Abstract
We describe a right external auditory canal (EAC) recurrent ceruminous pleomorphic adenoma (CPA) accompanied by itching and review the clinical features and histopathology of this disease. A female in her 70s presented with a right EAC mass accompanied by itching. We initially diagnosed the mass as a ceruminous gland adenoma (CGA) following excisional biopsy. Two years and nine months later, the tumor recurred at the same site. A preoperative computed tomography (CT) scan demonstrated no bone destruction, and magnetic resonance imaging (MRI) showed a 1 × 1 cm mass with a clearly defined margin in the right EAC. We completely excised the recurrent tumor using a transmeatal approach under general anesthesia. Histopathology demonstrated haphazard proliferation of tubule-glandular structures lined by two layers of the epithelium in the hypocellular stroma composed of a mucoid matrix. The recurring tumor was diagnosed as a CPA. Here, an EAC tumor—originally diagnosed as a CGA following excisional biopsy—recurred and was subsequently diagnosed as a CPA. CPA can be considered an unusual variant of CGA.
Introduction
Ceruminous gland neoplasms arising from the external auditory canal (EAC) are rare, constituting 2.4% of all ear neoplasms. 1 According to World Health Organization, ceruminous gland neoplasms include both benign and malignant tumors. 2 Benign ceruminous gland neoplasms include ceruminous gland adenoma (CGA), ceruminous pleomorphic adenoma (CPA), and ceruminous syringocystadenoma papilliferum. 2 While CGA is the EAC’s most common benign glandular neoplasms, CPA is second most common. 3 There are very few reported cases of recurrent ceruminous neoplasms,3-6 whether benign or malignant. Here, we report a case of a CPA originating from the site of a previously-excised CGA. We discuss the radiological and histopathological characteristics of these rare tumors and review the relevant literature.
Case Report
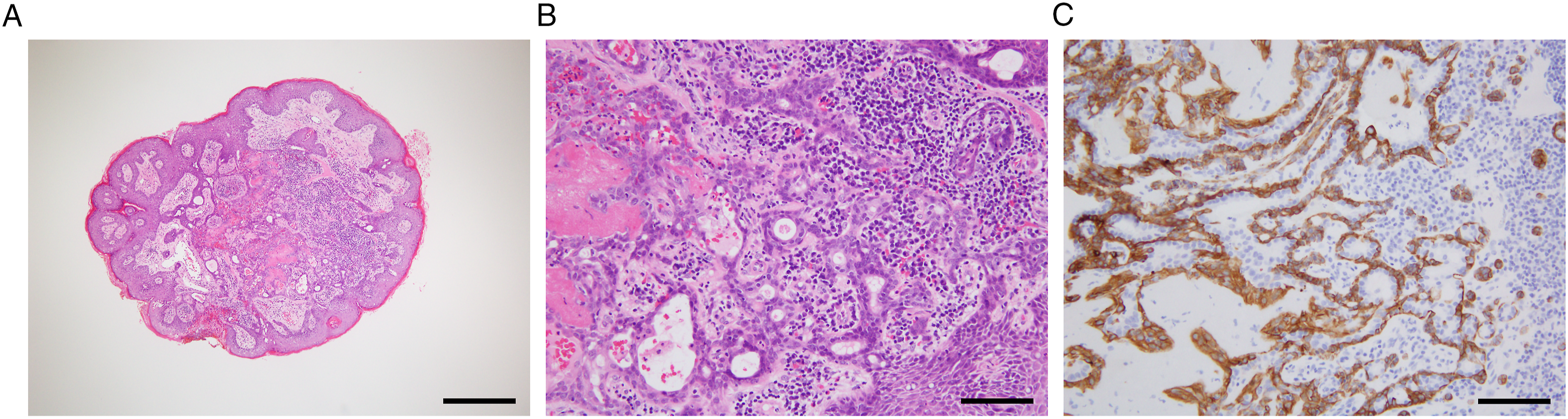
A female in her 70 s presented with a one-year history of a right EAC mass accompanied by itching. We made a preliminary diagnosis of furunculosis; however, treatment with oral antibiotics did not improve her symptoms. A physical examination showed a 0.4 × 0.4 cm lesion on the posterior cartilaginous part of the right EAC (Figure 1A) that was subsequently excised under local anesthesia. We diagnosed the mass as CGA based on biphasic glandular proliferation (Figure 2A, B) with the myoepithelial cells expressing cytokeratin (CK) 5/6 (Figure 2C). Because we felt the entire tumor had been excised, we did not perform any additional resection. Endoscopic view of the right external auditory canal (EAC). (A) A tumor protrudes from the posterior EAC wall (arrow). (B) The recurrent tumor 2 years and 9 months after initial resection (arrow). (C) The right EAC is free of disease 1 year after resection of the recurrent tumor. Histopathology of the tumor, including (A-B) hematoxylin and eosin (H and E) staining. (C) Immunohistochemistry for cytokeratin (CK) 5/6. A non-encapsulated but well-circumscribed mass (A) appears composed of luminal cells in the inner layer and myoepithelial cells in the outer layer (B). The basal myoepithelial cells are positive for CK5/6 (C). Scale bar = 500 μm (A), 100 μm (B,C).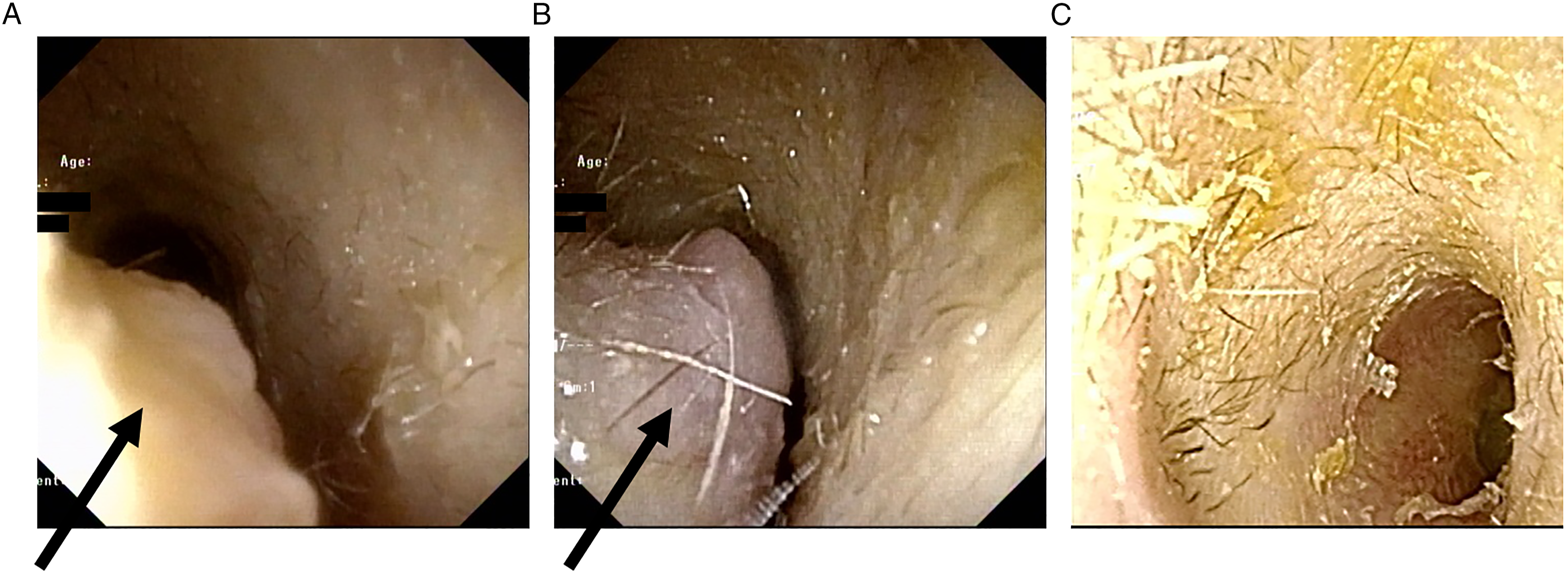
Two years and 9 months later, a protruding mass reappeared in the posterior wall of the right EAC (Figure 1B). A computed tomography (CT) examination showed a right EAC mass with no signs of bone destruction (Figure 3A-B). Magnetic resonance (MR) imaging revealed a 1 × 1 cm mass with a clearly defined margin within the right EAC. The mass lesion showed a low signal intensity on the pre-contrast T1-weighted image (Figure 3C), intermediate signal intensity on the T2-weighted image (Figure 3D, F), and enhancement on the gadolinium-enhanced T1-weighted image (Figure 3E, G). Based on the results of her physical examination, CT, and MR, we suspected recurrent CGA in the right EAC. We performed en bloc tumor resection under general anesthesia using a transmeatal approach (Figure 4A, B). The skin in the EAC remained intact and was replaced after tumor resection (Figure 4C). Axial (A) and coronal (B) computed tomography (CT) images of CGA recurring as a right EAC CPA (asterisk) without bone erosion. Axial (C-E) and coronal (F, G) MR images of the right EAC CPA. After dissection, the tumor presents as a lobulated 1-cm mass with a clearly defined margin and without invasion of surrounding tissues. The mass lesion showed low signal intensity on T1-weighted (T1-W1; C) and medium signal intensity on T2-weighted (T2-W1; D and F), and enhancement on gadolinium-enhanced T1-weighted MR images (GdE-T1- W1; E, G). The recurrent tumor is exposed following skin flap elevation (A). The tumor is removed using an en bloc resection (B). The right EAC skin remains intact and is replaced after tumor resection (C). The cut surface of the extracted specimen appears whitish (D). Histological examination (H and E staining) reveals encapsulation with proliferated ceruminous glands (E) lined by apocrine cells at the luminal site and myoepithelial cells on the basal layer (F). Additionally, a chondromyxoid stroma is noted within the specimen (G). Scale bar = 1 mm (E), 100 μm (F,G).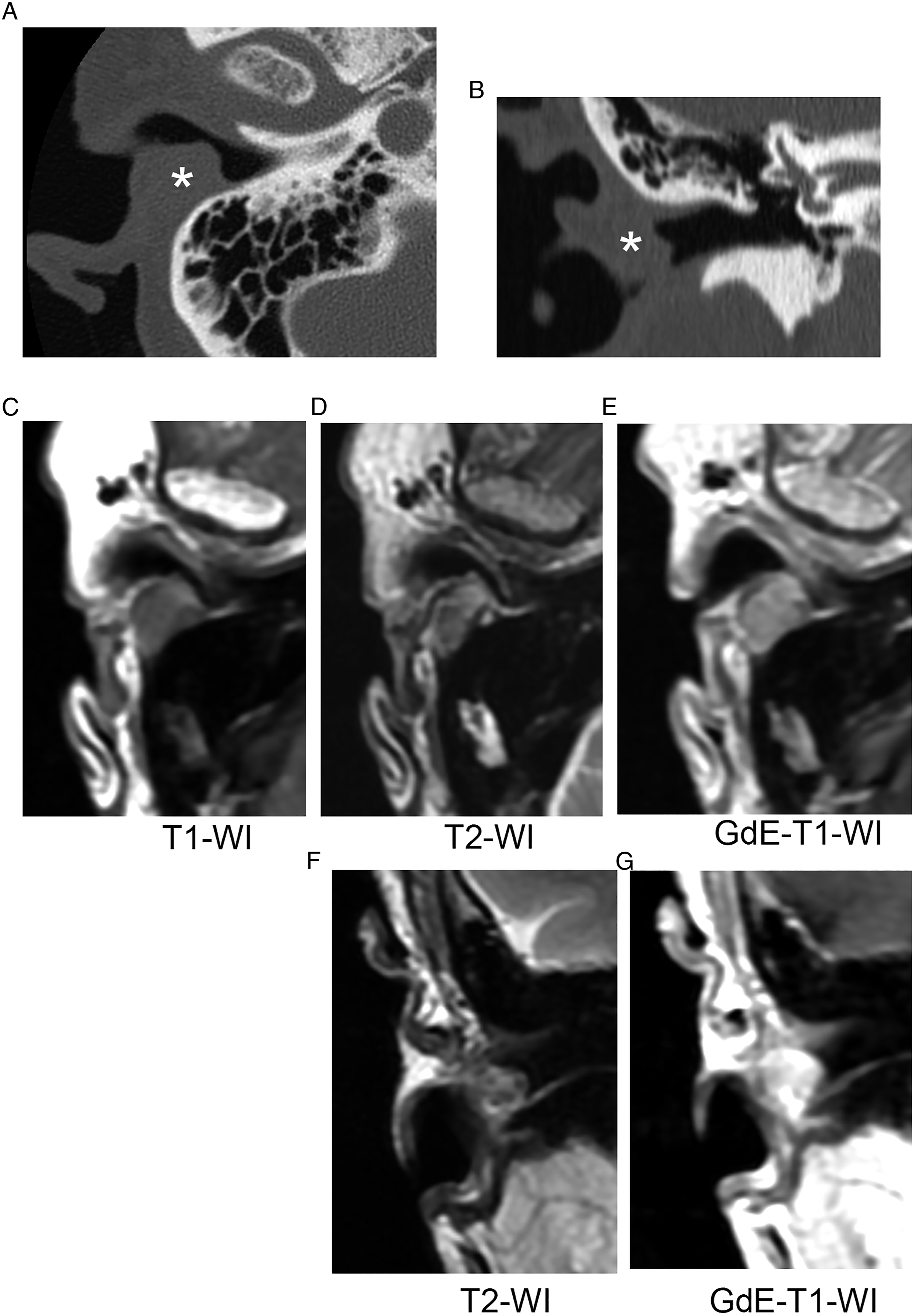
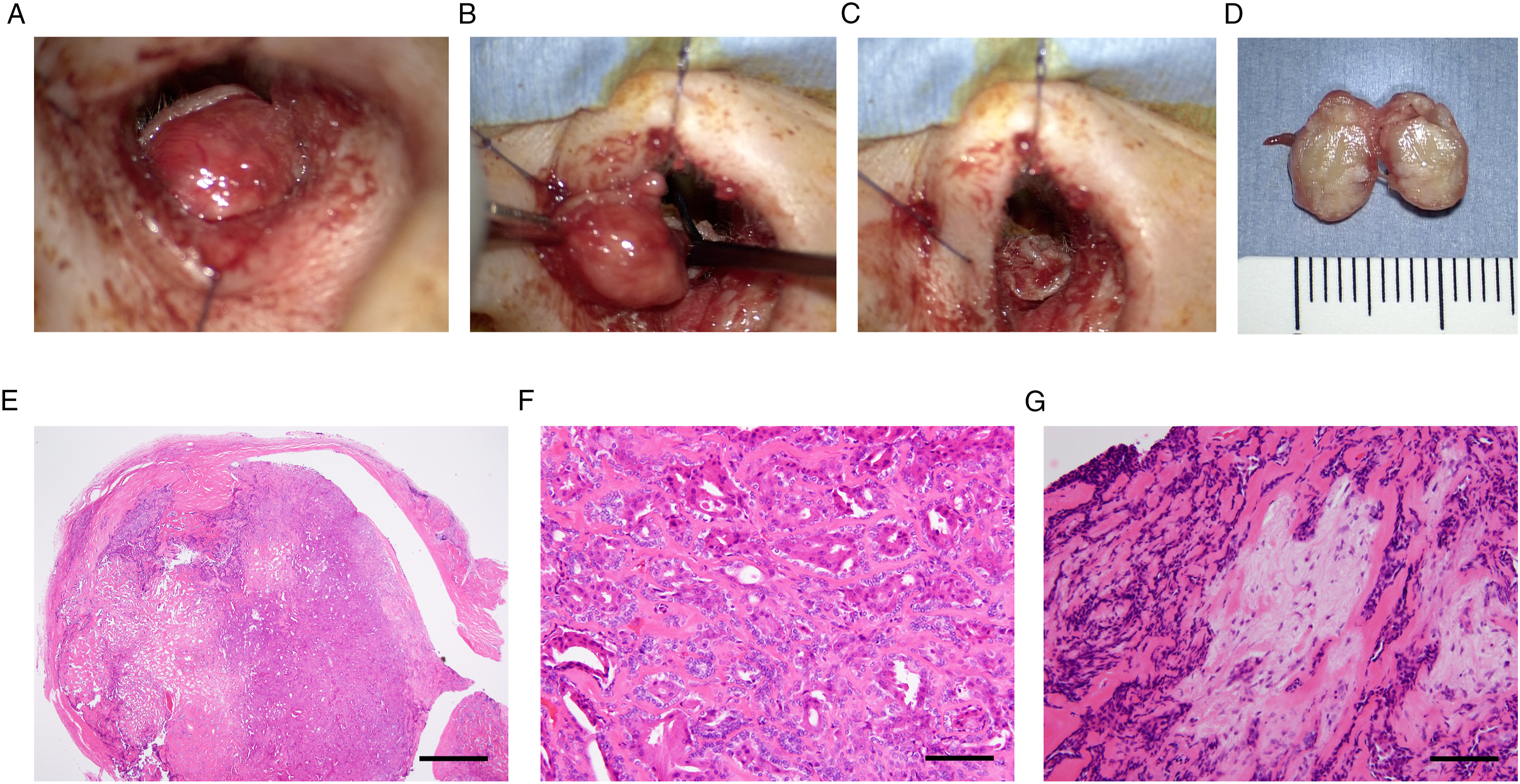
The cut surface of the specimen appeared whitish (Figure 4D). Histological examination revealed lesion encapsulation by ceruminous glands, which were lined by apocrine cells at the luminal site and myoepithelial cells on the basal layer (Figure 4E, F). Additionally, a chondromyxoid stroma was observed within the specimen (Figure 4E, G). Because of these findings, the tumor was diagnosed as CPA.
The patient’s postoperative course was uneventful. At her last follow-up visit one-year postoperatively, there was no evidence of recurrence (Figure 1C). After that, she was followed-up every 3 months to monitor for potential tumor recurrence.
Discussion
CGA is a benign glandular neoplasm of the ceruminous glands and accounts for <1% of all external ear tumors. CGA are typically diagnosed in middle-aged adults without gender predilection. 7 We based our diagnosis of CGA by excisional biopsy on the proliferation of well-differentiated ceruminous glands with a luminal epithelial cell layer and an outer myoepithelial cell layer. As immunohistochemistry highlights the biphasic nature of CGA, 7 the basal cells in our case were positive for CK5/6. Although we did not examine other markers except for CK 5/6, the luminal cells are strongly immunoreactive to CK7, and the basal myoepithelial cells are strongly and diffusely reactive to S-100 protein (in the cytoplasm only for CK 5/6 and in the nucleus only for p63). 3 Since CGA generally does not recur following complete local excision, 8 we concluded that the patient’s initial excisional biopsy was microscopically incomplete. While CGA typically arises in the outer one-third to one-half of the EAC, 3 where ceruminous glands reside, there are several reported cases of CGA originating in the osseous EAC.9-11 In the present case, CGA arose from the cartilaginous EAC, like most reported cases of CGA.3,12-15
Mark and Rothberg reported the first case of CPA in the EAC. 16 After that, a few cases were reported in the literature.8,17-29 As mentioned above, CPA is the EAC’s second most common benign glandular neoplasm. 3 It is typically non-capsulated and composed of a haphazard proliferation of tubular-glandular structures against a background of hypocellular chondromyxoid stroma. 30 While our patient’s recurrent tumor was glandular and mainly composed of two layers of epithelium, the background stroma consisted of a mucoid matrix. Thus, the tumor was diagnosed as a CPA. Since CPA can be considered an unusual variant of CGA, 24 we concluded that the recurrent CPA originated from the initially diagnosed CGA.
Some researchers believe that pleomorphic adenoma (PA) in the EAC arise from ectopic salivary gland tissues. Others argue PA arises from ceruminous glands within the EAC. Our case supports the latter view since the recurrent CPA originated from the initial CGA. Furthermore, neither tumor contained any salivary gland tissue. Therefore, capsule formation in the recurrent tumor does not necessarily exclude the diagnosis of CPA, although de novo CPA are usually non-capsulated. 30
MRI findings were reported in a few cases of CPA.18,29 Briefly, the tumor had a well-defined margin showing a low signal intensity on T1-weighted images 29 , an intermediate 18 or a high 29 signal intensity on T2-weighted images, and enhancement by gadolinium on T1-weighted images.18,29 These results are compatible with a PA originating from the salivary gland. The MRI findings in our case agreed with those reported by Koyuncu 18 in that the recurrent tumor demonstrated an intermediate signal intensity on the T2-weighted image (Figure 3D, F). Higher cellularity—mainly consisting of epithelial cells in the presented case (Figure 4E-F)—may have attenuated signal intensity on T2-weighted images similar to syringocystadenoma papilliferum of the EAC. 31 Local surgical excision is considered the gold standard treatment provided MRI and CT examinations indicate no invasion of the surrounding tissues. 18 Our patient had no bone erosion on her preoperative temporal bone CT and a well-defined margin on MRI, both of which suggest a benign mass. The recurrent tumor was therefore excised under general anesthesia using a transmeatal approach, which is considered a quick and generally safe procedure. 32 While complete surgical excision with negative margins is sufficient for treating benign ceruminous neoplasms, long-term follow-up is recommended.
Conclusions
We describe a recurrent CPA in the EAC. The tumor arose from a mass originated diagnosed as CGA following excisional biopsy. CPA should be considered an unusual variant of CGA. Therefore, complete surgical resection with negative surgical margins is necessary to prevent recurrence.
Footnotes
Acknowledgments
We thank Dr. Tatsuya Aso, MD, for pathological examinations.
Author contributions
KN conceived the idea of the study and drafted the original manuscript. NH supervised the conduct of this study. All authors reviewed the manuscript draft and revised its intellectual content critically. All authors approved the final version of the manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for this article’s research, authorship, and publication.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the author(s), without undue reservation.
Ethics Statement
This case report is in accordance with the Code of Ethics of the World Medical Association (Helsinki Declaration). The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individuals(s) for the publication of any potentially identifiable images or data included in this article.
