Abstract
Objective
Burns and injuries can lead to massive defects in the mastoid tissues, which increase the difficulty of ear reconstruction. It is crucial to choose an appropriate surgical method for these patients. Here, we introduce strategies for auricular reconstruction in patients without satisfactory mastoid tissues.
Methods
From April 2020 to July 2021, 12 men and 4 women were admitted to our institution. Twelve patients were severely burned, 3 patients experienced car accidents, and 1 patient had a tumor on his ear. The temporoparietal fascia was used for ear reconstruction in 10 cases, and the upper arm flap was used in 6 cases. All of the ear frameworks were made of costal cartilage.
Results
The location, size, and shape of both sides of the auricles were generally the same. Two patients needed further surgical repair because of cartilage exposure at the helix. All of the patients were satisfied with the outcome of the reconstructed ear.
Conclusion
For patients with ear deformity and poor skin coverage in the mastoid area, we can choose the temporoparietal fascia if the patient’s available superficial temporal artery is longer than 10 cm. If not, we can choose the upper arm flap. The latter needs a five-stage operation, which is more time consuming and difficult than the former. Moreover, the expanded upper arm flap is thinner and has better elasticity than the temporoparietal fascia, so the shape of the reconstructed ear is better. We need to evaluate the condition of the affected tissue and choose the appropriate surgical method to achieve a good result.
Keywords
Background
Ear reconstruction is the best method for the treatment of congenital microtia and acquired auricle defects. It is also one of the most complex procedures in plastic surgery. There are 2 main procedures for ear reconstruction. The first is to sculpt a vivid and exquisite ear scaffold that is similar to the normal ear. The second is to choose a suitable flap to cover the ear scaffold, such as the mastoid fascia flap. There are many methods for ear reconstruction, including Brent’s method, 1 Nagata’s technique, 2 and the expanded postauricular flap method. 3 Regardless of the kind of method we choose, sufficient and qualified mastoid tissue is needed.
Ear defects can be caused by trauma, burns, and other factors. If the fascia and skin coverage is poor in the mastoid area, it will be difficult to cover the ear framework during ear reconstruction. 4 Some scholars use expanded mastoid fascia flaps that contain scars, 5 but the elasticity of flaps with scars is too poor to be expanded, so it is difficult to obtain a good effect. Therefore, some surgeons choose a temporoparietal fascia flap to cover the ear scaffold. 6 There is no consensus on the treatment of such patients with ear deformity and poor skin coverage at the mastoid area, so it is difficult to obtain a good outcome. In this study, we discuss auricle reconstruction for patients without satisfactory mastoid tissues.
Methods
Clinical Data
Demographic Features of the Patients.
Patients with an acquired auricle defect instead of congenital microtia, massive damage of the mastoid area, a deep second-degree or third-degree burn if he or she experienced a burn and who underwent total auricle reconstruction were included in the study. All patients provided written consent. This research was reviewed and approved by the Ethics Committee of the Plastic Surgery Hospital, Chinese Academy of Medical Sciences. The principles of the 1975 Declaration of Helsinki were followed.
Surgical Procedures
1. Preoperative evaluation
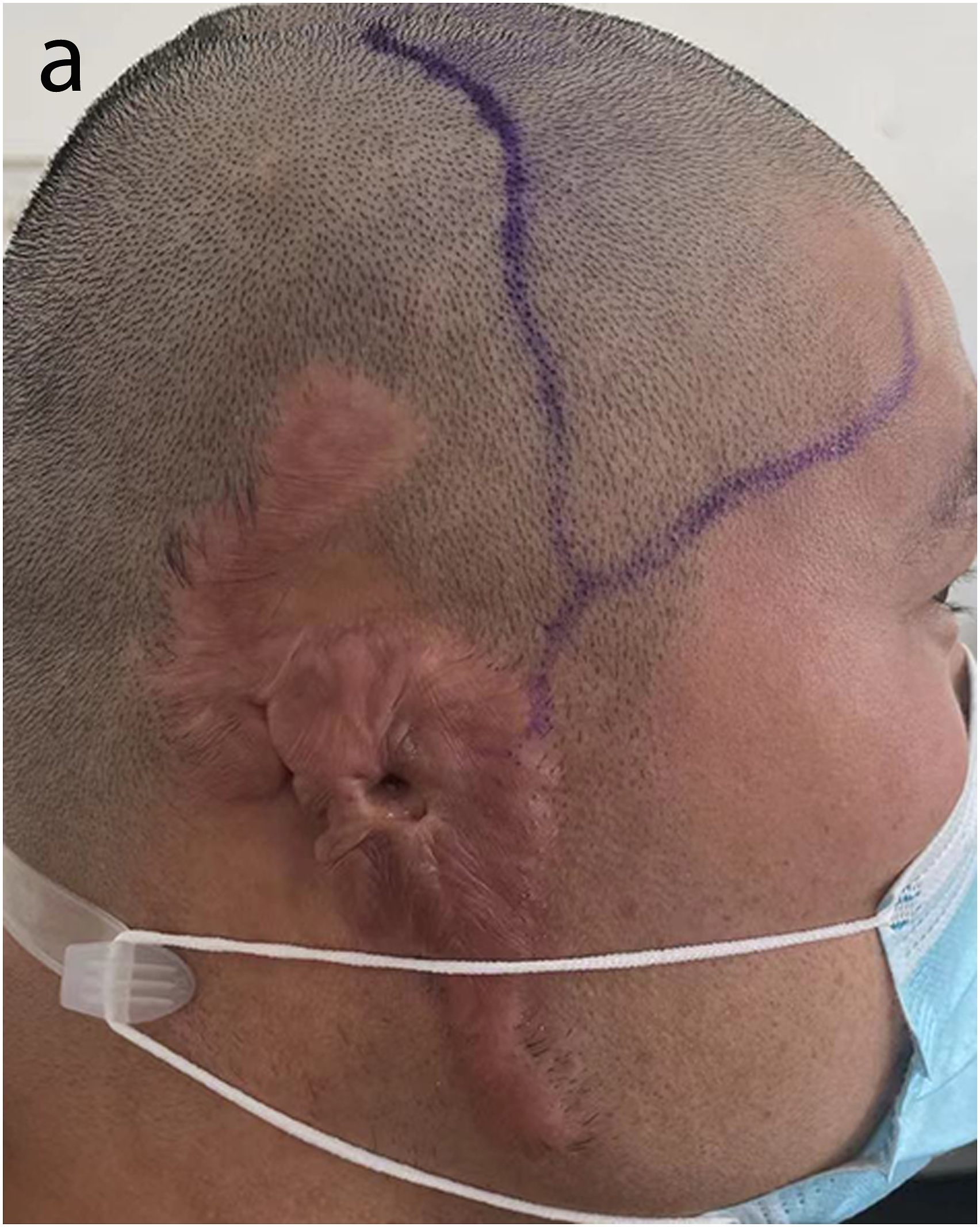
It is necessary to evaluate the quality of the patient’s superficial temporal artery before surgery. We used Doppler ultrasound (BC-520P+, Bestman, China) to detect the length of the superficial temporal artery (Figure 1). The patients were divided into 2 groups based on the length of the superficial temporal artery. If the length was more than 10 cm, the blood supply of the temporal-parietal fascia flap was rich, and we could use the flap for auricular reconstruction. If the length of the superficial temporal artery was less than 10 cm, we chose the upper arm flap. 2. Total auricle reconstruction with the temporal-parietal fascia flap Outline of the superficial temporal artery (a).
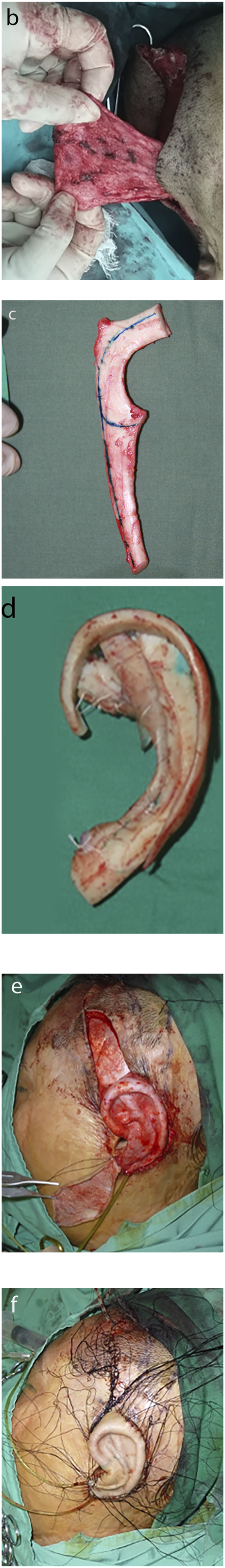
This method could complete total auricle reconstruction through a single-stage operation. We depicted the outline of the superficial temporal artery and made an “S”- or “C”-shaped incision. A 10 cm×10 cm fascial flap was separated and removed from the scalp and periosteum along with the superficial temporal artery so as to cover the whole ear framework. At the same time, the costal cartilage scaffold was sculptured. The 7th and part of the 6th costal cartilage (approximately 3–5 cm) were usually carved and spliced into a three-dimensional ear scaffold. The helix was usually formed by the outer edge of the seventh rib cartilage to avoid breaking the cartilage. The cartilage scaffold should have a fine morphology, such as a helix, crus, triangular fossa, and concha, to achieve a more realistic appearance. Cartilage strips were used to increase the thickness behind the framework to form the cranioauricular sulcus. Then, the scar and tissue of the residual ear were completely wiped off. The position of the reconstructed ear was according to the normal ear, and the ear framework was fixed to the periosteum. The framework was covered by the temporal-parietal fascia, and the fascia wound was covered by a split-thickness skin graft. The split-thickness skin was taken from the thigh, abdomen, or rib area. Finally, we placed a drainage tube that had negative pressure in the subcutaneous area and then bandaged the reconstructed ear (Figure 2). 3. Total auricle reconstruction with the upper arm flap The temporal parietal fascia flap (b). Ear framework carved out of the 6th and 7th costal cartilage (c, d). The temporal parietal fascia flap is used to cover the ear scaffold (e). The split-thickness skin covers the ear framework and the temporal parietal fascia flap (f).
There are 5 stages in this method. In the first stage, we placed a 150 mL kidney-shaped silicon expander into the subcutaneous layer at the outside of the upper arm. Ten days later, saline injections were administered to the patients 2–3 times a week. Approximately 4–5 weeks later, approximately 450–500 mL of saline was injected into the expander. We removed the expander and carried out skin tube formation for skin flap delay. Three weeks later, in the stage III operation, the proximal end of the tube was transferred to the auricle (Figure 3). Later, the blood circulation of the flap was trained until the blood supply was stable after being clamped for more than 1 hour. The 4th-stage operation was to perform pedicle amputation four weeks later, and we implanted an 80 mL kidney-shaped expander in the mastoid area. We started injecting saline into the expander on the 7th day after surgery, and the expanding period lasted for 6–10 weeks to reach a volume of 130 mL–160 mL. This condition was maintained for another 6–8 weeks to avoid potential retraction of the expanded flap. In the 5th stage operation, the ear framework was carved and put into the mastoid area where the expander existed. The ear scaffold was carved as mentioned above. The method for constructing an ear framework was the same as auricle reconstruction with the temporal-parietal fascia flap. A 150 ml expander in the subcutaneous layer at the upper arm (g, h). The expander was removed from the upper arm, and the skin tube in the left upper arm was formed as a delayed tube and transformed to ear (i).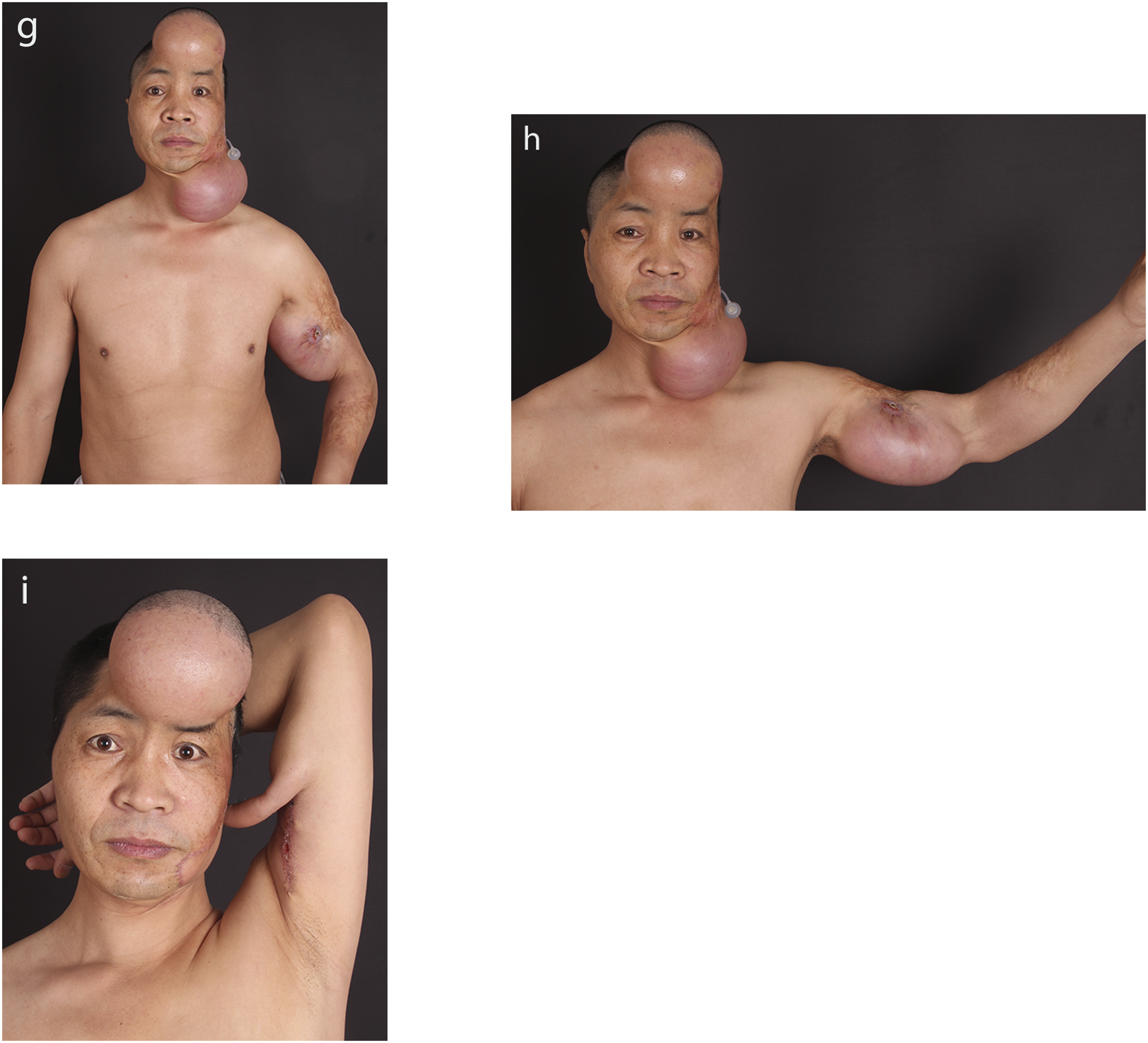
Results
All patients were followed up, and the median follow-up period was 7.5 months (range 6-18 months). The shape, color, texture, relative position, and structure of the reconstructed ear were satisfactory. One patient had cartilage exposure at the earlobe after the operation, which was repaired by covering the exposed cartilage with posterior auricular fascia. Two patients had poor earlobe morphology, which was corrected by earlobe repair. No other complications were found in this study. Nine patients (56.3%) considered the outcomes to be excellent, 4 patients (25%) considered the outcomes to be good, and 3 patients (18.7%) considered the outcomes fair.
Typical Cases
Patient 1
A 21-year-old female with flame-defection of her left ear was admitted to our hospital. The physical examination showed a complete defect of her left auricle and an atrophic scar in the mastoid region. The length of her superficial temporal artery is 13 cm, which is more than 10 cm. She was diagnosed with a burned deformity of the left auricle. Finally, she underwent total auricle reconstruction with a temporal-parietal fascia flap (Figure 4). Patient 1 (j, k, l). Preoperative appearance of a 21-year-old female with left ear deformity by a burn (j). The appearance after total auricle reconstruction with the temporal-parietal fascia flap (k, l).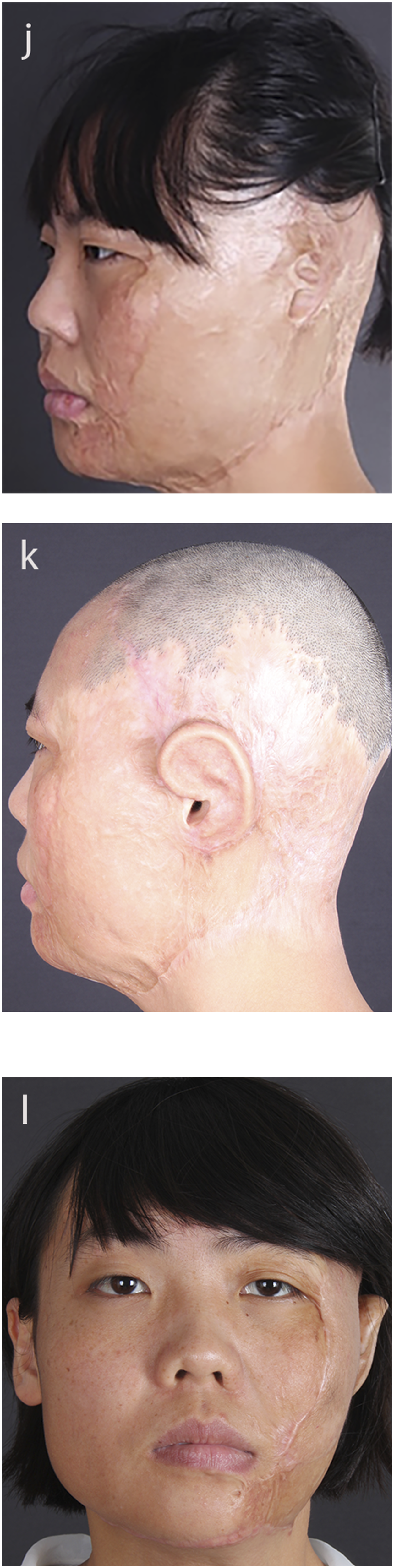
Patient 2
The patient is a 48-year-old male with a left-sided craniofacial postburn deformity. He had left-sided baldness and gross scars in the mastoid area. His left auricle was utterly damaged, and the skin around the ear was cicatricial contracture. The right ear was normal. He was diagnosed with a deformity of the left total auricle defect, which was classified as a grade IV ear defect, so we performed total auricle reconstruction with an upper arm flap (Figure 5). Patient 2 (m, n, o). Preoperative lateral view of a 48-year-old male with left ear deformity by a burn (m). The postoperative follow-up results showed that the outline of the outer ear was good (o).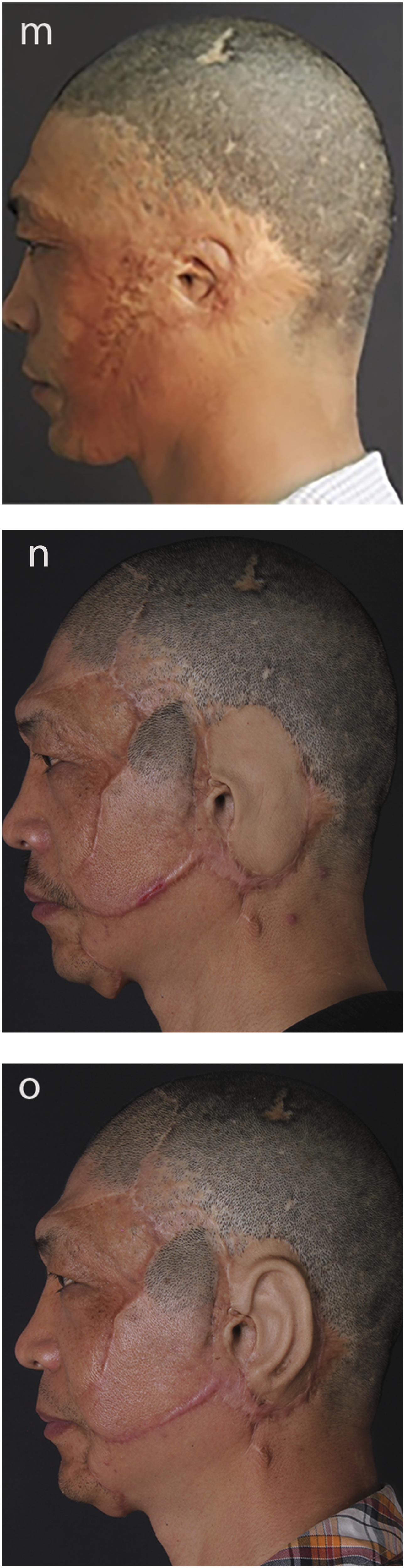
Discussion
Adequate mastoid skin is crucial for successful total auricular reconstruction. In previous studies, researchers have reported that mastoid skin is needed regardless of the type of total auricle reconstruction.7-9 However, some acquired auricle defects can lead to massive skin damage and poor skin coverage in the mastoid region. Such issues increase the difficulty of total ear reconstruction. 10 In conventional plastic surgery, skin insufficiency is usually resolved in 2 ways: skin grafting 11 and skin expansion. 12 The most important objective of skin transplantation is maintaining blood supply in the recipient area. 13 Therefore, according to the length of the superficial temporal artery, we classified the patients into 2 categories. A superficial temporal artery that is longer than 10 cm, and a temporoparietal fascial flap that can completely cover the ear scaffold. Therefore, we chose the temporal-parietal fascia flap to perform ear reconstruction. It is a single-stage reconstruction that is less time consuming and less complex. For patients with a superficial temporal artery that is less than 10 cm or a temporoparietal fascial flap is not enough to cover the ear framework, an expanded upper arm flap is chosen to cover the ear scaffold. It requires a five-stage operation, which is more time consuming and difficult than total auricular reconstruction with a temporal-parietal fascia flap.
The temporoparietal fascia flap is thin, and the blood supply is rich.14,15 It is the primary choice for patients with extensive mastoid region lesions. This method is relatively simple, less time consuming, and more acceptable for patients. However, this method depends on the blood supply of the temporoparietal fascia. 16 Therefore, it is essential to evaluate the length of the superficial temporal artery. 17 We can use Doppler ultrasound to measure its length and judge the blood supply during surgery. 18 We suggest that if the length of the superficial temporal artery is more than 10 cm and the blood supply is good, a temporal-parietal fascia flap should be used in total auricular reconstruction. To fully cover the auricle scaffold, the temporal fascia needs to reach a range of 10 cm×10 cm.
If the temporoparietal fascia is also damaged or the length of the superficial temporal artery is less than 10 cm, the temporal-parietal fascia flap cannot fully cover the auricle scaffold. An upper arm flap should be chosen for ear reconstruction. The upper arm flap also has a rich blood supply and is well anastomosed, concealed, soft, smooth, hairless, and has good elasticity. It has been widely applied for facial restoration. Therefore, it is also an ideal donor site for ear reconstruction. 19 We expanded it on the upper medial arm and transferred the flap from the medial arm to the mastoid region via a skin tube. At the same time, we suggest that the upper arm flap should be expanded to a suitable thickness. Compared with the temporoparietal fascial flap, the expanded upper arm flap is thinner and has better elasticity, so the shape of the ear scaffold can be better revealed. In addition, the donor site of the upper arm is hidden and has few scars. However, the disadvantage of this method is that the surgical procedure is time consuming and more complex than ear reconstruction using the temporal-parietal fascia flap. 20
For patients who need to improve their hearing, we implanted bone conduction devices for them. This procedure should be performed after the ear reconstruction. And it provided a good solution for patients with hearing loss. This study also requires carving of the rib cartilage. Most of the patients with traumatic auricle defects were adults. The costal cartilage is more difficult to carve in adults because it is harder and less elastic. 21 If we use the eighth rib for carving the ear scaffold, it is prone to break or be too sharp at the edge of the scaffold, and the shape of the auricle will be poor. 22 Therefore, we chose the seventh rib and used its outer edge as the helix. This method could make full use of the natural curvature of the seventh rib and prevent the ear bracket from breaking. At the same time, we chose the widest part of the sixth rib cartilage together with the seventh rib to make the triangular fossa, which was the base of the auricle. The remaining rib cartilage was carved into a “C” shape and spliced under the base of the ear to form the cranioauricular angle. This method can obtain a stable scaffold and a better effect of ear reconstruction. In recent years, with the development of the porous polyethylene, the ear reconstruction can be performed with the Medpor framework. The medpor has tough texture, good memory, strong plasticity, and we can obtain good postoperative result. However, as ear scaffold, the medpor material has poor histocompatibility and sometimes cause the stent exposure or infection. So it has not been used widely.
Conclusion
Ear reconstruction is very challenging if the mastoid region is damaged or the mastoid area has poor tissue coverage. We evaluated the temporoparietal fascial blood supply by measuring the length of the superficial temporal artery of these patients before surgery. We used the temporoparietal fascial flap to cover the ear scaffold if the length of the superficial temporal artery was longer than 10 cm and the blood supply of the temporoparietal region was good. If not, we chose the upper arm flap. The latter needs a five-stage operation, which is more time consuming and more difficult than the former. Moreover, the expanded upper arm flap is thinner and has better elasticity than the temporoparietal fascia, so the shape of the reconstructed ear is better. In summary, we need adequate preoperative evaluation, an appropriate amount of time, and suitable surgical strategies for these patients without satisfactory mastoid tissue to obtain a good result.
Footnotes
Acknowledgments
We would like to express my sincere thanks to the members of the Department of Ear Reconstruction, Plastic Surgery Hospital.
Authors’ Contributions
Tun Liu, Dejin Gao, and Bingqing Wang participated in the clinical application of the technique. Bingqing Wang and Dejin Gao participated in the follow-up of the patients, analysis of data and drawing pictures for the work. All authors participated in the conception and design of the study. Dejin Gao drafted the article. Bingqing Wang and Tun Liu critically revised the manuscript for intellectual content. All the authors approved the version to be submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Special Research Fund for Plastic Surgery Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (grant number YS202028 to Tun Liu) and the National Natural Science Foundation of China (81701930 to Bingqing Wang).
Ethical Approval
All procedures involving human participants were approved by the institutional ethics committee and conformed to the ethical standards of the institutional and national research committee and national research committee and the World Medical Association Declaration of Helsinki (June 1964).
Informed Consent
Informed consent was obtained from the subjects and their guardians as recommended by the ICMJE. All patients or their guardians provided written consent for using and publishing their images.
Data Availability
All data and figures generated and/or analyzed during the current study are included in this published article.
Patient Consent
All patients or their guardians provided written consent for the use of patients’ images.
Level of Evidence
Level IV, Cases Study.
