Abstract
Nocardia farcinica usually infects people with impaired immune status and usually manifests in the lungs. Otomastoiditis caused by Nocardia infection is extremely rare, with only 4 cases reported to date. This report describes a case of otomastoid N. farcinica infection in an immunocompetent patient. The case was a 10-month-old immunocompetent infant who presented with an approximately 3-month history of right ear discharge for which treatment with various antibiotics had not resulted in significant improvement. Multiple cultures of secretions and pathologic examination failed to identify the causative organism. The patient then underwent right mastoidectomy. Finally, metagenomic next-generation sequencing identified the pathogen to be N. farcinica. The patient was infection-free at the 6-month follow-up but had developed labyrinthitis ossificans. Otomastoid Nocardia infection has characteristic clinical features, namely, formation of a large amount of granulation tissue and coexistence of bone destruction and new bone formation. Traditionally, Nocardia is challenging to diagnose. Metagenomic next-generation sequencing of lesions is helpful. Complete local debridement and free drainage are key to treatment.
Introduction
Otitis media is a common childhood infection worldwide. Acute otitis media is clinically characterized by ear pain, discharge, tympanic perforation, and low-grade fever.
1
Acute suppurative otomastoiditis is mainly associated with bacterial infections in children. The main pathogens are Streptococcus pneumoniae and Hemophilus influenzae.
2
However, autoimmune disease or infection with a less common causative pathogen should be considered when long-term antibiotic therapy is ineffective. In this report, we describe the clinical course of a patient with no evidence of immunodeficiency who developed otomastoiditis caused by Nocardia farcinica, which was eventually detected by Metagenomic next-generation sequencing (Figure 1). Treatment timeline of the patient.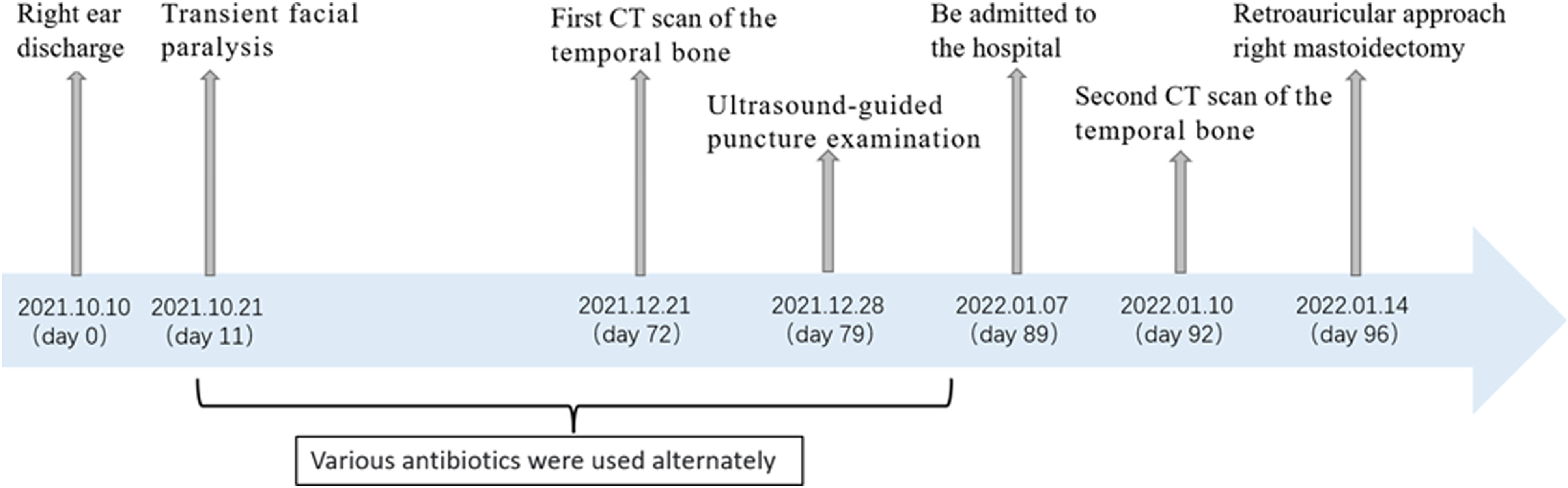
Case report
The patient was a 10-month-old Chinese infant who first developed a right ear discharge (day 0) and was subsequently noted to have transient facial paralysis (day 11). Various antibiotics were administered without significant improvement. Secretions from the right ear were cultured several times but grew no specific flora. The patient then presented to Shandong Provincial Hospital Affiliated to Shandong First Medical University (day 89) with a 3-month history of right ear discharge.
On examination, his nutrition status was average and his temperature was normal. Ultrasound-guided puncture examination identified an inflamed retroauricular abscess. Examinations of the chest, cardiovascular system, and central nervous system were normal. There were no hematologic abnormalities except for microcytic hypochromic anemia. Tests for the ANCA antibody spectrum, Mycobacterium tuberculosis, and human immunodeficiency virus were negative.
Endoscopic examination revealed a large amount of granulation tissue and pulsatile mucopurulent discharge in the deep portion of the right external auditory canal (Figure 2A). The left tympanic membrane and external auditory canal were normal. Audiologic examination showed an AC-ABR threshold of the right ear was 85 dB, and the ASSR threshold was 95-70-70-85 dB (0.5-1-2-4 kHz). (A) Endoscopic findings at the right tympanic membrane on January 10, 2022 (before surgery). (B) Computed tomography scan of the temporal bone obtained on December 20, 2021. (C) Computed tomography scan of the temporal bone acquired on January 10, 2022. (D) Fat-suppressed T1-weighted magnetic resonance image of the internal auditory canal. (E) Fat-suppressed T2-weighted magnetic resonance image of the internal auditory canal. (F) A large amount of granulation tissue mixed with a small amount of mucus and broken bone fragments in the mastoid cavity. (G) Part of the malleus handle (yellow arrow) combined with a large amount of granulation tissue in the external auditory canal. (H) The long foot of the anvil was damaged. (I) Microscopy demonstrated necrotic bone tissue (yellow arrow), multinucleated giant cells (black arrow), and the formation of micro-abscesses (green arrow), indicating a purulent lesion (hematoxylin and eosin staining, 100×). (J) Follow-up computed tomography scan obtained 6 months after surgery. (K) Endoscopic findings at the right tympanic membrane on June 20, 2022 (after surgery).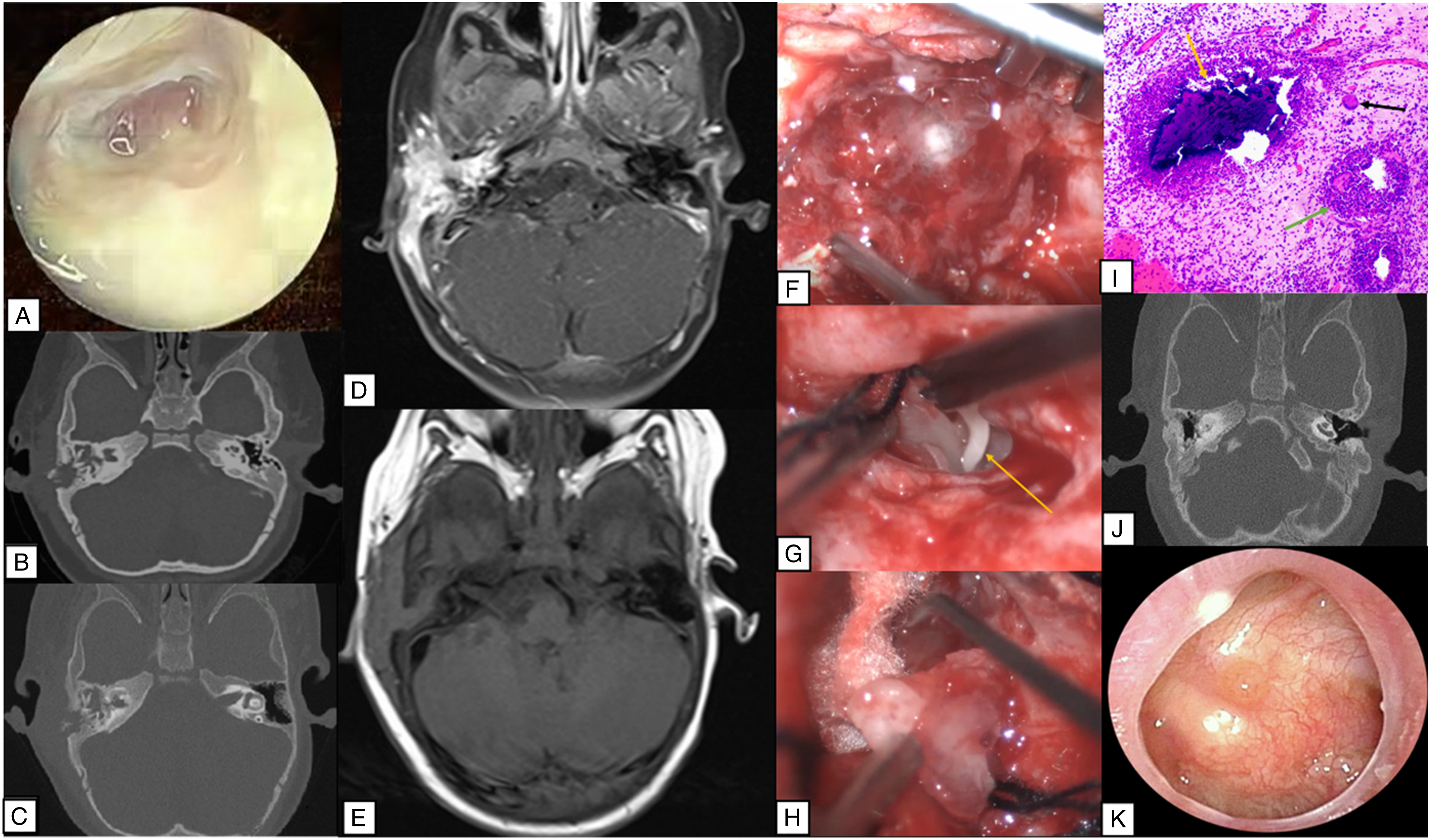
Computed tomography (CT) scans of the temporal bone obtained on day 72 (Figure 2B) and day 92 (Figure 2C) indicated worsening bone destruction with bone resorption at the base of the right cochlea and formation of punctate new bone. Fat-suppressed magnetic resonance images of the internal auditory canal showed hyperintense T1 signals and isointense T2 signals in the right mastoid process and adjacent soft tissue (Figure 2D and E). The imaging findings indicated gradual progression of the lesions. Right mastoidectomy was performed to remove the lesion (day 96), prevent disease progression, and secure a precise diagnosis.
A retroauricular transmastoid approach was used. Intraoperative examination revealed a large amount of granulation tissue containing a small amount of pus and bone fragments in the mastoid air chamber (Figure 2F). Full exposure revealed damage to the chorda tympani and part of the handle of the malleus combined with a large amount of granulation tissue in the external auditory canal (Figure 2G). The long foot of the anvil was also damaged (Figure 2H) but the stapes was intact. We removed all the damaged ossicles and granulation tissue to facilitate drainage. Artificial ossicle reconstruction was not performed, considering the patient’s infected status and young age. An intraoperative frozen section recommended consideration of inflammatory lesions. A sample of pus from the mastoid sent for culture showed no growth after 48 h of aerobic incubation. Pathologic examination revealed infiltration of a large number of plasma cells, lymphocytes, and histiocytes into the blood vessels with micro-abscesses and a multinucleated giant cell reaction (Figure 2I). Histopathologic analysis showed the following: CD38(portion+), CD20(portion+), CD68(portion+), CD163(portion+), Langerin(-), S-100(-), CD1a(-), CMV(-), and PAS(-). Considering that none of the previous investigations had identified a causative pathogen, we sent granulation tissue samples for metagenomic next-generation sequencing, which revealed the culprit microorganism to be N. farcinica (reported by Guangzhou Kingmed Center for Clinical Laboratory).
At follow-up 6 months later, CT of the temporal bone confirmed that there was no infection but identified labyrinthitis ossificans (Figure 2J). On reexamination of the right ear, the BC-ABR threshold was 40 dB, the AC-ABR threshold was 96 dB, and the ASSR threshold was 100-90-90-100 dB (0.5-1-2-4 kHz). Endoscopic examination revealed a large central perforation in the tense part of the right tympanic membrane, no malleus stalk and incus, and a dry tympanic cavity with no signs of infection (Figure 2K).
Discussion
Nocardia is an aerobic, gram-positive, opportunistic pathogen and a slow-growing actinomycete. 3 About 40% of the reported cases of Nocardiosis have involved the lungs. 4 Otomastoid Nocardia infection is exceedingly rare, with only 4 reported cases. We have encountered such a patient, whom we treated by mastoidectomy.
Our patient had the common clinical manifestations of otomastoid infection but also had bone destruction and new bone formation, likely reflecting formation of new bone to replace the tissue destroyed by Nocardia.
It has been suggested that Nocardiosis is an opportunistic infection 5 and that only immunodeficient patients are susceptible. Common risk factors are human immunodeficiency virus, neoplastic disease, diabetes mellitus, systemic corticosteroids, and immunosuppressive agents. 6 However, our patient did not have a significantly impaired immune system or any other known risk factors. Three of the four previously reported cases of otomastoiditis caused by Nocardia infection occurred in immunocompromised patients. However, among the 1,000 cases of Nocardia infection published between 1950 and 1991, about 38% had neither impaired immune status nor risk factors. 7 These seemingly immunocompetent individuals might have had unrecognized primary or inherited immunodeficiency. 8 Our 10-month-old patient, despite not having an immunosuppressive disease, was in a state of immune development, which would have put him at risk for infection.
Traditionally, Nocardia is diagnosed based on a combination of pathology and bacterial culture. Although Nocardia has no particular selectivity for culture medium, its slow growth means that a culture time of 2-3 weeks is often required and may be affected by antibiotic therapy. 9 On Gram staining, Nocardia appears as noncontiguous gram-positive beads of varying sizes. 10 Owing to its unique morphologic characteristics, Nocardia infection can be misdiagnosed as M. tuberculosis infection. 11 A short culture time and the non-specific morphology in secretions can result in a delayed diagnosis. However, metagenomic next-generation sequencing can detect pathogens regardless of whether they are bacteria, viruses, or fungi, and is less affected by previous antibiotic therapy. 12 Moreover, unlike traditional methods, metagenomic next-generation sequencing can report antibiotic sensitivity and type a strain within 24-48 hours after the sample is received, which can significantly shorten the time to diagnosis and improve the prognosis. 12
Complete local debridement and appropriate antibiotic therapy are essential in the treatment of Nocardia infection. 13 Several antibiotics had been ineffective in our patient. Nocardia infection can result in formation of many granulomas deep within the mastoid that would be challenging to remove entirely by pharmacotherapy alone. Hence, drainage is the treatment of choice for otomastoiditis caused by Nocardia. Our patient was treated by mastoidectomy, and there was no recurrence of infection during 6 months of follow-up.
Nocardia infection should be considered in a patient who presents with otitis media unresponsive to empirical broad-spectrum antibiotic therapy, a large amount of granulation tissue, and coexistence of bone destruction and new bone formation. Misdiagnosis and inappropriate therapy may have serious consequences. This case also highlights the importance of both metagenomic next-generation sequencing in the diagnosis of Nocardia infection and the good outcome that can be achieved by mastoidectomy.
Footnotes
Contributorship
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Shandong Foundation Project of Natural Science (grant number ZR2020MH376).
Ethical approval
This case report was approved by the ethics committee of the Ophthalmic Hospital affiliated with Shandong Provincial Hospital Affiliated to Shandong First Medical University.
Informed consent
The patient’s parents were informed about the purposes of the report and signed a consent form. A copy of the written consent is available for review by the editor of this journal.
