Abstract
Background
Cerebrospinal fluid (CSF) leakage from the lateral recess of the sphenoid sinus (LRSS) is usually repaired using endoscopic endonasal approaches, which can be challenging. Various surgical techniques have been developed for the disease.
Objective
To report our experience with repairing CSF leak from the LRSS via transethmoid sphenoidotomy approach (TESA) and transprelacrimal recess pterygoid root approach (TPLRA), to assess the efficiency of TPLRA by comparing it with TESA.
Methods
This retrospective study included patients with LRSS CSF rhinorrhea who underwent TESA (n = 10) or TPLRA (n = 5) from January 2011 to December 2020. Demographic characteristics and operation-related parameters were recorded.
Results
The mean operation time was 169.5 and 225.0 mins in the TESA and TPLRA groups, respectively, with a mean blood loss of 65 mL and 68 mL, respectively. Histopathological examinations confirmed encephalocele in 11 (73.33%) and 4 (26.67%) cases with meningocele, respectively. CSF rhinorrhea was successfully repaired in the first attempt in both groups during the mean follow-up time of 54 months. Postoperative permanent numbness of the cheek was observed in two patients in the TESA group. No cases of lacrimal overflow or subjective dry eye were observed.
Conclusions
The TPLRA, which could be an alternative procedure to treat CSF rhinorrhea in the LRSS, provides a straight-line trajectory and effective maneuverability. We also found that CSF rhinorrhea in the LRSS was accompanied by encephalocele or meningocele, with encephalocele presenting more commonly.
Keywords
Introduction
The most common location of cerebrospinal fluid (CSF) rhinorrhea is the cribriform plate. CSF rhinorrhea arising from the sphenoid sinus is less frequent.1-3 However, the rate of CSF rhinorrhea originating in the lateral recess of the sphenoid sinus (LRSS) accompanied by meningoencephalocele was higher than that from the sella. 4 Resulting from excessive pneumatization of the sphenoid sinus, the LRSS leads to thinning of the bone of the lateral wall of the sphenoid sinus. It is challenging to treat LRSS CSF rhinorrhea through the endoscopic endonasal corridor because the LRSS is located far from the midline of the skull base, either in the horizontal or vertical direction, and is also adjacent to the neurovascular structure.
Transpterional and LeFort osteotomy approaches were widely applied to treat LRSS CSF rhinorrhea in the early years of surgical intervention for this condition, which later developed into the nasal endoscopic approach. It is not always feasible to manage the LRSS disease via the standard transsphenoidal approach. More recently, the endoscopic transpterygoid approach (ETPA) to the LRSS was first reported by Bolger et al. 5 Subsequently, this approach was used intensively, although it required complex and time-consuming dissection of the pterygopalatine fossa (PPF) and placed the neurovascular structures of the PPF at risk. However, it remains the classic approach to treat LRSS disease. Meanwhile, complications related to this surgical method, such as ipsilateral cheek numbness and/or dry eyes, gradually arose. Over the past decade, physicians worldwide have modified the approach to improve maneuverability and reduce complications, focusing on allowing easier dissection of the PPF.
Zhou et al. first reported the endoscopic prelacrimal recess approach (PLRA) in the treatment of complex maxillary sinus lesions. Subsequently, this procedure was widely applied to get the skull base including the area of PPF and infratemporal fossa (ITF), as well as the lateral skull base. 6 In the last 10 years, we used transethmoid sphenoidotomy (TESA) to treat LRSS CSF rhinorrhea. We first attempted the transprelacrimal recess pterygoid root approach (TPLRA) to manage LRSS CSF rhinorrhea in 2013 for its ability to preserve the sphenopalatine artery and the integrity of nasal structures.
This study aimed to present our experience in repairing LRSS CSF rhinorrhea using two separate surgical approaches and compare the results between the corresponding patient groups over an average 54-month follow-up.
Patients and Methods
Clinical Data
This is a retrospective study based on medical records of 15 cases of LRSS CSF rhinorrhea, treated from January 2011 to December 2020, at the Department of Otolaryngology - Head and Neck Surgery, Xijing Hospital, Air Force Medical University. The patients were divided into two groups according to the surgical approach. Nasal fluid test was performed to confirm the presence of active CSF rhinorrhea. High-resolution guided computed tomography (CT) images (with slice thickness of 0.625 mm) identified the bony defects and dehiscence in the skull base. The study protocol was approved by the research committee of Xijing Hospital, Fourth Military Medical University, Shaanxi, China (XJLL-KY20212154).
Surgical Procedure
Patients were placed in the supine position with a 10° adduction to the surgeon. The nasal cavity mucosa was sterilized with 0.5% povidone-iodine solution. The nose was then packed with cotton pledget soaked in an adrenaline hydrochloride solution (0.01%).
TESA was initiated by opening the ethmoid sinus and sphenoid sinus widely and confirming a lesion in the LRSS (Figure 1B). Meningoencephalocele was then treated with 70° endoscope and auxiliary angled instruments (Figure 1C). The bipolar forceps were coagulated to help remove the prolapsed soft tissue. The LRSS mucosa was completely removed (Figure 1D). The bone was smoothed with a diamond burr around the defect to support nourishment and stability. (Figure 1, from patient No. 6, March 2018). CT imaging and intraoperative snapshot of the case treated via TESA approach (from patient No.6, March 2018) 1a. present the bone defect (red arrow) in the left LRSS in coronal/axial CT imaging; 1b. The meningoencephalocele (white star) of 0° endoscopic view; 1c-e intraoperative snapshot with 70° endoscope; 1c. The same meningoencephalocele (white star); 1d. Expose the skull bone defect (blue triangle) after resecting the meningoencephalocele and trim the bone from the middle turbinate bone (blue star);1e. Inlay the bone to shore up the defect (blue star).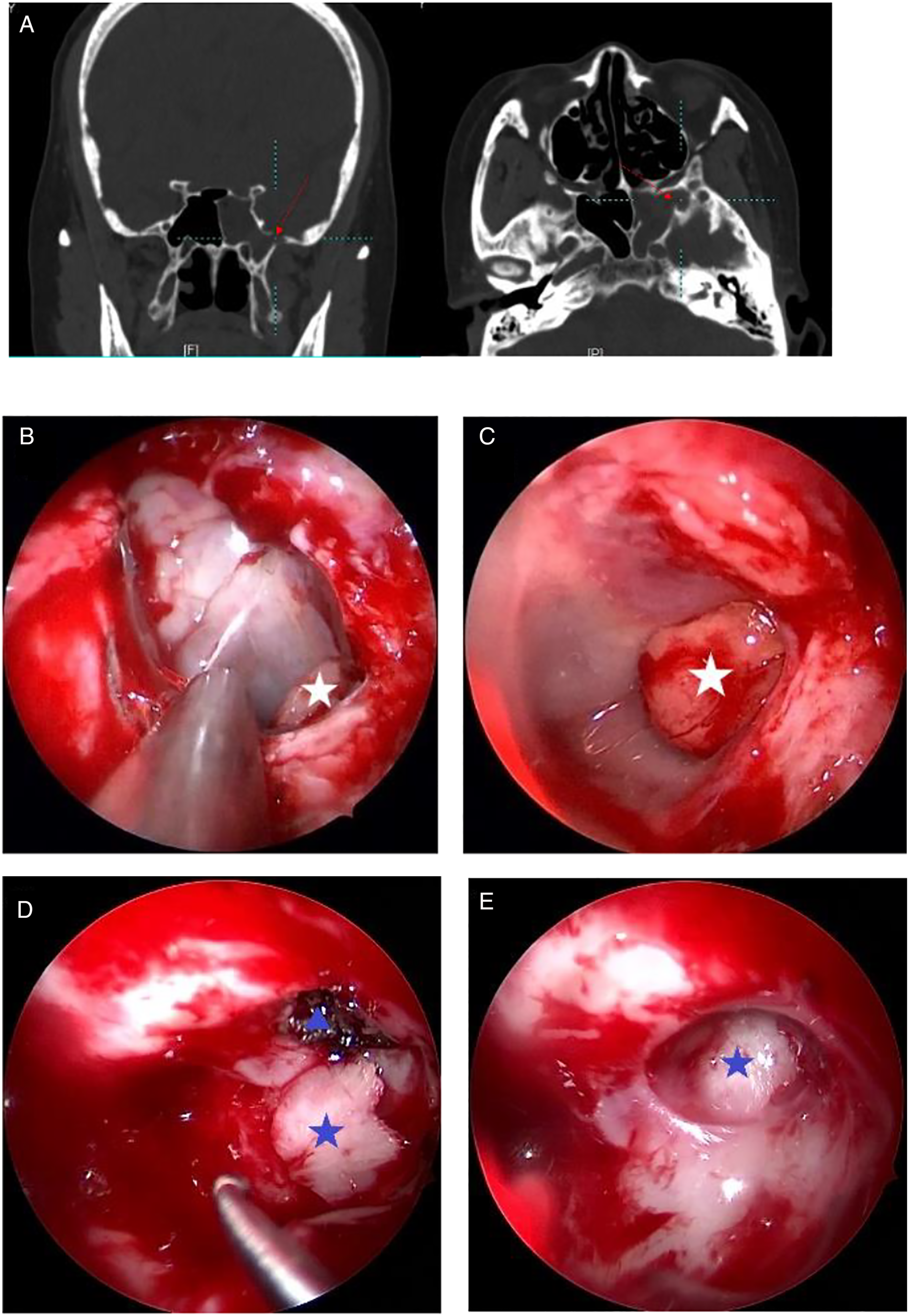
TPLRA involved a curved vertical mucosal incision on the lateral wall of the nasal cavity (Figure 2B). The mucosal flap was separated from the bony wall backward to the ostium of the maxillary sinus and downward to the inferior nasal meatus. The nasolacrimal duct (NLD) should be carefully protected at the same time. Subsequently, the bony attachment of the inferior turbinate (IT) was exposed and bone in the medial wall of the maxillary sinus was removed, thereby creating the IT-NLD flap (Figure 2C). CT imaging and intraoperative snapshot of the case treated via TPLRA approach (from patient No. 5, January 2019) 2a. The bone defects (red arrow) in left LRSS in axial/sagittal/coronal CT imaging; 2b-g intraoperative snapshot with 0° endoscope; 2b. The incision, SP (septum)&IT (inferior turbinate); 2c. The nasolacrimal duct (white star); 2d. PWMS (the posterior wall of maxillary sinus); 2e. BP (the base of the pterygoid process), meningoencephalocele (blue star), the sphenopalatine artery (blue triangle); 2f. The bone defect of skull base (blue star); 2g. Inlay bone graft (blue star) and artificial dura (blue triangle).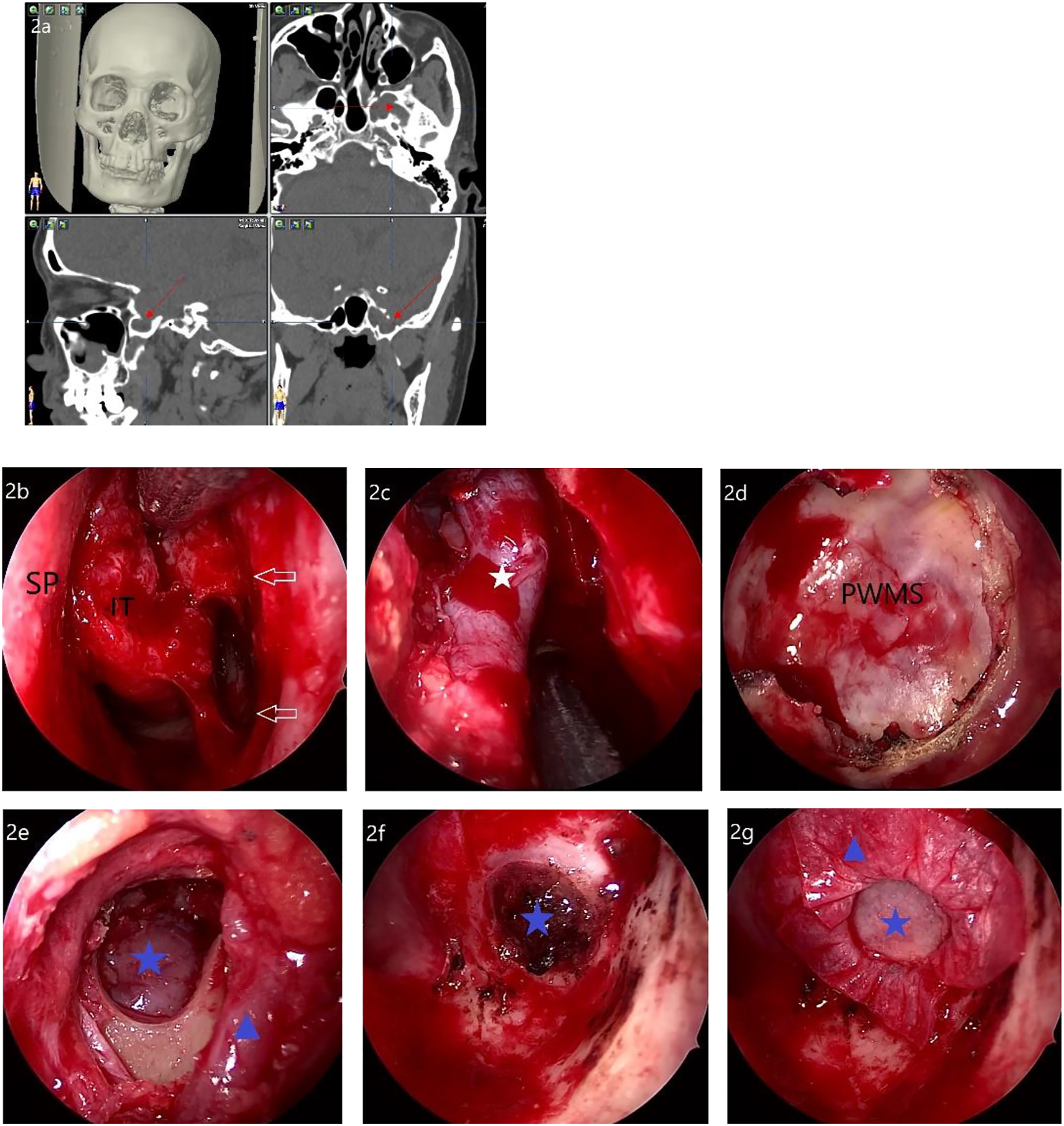
Once the posterolateral wall of the maxillary sinus between the vertical palatine plate and the suborbital nerve was opened to expose the PPF and ITF (Figure 2D), the connective tissue was bluntly separated to preserve the neurovascular structures with a Freer elevator along the upper fusion of the medial and lateral pterygoid process plate. Electrocautery was used to control bleeding from the small vessels within the adipose tissue of the pterygopalatine fossa.
After revealing the base of the pterygoid process, the vidian canal (VC) and foramen rotundum (FR) were identified. The area planned for resection was identified as inferior to the imaginary line from VC to FR (V-R line). The bone window was made at least 1 cm in diameter using a diamond burr combined with a Kerrison rongeur, enabling entry into the LRSS to expose the hernia tissue (Figure 2E). Finally, removal of the hernia by bipolar cautery and the LRSS mucosa was performed (Figure 2F). (Figure 2, from patient No. 5, January 2019).
We also depicted the trajectory in the same axial image to show the differences of the two endoscopic approaches to the LRSS (Figure 3A–B). The diagram of two endoscopic approaches to the LRSS. 3a. TESA approach via 70° endoscope, 3b. TPLRA approach via 0° endoscope.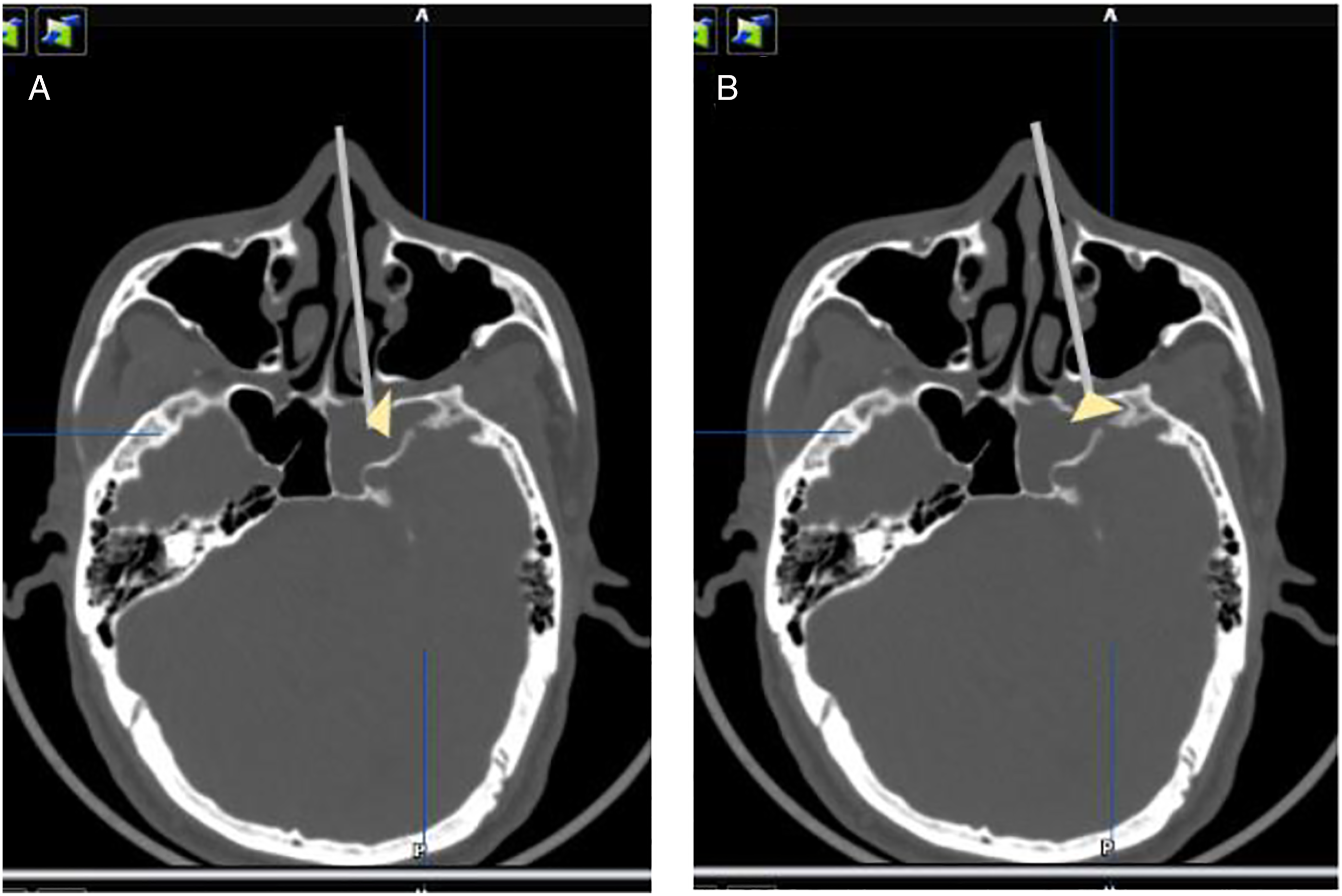
Repair Methods and Materials for Skull Base Reconstruction
Before repair, 125 mL of mannitol and 10 mg of furosemide were injected to reduce intracranial pressure. Multilayer repair was preferred to stop the CSF leak and “reattach” the thin skull base, thus preventing encephalocele recurrence. Custom-shaped bone was widely used (Figure 1E, Figure 2G), which was placed in the epidural space as the key layer. The inner and outer lining material consisted of an artificial dura, fascia, and free mucosal flap. Gel-foam and iodoform gauze strips were applied to hold the grafts in place.
Postoperatively, all patients were on bed rest and received antibiotics and 20% mannitol solution for 5–7 days. It was necessary to prevent deep vein thrombosis and constipation, and to maintain the balance of patient intake and output. Iodoform gauze strips were removed 7–10 days after the surgery. Patients were instructed to avoid blowing their nose and strenuous activity for 3 months after the surgery.
Statistical Analysis
Continuous variables were analyzed using the t-test, while categorical variables were analyzed using the Fisher exact method, for the total number of cases was less than 40. All p values were two-sided, and statistical significance was set at p < .05. Statistical analysis was performed using SPSS software (IBM SPSS Statistics for Windows, Version 25.0 Armonk, NY: IBM Corp).
Results
We retrospectively reviewed 15 patients with CSF rhinorrhea originating from the LRSS admitted to our department in the last 10 years. Among these 15 patients, clear inducement was identified in only one who had a history of benign cranial hypertension and head trauma. Two patients with recurrent CSF rhinorrhea had undergone endoscopic surgery in other hospitals before.
Demographic data of patients with sphenoid sinus lateral recess cerebrospinal fluid rhinorrhea.
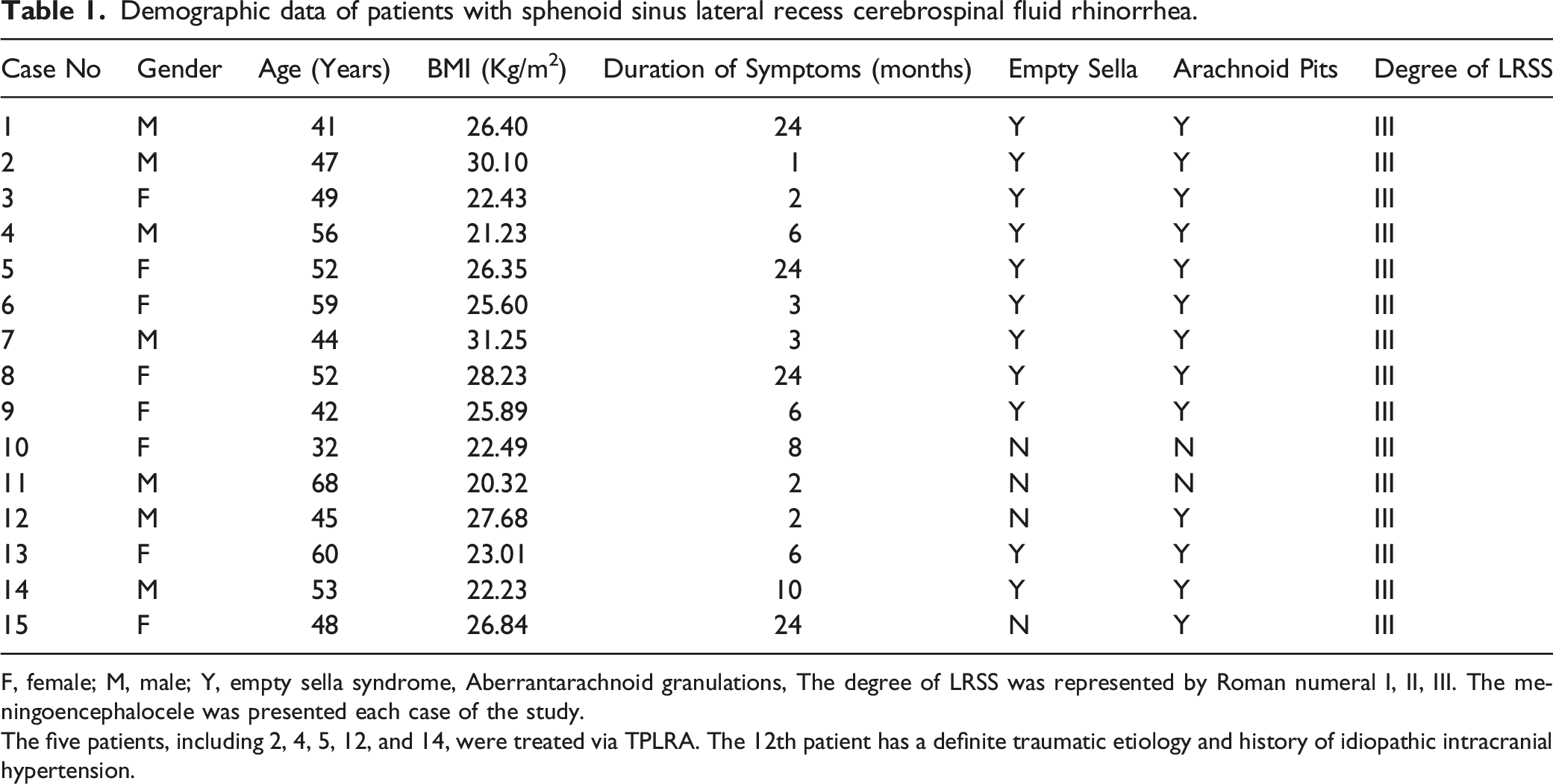
F, female; M, male; Y, empty sella syndrome, Aberrantarachnoid granulations, The degree of LRSS was represented by Roman numeral I, II, III. The meningoencephalocele was presented each case of the study.
The five patients, including 2, 4, 5, 12, and 14, were treated via TPLRA. The 12th patient has a definite traumatic etiology and history of idiopathic intracranial hypertension.
The bone defect in the skull base was confirmed and located inferior to the FR in the operation (Figure 1A, Figure 2A). Thirteen of 15 (86.67%) cases showed signs of arachnoid granulation depression at the base of the skull and 11 of 15 (73.33%) cases were accompanied by partial or completely empty sella. According to the Vaezi classification standard, the LRSS of these 15 cases were categorized into type Ⅲ. 7 The postoperative pathology confirmed 4 cases with meningocele and 11 cases with encephalocele.
Compard the two groups of transethmoid sphenoidotomy approach and transprelacrimal recess pterygoid root approach.
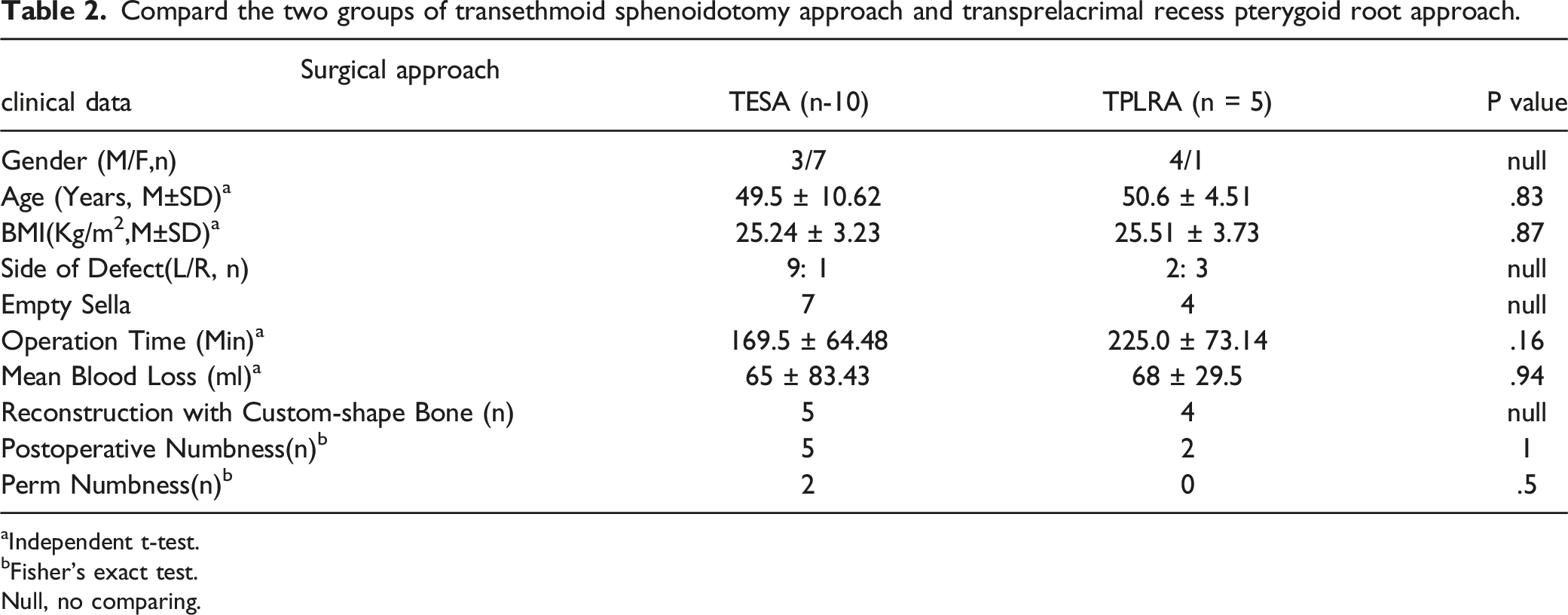
Independent t-test.
Fisher’s exact test.
Null, no comparing.
Discussion
Middle-age, female sex, and obesity were previously reported to be independent risk factors for CSF rhinorrhea due to correlation with elevated intracranial pressure (ICP). In this study, most patients were overweight; however, there was no significant sex difference. Arachnoid cavities and empty sella observed in most patients were associated with radiographic signs of elevated ICP, which could be found in previous studies.1-3,8-11 The underlying etiology of LRSS CSF leak is widely considered to be the combination of pathologic and anatomic factors.
CSF rhinorrhea patients with meningoencephalocele were reported in Bloger et al. (6 cases), 12 Zhou et al. (9 cases), 13 and Tomazic et al. (5 patients). 14 Alaxender et al. described an empty sella in 7 of 11 patients with meningocele in all of them. 9 Settecase et al. reported 26 cases of meningocele with imaging, among which 61% had an empty sella or partial empty sella. 15 In addition, Gabriel et al. noted that of 35 patients, 62% had an empty sella, and up to 72% had associated meningocele/encephalocele. 16 Ulu et al. reported 13 cases in a single center study, including 8 cases (62%) of empty sella and 11 cases (84.62%) of meningocele. 11 Zheng et al. reported that 50% of the cases were accompanied by meningocele. 17 Furthermore, a multicenter research published in laryngoscopy indicated that 23 of 33 patients (69.7%) had meningocele, 18 and Zhou et al. reported that most patients (44/49, 89.8%) had meningoencephalocele. 19 All of the above studies showed that the LRSS had a high incidence of meningoencephalocele. 20 All patients in our study had hernia, of which encephalocele accounted for 73.33%.
TESA is considered a basic and effective approach for appropriate cases, in which angled endoscope and some curved instruments always need to be used. Though this approach consumes less time because only a small amount of bone needs to be removed, it is not easy to manipulate the instruments under the angled endoscope. Tarabishi et al., who conducted a prospective study with the bi-nostril retrograde transsphenoid approach, stressed that this way could allow access to the blind recess and angle of the LRSS with minimal bone removal. 10
The transpterygoidal approach is recognized as a classic procedure for excessive LRSS. Bolger et al. reported ETPA used in the treatment of meningocele in the LRSS in 2002. Subsequently, in 2005, the authors described specific surgical procedure step by step, which allowed directly approach to the LRSS.4,5 Al-Nashar et al. considered that this approach was favorable in 12 patients with similar diseases. 21 Tanriover et al. reported the use of the bi-nostril minimal ETPA with the application of Foley packing. 11 Zhen et al. also use the ETPA and performed multilayer repair and obliteration of the LRSS instead. 17 Sreenath et al. shared the modified ETPA and considered the pterygoid wedge bone as a landmark, with 100% success rate for skull base repair; meanwhile, postoperative complications included V2 anesthesia and subjective eye dryness were observed in a few cases. 18
Zhou et al. proposed the prelacrimal recess approach (PLRA) for the treatment of complex maxillary sinus lesions and developed its clinical application in the PPF and ITF, as well as the lateral skull base. 22 In 2017, Huang et al. conducted cadaver studies, showing that TPLRA could provide a clear view and wide exposure of the route, and preserve the integrity of the nasal structures. 23 In 2020, Li et al. measured the PLRA operative parameters based on cadaveric study, and determined that the vidian nerve may be an obstacle in the transpterygoid approach route when it is higher than the floor of the LRSS. 24 TPLRA emerged as a reasonable alternative to access the LRSS, while protecting the sphenopalatine ganglion. Zhou et al. confirmed that the endoscopic route of PLRA is more direct than that of the transpterygoid approach for selective cases. 19
Our team had performed TPLRA in five cases since 2013. Compared with TESA, TPLRA had a longer operation time. The most time-consuming step was separating the neurovascular structures of the PPF, the preservation of which requires caution. With the advantage of preserving nasal integrity, exposure of PPF via TPLRA does not take much time. TPLRA facilitated the use of instruments by providing a direct access to the LRSS and space for better manipulation of the instruments. It is suitable for excessive pneumatization of the LRSS, especially pneumatization of the base of the pterygoid process. However, this technique comes with a steep learning curve.
In this study, two patients in TESA group experienced permanent numbness, whereas none did in the TPLRA group. Though it is simple to access the LRSS through TESA, visual distortion under 70° endoscope and the lack of suitable instruments, which may be the possible reason for the relatively high incidence of postoperative numbness in the TESA group, not only makes the process more difficult, but also increases time-consumption and risk of soft tissue injury. There were no significant statistical differences between the two groups, which may be due to the limited sample size.
Conclusion
TPLRA, which is suitable for excessive pneumatization of the LRSS, especially pneumatization of the base of the pterygoid process, could be another route to treat CSF rhinorrhea in the LRSS with less risk of numbness. It provides a straight-line trajectory and allows effective maneuverability. Every step taken intraoperatively and postoperatively affects the success rate of the operation, which must be taken into consideration. Although our cases of the LRSS all classified into type III, there are still differences in the pneumatization direction and degree. Therefore, preoperative imaging evaluation can help us select the appropriate surgical approach. Meanwhile, we found that CSF rhinorrhea in the LRSS was accompanied by encephalocele or meningocele, with encephalocele presenting more commonly.
Footnotes
Declaration of conflict of interests
The author(s) declared that they have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Shaanxi Provincial Department of Science and Technology Key Industry Innovation Chain (Group) social development field. Fund serial number: 2021ZDLSF02-12.
