Abstract
Importance:
Operative laryngoscopy is a commonly performed ambulatory procedure in patients with significant co-morbidity. Optimal anesthetics for surgical exposure with rapid return to baseline after the procedure enhances postoperative patient safety.
Objective
To determine whether sugammadex hastens recovery in patients undergoing operative laryngoscopy under general anesthesia with rocuronium-induced paralysis.
Design
Prospective clinical intervention randomized single-blinded, single-center study in an academic tertiary care center. Approved by the institutional review board and registered with ClinicalTrials.gov
Settings
Single center tertiary care academic institution.
Participants
18 years or older, American Society of Anesthesiology physical status I–III with ability to give written informed consent undergoing operative laryngoscopy.
Intervention
Participants were randomized into two groups. Both groups received inhaled anesthetic: sevoflurane, remifentanil, and rocuronium at 0.6–1.2 mg/kg for intubation and anti-nausea prophylaxis. Group 1 received reversal with neostigmine (0.04 mg/kg) and glycopyrrolate (0.01 mg/kg). Group 2 received reversal with sugammadex (4 mg/kg). Vital signs were maintained at 20% of baseline in both groups. Post anesthesia care unit nurses were blinded to the reversal agent and were the evaluators of the discharge criteria and times. Primary end point was time to extubation after the procedures and secondary end points were: Subjective interpretation of surgical conditions by the surgeon, hemodynamic, respiratory parameters, anesthetics, and opioids used, operative time, and duration to achieve discharge readiness.
Results
A total of eighty-four participants, who were similar in age, sex, and weight in both groups. The primary end point and secondary end points were similar except time to meet discharge criteria in the two groups. 65% in the sugammadex versus 35% in the neostigmine group met Aldrete criteria of 18 or higher on arrival at the post anesthesia care unit.
Conclusions
Optimizing the anesthetic regimen, along with stable intraoperative hemodynamics and reversal with sugammadex improves discharge readiness in patients undergoing operative laryngoscopy.
Introduction
Operative laryngoscopy (OL) for oropharyngeal, laryngeal, and tracheal procedures directly stimulates visceral afferent fibers in the pharyngeal plexus of the glossopharyngeal nerve and the internal laryngeal branch of the vagal nerve. This can elicit an intense sympathetic response or patient movement which can cause physical or physiological harm to the patient. 1 An optimal anesthetic for OL would provide patient comfort, safety, and paralysis of the masticatory and laryngeal muscles during the procedure, followed by rapid extubation at the conclusion of the procedure to aid in operative room turnover. Due to the pharmacodynamics and pharmacokinetics of older anesthetic drugs, the above goals were often difficult to achieve, and an optimal regimen has remained elusive. Previous anesthetic approaches to achieve akinesis and rapid recovery have included deep sedation, nondepolarizing muscular blockade, and depolarizing muscular blockade with a succinylcholine drip. 2,3 However, the duration of action of nondepolarizing muscle blockade (NMBD) medications was often longer than the operative laryngoscopy procedure itself, even in spite of reversal of neuromuscular blockade (NMB) with an acetylcholinesterase inhibitor such as neostigmine. 5 More recent approaches have attempted to optimize exposure and decrease time to extubation by eliminating the need for endotracheal intubation and NMB through the use of total intravenous anesthesia (TIVA) and the maintenance of spontaneous ventilation. 4 While this technique decreased time to extubation and operating turnover, surgical exposure was less than optimal due to inadequate paralysis of laryngeal and masticatory muscles.
Sugammadex (Bridion) is a new pharmacologic agent that provides rapid reversal of rocuronium even after high dose administration used for rapid sequence intubation. Its rapid reversal of NMB makes it an attractive option in the anesthetic care of patients undergoing OL. The aim of this study was to compare time to extubation in patients randomly assigned to the use of sugammadex or neostigmine for the reversal of neuromuscular blockade. Our hypothesis was that patients receiving sugammadex for NMB reversal would have significantly lower mean time to extubation compared to patients receiving neostigmine.
Methods
A prospective clinical intervention randomized single center was employed. The institutional review board (IRB)-approved study is registered with clinicaltrials.gov. After IRB approval, a written IRB-approved consent form was used to obtain consent from participants who met the inclusion criteria. Inclusion criteria included participants who underwent operative laryngoscopy, had American Society of Anesthesiology (ASA) physical status I–III, were able to provide written informed consent, and were 18 years or older. The exclusion criteria were as follows: • Known or suspected neuromuscular disease/pre-existing weakness • Creatinine clearance less than 30 ml/min • Bradycardia of fewer than forty beats/min • Pregnant or breastfeeding women • Known or suspected allergy to sugammadex, neostigmine, or rocuronium • Participants with contraindications toward sugammadex, neostigmine, or rocuronium • Participants included in another trial within the last 30 days • Participants with legal guardians or surrogate decision-making • Participants who refused to use the non-hormonal contraceptive method or backup method of contraception (such as condoms and spermicides) for the next seven days.
Participants were drawn from among patients of the otolaryngologists participating in the study and the treatment was offered to all eligible participants.
The participants were placed in one of two anesthetic groups using a block randomization scheme with block sizes varying from two to six participants per block. Participants received a unique study ID number (participant 1: i.e., 0001- {3 initial, i.e., A-B-C}). The statistician developed the randomization scheme; the master list of study IDs and reversal treatment allocations were held by the research pharmacist and research coordinator. Strict adherence to the sequence of treatment allocation was maintained.
Group 1: Inhaled anesthetics: sevoflurane at 1 MAC, remifentanil, and intubation with rocuronium at 0.6–1.2 mg/kg (vitals maintained within 20% of baseline). Group 1 received reversal with neostigmine (0.04 mg/kg and glycopyrrolate (0.01 mg/kg)).
Group 2: Inhaled anesthetics: sevoflurane at 1 MAC, remifentanil, and intubation with rocuronium at 0.6–1.2 mg/kg (vitals maintained within 20% of baseline). Group 2 received reversal with sugammadex 4 mg/kg. Both groups received a standard anti-nausea prophylaxis of Ondansetron and dexamethasone intraoperatively. After induction, the quantum of inhaled anesthetic and remifentanil was titrated based on hemodynamic parameters (maintained within 20% of baseline) a Bispectral Index (BIS) monitor and a train of four monitor was placed. All participants underwent train of four testing with a peripheral nerve stimulator every five minutes throughout the procedure and were tabulated in the Electronic Medical Record (EMR). The train of four count was maintained at two and under for optimal surgical exposure. The anesthetic was titrated to maintain a BIS between 45 and 55 during the procedure. At the end of the procedure, the participants were extubated when they meet the following criteria: tidal volume, > 5 cc/kg; respiratory rate: >8 /min, O2sat > 95%; ON, 100% inspired oxygen; and vitals, 20% of baseline. Extubation process began when the surgeon stated, “We are done,” which usually coincided with the withdrawal of the laryngoscope. The start and end times for extubation were recorded in the EMR. Reversal agent was administered when patients had a minimum of one out of four in the train of four count peripheral nerve stimulator. All patients were extubated after they had all four counts on the train of four monitor.
The post anesthesia care unit (PACU) evaluated Aldrete discharge criteria and noted the participants discharge times from PACU in the EMRs. The PACU nurses were the only patient evaluators and were blinded to the two groups.
The following outcomes were evaluated
Primary End point
• Time to extubation after the end of the procedure. Our institution utilizes EMRs to chart the end of the procedure. The emergence and extubation process always begins with the withdrawal of surgical instruments. The start and end times of extubation were recorded on the EMR.
Secondary End points
• Subjective interpretation of optimal surgical conditions as evaluated by the surgeon for ease of exposure were graded within 1–10 (1 being difficult and 10 being the best surgical conditions). • Time taken to meet Aldrete criteria in PACU.
Peri-Operative End points:
• Hemodynamic parameters: Intraoperative and postoperative changes in blood pressure, heart rate, and carbon dioxide levels from the baseline were evaluated. Any variations greater than a 20% change from the preoperative baseline vital signs were considered significant. • Respiratory parameters: intraoperative and postoperative. • Inhaled anesthetic concentration intraoperative. • Cumulative opioids required. • Total operative time. • Total PACU time.
Statistical Analysis
Preoperative and intraoperative characteristics by group.
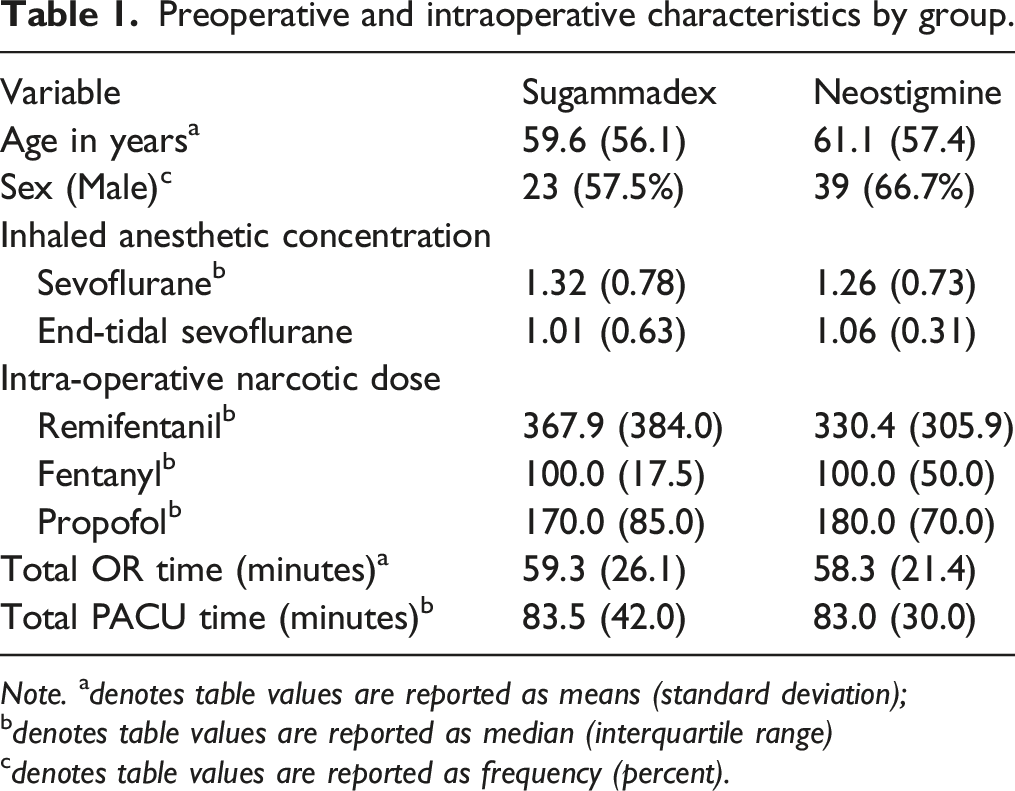
Note. a denotes table values are reported as means (standard deviation);
b denotes table values are reported as median (interquartile range)
c denotes table values are reported as frequency (percent).
We used the Kaplan–Meier method and log-rank test to assess the extubation time between the two groups after the procedure ended. Statistical significance was assessed using a two-sided test with an alpha-level of 0.05. All statistical analysis was performed using SAS v. 9.4 (SAS Institute, Cary, NC).
Results
A total of 84 participants were enrolled in the trial and were randomized to one of two groups, the sugammadex and neostigmine groups, using a blocked randomization scheme. Five participants were excluded due to (a) refusal day of surgery, (b) change of procedure, (c) lack of intubation, or (d) did not receive rocuronium for the procedure. The final analytic sample consisted of 79 participants, of whom 40 (50.6%) received sugammadex and 39 (49.4%) received neostigmine.
The sugammadex and neostigmine groups did not differ significantly by age. The mean age of participants who received sugammadex was 61.1 compared to 59.6 for participants who received neostigmine. The groups also did not differ significantly by gender. In the sugammadex group, 23 of 40 (57.5%) participants were male, and in the neostigmine group, 26 of 39 (66.7%) participants were male.
Participants in the sugammadex and neostigmine groups were equally balanced. We found no significant differences between the sugammadex and neostigmine groups in terms of intraoperative and postoperative hemodynamic parameters, respiratory parameters, or opioid usage. Similarly, we found no significant differences between the groups in total OR time, total PACU time, or intraoperative inhaled anesthetic concentrations (Table 1). No significant differences were found in weight and total amount of rocuronium administered in both groups were similar with an average of 0.71mg/kg in sugammadex group and 0.68 mg/kg in neostigmine group. The average train of four count at the end of the procedure was 2 in both groups and the count through the procedure measured every 5 minutes were similar in both groups.
For our primary outcome, we did not find a significant difference between participants in the sugammadex and neostigmine group in extubation time after the procedure ended (LR = .16, p = 0.6906 (see Figure 1). For our secondary outcome, we found a significant association between groups and Aldrete score ≥ 18 upon arrival in the PACU. Participants in the sugammadex group were 2.97 times more likely to have an Aldrete score of 18 or higher upon arrival in the PACU than were participants in the neostigmine group, X2 = 5.57, p = 0.02, OR = 2.97 (95% CI: 1.07, 2.67). For the secondary outcome of exposure rating by surgeon, we did not find a significant difference between the two groups. Both the sugammadex and Neostigmine groups had a median rating of 10 on a 1–10 scale (see Table 2). Kaplan–Meier curve by group for time in minutes from end of procedure to extubation. Secondary End points. Note. adenotes table values are reported as median (interquartile range)
bdenotes table values are reported as frequency (percent). An effect size was not provided for surgeon exposure scale rating as this was a non-significant finding.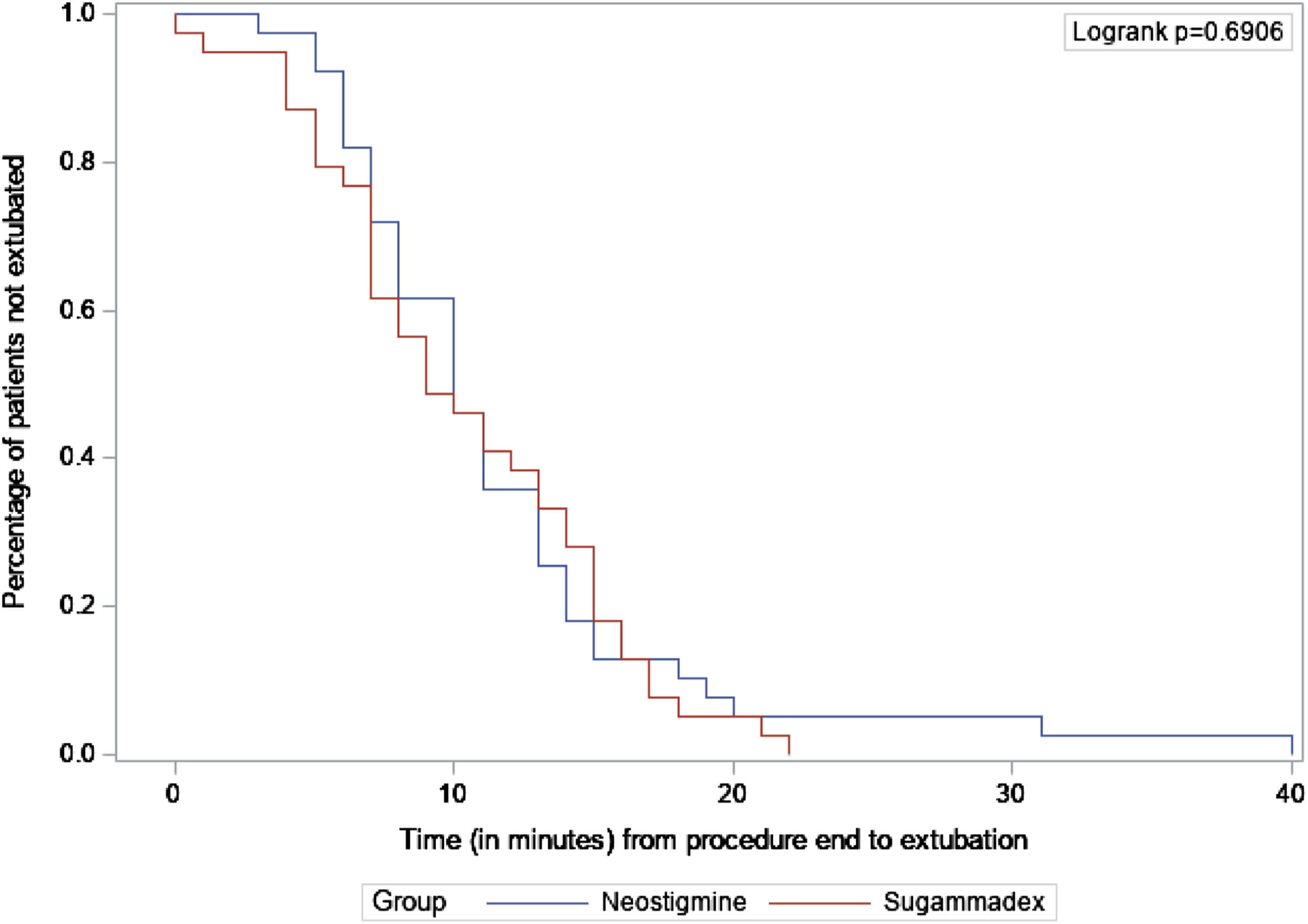

Discussion
Anesthetic technique for any surgical procedure should attempt to facilitate surgical exposure, promote efficient utilization of peri-operative care, and maximize patient safety. Operative laryngoscopy is often a brief procedure with a potential for adverse airway events. Consequently, neuromuscular blockade with nondepolarizing neuromuscular blocking drugs is often avoided because of the need for rapid reversal of neuromuscular blockade after the procedure. 6 However, neuromuscular blockade is desirable to facilitate surgical exposure during the stimulation procedure. Our study sought to determine whether using sugammadex to reverse neuromuscular blockade would help meet the challenges of providing anesthesia for operative laryngoscopy.
Our study aimed to determine if sugammadex use resulted in a faster return to baseline and improved times to discharge. This allows for more practical use of neuromuscular blockade during operative laryngoscopy.
On standardizing the time to extubation by using a remifentanil infusion with a constant half-life of 10 minutes, differences in times to meeting discharge criteria can be clearly attributable to the reversal agent utilized. Remifentanil infusion was chosen as part of a balanced anesthetic because it is an opioid that can be administered as a standardized weight-based infusion. In addition, it is metabolized by blood and tissue esterase’s and has a predictable elimination half-life in all participants within 10 minutes. 7 In our study, remifentanil infusion was discontinued when the surgeon announced the end of the procedure. Future studies using a multimodal pain control regimen that exclude opioids may also help validate our conclusions.
The use of a nondepolarizing neuromuscular blocking drug for operative laryngoscopy helps reduce the quantum of inhaled anesthetics and opioids needed. 1 -5 An overall decrease in anesthetic requirements is a desirable goal for a patient population with significant co-morbidities, as is often the case for those undergoing operative laryngoscopy. Post-operative cognitive dysfunction and hemodynamic instability can cause significant long-term effects in participants.
We found that a significant number of participants in the sugammadex group received an Aldrete score of 18 or higher on arrival to the PACU, indicating that they were ready for discharge. The Aldrete score determines readiness for discharge based on the following criteria: activity, respiration, circulation, consciousness, Oxygen saturation, dressing, pain, ambulation, fasting feeding, and urine output. 8,9 Decreased time in PACU results in improved processes, while shortening the time of return to baseline in these participants undergoing operative laryngoscopy increases satisfaction for participants, families, and the perioperative surgical team. Deep neuromuscular blockade with rocuronium provides superior surgical exposure 3 while allowing for anesthetic requirements to be lower than when participants do not receive neuromuscular blockade or when the blockade wears off as in the case of succinylcholine.
Operative laryngoscopy is performed on benign and malignant conditions from vocal cord nodules to laryngeal papilloma and carcinoma of the vocal cords. 10 . One study reported 33% of cases as being performed on laryngeal malignancy, indicating a high population of participants with significant co-morbidity. 11 In instances with significant co-morbidities, deep sedation is not ideal as deeply anesthetizing these participants can cause profound hypotension, worsening the patient’s medical status and resulting in longer PACU stays, leading to increased postoperative complications.
Cases of those with significant co-morbidities shed light on the argument for the use of muscle relaxants. Muscle relaxants are synergistic with inhaled anesthetics and opioids. Hence, the total amount of anesthetics and opioids used can be significantly reduced when combined with muscle relaxants. 1 If it is possible to fully reverse the paralysis at the end of the procedure efficiently, then the ideal approach would be the utilization of a multimodal regimen for anesthesia, thereby reducing the quantum of anesthetics and opioids when used in conjunction with muscle relaxants. This can potentially decrease the incidence of post-operative cognitive dysfunction especially associated with the elderly population. 10 Post-operative cognitive dysfunction can cause significant morbidity in patients with preexisting conditions that can increase the risk of post-operative cognitive dysfunction. 11 Reducing the quantum of anesthetics enhances hemodynamic stability during the procedure. In addition, muscle relaxants should effectively blunt the airway response, keep the patient akinetic, and optimize the surgical exposure. This “balanced” anesthetic would be ideal for surgical exposure, hemodynamic stability, and reduced post-operative cognitive dysfunction especially in patients with multiple medical conditions.
Sugammadex is a collection of γ-cyclodextrins fitted with eight identically modified side chains containing propionic acid.12 This allows for a hydrophobic inner cavity that chelates the muscle relaxant and a hydrophilic exterior to maintain the structure. 12 This structure makes Sugammadex unique and the first approved direct reversal of vecuronium and rocuronium. A Cochrane systematic review found that sugammadex reversed neuromuscular blockade among adults 10.22 minutes faster than neostigmine. 13 -17 Therefore, it seems logical that the use of depolarizing muscular relaxants such as rocuronium and reversal with Sugammadex will allow for optimal surgical exposure and immobility, while maintaining more hemodynamic stability in participants with significant co-morbidities and attenuating the risk of post-operative cognitive dysfunction.
The limitations of this study include the lack of an additional arm using succinylcholine and the use of an acceleromyograph. In future studies, inclusion of a third group with succinylcholine may be helpful as a comparator. In addition, utilizing an acceleromyograph rather than the standard train of four twitch monitor may provide a better understanding of the real time depth of neuromuscular blockade. The average PACU discharge time in both groups, irrespective of Aldrete criteria, was 81 minutes. The average discharge time in our institution was 82 mins during the period of our study, regardless of the type of surgical procedure. The lack of variance in discharge time is indicative of the impact of perioperative culture and tradition at each institution on operational efficiency.
Conclusion Paragraph
Anesthetic optimization and reversal with Sugammadex accelerate discharge readiness in participants undergoing operative laryngoscopy.
Footnotes
Author’s Note
Question: Does reversal of neuromuscular blockade using Sugammadex during operative laryngoscopies hasten discharge readiness after operative laryngoscopy. Findings: In this randomized clinical trial, more participants in the Sugammadex group met the postoperative discharge criteria on arrival to the recovery room than the group that received neostigmine and glycopyrrolate for reversal of neuromuscular blockade with rocuronium.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Merck Sharp and Dohme, 006629-00001
