfactory dysfunction can significantly disrupt daily life and occupational function. Furthermore, permanent olfactory loss may be associated with depression, impaired cognition, and a higher risk of premature death. 10
The prevalence of smell dysfunction and its associated risk factors have been examined in previous literature.11-13 However, little has been established regarding its relationship with other factors, such as disease severity, history of COVID-19 vaccinations, and medical treatment for COVID-19 infection. These factors could potentially impact the overall recovery of olfaction in COVID-19 patients. Furthermore, providing patients with counseling regarding prognostic factors for symptom recovery is an essential aspect of the treatment plan and may help alleviate their concerns. Therefore, our study aims to investigate the prevalence of early olfactory recovery from COVID-19 infection and identify its associated factors.
Patients and Methods
This study was a prospective cohort study conducted at the isolation unit for COVID-19 patients, which falls under the service of Ramathibodi Hospital, Faculty of Medicine, Mahidol University. The study took place from August 2021 to December 2021. Ethical approval for the study (COA.MURA2021/593) was obtained from the ethical committee, and the research was conducted following the ethical standards outlined in the Declaration of Helsinki. Before enrolling in the study, all participants provided electronic signatures as informed consent.
The inclusion criteria for this study consisted of COVID-19 patients aged 18 years and above who experienced a new onset of anosmia or hyposmia, which was confirmed through reduced smell sensation scores on the olfactory self-assessment test. Additionally, the diagnosis of COVID-19 infection was confirmed using the reverse transcription-polymerase chain reaction technique. On the other hand, the exclusion criteria included patients with a history of smell loss before contracting COVID-19 and those with a history of rhinological diseases such as chronic rhinosinusitis, nasal tumor, or previous sinonasal surgery. Pregnant women were also excluded, along with patients who could not complete the study or refused to participate.
All participants who met the eligibility criteria received 7 disposable smell test kits and a quick response code to access an instructional video for odor testing. The odors used in the study were selected based on their proven cultural recognition among the local normosmia population, as indicated by a previous study. 14 The smell sensation of the 4 specific odors was assessed using a visual analog scale (VAS) score, which was recorded instantly at baseline and every week until the 6th-week follow-up. The grading scale for each odor ranged from 0 to 10, with 0 indicating no odor perception and 10 representing the best perception.
History-taking regarding olfactory dysfunction and concomitant symptoms, such as fever, upper respiratory tract infection (URI) symptoms, and taste alterations, was conducted on the first admission date. A clear explanation was provided to avoid confusion between taste perceptions (salty, sweet, bitter, and sour/acidic) and smell perceptions. Other data, including age, sex, weight, height, underlying diseases, current medications, smoking status, COVID-19 vaccination status, presence of COVID-19 pneumonia from chest radiographs, oxygen supplementation, the severity of COVID-19 infection classified as mild, moderate, or severe disease, and specific drugs administered for COVID-19 treatment, including antiviral drugs and systemic corticosteroids, were extracted from the medical records. The severity of the disease was classified based on the National Institutes of Health’s Coronavirus Disease (COVID-19) Treatment Guidelines. Mild illness was characterized by any signs and symptoms of COVID-19 but without dyspnea or abnormal chest imaging. Moderate illness referred to patients with evidence of lower respiratory disease but a normal pulse oximetry (SpO2) reading of ≥94%. Severe illness encompassed individuals with SpO2 <94%, lung infiltrates >50%, or respiratory failure. 15
The primary outcome of this study was the prevalence of early olfactory recovery, which was defined as the complete improvement of the VAS score for smell sensation across all 4 odors within the first 2 weeks of follow-up. The choice of a 2-week cutoff for early recovery from olfactory dysfunction was based on a previous study conducted by Lechien et al 16 The secondary outcomes included the analysis of factors associated with early olfactory recovery and determining the median time for complete recovery of smell loss. Complete recovery was evaluated by assessing the time participants reached the total smell score for all 4 specific odors.
The sample size was calculated using the one-proportion formula based on a referenced study by Lechien et al 16 The calculations utilized a type I error (α) of 0.05, a proportion (p) of 0.25, and a confidence interval (d) of 0.05. The authors determined that a sample size of at least 290 was most appropriate to detect statistically significant results. An additional 20% was factored into the calculation to account for potential data loss, resulting in a final sample size of approximately 348.
Baseline characteristics were presented using the mean, standard deviation, or median and interquartile range (IQR) for continuous variables, and percentages were used for categorical variables. The prevalence of early olfactory dysfunction was reported as a proportion (percentage). A logistic regression model was employed to analyze the factors associated with the early recovery of olfactory dysfunction, reporting both crude and adjusted odds ratios (OR) with 95% confidence intervals (CI). The median time for complete recovery from olfactory dysfunction was determined using survival analysis. The statistical analyses were performed using Stata version 17.0, (StataCorp, TX, USA) and the results were considered statistically significant when the P-value was less than .05.
Results
Baseline Characteristics
A total of 348 participants enrolled in the study, with 8 lost to follow-up and 1 died from COVID-19 respiratory failure; thus, 339 completed the outcome measurements during the follow-up periods. Of the 339 participants, 120 were females (35.4%), and the mean age was 39.9 ± 13.5 years. The baseline demographic data and the variables of interest have been reported in Table 1.
Baseline Characteristics for All Participants.
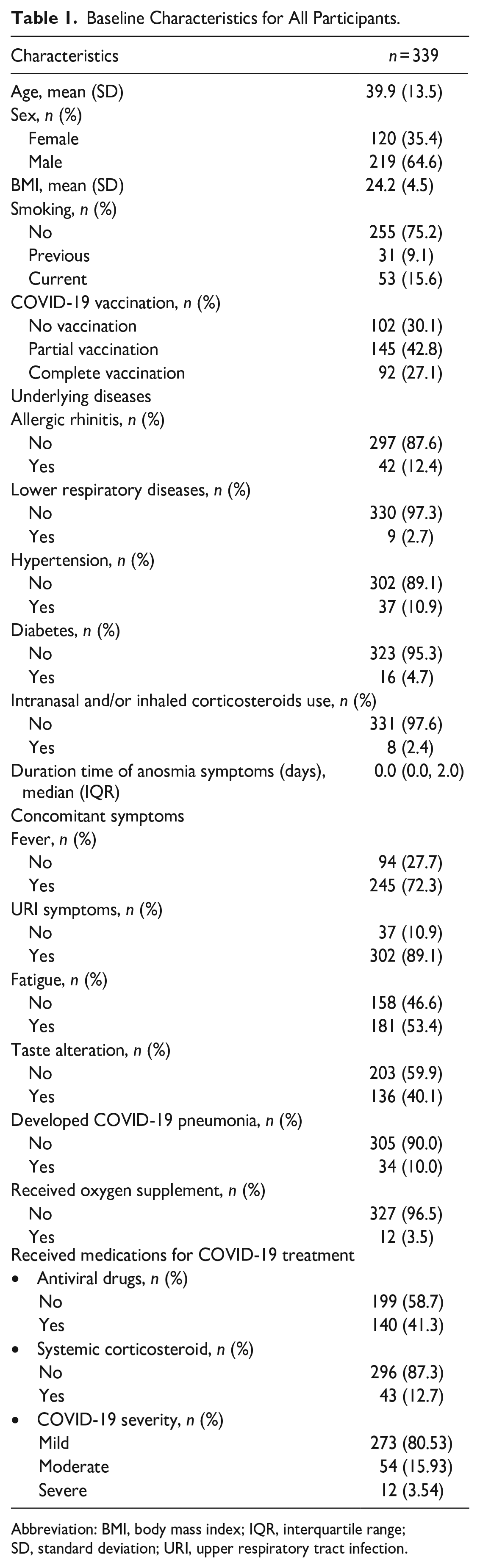
Abbreviation: BMI, body mass index; IQR, interquartile range; SD, standard deviation; URI, upper respiratory tract infection.
Primary Outcome
Of the 339 patients, 214 (63.1%, 95% CI: 0.58-0.68) experienced complete recovery from olfactory dysfunction within 2 weeks.
Secondary Outcome
The univariate analysis revealed significant associations with early olfactory recovery for patients with a higher body mass index (BMI), with an OR of 1.06 (95% CI: 1.01-1.12, P = .021), patients without allergic rhinitis, with an OR of 2.59 (95% CI: 1.34-4.99, P = .005), patients without clinical presentation of URI symptoms, with an OR of 2.29 (95% CI: 1.01-5.19, P = .046), patients without alteration of taste perception, with an OR of 1.68 (95% CI: 1.07-2.63, P = .024), and patients who developed COVID-19 pneumonia, with an OR of 0.41 (95% CI: 0.17-0.97, P = .043) (Table 2).
Univariate Analysis of Variables Associated With Early Olfactory Recovery Using a Logistic Regression Model.
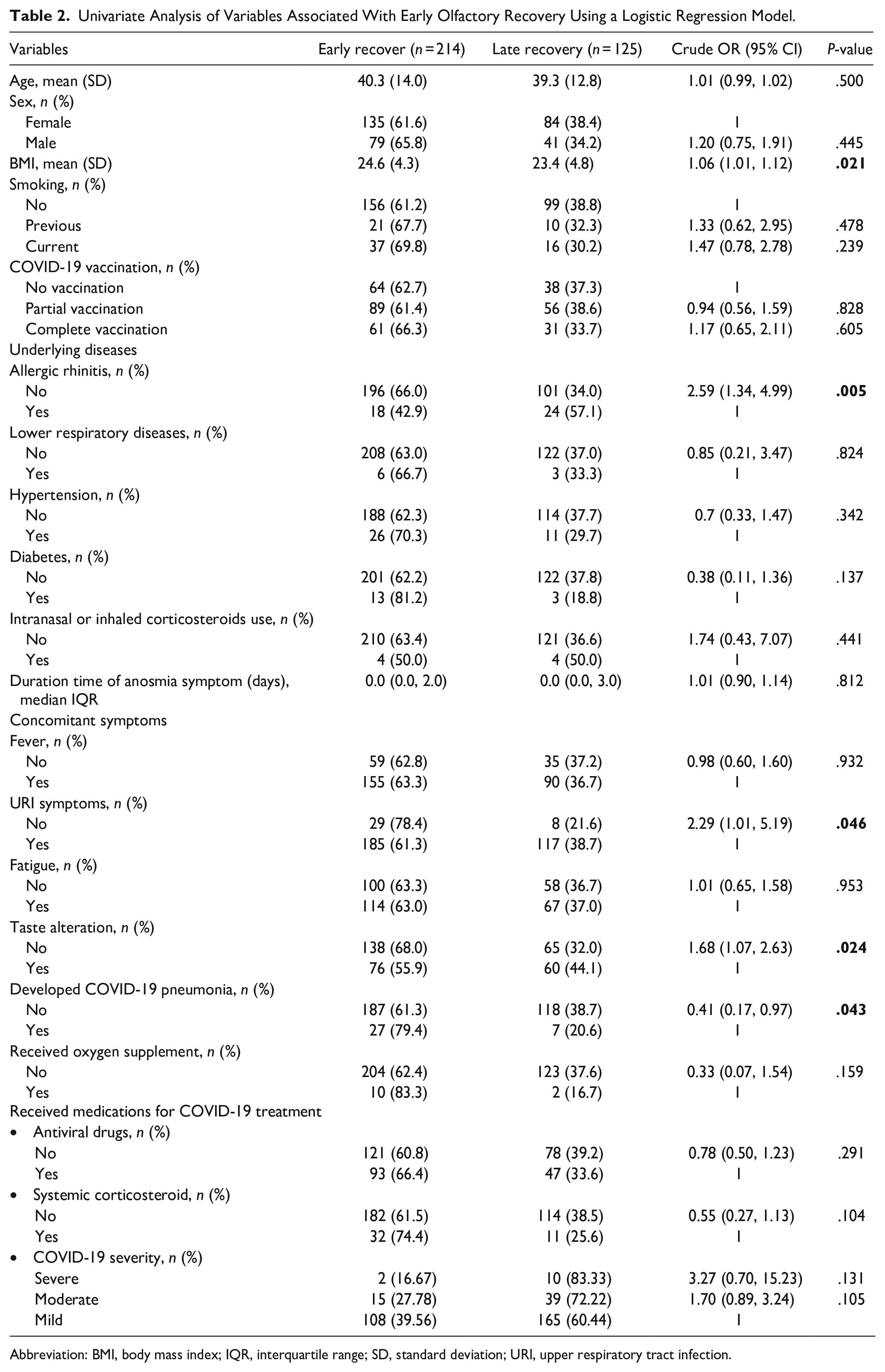
Abbreviation: BMI, body mass index; IQR, interquartile range; SD, standard deviation; URI, upper respiratory tract infection.
In the multivariate logistic regression model analyzing the significant variables, factors associated with early recovery of olfactory dysfunction were observed for patients without allergic rhinitis, with an OR of 2.19 (95% CI: 1.11-4.33, P = .024), and patients without alteration of taste perception, with an OR of 1.61 (95% CI: 1.01-2.55, P = .043) (Table 3).
Multivariate Analysis of Variables Associated With Early Olfactory Recovery Using a Logistic Regression Model.
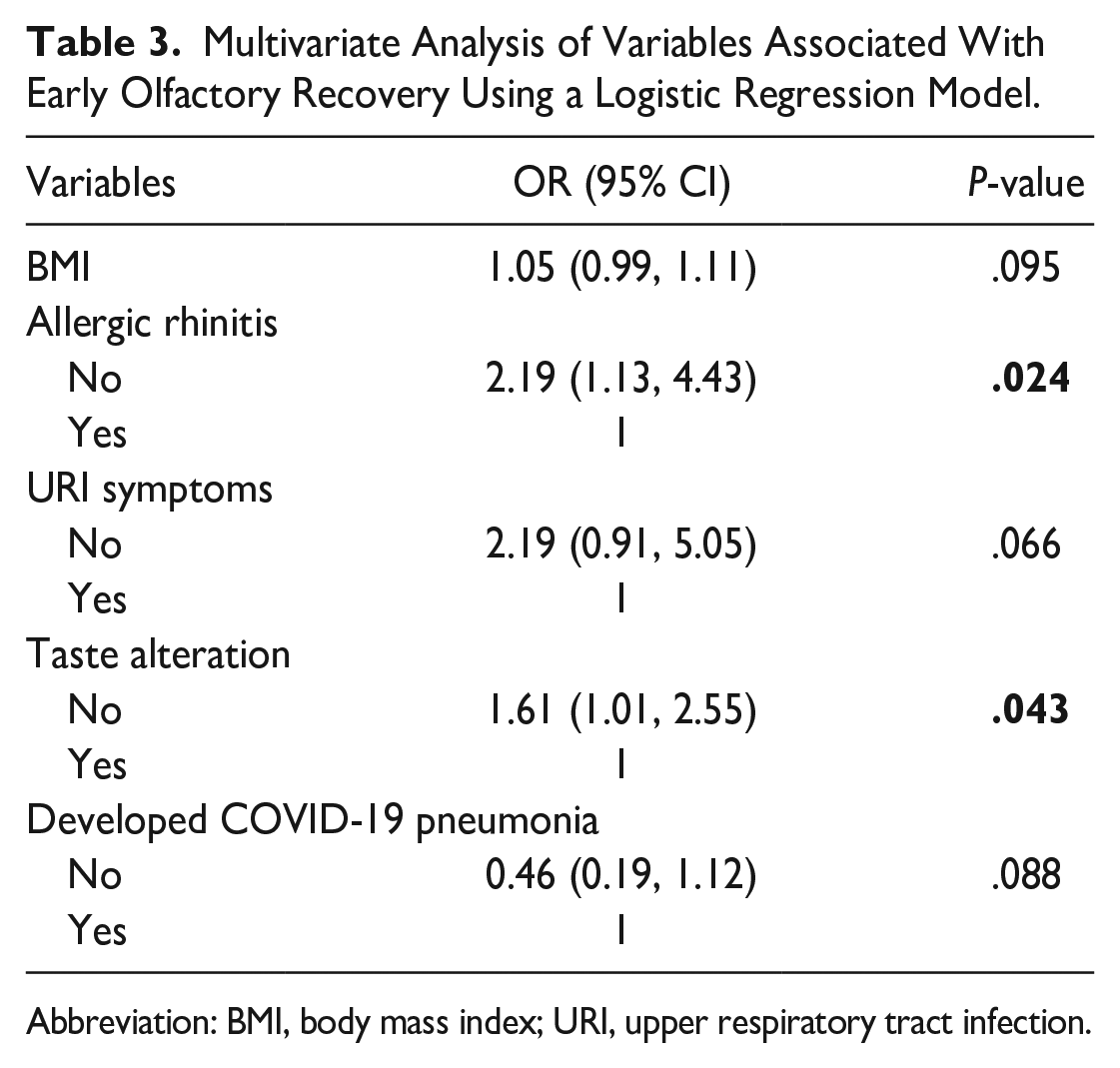
Abbreviation: BMI, body mass index; URI, upper respiratory tract infection.
Furthermore, after additional estimation from a multivariate logistic regression model adjusted for age, sex, and systemic corticosteroid treatment for COVID-19 pneumonia, both factors remained significant (Table 4).
Multivariate Analysis of Variables Associated With Early Olfactory Recovery Using a Logistic Regression Model, Adjusted for Age, Sex, and Systemic Corticosteroid Treatment for COVID-19 Pneumonia.
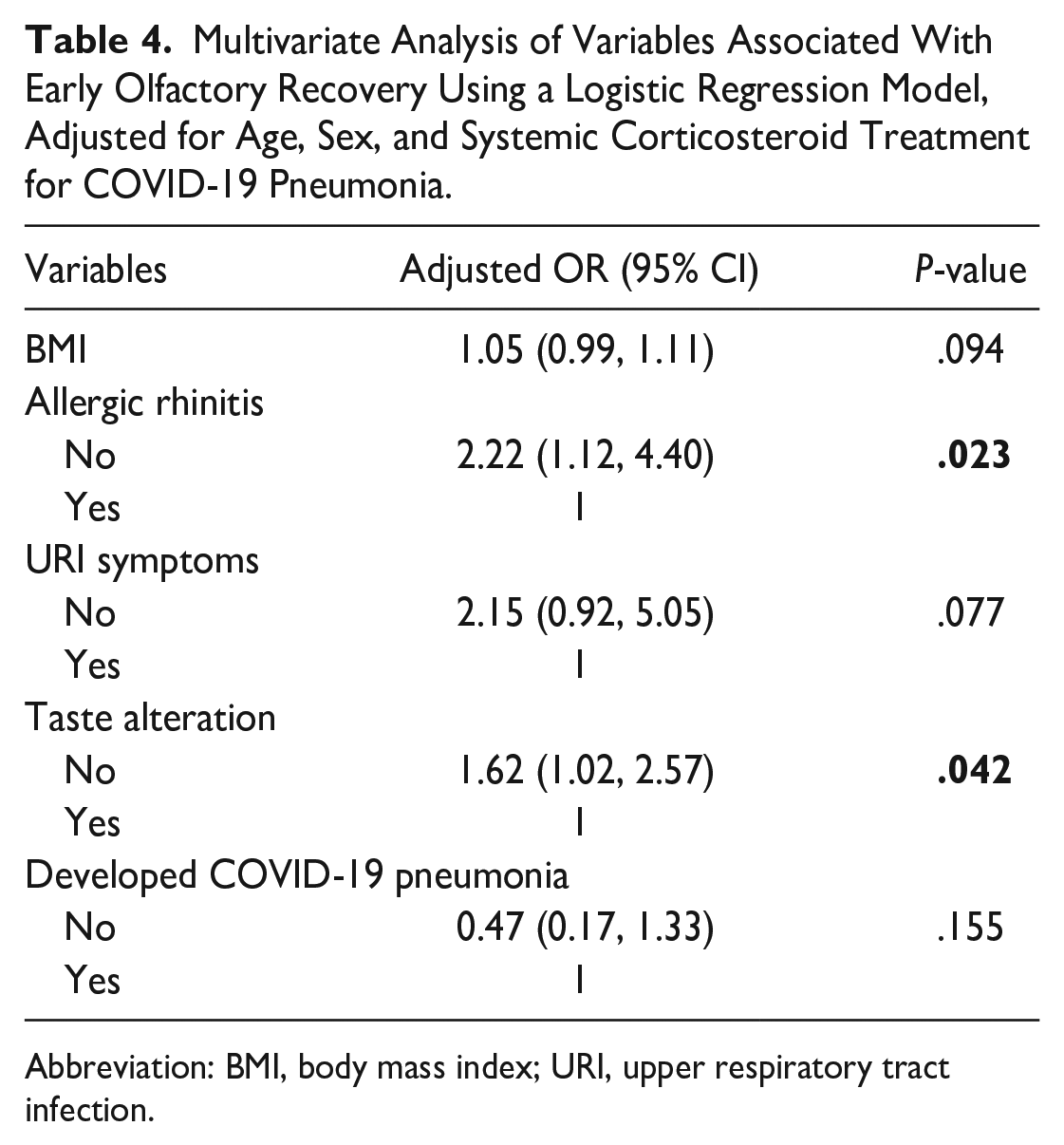
Abbreviation: BMI, body mass index; URI, upper respiratory tract infection.
The median time to recover from smell loss was 2 weeks, as depicted in Figure 1.
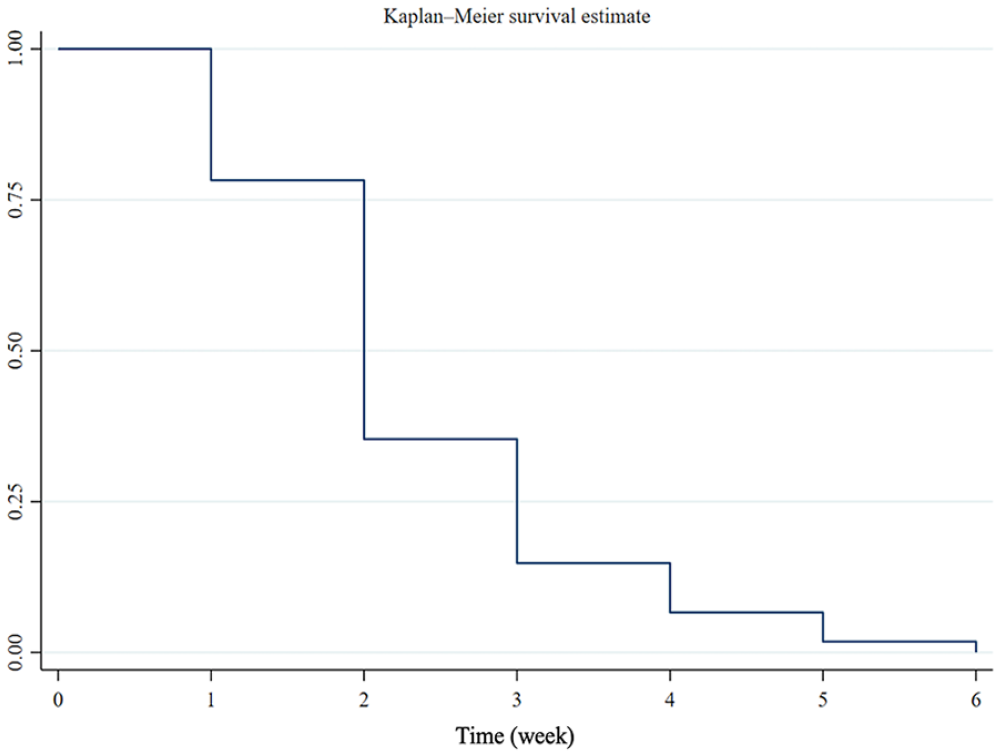
Kaplan–Meier curves demonstrated that the median time to complete recovery of smell loss in the study group was at 2 weeks.
Discussion
Olfactory dysfunction has been a hallmark of COVID-19 infection, particularly in the early stages of the pandemic. The prevalence of olfactory dysfunction in COVID-19 patients has been reported to be between 48% and 74% during the earlier period of the pandemic, with a decline in the numbers following the emergence of various new strains.2-7
The pathophysiology of olfactory dysfunction has been hypothesized to be linked to the SARS-CoV-2 virus invading cells through the Angiotensin-Converting Enzyme 2 receptor (ACE2), which combines with the transmembrane serine protease 2 (TMPRSS2) enzyme on sustentacular cells. 17 This virus invasion into the olfactory neurons can cause loss of smell due to inflammation of the peripheral and central olfactory systems. The natural course of olfactory loss is often self-limited, with symptoms improving over time, even without specific treatment.13,18 A previous systematic review reported recovery times ranging from less than 2 weeks to more than 6 months. 19 A recent study indicated that around 7% of patients remained anosmic following COVID-19 infection. 9 The mechanisms underlying persistent olfactory loss remain unclear, but a recent study proposed that patients with persistent olfactory loss had fewer olfactory sensory neurons and unresolved local immune cell alterations in their olfactory mucosa. 20 Lechien et al 13 proposed the duration of early recovery of smell loss, defined as the improvement of smell function within 2 weeks, and reported a prevalence of 25%, which differs from the findings of the present study, where the prevalence was 63.1%. This difference in proportions could be attributed to variations in ethnicity, virus variants, and the type of olfactory test used. The former study was conducted on Caucasians during the earliest variant outbreak and utilized an objective olfactory measurement.
During the early pandemic, when the Alpha, Beta, and Gamma variants of SARS-CoV-2 were predominant, several publications reported factors related to olfactory dysfunction. These factors included being female,2,21,22 young patients,3,21 Caucasian ethnicity, 21 having a mild severity of COVID-19 disease, 2 and being anosmic.3,22 The present study aimed to identify factors associated with early olfactory recovery during the outbreak of the Delta variant. In the univariate analysis, the authors found that several factors were linked to early olfactory recovery, including higher BMI, the absence of allergic rhinitis, normal taste perception, no URI symptoms, and the presence of COVID-19 pneumonia. However, in the multivariate analysis, only the absence of allergic rhinitis and normal taste perception was significantly associated with early olfactory recovery.
Previous randomized trials and reviews have recommended systemic corticosteroid therapy to improve clinical outcomes and reduce the mortality rate in potentially high-risk COVID-19 patients, such as those who are obese and those who have COVID-19 pneumonia.23-25 Current evidence also supports some benefits of systemic corticosteroids in restoring COVID-19 smell loss. 26 Our study observed that patients with a higher BMI and those who developed COVID-19 pneumonia experienced early olfactory recovery, possibly due to receiving systemic corticosteroids to treat COVID-19 pneumonia.
Patients without allergic rhinitis showed a significant association with early olfactory recovery, consistent with a previous study where patients with allergic rhinitis had a significantly higher rate of olfactory dysfunction. 11 Allergic patients may be more susceptible to viral infections due to deficient innate type 1 and 3 interferons and epithelial barrier dysfunction. Viral infections, including SARS-CoV-2, can also stimulate a type 2 immune response, exacerbating allergy symptoms. 27
Our study also observed less favorable olfactory recovery in the early period in patients with predominant URI symptoms. We speculate that the nasal blockage caused by swollen mucosa and nasal discharge may create conductive barriers that prevent odors from reaching the receptors. Another contributing factor could be the elevation of inflammatory cytokines in the nasal mucosa, which may affect the function of olfactory neurons. 28
The taste sensation is innervated by cranial nerves VII, IX, and X, and it is recognized as sweet, bitter, salty, sour, and umami. A previous study found a positive correlation between the amount of SARS-CoV-2 virus in saliva and the severity of taste alteration. 29 The hypothesized pathophysiology for taste abnormalities in COVID-19 is possibly due to cranial nerve impairment or direct attack by the virus on taste buds, buccal mucosa, and salivary glands. 30 The loss of various special sensations may indicate the involvement of multiple organs by the virus, which could potentially delay recovery. Our present study supports the evidence that the absence of taste alteration significantly and positively predicted early olfactory recovery in COVID-19 patients.
The study has several limitations that should be acknowledged. First, the population was restricted to mild-moderate severity COVID-19 patients, and the study was conducted during the outbreak of the Delta strain. As a result, the generalizability of the study results may be limited to this specific population and time frame, potentially impacting the external validity of the findings. Second, the lack of availability of standardized psychophysical olfactory tests and taste sensation assessments in our country posed challenges to the reliability of olfactory and taste assessments in this study. The absence of these standardized measures may affect the accuracy and consistency of the results. It is essential to consider that when utilizing standardized psychophysical olfactory tests and taste sensation assessments in future studies, the outcomes might not necessarily align with the findings reported in this study.
Conclusion
Olfactory dysfunction is a frequently encountered symptom in COVID-19 patients, especially in the early stages of the pandemic. Fortunately, the natural course of this symptom tends to lead to favorable outcomes with recovery over time. Our study observed that 63.1% of patients experienced early olfactory recovery within 2 weeks. Furthermore, we found that patients without allergic rhinitis and taste alteration had a significantly higher likelihood of early recovery of olfactory function. These factors appear crucial in determining the speed of olfactory function restoration in COVID-19 patients.
Footnotes
Acknowledgements
The authors thank Patima Noonoi, registered nurse at Ramathibodi Hospital, Faculty of Medicine, Mahidol University, for assistance with the data collection and Sukanya Siriyotha, biostatistician at Department of Clinical Epidemiology and Biostatistics, Ramathibodi Hospital, Faculty of Medicine, Mahidol University, for assistance with statistical analysis in this study.
Data Availability Statement
The data generated and analyzed during the study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a research grant from Faculty of Medicine, Ramathibodi Hospital, Mahidol University, a not-for-profit sector (RF_65010).
Ethical Approval
Ethical approval to report this case was obtained from Human Research Ethics Committee, Faculty of Medicine Ramathibodi Hospital, Mahidol University (COA.MURA2021/593) and was conducted following the ethical standards of the Declaration of Helsinki
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Human Research Ethics Committee, Faculty of Medicine Ramathibodi Hospital, Mahidol University approved protocols.
Statement of Informed Consent
Electronically signed informed consent was obtained from the patients for their anonymized information to be published in this article.
