Abstract
Objectives
Pharyngocutaneous fistula (PCF) formation following open surgical treatment of hypopharyngeal cancer (HPC) is a common and troublesome complication. To date, the postoperative protocol of restarting oral intake is not clear, and vast discrepancies exist in the literature and among institutions. This study aimed to explore the impact of a postoperative protocol of restarting oral intake on PCF formation after open surgical treatment of primary HPC, and its impact on overall survival (OS) and swallowing function based on the functional outcome swallowing scale (FOSS).
Materials and Methods
This was a prospective observational study of 42 patients who received open surgical treatment for primary HPC at Beijing Friendship Hospital between April 2019 and August 2021. This cohort included two groups: patients who restarted oral intake on the 10th postoperative day (Group 1), and those who started on the 20th (Group 2). The Chi-square test and Fisher’s exact chi-squared test were used for comparing qualitative data among the groups.
Results
Group 1 (n = 27) and Group 2 (n = 15) were comparable in clinical characteristics. PCF occurred in 7 (25.9%) patients in Group 1, while none occurred in Group 2 (P = 0.038). The 2-year OS of all 42 patients was 75.6%; 65.8% and 93.3% for Groups 1 and 2, respectively (P = 0.07). The swallowing function was satisfactory (FOSS Grades 0–III) for 19 (70.4%) patients in Group 1 and 15 (100%) patients in Group 2 (P = 0.035). Laryngeal preservation was achieved in 25 (59.5%) patients, while decannulation was successful in 22 (88.0%) patients.
Conclusions
Delayed oral feeding significantly reduces PCF after open surgical treatment of primary HPC, and improves the swallowing function outcome without jeopardizing the OS.
Keywords
Introduction
Pharyngocutaneous fistula (PCF) is a common complication that occurs after primary or salvage surgical treatment of laryngeal cancer or hypopharyngeal cancer (HPC). Leakage of saliva through the PCF into the surgical field increases the length of hospital stay, delays oral intake (tripled time) and adjuvant therapy, and increases the need for complex wound care and additional procedures for PCF closure.1,2 Untreated or unsuccessfully treated PCF can lead to fatal complications such as carotid artery rupture and sepsis. 3 It is thus important to identify the potential risk factors and incorporate these conditions into preoperative surgical planning to minimize the occurrence of PCF because of its high incidence, significant morbidity, and adverse effect on the patient’s outcome. 1
Comorbidity including hypertension, diabetes, cardiovascular disease, hypoalbuminemia, anemia, thrombocytosis, poor nutritional status, liver disease, peripheral vascular disease, chronic obstructive pulmonary disease, and immunosuppressive medication can predispose patients to PCF.1,3-6 Prolonged operative time (>10 hours) is independently associated with postoperative wound complications. 4 Local factors, including preoperative tracheostomy, tumor site and stage, the extent of neck dissection, and technique of hypopharyngeal closure, which affect wound healing can also increase PCF formation.4,5 Notably, index treatment with radiotherapy (RT) or chemoraidotherapy (CRT) is an independent predictor of PCF development. The addition of chemotherapy enhances the damaging effects of RT on normal tissues, leading to impaired wound healing and increased wound infection, dehiscence, and PCF formation.2,3,7 In the same line, pulmonary diseases, a Washington University Head and Neck Cancer Comorbidity Index (WUHNC Index) score ≥1, high T stage, and overall stage, are significantly associated with increased risk of PCF for salvage cases. 1
In China, surgery followed by RT is the mainstream treatment for primary HPC. Maximal restoration of pharyngoesophageal continuity and function improves survival in patients whose tumors are excised completely. 8 Hypopharyngeal surgical defect closure methods should be chosen carefully because they are closely associated with PCF development. Generally, residual pyriform fossa mucosa is relatively abundant for linear closure of surgical defects in laryngeal cancer. However, the wide surgical margin around the primary lesion is usually troublesome for closure in HPC. The primary lesion is usually advanced because HPC is difficult to detect during the early stages, 9 rendering hypopharyngeal surgical defect generally difficult to close. The risk of PCF is thus significantly higher for open surgical treatment of HPC than for laryngeal cancer.10,11 This study reports the prevention and management of PCF after open surgical treatment of primary HPC, especially after delayed oral feeding which significantly reduces PCF formation.
Materials and methods
Patients’ characteristics and staging
This study adopted a prospective observational approach of patients who received open surgical treatment for primary HPC at Beijing Friendship Hospital between April 2019 and August 2021. Patients who received induction chemotherapy, induction CRT and those with salvage cases were excluded from the study. Patients’ demographics, medical history, preoperative examination, treatment, complications, functional outcome (especially the swallowing function), and survival (assessed using Kaplan–Meier analysis) were analyzed and summarized.
TNM staging was in accordance with the American Joint Committee on Cancer (AJCC, 8th edition, 2017). The preoperative risk factors of patients, including hypertension, diabetes, cardiovascular disease, hypoalbuminemia, anemia, thrombocytosis, hypothyroidism, vascular diseases, liver disease, and pulmonary diseases, were exterminated or suppressed before the surgical procedure.
A precise estimation of the surgical defect was always carried out through preoperative imaging examinations using contrast computed tomography (CT) scan, contrast magnetic resonance imaging (MRI) scan, barium swallow X-ray, gastro-endoscope, and positron emission tomography-CT (PET-CT) scan.
Surgical protocol
An excision margin of ≥5 mm circumference around the primary tumor was made, after which 2–3 mm of the circumferential residual mucosa was resected as surgical margins. The resected residual mucosa was divided into smaller pieces (labeled individually according to anatomic sites) and subjected to frozen section evaluation. Positive margins required further excision of the corresponding residual mucosa, which was continued until tumor-free margins were microscopically confirmed.
A nasogastric feeding tube (NG-FT) was introduced through the nostril and the hypopharyngeal surgical defect into the stomach when the excision of the primary lesion was completed. The NG-FT contaminated surgical field was then irrigated with hydrogen peroxide, diluted iodophor, and saline, before commencing hypopharyngeal closure.
The closure methods, including primary closure of residual healthy mucosa, regional pedicled flap, and free tissue flaps among other methods, were decided based on the final surgical defect. The choice of the closure method was systematic; a flap was not used if the primary closure could be achieved, while a free tissue flap, which would need microscopic vessel anastomosis, was not used if a pedicled flap could be used. Important surgical techniques included meticulous hemostasis with preserved viability of the hypopharyngeal mucosa, atraumatic handling of the mucosa, linear closure type, the two-layer technique, water-tight continuous submucosal suture line without tension, and adequate use of drains to eliminate dead space. 5 First-line submucosal suture stitches were not extremely tightened to avoid mucosal ischemia. The multilayer fascial underlay (MLFU) technique was adopted for reconstruction utilizing the anterolateral thigh (ALT) free flap. A patch of fascia lata wider than skin paddle was harvested and was sutured to neck soft tissues and prevertebral fascia beside the standard closure of flap epithelium to pharyngeal mucosa. 12
Inferior constrictor muscles and strap muscles were used to strengthen the newly established hypopharyngeal wall, especially the suture line, after the primary mucosal closure of the hypopharynx. Muscles were not too tightly reapproximated around the hypopharynx to avoid hypopharyngeal stenosis, which would jeopardize the swallowing function outcome.
A cuffed tracheotomy tube was introduced for patients with larynx preservation to help reduce aspiration from saliva and oral feeding contents. The tube was kept in place until oral feeding was regained without aspiration and was then deflated.
Postoperative management
Prophylactic broad-spectrum antibiotics were administered, and intravenous proton pump inhibitors 13 were used during the postoperative period until oral feeding without NG-FT could satisfy patients’ nutritional demands. Drainage tubes were constantly maintained to avoid blockage and retention of fluid inside the surgical field, which would predispose patients to infection. NG-FT feeding was started on the 2nd postoperative day.
The first clinical signs of PCF were wound erythema and swelling, especially those anteroinferior to the hypopharyngeal anastomosis. Tenderness and fluctuation could also be felt on palpation and increased turbid discharge from drainage tubes, 3 and fever, usually between 37.5°C and 38.5°C.
Swallow X-ray studies were performed on the 9th postoperative day. The patients were not grouped until their hypopharyngeal anastomoses were well healed based on swallow X-ray studies. This timeline was set as a starting line for all involved patients regardless of their former conditions. Patients were consulted about the benefits and disadvantages of early oral feeding (10th postoperative day) and delayed oral feeding (20th postoperative day) in November 2018. Only patients who complied with delayed oral feeding were included in experimental group (Group 2), otherwise, patients started oral feeding on the 10th postoperative day and were included in control group (Group 1).
Patients with PCF onset before oral feeding were excluded from the study. Conservative treatment or surgical repairment resulted in the successful closure of all PCF after oral feeding.
All patients were scheduled to receive postoperative RT, preferably commencing within 6 weeks. The RT was intensity-modulated: 5 days a week, 2 Gy/day over 7 weeks, reaching a total dose of 60–70 Gy. Patients with PCF received delayed RT after fistula closure. Other modalities of treatment, including chemotherapy, targeted therapy, immune therapy, or a combination of various treatment modalities were administered based on the presence of clinicopathological risk factors, such as N stage, extra-nodal extension, perineural, and perivascular invasion among other factors.
Follow-up
Post-operation follow-ups were done regularly at 1, 3, and 6 months, and every 6 months after that. Swallowing function outcomes were routinely evaluated using a functional outcome swallowing scale (FOSS) during the follow-up sessions. 14
Statistical analysis
All statistical analyses were performed using SPSS 26.0 (IBM Corp, Armonk, NY) and GraphPad Prism 9.0 (GraphPad Software, Inc). The Kaplan–Meier analysis and Log-rank test were used to calculate the OS and compare survival statistically. The Chi-square test, Fisher’s exact chi-square test, and Continuity (Yates) correction were used to compare qualitative data. The two-tailed Student's t-test was used to compare differences in continuous variables at a significance threshold of P < 0.05.
Results
Characteristics of our cohort of patients with hypopharyngeal cancer.
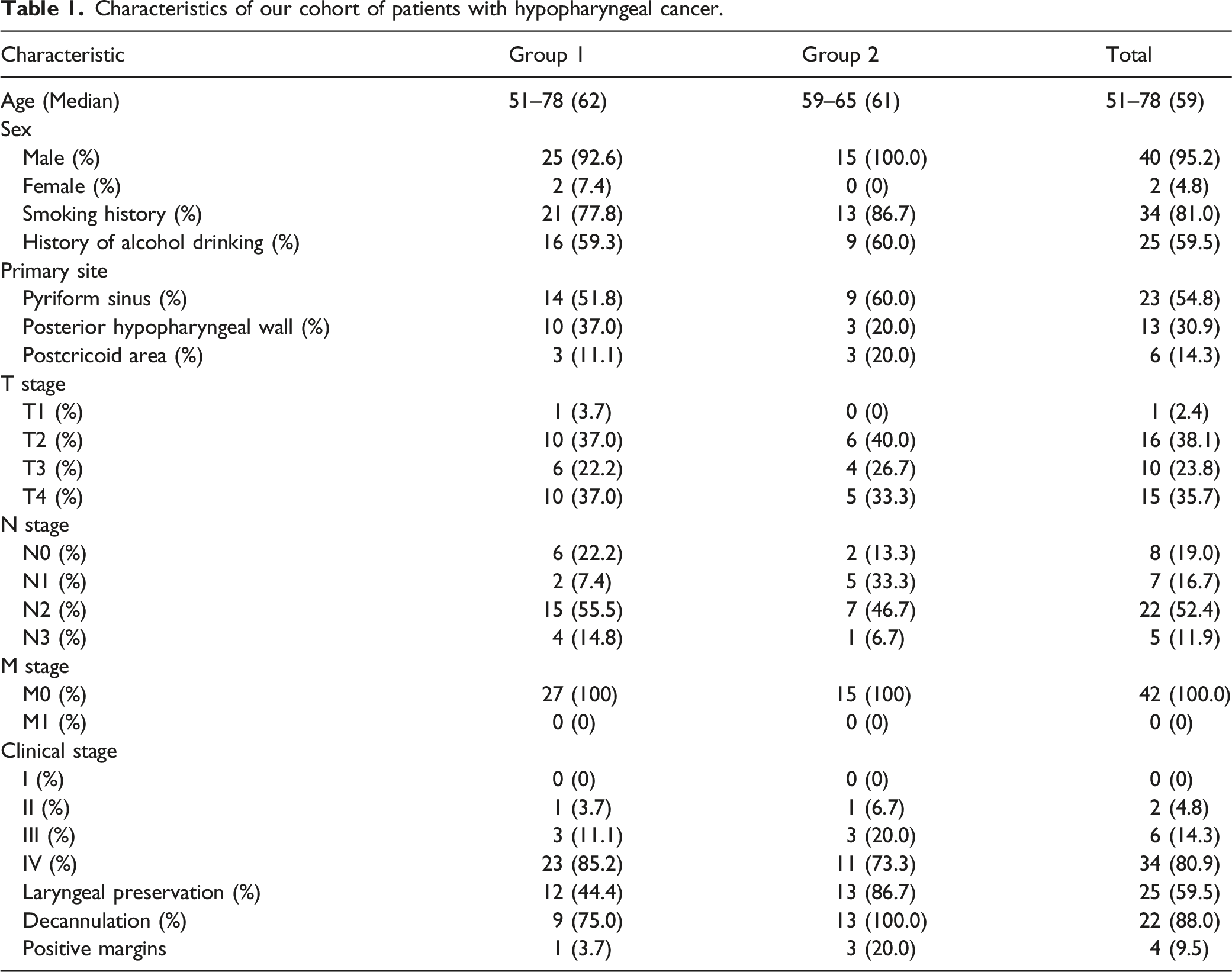
Methods of closure of hypopharyngeal defects in our cohort.
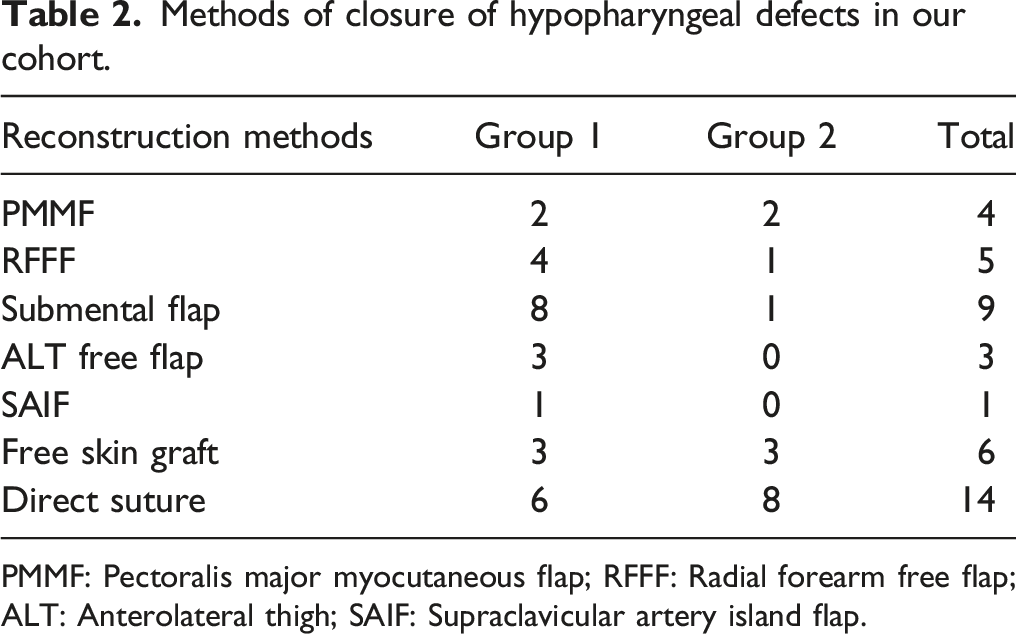
PMMF: Pectoralis major myocutaneous flap; RFFF: Radial forearm free flap; ALT: Anterolateral thigh; SAIF: Supraclavicular artery island flap.
Clinical data of pharyngocutaneous fistulas (PCF).
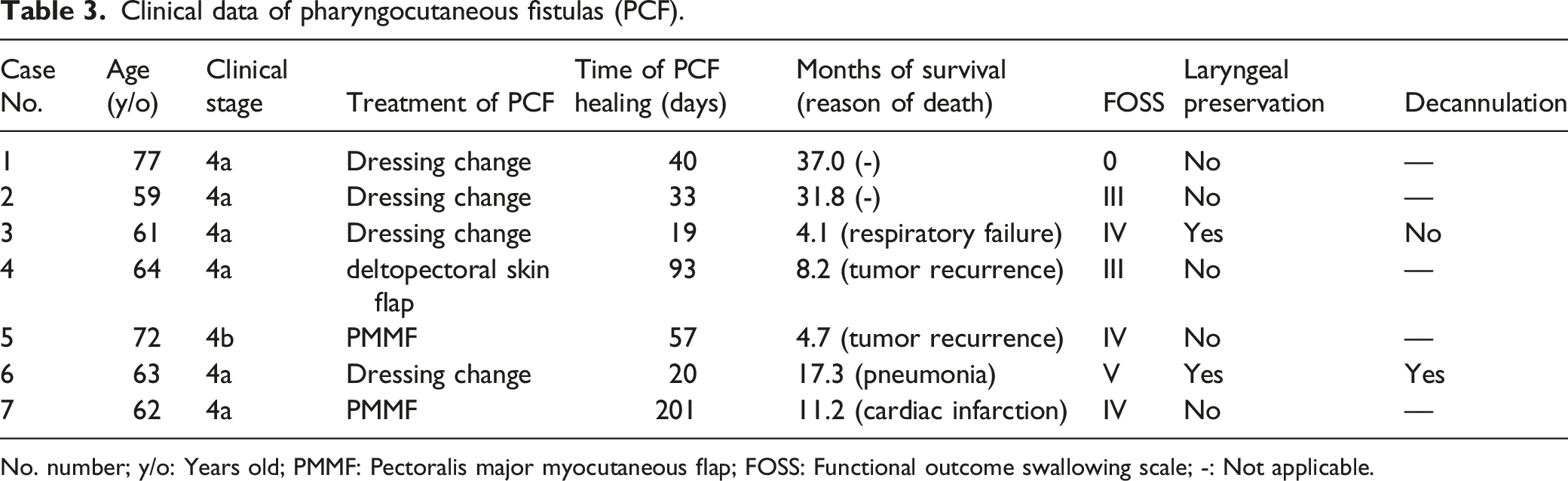
No. number; y/o: Years old; PMMF: Pectoralis major myocutaneous flap; FOSS: Functional outcome swallowing scale; -: Not applicable.
The two-year OS of all 42 patients was 75.6%; 65.8% and 93.3% for Group 1 and 2, respectively (P = 0.07) (Figure 1). Laryngeal preservation was achieved in 25 (59.5%) patients, while decannulation was successful in 22 (88.0%) patients. Overall survival (OS) of this cohort of patients. Kaplan–Meier analysis was performed for the two Groups, and the difference in OS between them was not statistically significant.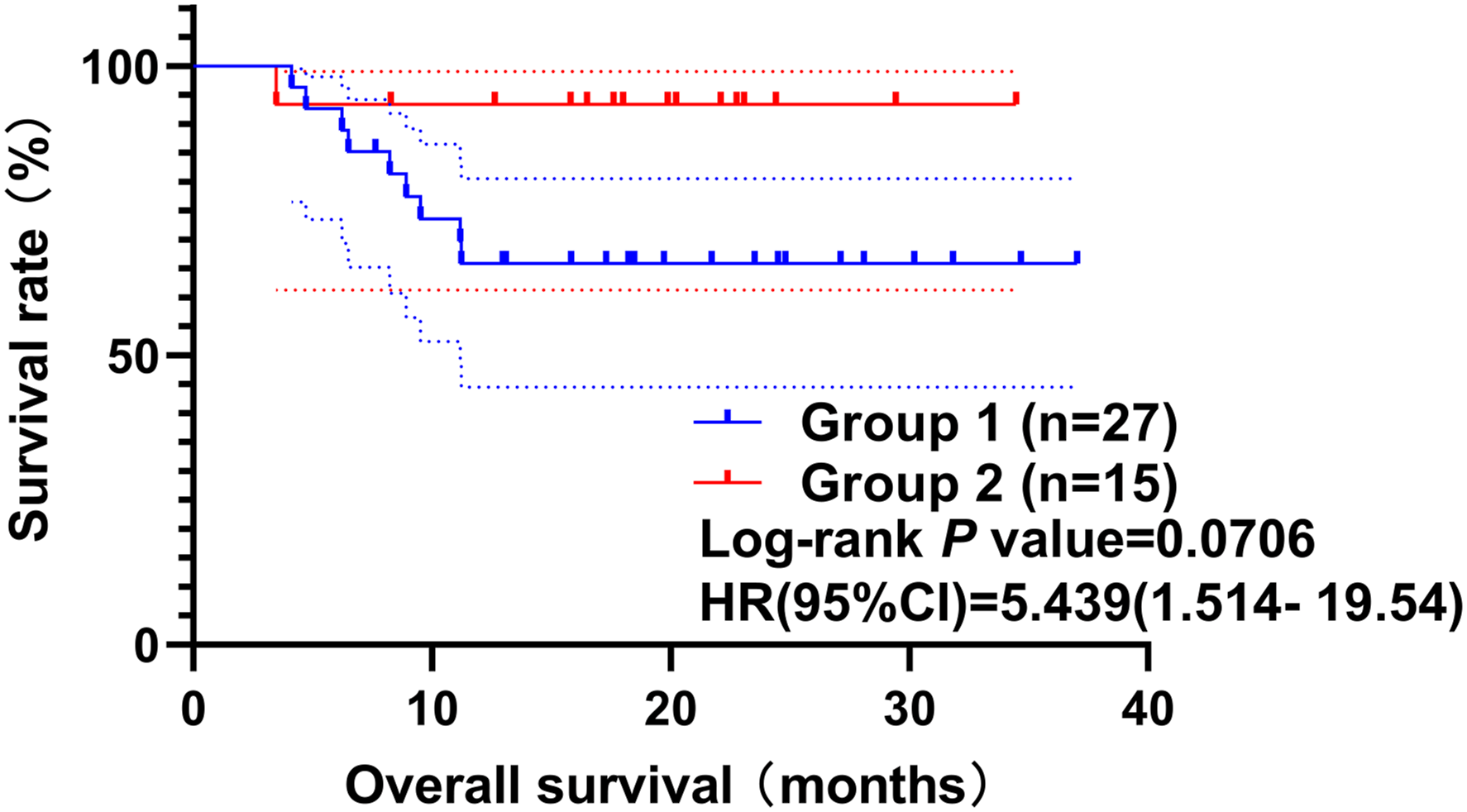
Swallowing function outcome of the two groups.
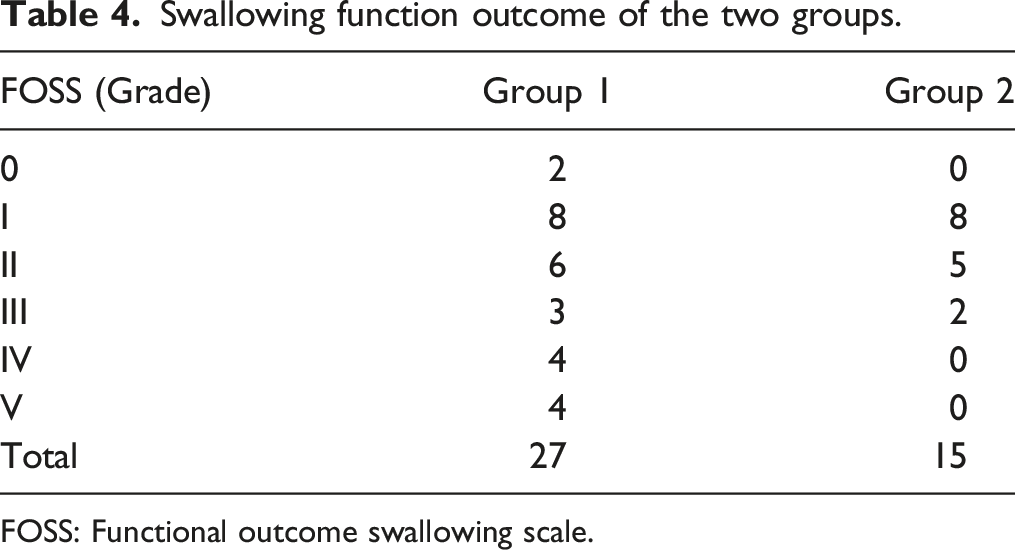
FOSS: Functional outcome swallowing scale.
Discussion
A relatively high proportion of PCF has been found in smokers among the surgically treated HPC patients, though the proportion is not statistically significant. Similarly, underweight and overweight patients have relatively high proportions of PCF but are still not statistically significant. 2 It is widely stated that postoperative incidence of PCF is not influenced by age and gender. 11
Surgical treatment is one of the most important factors in the occurrence of PCF. Nonetheless, no specific technique or donor site has emerged as a clear best option to date. 15 Having a submucosal continuous suture as the first suture line between realigned healthy residual mucosal edges, or between residual mucosa and epithelium of various kinds of flaps (pedicled or free) is the most important technique of hypopharyngeal surgical defect closure. Notably, there should be no tension on the first suture line. Though a modified continuous Connell suture significantly reduces PCF incidence to 16% after total laryngectomy (TL), 16 it was not preferred because it is time-consuming and inverts mucosal edges, which hinders mucosal healing. Herein, the MLFU technique in ALT reconstruction was especially useful because it significantly reduced PCF incidence for salvage cases to 3%. The technique strengthens the structural integrity of wound closure, provides a second water-tight closure, and suspends flap reconstruction in place, preventing gravitational or other movement-related forces from insulting the pharyngeal mucosal-epithelial closure. 12
The protocol of restarting oral intake is another major issue regarding PCF formation after hypopharyngeal reestablishment. In a 2018 Dutch study on PCF after TL, the grouping of the patients was done based on the oral feeding time: an early group (within 3 days postoperatively) and a late group (6–12 days postoperatively). Notably, there was no significant difference in PCF development between the two groups (OR 1.11; 95% CI: 0.57–2.17; P = 0.752). 17 In the same line, a 2021 review, based on multiple database searches, compared the outcomes of early (≤5 days) versus late (>5 days) oral feeding after TL and total pharyngolaryngectomy. The rates of PCF in randomized control trials (RCTs) in early versus late feeding were 15.2% versus 11.7% (RR 1.35, 95% CI: [0.68–2.7], P = 0.40), whereas the corresponding PCF rates in cohort studies were 14.1% versus 20.5% (RR 1.0, 95% CI: [0.76–1.3], P = 0.98). 18 The study concluded that the difference between early and late oral feeding was not statistically significant. However, there could have been selection bias, and investigators may have delayed oral feeding in patients who were thought to be at higher risk of PCF, such as those with malnutrition. Notably, the incidence of PCF was higher in cohort studies in the late oral feeding group than that reported in RCTs. 18
In another 2021 meta-analysis comparing early oral feeding (before 7 days) and late feeding oral feeding (after 7 days), there was a higher risk of PCF for early feeders compared to late feeders (RR = 1.56, 95% CI: [1.15–2.11]). A higher risk was also observed for RCT but was not significant (RR = 1.40, 95% CI: [0.85–2.30]), strongly suggesting that there was an overall 50% greater risk of PCF for early oral feeding compared to late oral feeding (RR = 1.51, 95% CI: [1.17–1.96]). 19 Based on the findings of this study, it was assumed that restarting oral feeding might be a major test for hypopharyngeal closure because swallowing results in the movement of the entire neo-hypopharynx, and any vulnerability would jeopardize mucosal healing. Longer healing time would thus lead to safer oral intake, especially for the surgical defects after HPC extirpation, which would be harder to close than those after TL. In this study, delayed oral feeding resulted in a significantly lower rate of PCF although the study sample was relatively small. This finding should thus be validated using larger studies.
Notably, in the 2021 Italian endeavor (“Fistula zero” project after TL) which achieved excellent results, initiation of oral feeding was on the 10th postoperative day for TL only cases, and on the 13th for cases with TL and reconstruction. 20 Hence, it might not be in the best of the patient to restart oral feeding too early, especially after HPC surgeries. A study based on the Japanese national inpatient database reported 20 days as the median for oral feeding after pharyngolaryngectomy for HPC patients without PCF. 2
Some institutions do not do any test before restarting oral intake,16,21 while others perform a methylene blue test first. 22 Based on the findings of this study, a swallow X-ray is ideal before initiation of oral intake. 20 In some institutions, NG-FT would be removed as soon as there is confirmation of no sign of PCF and oral intake is restarted. 22 In contrast, it is the practice of our institution and others that an oral liquid intake would be initiated first and gradually increased, while still using NG-FT to replenish the deficiency of oral intake. The NG-FT would not be removed until oral intake can maintain sufficient calories to sustain the patient. 21
Despite the noticeable insights revealed by this study, it was limited by several factors. First, this was a relatively small case series, whose conclusions require further validation in future studies with a larger number of patients. Second, the grouping of patients could have been randomly performed despite adopting a more ethical approach. Notably, all 42 patients achieved satisfactory healing of hypopharyngeal anastomoses on the 10th postoperative day. About 20% of these patients would have suffered from PCF if oral intake was initiated at this time point based on previous experience. As such, we tried to explore whether such patients would benefit from delayed oral feeding. The results could be more easily interpreted and convincing if such patients were randomly grouped. In contrast, the approach adopted herein was more ethical and respectful for the patients who had already undergone a lot of suffering from the management process, especially the NPO experience following surgery. All the patients complied well and the only comparing factor was the time point of initiation of postoperative oral feeding, which was the essence of a controlled study. As such, we believe that the conclusion of this study is well founded, statistically sound, and might contribute to the literature.
Conclusion
Delayed oral feeding significantly reduces PCF after open surgical treatment of primary HPC, and greatly improves swallowing function outcome. The overall survival for patients with delayed oral feeding seemed to be better than that of patients with early oral feeding though not statistically significant.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Beijing Hospitals Authority’s Ascent Plan (DFL20220102), Beijing Friendship Hospital Seed Project, Capital Medical University (YYZZ202125), and a grant from Capital Medical University (2020YGS02).
