Abstract
Introduction
Sinonasal inverted papilloma (SNIP) is one of the most common benign tumors of the nasal cavity and sinuses, responsible for about .5–4.7% of incidences of all sinonasal tumors. 1 Sinonasal inverted papilloma is more prevalent in men aged 50 to 60 and is predominantly unilateral; its clinical symptoms are complex, and it can recur and become malignant. 2 Sinonasal inverted papilloma is differentiated primarily from unilateral sinonasal disorders, particularly unilateral CRS with/without nasal polyps, the most prevalent unilateral sinus condition.
Imaging is one of the most efficient techniques for differentiating SNIP from other unilateral sinonasal disorders. Characteristics of SNIP on sinus CT include the presence of lobulated edge, air sign within the lesion or between the lesion and the nasal cavity/sinus, and focal hyperostosis or focal osseous erosion.3-8 An MRI scan, and specifically the convoluted cerebriform pattern (CCP), can help with the diagnosis of SNIP.9,10 However, MRI scans can be noisy, complicated, time-consuming to schedule and review, and prohibitively expensive for certain primary care facilities. Furthermore, its clinical application is limited by the inability to assess patients with cardiac pacemakers, implanted metal, metal for internal fixation, therapeutic pumps, and claustrophobia.
Although sinus CT is the preferred imaging examination method for nasal diseases, currently, sinus CT-based methods for making a preliminary clinical diagnosis of SNIP are not as simple, quick, inexpensive, effective, noninvasive, or easy to use as they could be. Consequently, it is essential to determine the diagnostic significance of SNIP using easily accessible clinical data, such as CT findings, demographics, and clinical characteristics, and to develop a clinical prediction model to facilitate diagnosis.
Patients and Methods
Study Description and Subjects
The Medical Ethics Committee of the Affiliated Hospital of Qingdao University approved this single-center retrospective study of our hospital. A total of 267 consecutive confirmed cases with SNIP and 273 consecutive patients with unilateral CRS with/without nasal polyps were enrolled by searching our pathology database from November 2016 to December 2021. The inclusion criteria were as follows: (1) patients diagnosed with SNIP or unilateral CRS with/without nasal polyps with surgical pathology, and all the patients met the diagnostic criteria for SNIP as recommended by the European position paper on endoscopic management of tumors of the nose, paranasal sinuses and skull base (2010) 1 or CRS as recommended by the European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS) 2012 11 ; (2) a non-contrast sinus CT scan must have been performed within 14 days before surgery; (3) patient demographic and clinical data must be complete. Exclusion criteria were as follows: (1) patients with bilateral sinonasal lesions on CT scan; (2) coexistence of other sinus diseases such as fungal sinusitis, cystic fibrosis, primary ciliary dyskinesia, or other sinonasal tumors; (3) immunodeficiency; (4) pregnancy; and (5) inability to obtain CT images or poor CT image quality such as artifact presence.
Two hundred patients with SNIP and 200 patients with unilateral CRS with/without nasal polyps were randomly assigned to the training set using a randomized block procedure to ensure that the ratio of the training group to the validation group was approximately 7:3. Moreover, the remaining 67 patients with SNIP and 73 patients with unilateral CRS with/without nasal polyps were randomly assigned to the validation set. Demographic and clinical characteristics, including gender, age, nasal symptoms (nasal obstruction, nasal discharge, facial pain/headache, reduction or loss of smell, epistaxis/blood tinged mucus), history of sinus surgery, smoking, and alcohol dependence were obtained from admission records.
Computed Tomography Evaluation
Computed Tomography images were obtained using 2 64-slice CT scanners (without contrast), a GE Healthcare Discovery 750 and a Siemens Healthcare Somatom Sensation 64.
Two radiologists with 15 and 25 years of experience evaluated the CT image features in consensus, blinded to pathology and surgical conditions. Disputes were resolved by consultation. If disagreements persisted after consultation, a radiologist with 30 years of expertise was consulted to reach a conclusion. The CT features were evaluated as follows: (1) lobulated/wavy edge (absent or present); (2) air sign (absent or present); (3) focal hyperostosis (absent or present); (4) diffuse hyperostosis (absent or present); (5) focal osseous erosion (absent or present); and (6) CT values, as shown in Figure 1. The CT image evaluation. (A) Lobulated edge. (B) Wavy edge. (C) Air sign. (D) Focal hyperostosis. (E) Diffuse hyperostosis. (F) Focal osseous erosion.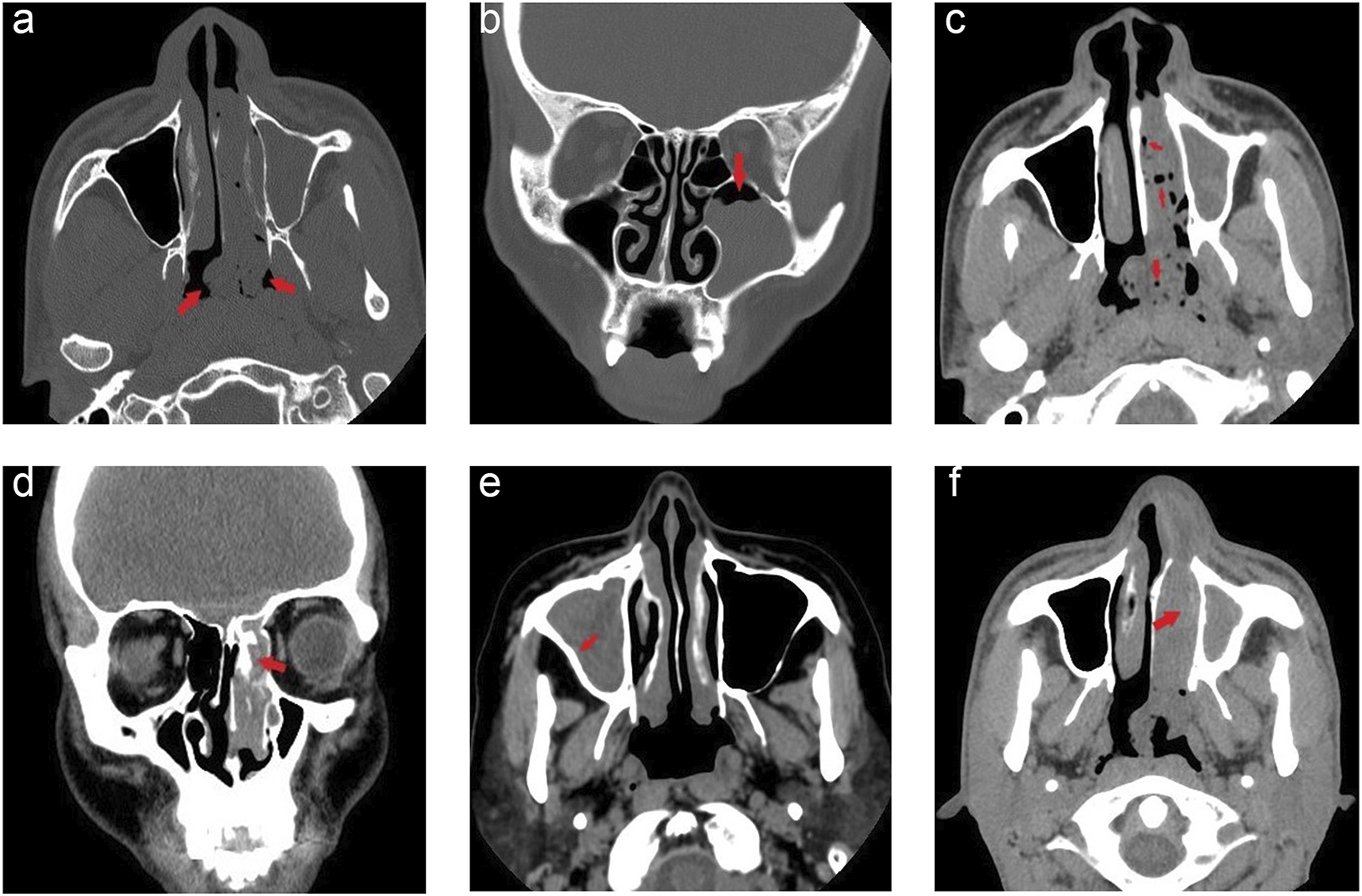
A lobulated edge was defined as the edge of the lesion lobulated in at least three slices. 3 (Figure 1A) A wavy edge was defined as two or more contiguous wavy protrusions on the edge of the lesion in at least three slices. (Figure 1B)
The air sign was defined as two or more bubbly air shadows within the lesion or between the lesion and the nasal cavity and sinus walls in at least three slices. 5 (Figure 1C)
The focal hyperostosis was defined as a focal and eccentric bone thickening or new bone growth in the bony walls of the nasal cavity and paranasal sinus cavities in at least three slices compared to the contralateral side. 8 (Figure 1D)
Diffuse hyperostosis was defined as an apparent diffuse and concentric thickening of bone in the nasal cavity and paranasal sinus bone walls in at least three slices compared to the contralateral side. 7 (Figure 1E)
Focal osseous erosion was defined as apparent focal bone destruction or bone erosion in at least three or more slices in the nasal cavity and paranasal sinus bone walls compared to the contralateral side.6,8 (Figure 1F)
Computed Tomography value measurement of the lesion; from axial CT images, at least 6 slices with clear lesions and high image quality were selected. The region of interest was drawn as large as possible in each slice away from the lesion’s margins to avoid bone and calcification, and the lesion’s average CT values were assessed. 12 The average CT values of the lesions are considered the final observation.
Statistical Analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences version 25.0 (IBM, Corp, Armonk, NY, USA) and R software version 4.1.3 (R Foundation, Vienna, Austria). Additionally, the Student’s t-test was used to make inferential inferences between groups. Furthermore, quantitative measures of normal distribution and quantitative measures of non-normal distribution were expressed as mean ± standard deviation (SD) and median ± interquartile range, accordingly. Moreover, the comparison between groups was analyzed using the Kruskal–Wallis rank-sum test. Frequencies and proportions were used to indicate qualitative indices, and the Chi-square test or Fisher’s exact test was used to compare groups. Univariate and multivariate logistic regression analyses were employed to identify independent risk factors for SNIP, and odds ratios (OR) and 95% confidence intervals (CI) were computed. A nomogram was established, and receiver operating characteristic (ROC) curves and calibration curves of the training and validation sets were formed to examine the diagnostic capacity of the nomogram model. These tests were two-tailed, with P < .05 being considered statistically significant.
Results
Baseline Characteristics of Training and Validation Sets
Baseline Characteristics of the Training (n = 400) and Validation Sets (n = 140).

Abbreviations: SD, standard deviation.
Demographic and Clinical Characteristics of the Training Set
Demographic and Clinical Characteristics of the Training Set.
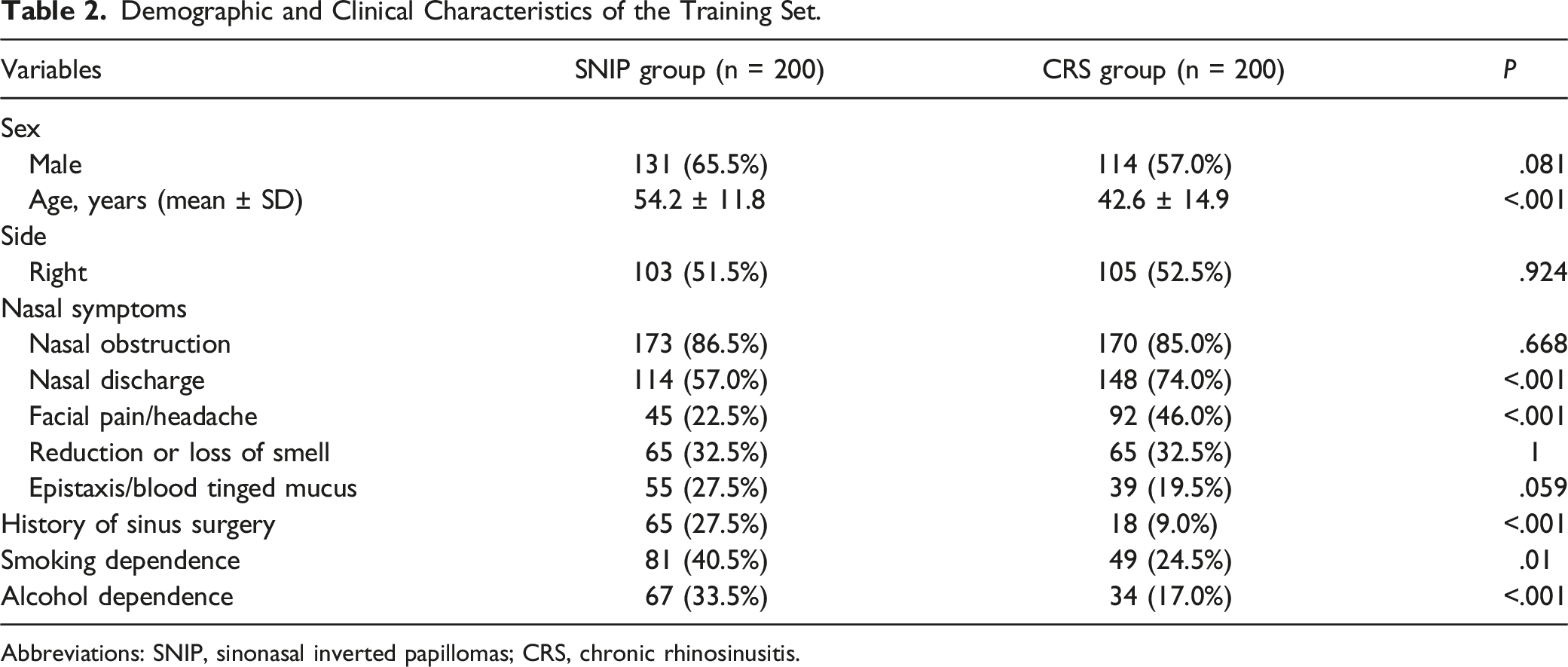
Abbreviations: SNIP, sinonasal inverted papillomas; CRS, chronic rhinosinusitis.
Computed Tomography Features of the Training Set
The CT Features of the Training Set.
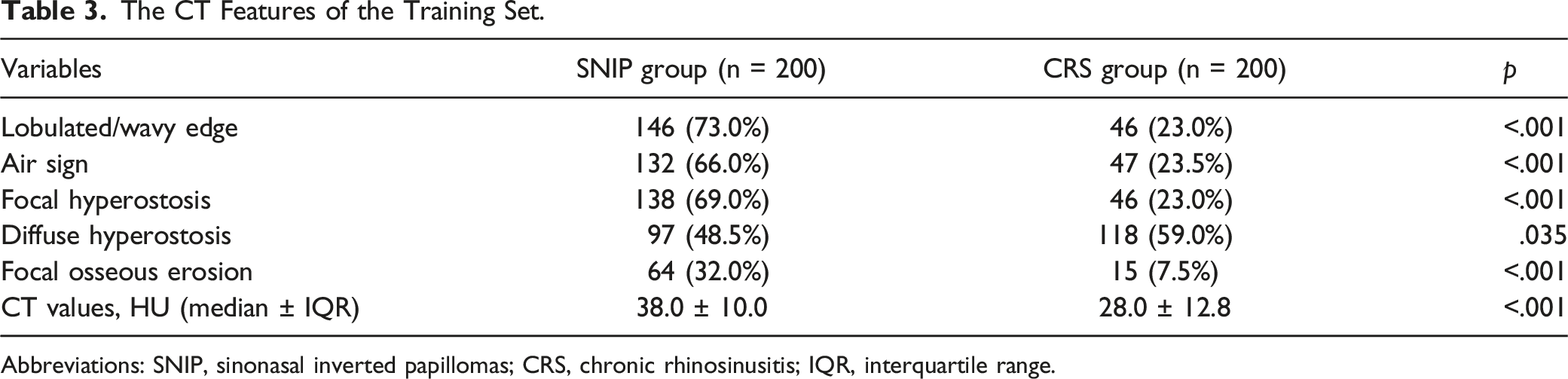
Abbreviations: SNIP, sinonasal inverted papillomas; CRS, chronic rhinosinusitis; IQR, interquartile range.
Univariate and Multivariate Logistic Analyses of the Training Set
Univariate Logistic Regression Analysis for Identification of Factors Associated with SNIP.
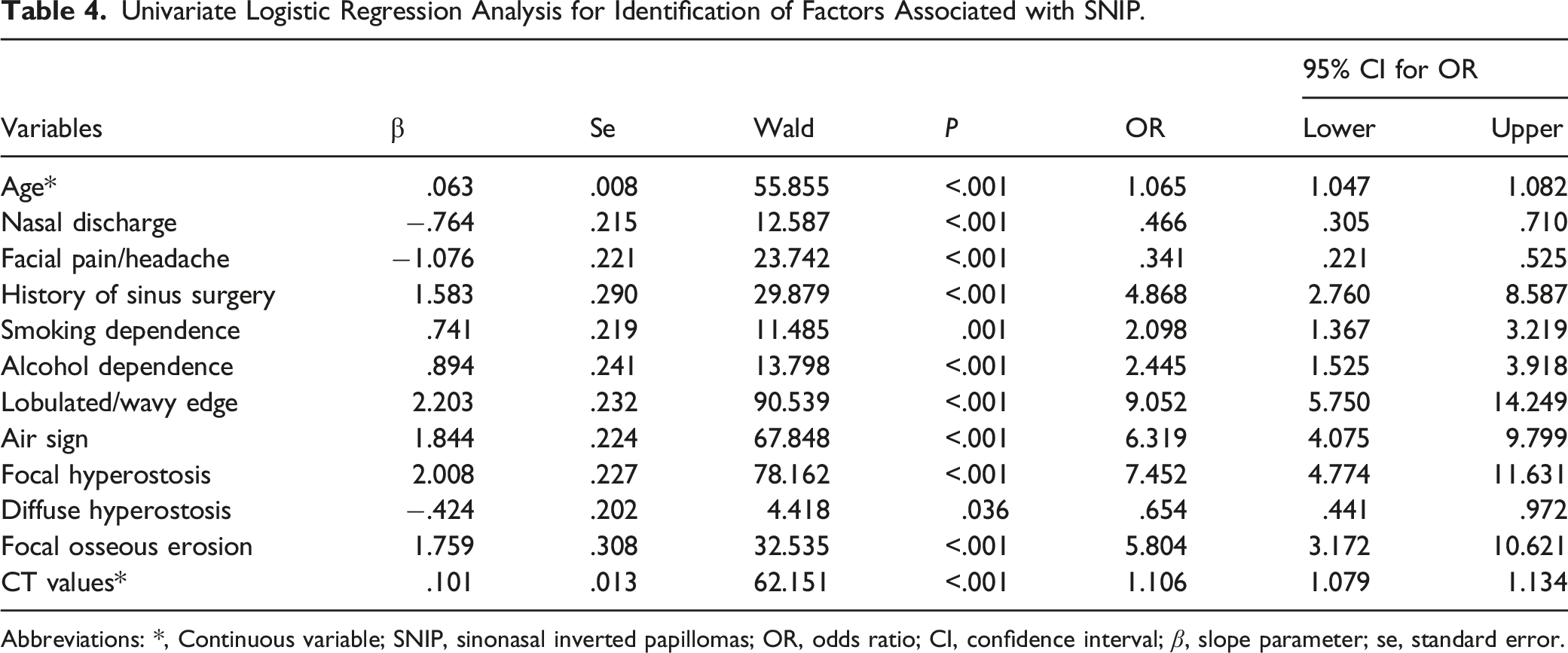
Abbreviations: *, Continuous variable; SNIP, sinonasal inverted papillomas; OR, odds ratio; CI, confidence interval; β, slope parameter; se, standard error.
Multivariate Logistic Regression Analysis for Identification of Factors Associated with SNIP.
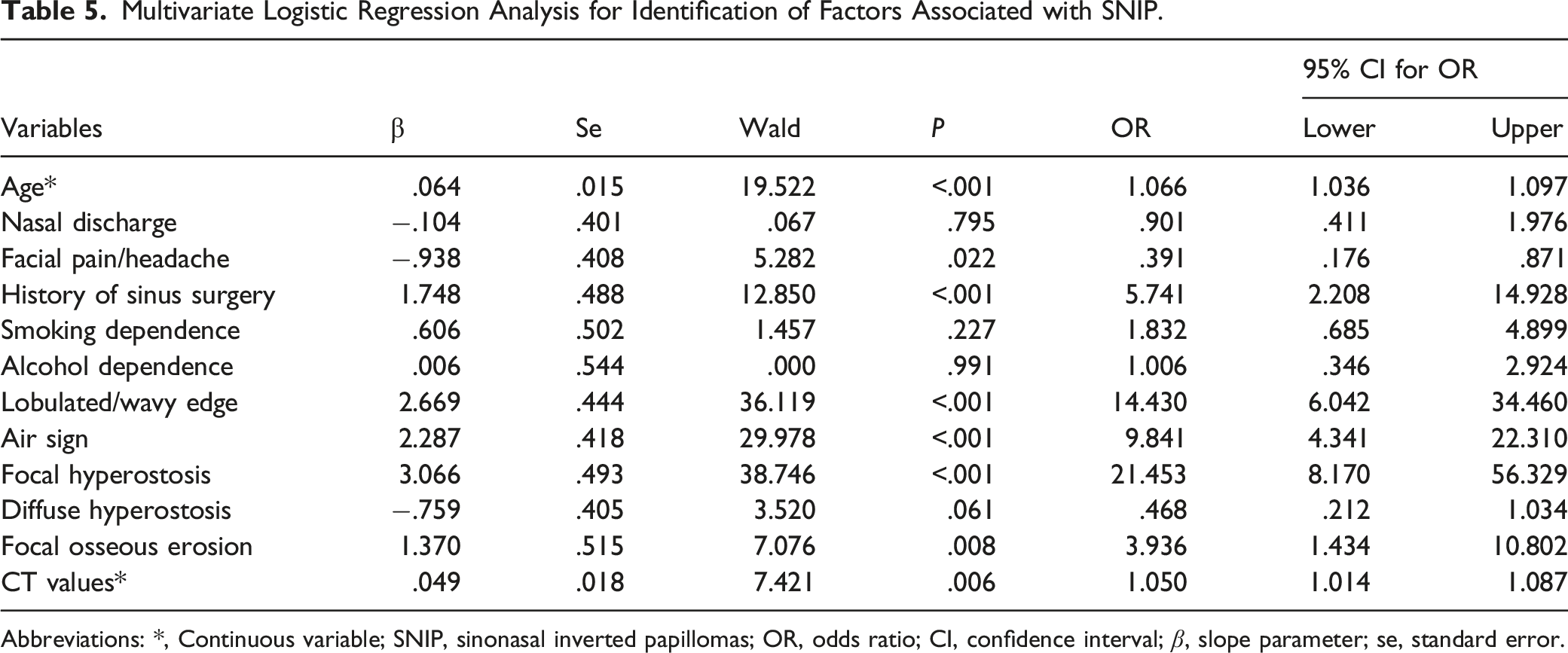
Abbreviations: *, Continuous variable; SNIP, sinonasal inverted papillomas; OR, odds ratio; CI, confidence interval; β, slope parameter; se, standard error.
Comparison of SNIP Patients With/Without Malignant Transformation in the Training Set
In the training set, age (P = .020), rates of epistaxis/blood tinged mucus (P = .009) and focal osseous erosion (P < .001) were significantly higher in SNIP patients with malignant transformation than those without malignant transformation. The rates of lobulated/wavy edge (P = .008) and air sign (P = .027) were considerably lower in SNIP patients with malignant transformation than those without malignant transformation. There were no significant differences in other clinical characteristics and CT features between SNIP patients with malignant transformation and those without transformation. (Supplementary Tables S1-S2)
According to the univariate logistic analysis, age (OR = 1.048, 95% CI, 1.007−1.091, P = .022), epistaxis/blood tinged mucus (OR = 3.143, 95% CI, 1.352−7.306, P = .008), lobulated/wavy edge (OR = .308, 95% CI, .132−.717, P = .006), air sign (OR = .386, 95% CI, .167−.889, P = .025), and focal osseous erosion (OR = 40.200, 95% CI, 9.105−177.498, P < .001) were significantly associated with malignant transformation in SNIP patients. Multivariate logistic regression showed that age (OR = 1.053, 95% CI, 1.001−1.108, P = .044), epistaxis/blood tinged mucus (OR = 4.510, 95% CI, 1.416−14.364, P = .011), lobulated/wavy edge (OR = .306, 95% CI, .102−.918, P = .035), and focal osseous erosion (OR = 38.411, 95% CI, 8.136−181.334, P < .001) were risk factors for malignant transformation in SNIP patients. (Supplementary Table S3)
Nomogram Construction and Validation
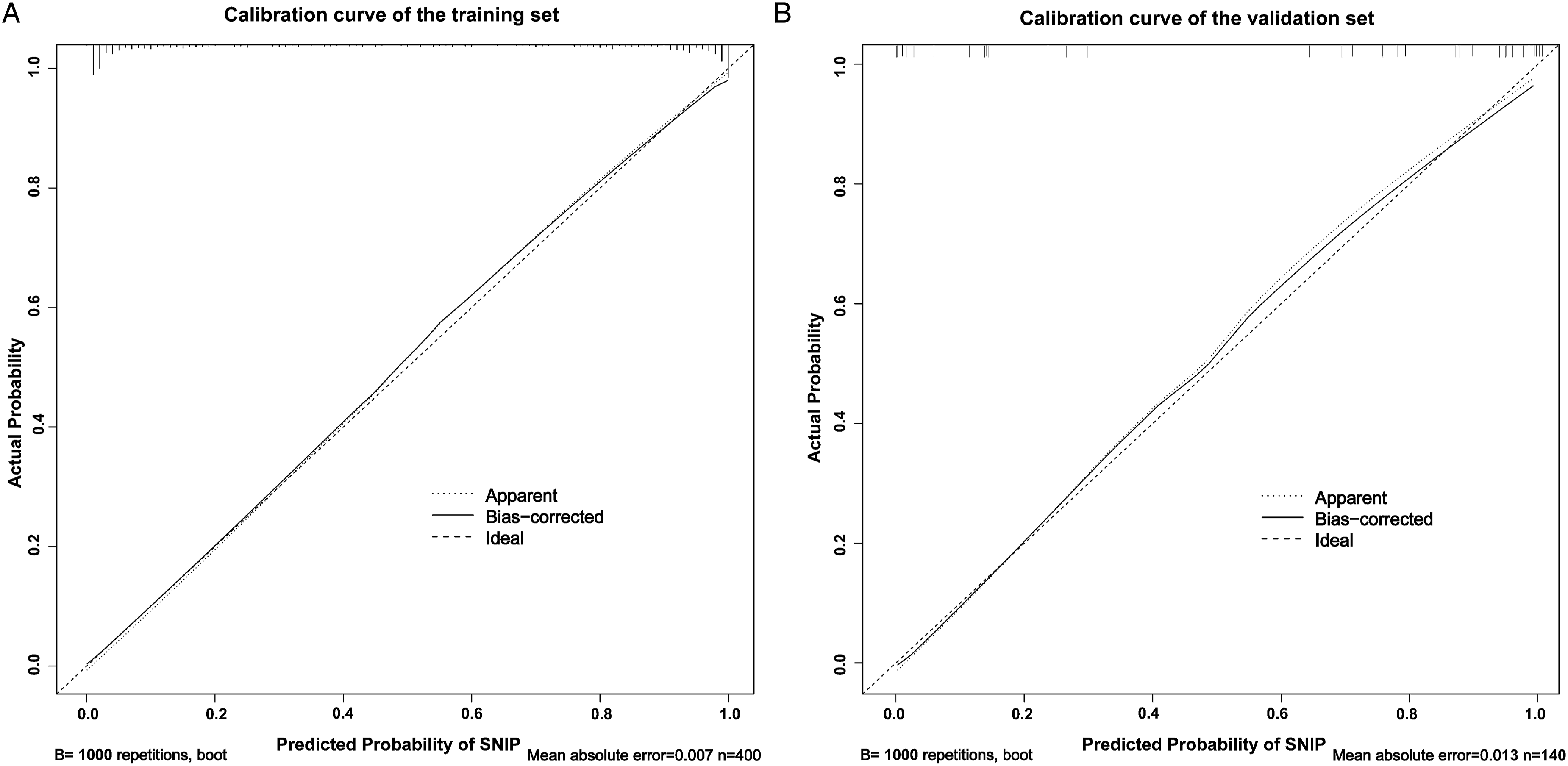
A nomogram was created based on a multivariate logistic analysis of linked factors (age, facial pain/headache, history of sinus surgery, lobulated/wavy edge, air sign, focal hyperostosis, focal osseous erosion, and CT values). Each variable in the nomogram model was assigned by drawing a vertical line from the corresponding point on each variable scale to the highest scale. The scores for all variables were added to obtain total points, and a vertical line was drawn from the corresponding position on the total points scale to the lowest scale to predict the risk of SNIP. Total points of 73, 122, 165, 208, and 256 correspond to 1%, 10%, 50%, 90%, and 99% risk of SNIP, respectively (Figure 2). The nomogram was well-calibrated in the training and validation sets, as shown in the calibration curves (Figure 3). Nomogram for predicting sinonasal inverted papilloma (SNIP). Points are assigned for each factor by drawing a perpendicular line from each factor’s respective value to the uppermost line. The scores for all factors were added to obtain total points, and a vertical line was drawn from the corresponding position on the total points scale to the lowest scale to predict the risk of SNIP. (A) Calibration curve of the training set for predicting sinonasal inverted papillomas. (B) Calibration curve of the validation set for predicting sinonasal inverted papillomas.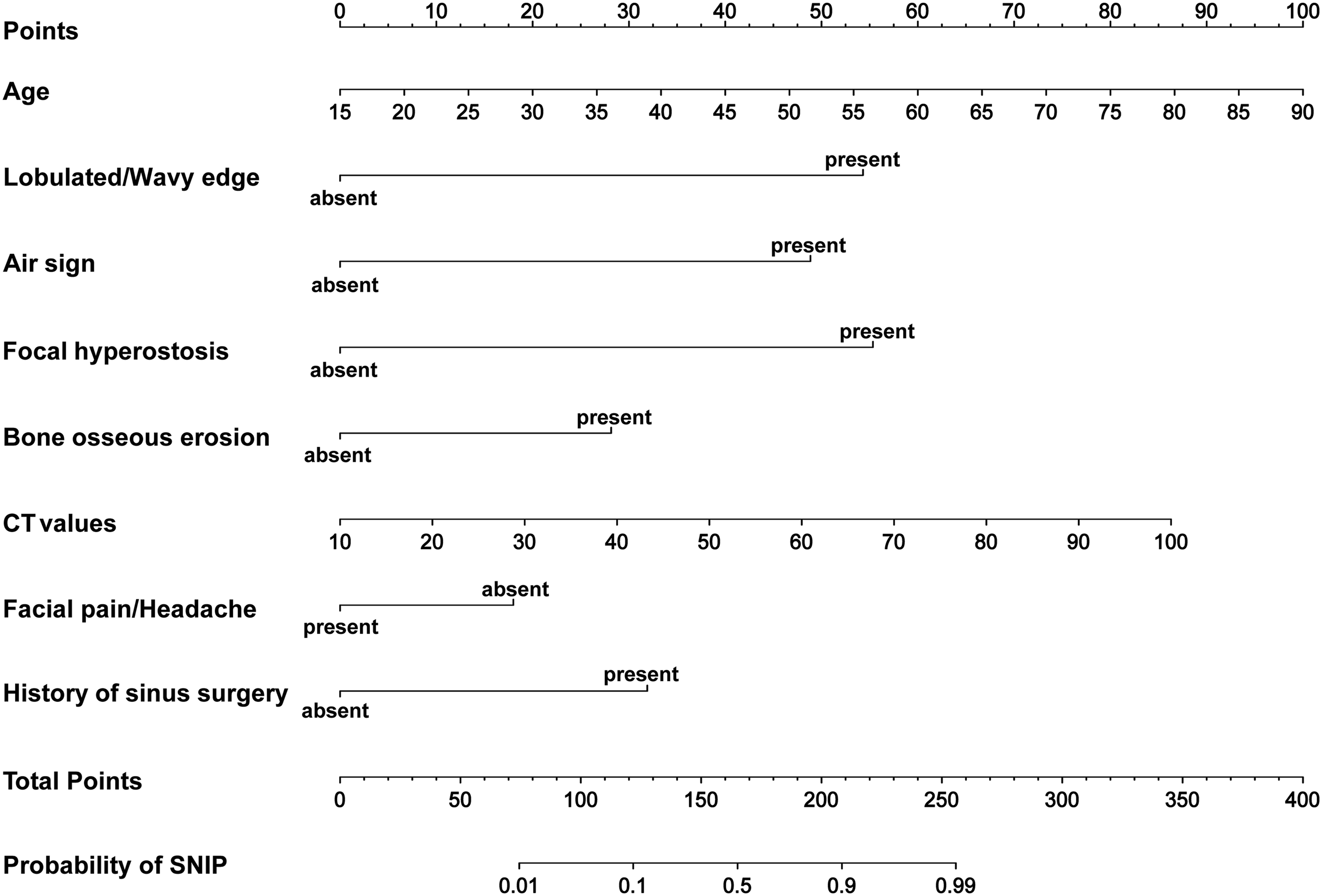
In the training set and the validation set, the area under the curve (AUC) of the nomogram model was .960 (95% CI, .942-.978) and .951 (95% CI, .929-.971), respectively, higher than the AUC of other single variables and showed a better predictive capability. In this view, the nomogram model demonstrated great clinical applicability in SNIP prediction, shown in Figure 4. (A) The receiver operating characteristic curve of the training set for predicting sinonasal inverted papillomas. (B) The receiver operating characteristic curve of the validation set for predicting sinonasal inverted papillomas. AUC, area under the curve.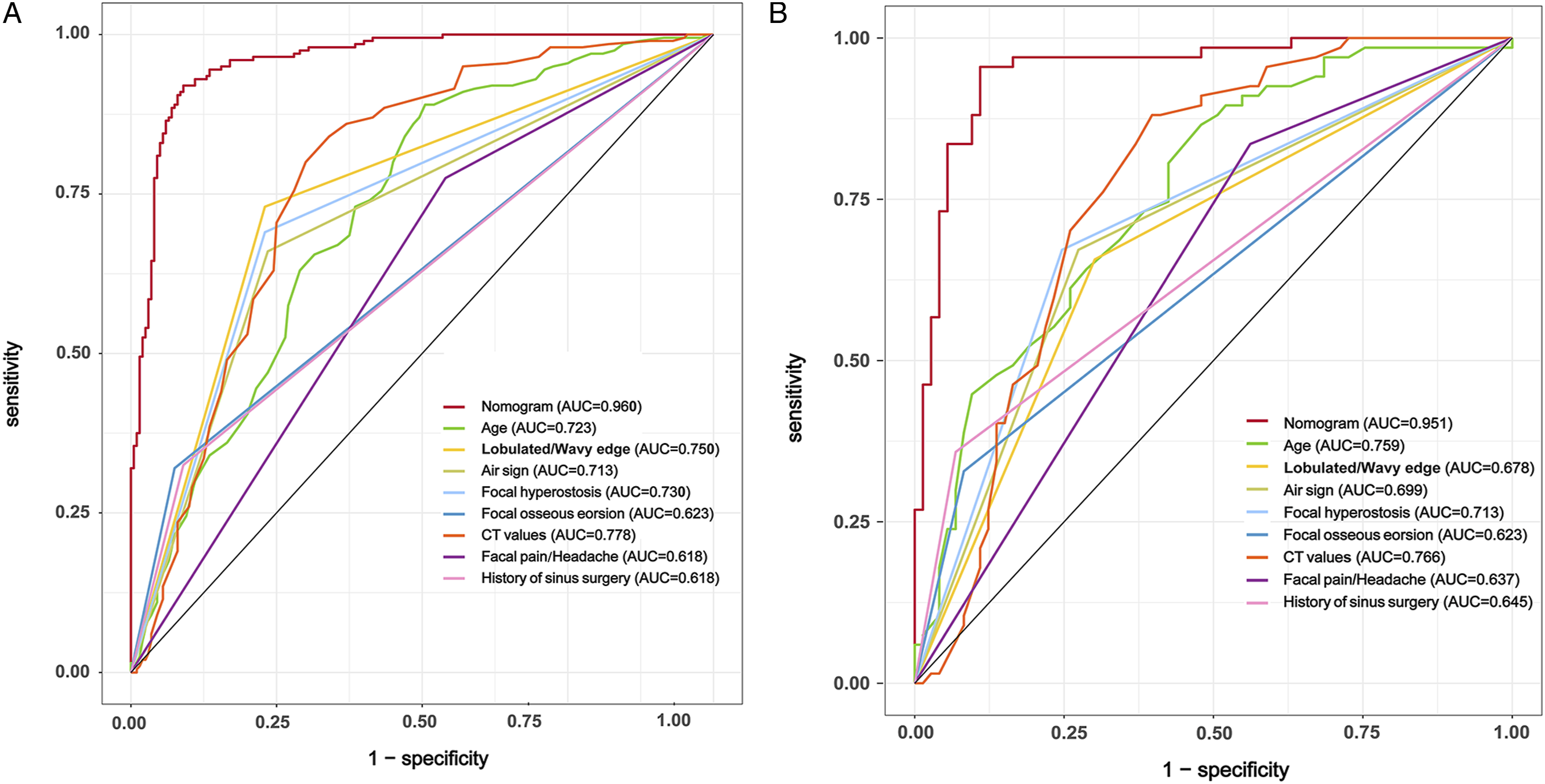
Discussion
Sinonasal inverted papilloma is a benign tumor arising in the Schniderian mucosa, 1 which is primarily treated by endoscopic transnasal approach or combined approach surgery.13-15 Its recurrence rate reaches 12.8% to 44%,15-17 and the incidence of SNIP with malignancy is approximately 2–27%. 18 Thus, early detection and diagnosis of SNIP can serve as the foundation for developing various treatment and follow-up strategies for patients, providing them with early surgical intervention and reasonable surgical techniques, preventing malignancy due to delayed treatment, and avoiding tumor recurrence due to inappropriate selection of surgical procedures. 19
Unilateral CRS with/without nasal polyps is the most common unilateral sinonasal disease, and its clinical signs and imaging examinations are comparable to those of SNIP, resulting in the misdiagnosis of SNIP and unilateral CRS with/without nasal polyps. 20 Studies have confirmed the diagnostic utility of MRI for SNIP: On T2WI and contrast-enhanced T1WI images, tumors display alternating hyperintense and hypointense bands, this characteristic CCP is a reliable indicator of SNIP,9,21 however, many patients did not get MRI testing due to MRI’s limitations, the institution’s limited medical resources, or patient contraindications. Pathology is used to diagnose SNIP as an invasive method, but it might be difficult to identify the kind of lesion on biopsy if the tumor is restricted to the sinus cavity, the lesion is contaminated with inflammatory tissue, or the lesion is deformed. Therefore, there is a need to establish a cost-effective, rapid, noninvasive, and efficient diagnostic method based on sinus CT to predict SNIP, particularly in outpatient and primary healthcare settings.
Sinus CT is an effective diagnostic technique for initial diagnosis and pre-operative SNIP evaluation. Nasal endoscopy and MRI reveal a lobulated and granular proliferation of SNIP, while sinus CT reveals unique signals of SNIP because of this feature. Tumor edges can be lobulated. 3 In our clinical observation, if the lobulated/granular SNIP tissue is densely grouped, the tumor edges may look wavy on sinus CT, which can be an additional indicator of the lobulated lesion edge. An air sign is seen when lobulated/granulated tumor tissue forms an air-filled cavity within the tumor or between the tumor and the nasal cavity and sinus structures. 5 Previous studies only evaluated the sensitivity of these CT signs of SNIP but did not evaluate their value in the differential diagnosis of SNIP and CRS. In our study, we found that CT features can be used to distinguish SNIP from CRS, and CT features of SNIP were incorporated utilizing a nomogram-based variable assignment model to further improve the diagnostic value of sinus CT for SNIP.
Furthermore, SNIP often shows focal hyperostosis and focal osseous erosion on sinus CT.6,8 Lee et al. showed a correlation between focal hyperostosis and the development of SNIP using sinus CT. 22 Unlike CRS’s diffuse, concentric bone formation pattern, SNIP usually shows focal, eccentric bone hyperostosis.8,23 Accumulating evidence suggests that SNIP infiltration into underlying bone tissue causes focal hyperostosis or focal osseous erosion. The study reported by Liang et al. revealed pathologic evidence of SNIP infiltration into bone tissue. 7 Herein, the multivariate logistic analysis showed that focal hyperostosis and focal osseous erosion were associated with the SNIP group. However, diffuse hyperostosis was not significantly correlated with the CRS group, indicating that the obstructive inflammation caused by SNIP may induce a CRS-like hyperostosis pattern in SNIP patients.
CT value corresponds to the X-ray attenuation coefficient of each tissue in a CT scan, enabling the detection of minute variations in human tissue density. 24 According to the reported study by Sano et al., SNIP had higher CT values than nasal polyps. 12 In the present study, CT values of the SNIP group and CRS group were 38.0 ± 10.0 HU and 28.0 ± 12.8 HU, respectively. The SNIP group showed higher CT values than the CRS group, and the ROC curve showed that the CT value at the cut-point was 31.5 HU. It could provide a simple reference parameter for SNIP diagnosis, which is innovative. The reason for this discrepancy may be due to differences in tissue composition: in the case of SNIP, tumors derived from the Schneiderian membrane grow into the stroma and replace the mucous, serous glands, and ducts, 25 whereas in nasal polyps the main components are fibrin and water. 26 CRS, which consists mostly of inflammatory retention and polyp development, contains more water than SNIP, resulting in lower CT values for CRS than for SNIP.
Additionally, the diagnostic values of the demographic and clinical characteristics of SNIP were evaluated. The mean age of onset of SNIP was found to be approximately 55 years, 1 whereas the mean age of onset of CRS was found to be mostly 40-50 years 27 ; SNIP patients have a higher mean age of onset, which is consistent with our study, and multivariate logistic analysis showed that the risk of being diagnosed with SNIP was shown to increase with age.
Facial pain/headache and a history of sinus surgery were other risk factors for a SNIP diagnosis. Common SNIP symptoms are nonspecific and include nasal obstruction, nasal discharge, facial pain/headache, reduction or loss of smell, and epistaxis/blood tinged mucus25,28 These symptoms are similar to CRS and are an important reason SNIP is often misdiagnosed as CRS.29,30 In the present study, the multivariate logistic analysis indicated that facial pain/headache was significantly associated with CRS, which may be due to the pathophysiological mechanism of “sinus headache”: inflammation of the sinus mucosa stimulates neuropeptide release, induces histamine release from mast cells, and activates sympathetic and parasympathetic autonomic pathways, leading to facial pain/headache.31,32 In the SNIP group, the facial pain/headache symptoms were less severe than in the CRS group because the inflammation of the sinus mucosa was milder than in the CRS group.
Some SNIP patients may have undergone one or more sinus surgeries in the past. Even if the patient reports having undergone a nasal polypectomy in the past, extra consideration must be taken. According to the reported studies, 16% of patients diagnosed with nasal polyps before surgery were eventually diagnosed with SNIP on pathology. 20 In early diagnosis and treatment mode, or some areas with poor medical resources, SNIP may be misidentified as nasal polyps and excised without pathological examination, 16 or the diagnosis may be misdiagnosed due to insufficient pathological tissue for examination. 33 Furthermore, some outpatients reported ambiguity in the pathology diagnosis of past sinus procedures. As a result, if there is a history of one or more sinus surgeries and imaging findings suggesting sinus lesions, the possibility of SNIP should be considered.
Furthermore, the statistical differences between SNIP patients with or without malignant transformation were examined. Multivariate logistic regression revealed that age, epistaxis/blood tinged mucus, lobulated/wavy edge, and focal osseous erosion were risk factors for malignant transformation in SNIP patients. Malignant tumors tend to have a higher age of onset and a more pronounced tendency for osseous erosion, and bleeding from tumor tissue can result in epistaxis/blood tinged mucus. Moreover, since SNIP with malignant transformation grow faster and have larger tumor sizes, resulting in complete contact of the border with the bone wall without assessment of the border states, this may contribute to the disappearance of the lobulated/wavy edge and the presence of the air sign.
The current study is associated with several limitations. First, this investigation was carried out from a single institution. Future multicenter, large sample studies are needed to validate this nomogram model further. Second, although the nomogram established based on non-contrast sinus CT findings has high predictive value, future studies on contrast-enhanced CT or MRI are needed to optimize the SNIP diagnostic model. Third, although unilateral CRS with/without nasal polyps is the disease most in need of identification for SNIP, other unilateral benign and malignant sinonasal lesions should be included in the future to expand the applicability of this nomogram. Furthermore, to obtain predictive models with high diagnostic power, eight parameters were included in our nomogram, doctors may spend several minutes calculating nomogram scores, which can be solved by automated approaches in the future. Although this nomogram enrolled common CT signs, it still requires some experience from the radiologist.
Supplemental Material
Supplemental Material - Development and Validation of a Clinical Prediction Model to Diagnose Sinonasal Inverted Papilloma Based on Computed Tomography Features and Clinical Characteristics
Supplemental Material for Development and Validation of a Clinical Prediction Model to Diagnose Sinonasal Inverted Papilloma Based on Computed Tomography Features and Clinical Characteristics by Zengxiao Zhang, Longgang Yu, Jiaxin Jiang, Lin Wang, Shizhe Zhou, Dapeng Hao, and Yan Jiang in Ear, Nose & Throat Journal
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (81770978), Key Technology Research and Development Program of Shandong (2018GSF118012), Project of Medical and Health Technology Development Program of Shandong Province (2016WS0268).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
