Abstract
Background
More patients with oropharyngeal squamous cell carcinoma (OPSCC) in Eastern countries receive surgically inclusive treatment (SIT), while most patients in Western countries receive nonsurgical treatment (NST). The optimal treatment modality for OPSCC patients remains controversial.
Methods
A total of 153 consecutive OPSCC cases diagnosed between 2009 and 2019 in West China Hospital (WCH) and 15,400 OPSCC cases from the Surveillance, Epidemiology, and End Results (SEER) database (2000–2017) were obtained. Clinical characteristics, treatments, and survival outcomes were retrospectively collected. We constructed Kaplan–Meier curves and performed univariate (UVA) and multivariate (MVA) analyses to compare the prognosis of OPSCC patients among the WCH, SEER Asian, and SEER all ethnic populations by different treatment modality, human papilloma virus (HPV) infection status, age, and tumor stage.
Results
Overall, the proportions of patients with younger age, advanced tumors and HPV-negative status, and receiving SIT in WCH population were higher than those in the SEER all ethnic population, while the proportions in the SEER Asian population were between those of the other two populations. We observed consistent beneficial effects of SIT on the overall survival (OS) in OPSCC patients in all three populations (SEER Asian: MVA, hazard ratio (HR): 0.2, p < .001; SEER all ethnic: MVA, HR: 0.46, p < .001; WCH: UVA, HR: 0.62, p = .071), and HPV-negative Asian patients showed greater benefits from the SIT than HPV-positive Asian patients (HPV Negative: HR: 0.16, p = .005; HPV positive: HR = 0.28, p = .059). Male was a risk factor for reduced OS in OPSCC patients in the WCH population (HR: 3.17, p = .043), but was a protective factor in the SEER population (HR: 0.8, p = .002), which might be related to the differences of HPV infection status.
Conclusions
Even though differences in patient characteristics existed between the Chinese, American, and Asian American populations, our ten-year real-world data and SEER data suggested that patients with OPSCC who received SIT had a better prognosis than those who received NST.
Introduction
The incidence of oropharyngeal squamous cell carcinoma (OPSCC) has been increasing since the twentieth century, with 92,877 new cases and 51,005 deaths reported globally in 2018. 1 The epidemiology and prognosis of OPSCC have changed dramatically over decades, with fewer tobacco-related cases and more human papillomavirus (HPV)-related cases.2,3 Because HPV-positive patients are more sensitive to radiotherapy and HPV infection rates are higher in Western countries, 4 the treatment preferences for OPSCC patients vary between Eastern and Western countries, and more patients with OPSCC in Western countries received nonsurgical treatment (NST).5,6
Since open surgery is associated with serious complications, such as long-term tracheal cannulas use, gastric tube feeding, and high recurrence, as well as a high mortality rate, the primary management strategy for OPSCC patients has gradually shifted toward NSTs 7 such as radiotherapy (RT) and chemoradiotherapy (CRT). 8 Although chemotherapy and radiation can induce toxicity 7 in OPSCC patients, some studies have demonstrated that radiation and/or chemotherapy leads to better survival outcomes and organ prevention in OPSCC patients in Western countries.9,10 In China, however, OPSCC patients are more likely to undergo surgically inclusive treatment (SIT) than NSTs.3,11,12
Due to the lack of adequately robust randomized trials, the best standard treatments for OPSCC patients remains controversial, and the preferred treatment is usually based on institutional preference13–16 and patients’ overall health status. In this study, we herein analyzed whether SIT was beneficial for the survival outcomes in OPSCC patients by comparing patients in West China Hospital (WCH) to those in the Surveillance, Epidemiology, and End Results (SEER) database.
Method
Data Collection From West China Hospital
This study was approved by the Ethics Committee on Biomedical Research, West China Hospital of Sichuan University (No. 2019-357). Patients diagnosed with primary OPSCC and treated in WCH between January 1, 2009 and December 31, 2019 were retrospectively and consecutively enrolled in this real-world study. Age, gender, TNM classifications, tumor stages, and treatments (surgery, radiotherapy, chemotherapy, and chemoradiotherapy) data were collected from electronic medical records (EMRs). Data on recurrence, survival status, and survival months were obtained from patients by regular follow-up visits or telephone visits. Aged patients were defined as those over 60 years of age at the time of diagnosis.
P16 Immunohistochemical Staining
The HPV status of OPSCC patients in WCH was determined by p16 immunohistochemical (IHC) staining of tumor tissue. Immunohistochemical staining of formalin-fixed paraffin-embedded (FFPE) tumor tissues from OPSCC patients with mouse anti-human p16 monoclonal antibodies (BD Pharmingen™, Cat.No.550834) was conducted. The IHC scoring results were independently analyzed by two pathologists. Tumor p16 expression was scored as positive if strong and diffuse nuclear and cytoplasmic staining was present in 70% of the tumor cells. 17
Data Collection From the SEER Database
Patients diagnosed with OPSCC between January 1, 2000 and December 31, 2017 in the SEER database were identified. The codes of the primary site of OPSCC (International Classification of Diseases for Oncology, 3rd edition, ICD-O-3) included the base of tongue (C019 and C024), soft palate (C051), uvula (C052), tonsil (C090-091 and C098-C099), and oropharynx (C100-104 and C108-109). Histology was restricted to squamous cell carcinoma (morphology codes 8070/2-8078/3 in the ICD-O-3). Tumor stages were classified by the American Joint Committee on Cancer (AJCC) 7th edition staging system. Patients without pathological, T stage, or N stage data were excluded. Data on age, gender, ethnicity, TNM classifications, tumor stages, treatments, and survival status were collected. Considering the possible impacts of ethnic differences on survival outcomes and treatment selection, we performed further comparisons of OPSCC patients in the WCH population, SEER Asian population, and SEER all ethnic population.
Treatments and Outcomes
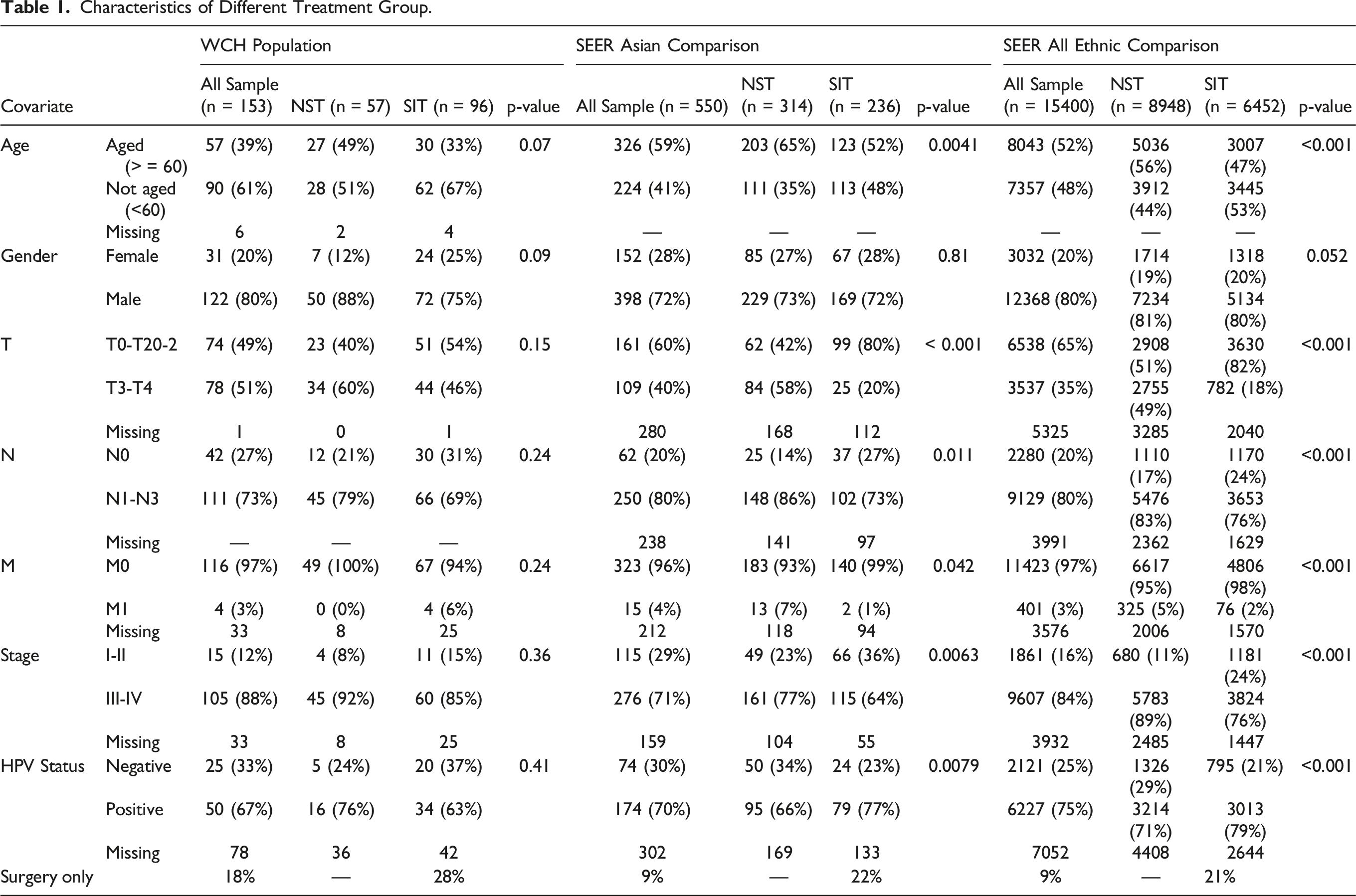
Characteristics of Different Treatment Group.
Subgroup Analysis
We further conducted subgroup analyses according to HPV status (positive vs. negative), gender (male vs. female), and tumor stage (stage I-II vs. stage III-IV) to compare differences in clinical variables and their correlations with treatment modalities.
Statistical Analysis
All analyses were carried out using R (version 3.6.3). The Kruskal–Wallis test, chi-square test, or Fisher exact test were used to compare baseline characteristics among different treatment groups in both the WCH and SEER populations. OS and progression-free survival (PFS) were calculated using the Kaplan–Meier method. The log-rank test was utilized to compare survival outcomes among different groups. The hazard ratio (HR), adjusted HR (aHR), and 95% confidential interval (CI) for different clinical variables were calculated using univariate (UVA) and multivariate (MVA) Cox proportional hazards models. A two-sided test was conducted, and significance was attributed to a P value of < .05.
Results
Patient Population and Clinical Characteristics
A total of 176 consecutive OPSCC patients diagnosed between 2009 and 2019 in WCH were identified. After removing patients with non-squamous carcinoma and those without treatment records, 153 OPSCC patients were finally included. A total of 15,400 OPSCC cases (all ethnic) and 500 Asian OPSCC cases diagnosed between 2000 and 2017 in the SEER database were included for comparison. As shown in Table 1, male patients predominated in all three populations (80% in the WCH group; 72% in the SEER Asian group; and 80% in the SEER all ethnic group). There were more young patients (61%) in the WCH population, whereas older patients (59% in SEER Asian, p = .0041; 52% in SEER all ethnic group, p < .001) were the majority in the SEER population. In the WCH population, approximately 51% of OPSCC patients had advanced T classifications (T3-T4), while more OPSCC patients had early T classification (T0-T2) in both the SEER Asian (60%, p < .001) and SEER all ethnic (65%, p < .001) populations. Human papilloma virus positive rates were the highest in the SEER all ethnic population (75%, p < .001), followed by the SEER Asian population (70%, p = .0079), and the WCH population was the lowest (67%).
Treatment-Related Characteristics and Overall Survival
Surgically inclusive treatment was the primary treatment modality for OPSCC patients in the WCH population (n = 96, 63%), while NST (RT, CT, and CRT) was more common in both the SEER Asian (n = 314, 57%) and SEER all ethnic (n = 8,948, 58%) populations (Table 1). In the WCH population, the baseline characteristics (e.g., gender, lymph node involvement, metastasis, clinical stage, and HPV status) were not significantly different between the SIT and NST groups. However, among the SEER Asian and SEER all ethnic populations (Table 1), a higher proportion of patients who received SIT had early T classifications (T0-T2, 80% and 82%, p < .001, respectively) and more were HPV positive (77%, p = .0079, and 79%, p < .001).
The 5-year OS rate in the WCH population was 59%, which was slightly lower than that in the SEER Asian (64%) and the SEER all ethnic populations (67%). As shown in Figure 1, Table 2, and Sup Table 1, SIT was significantly associated with superior OS in the SEER Asian (MVA, HR: 0.2, 95% CI: 0.08∼0.52, p < .001) and SEER all ethnic populations (MVA, HR: 0.46, 95% CI: 0.41∼0.52, p < .001). Surgically inclusive treatment also showed a trend toward a beneficial prognosis in the WCH population (UVA, HR: 0.62; 95% CI: 0.37∼1.04, p = .0071; MVA, HR: 0.8, 95% CI: 0.46∼1.37, p = .41), while the multivariate-adjustment results did not show statistical significance. Main Outcomes of West China Hospital population and SEER population. A. Overall survival assorted by treatments in West China Hospital group; B. Overall survival assorted by treatments in SEER Asian group;. C. Overall survival assorted by treatments in SEER all ethnic group. Multivariable Cox Model Regression Analysis on Overall Survival.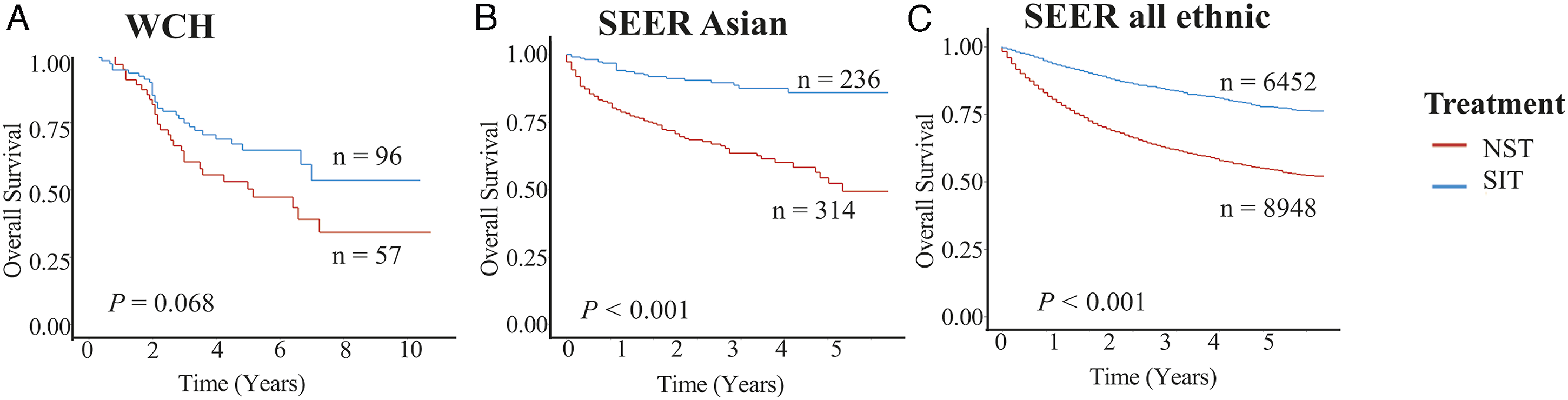
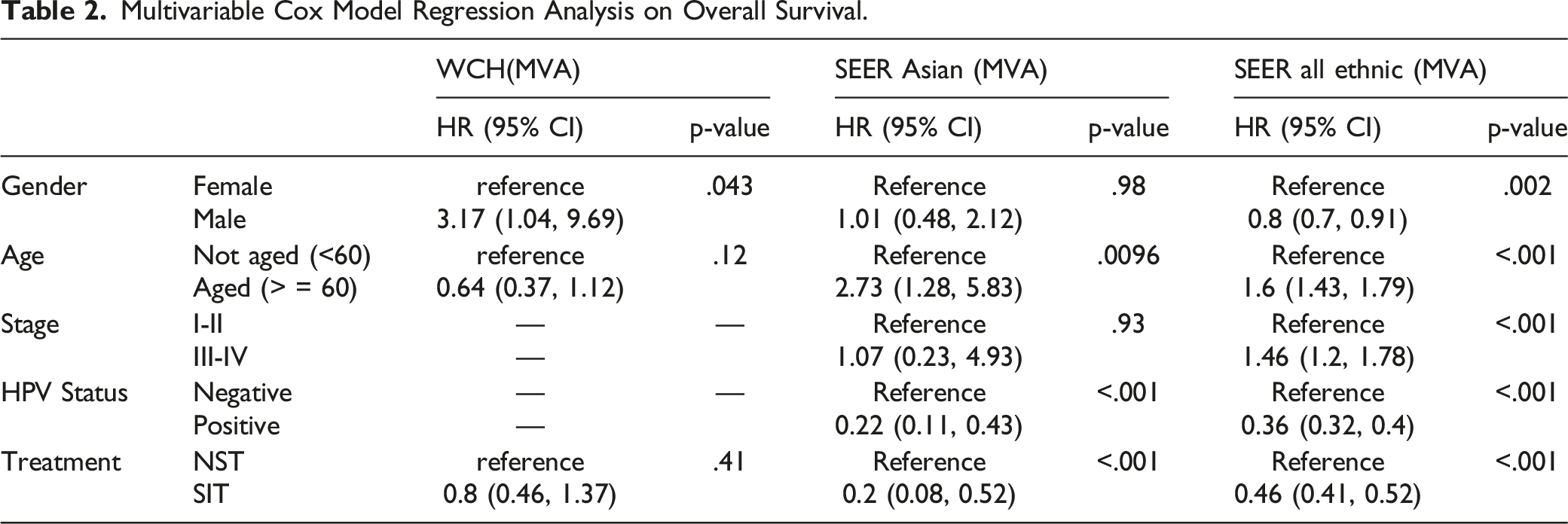
Interestingly, male was a risk factor for reduced OS in OPSCC patients in the WCH population, but was a protective factor in the SEER population. In addition, HPV-positive OPSCC patients in the SEER Asian (MVA, HR: 0.22, 95% CI: 0.11∼0.43, p < .001) and SEER all ethnic populations (MVA, HR: 0.36, 95% CI: 0.32∼0.4, p < .001) experienced better survival than HPV-negative patients. However, this trend was not significant in the WCH population (UVA, HR: 0.77, 95% CI: 0.31∼1.90, p = .56) (Figure 2, Table 2, and Sup Table 1). Secondary Predictors in West China population and SEER population. A. Overall survival assorted by gender in West China Hospital group, SEER Asian group, and SEER all ethnic group; B. Overall survival assorted by age in West China Hospital group, SEER Asian group and SEER all ethnic group; C.Overall survival assorted by P16 test in SEER Asian group and SEER all ethnic group.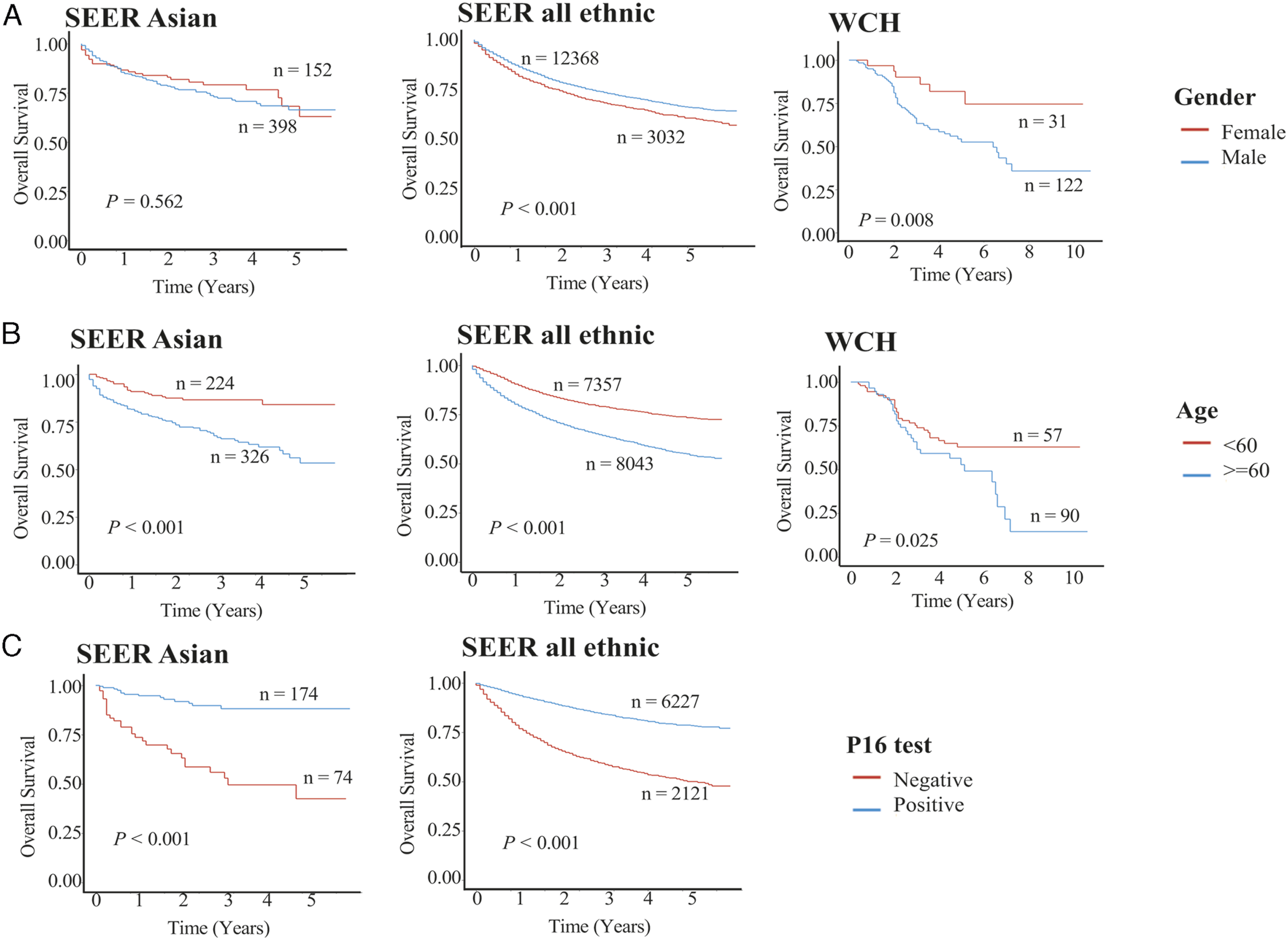
Subgroup Analysis
We further performed subgroup analyses by gender, tumor stage, and HPV status, which were important prognostic factors, to assess patient survival. Due to the limited sample size of WCH patients, we performed the subgroup analyses in only two SEER populations, where SIT and HPV-positive status were consistently observed to be beneficial factors in OPSCC patients.
In the HPV subgroup analysis (Sup Table 2–3), more aged HPV-negative patients received SIT in the SEER Asian population (aged: 71%, younger: 29%) than in the SEER all ethnic population (aged: 54%, younger: 46%). However, the age distribution of HPV-positive patients receiving SIT was similar in the SEER Asian (aged: 54%, younger: 46%, p = .0093) and SEER all ethnic populations (aged: 43%, younger: 57%, p < .001). Moreover, regardless of the HPV status, the positive effect of SIT on OS was more obvious in the SEER Asian population (HPV positive, aHR:0.28; HPV negative, aHR: 0.16, p = .005) than in the SEER all ethnic population (HPV positive, aHR:0.47; HPV negative, aHR: 0.47, p < .001).
In the gender subgroup analysis (Sup Table 4–5), the beneficial effect of SIT on OS was higher in the male SEER Asian population (male, aHR:0.21, p = .043) than in male SEER all ethnic population (male, aHR:0.46, p < .001). In the sub-analysis of tumor stages (Sup Tables 6–7), a larger number of advanced OPSCC patients received SIT in the SEER Asian population than in the SEER all ethnic population (42% vs 28%).
Discussion
In our study, OPSCC patients receiving SIT had better survival outcomes than those receiving NST in all three populations. Among patients in the WCH population, more underwent SIT than received NST, while in SEER Asian and SEER all ethnic populations, more patients received NST than underwent SIT. The HPV infection rate was the highest in the SEER all ethnic population, followed by the SEER Asian population, and the WCH population was the lowest. Overall, the OS of patients with OPSCC in the SEER database, including Asian-Americans, was higher than that of patients in WCH population, mainly due to the less advanced cancer stages and higher HPV infection rate of Western populations. Moreover, male was a risk factor for reduced OS in OPSCC patients in the WCH population, consistent with previous reports 18 ; however, in the SEER population, male was a protective factor, which was mainly related to the high HPV positivity rate in male OPSCC patients. 19
In accordance with the American National Comprehensive Cancer Network (NCCN) Guidelines (Version 1.2021), definitive RT or primary site resection is recommended for OPSCC patients with early-stage cancer; induction chemotherapy followed by RT, concurrent systemic therapy, or surgery with adjuvant therapy is recommended for the treatments of OPSCC patients with advanced-stage tumor. 20 However, the Chinese Society of Clinical Oncology (CSCO) Diagnosis and Treatment Guidelines for Head and Neck Cancer (2018) 21 recommended surgery as the primary treatment for OPSCC patients. It has been reported that HPV-positive OPSCC patients are more sensitive to radiotherapy than their HPV-negative counterparts. 22 A larger number of HPV-positive OPSCC patients (66.7%) than HPV-negative patients received NST between 2010 and 2014 according to the results of the National Cancer Data Base (NCDB), 23 which is consistent with our SEER database results. In contrast, due to a relatively lower HPV infection rate, SIT is still the main treatment in Chinese OPSCC patients.3,11 Our study showed that SIT was more beneficial for OS in OPSCC patients than NST in all three populations, and consistent results were observed in the subgroup analyses by tumor stage, gender, age, and HPV status. Both HPV-positive and HPV-negative OPSCC patients treated with SIT had better prognoses than those treated with NST.
Even though more OPSCC patients in the WCH population received SIT, OS in the WCH population treated with SIT was poorer than that in the SEER population, which might be largely due to the higher proportion of patients in the WCH population were HPV negative and had more with advanced tumor stage. In addition, compared with OPSCC patients in the SEER population, a larger number of OPSCC patients in the WCH population were treated with only surgery without adjuvant therapies (Table 1), which also reduced the survival rate, as OPSCC patients who received surgery plus adjuvant therapies had better survival outcomes than those who received surgery alone (Sup Table 15–16). Moreover, in terms of surgical approaches, transoral robotic surgery, a minimally invasive and favorable treatment for early OPSCC tumors,24–26 has been widely applied in Western countries but not in China due to its high cost, which might also be one of the reasons for the worse prognosis in the WCH population.
In addition to comparing the ethnicity-related differences in patient characteristics and prognoses between the Eastern and the Western populations, this study explored differences in patient characteristics and prognosis among Asian population due to the acquired cultural differences in emigration. We observed consistent positive effects of SIT on OS in OPSCC patients in the SEER Asian and SEER all ethnic populations after multivariate adjustment. The baseline characteristics of the Asian-American population, such as the proportions of age at diagnosis, cancer T stage, and HPV infection rate, were between those of the Chinese and American populations, and the OS rate in Asian-Americans was also between those in the Chinese and American populations, indicating that cultural differences associated with immigration status, for example, early cancer screening and sexual behaviors, lead to differences in OPSCC cancer survival.
This study has some limitations. First, the sample size of the patients recruited from the WCH was small. Even though some of the statistical results were not significant in the WCH population, the main trend was consistent with that of the SEER data. Second, due to the limited alcohol consumption and smoking data obtained from the SEER database, comparisons with alcohol consumption- and smoking-related prognoses could not be performed. Third, recurrence data were unavailable in the SEER database; therefore, PFS could not be calculated in this study. Fourth, since “no treatment” in the SEER database referred to “no treatment or missing treatment information,” a separate detailed treatment analysis of the SEER data was not possible.
In summary, our ten-year real-world retrospective study demonstrated that patients with OPSCC who received SIT had a much better prognosis than those who received NST in all three populations. Even though a higher proportion of OPSCC patients in the WCH population received SIT, the OS rate was lower than that in the SEER populations, including SEER Asian population, which was mainly because that more OPSCC patients in the SEER population had earlier cancer stages and HPV-positive status, and received surgery plus adjuvant therapies.
Supplemental Material
Supplemental Material—Survival Outcomes Related to Treatment Modalities in Patients With Oropharyngeal Squamous Cell Carcinoma
Supplemental Material for Survival Outcomes Related to Treatment Modalities in Patients With Oropharyngeal Squamous Cell Carcinoma by Danni Cheng, Yufang Rao, Jianqing Qiu, Yao Song, Wendu Pang, Ke Qiu, Yijun Dong, Qiurui Liu, Yu Zhao, Jun Liu, Wei Xu and Jianjun Ren in Ear, Nose & Throat Journal
Footnotes
Author’s Contributions
Danni Cheng: contributions to design, acquisition and analysis of data, and drafting the article. Yufang Rao: collection and interpretation the clinical data, and revising the article. Jianqing Qiu: analysis and interpretation of data, and revising the article. Yao Song: collection and interpretation the clinical data, and revising the article. Wendu Pang: collection, analysis and interpretation the follow-up data of patients. Ke Qiu: collection, analysis and interpretation the follow-up data of patients. Yijun Dong: collection and interpretation the clinical data. Qiurui Liu: collection and interpretation the clinical data. Jun Liu: acknowledgement of drafting the article and revising it critically for important intellectual content. Yu Zhao: acknowledgement of drafting the article and revising it critically for important intellectual content. Wei Xu: acknowledgement of drafting the article and revising it critically for important intellectual content. Jianjun Ren: agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by West China Hospital, Sichuan University (ZY, grant #2019HXFH003, grant #ZYJC21027, RJJ, grant #2019HXBH079); Chengdu Science and Technology Bureau (RJJ, grant #2019-YF05-00461-SN); Sichuan University (ZY, grant #GSALK2020021, RJJ, grant #2020SCU12049); The Science and Technology Department of Sichuan Province (ZY, grant #2020YFH0090, RJJ, grant #2020YFS0111, ZYB, grant# 2022YFS0066); The Health Department of Sichuan Province (RJJ, grant #20PJ030); China Postdoctoral Science Foundation (RJJ, grant#2020M673250); The Foundation of National Clinical Research Center for Geriatrics (SH, grant #Z20201013); National Natural Youth Science Foundation of China (RJJ, grant #82002868).
Data Availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
Ethics Approval
This study was conducted at Department of Otolaryngology head and Neck Surgery, West China Hospital, Sichuan University. This study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee on Biomedical Research, West China Hospital of Sichuan University (reference number: 2019-357).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
