Abstract
Objective
To analyze the pattern of lymph node (LN) metastasis and its effect on prognosis in sinonasal mucosal melanoma (SNMM).
Methods
This retrospective study was conducted based on the Surveillance, Epidemiology, and End Results (SEER) Program data. Survival outcomes were analyzed using the Kaplan-Meier method. Factors were compared between groups using log-rank test and Fisher’s exact test, and prognostic factors were screened using the Cox proportional hazards model. Propensity score matching (PSM) was conducted to examine the treatment differences after accounting for sex, age, race, T stage, N stage, and M stage.
Results
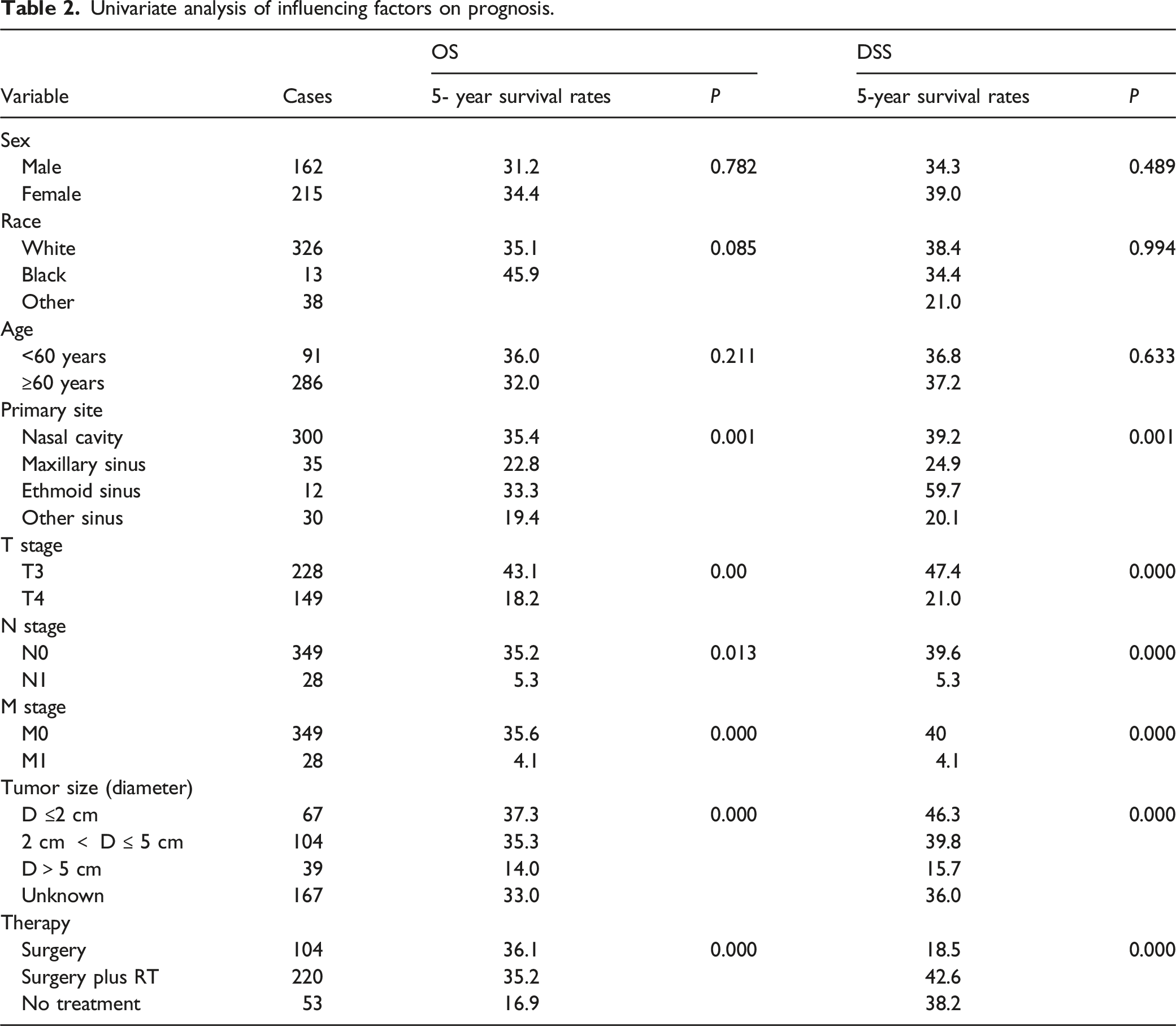
Level I (57.1%) and level II (53.6%) nodes were the most common sites of lymph node metastasis, followed by level III (17.9%) and IV (17.9%) nodes. T stage, M stage, and tumor size were associated with LN metastasis. The 5-year overall survival rates for patients without and with LN metastasis were 35.2% and 5.3%, respectively.
Conclusions
Level I and II lymph nodes may be the sentinel nodes of SNMM, Advanced T stage and increasing tumor size could promote LN metastasis. LN metastasis may promote distant metastasis and remains an important prognostic factor for patients with SNMM.
Introduction
Sinonasal mucosal melanoma (SNMM) is an uncommon malignancy, accounting for approximately 1% of all melanomas and 4-8% of sinonasal malignancies.1-3 The incidence rates of SNMM are evenly distributed in men and women, and peaks in populations aged 60-70 years.2,4 The tumor may originate from noncutaneous melanocytes of the nasal cavity and paranasal sinuses, 5 and accordingly, the nasal cavity and maxillary sinus are the most common sites of SNMM. 6
SNMM frequently are diagnosed with advanced stage, which is associated with poor survival. Previous studies reported a 5-year survival rate of less than 30% in patients with SNNM.7-9 The primary treatment method for early SNMM is surgical resection, and the role of radiotherapy in the treatment of SNMM is controversial. Some studies have suggested that radiotherapy improves prognosis, while other studies have indicated that SNMM is resistant to irradiation and postoperative radiotherapy does not improve survival.10-12 Currently, the pattern of LN metastasis and its influencing factors in SNMM has not been thoroughly explored, and the effectiveness of radiotherapy in SNMM patients with LN metastasis has yet to be determined.8,13 Therefore, we aimed to investigate the pattern and effect of LN metastasis in SNMM and its association with the clinical characteristics of the disease.
Patients and methods
Study cohort
A retrospective study was performed by extracting the data from the Surveillance, Epidemiology, and End Results (SEER) database from 2004 to 2015. Cases with the site codes of the nasal cavity (C30.0) and the paranasal sinuses (C31.0-31.3, C31.8-31.9) of ICD-0-3 were collected. The data were extracted for the histologic category of melanoma (8720/3-8799/3). Cases with a second primary tumor or death of missing/unknown cause were not included; codes of 999 or 99 in T, N, M stage were also excluded. In total, 377 patients were included in this cohort. All patients were staged according to the guidelines of the American Joint Committee on Cancer (AJCC), 8th Edition.
Statistical methods
Pearson Chi-square test and Fisher’s exact test were used to analyze the associations between lymph node metastasis and clinical variables of SNMM. The log-rank test was used to compare survival curves between groups. The effects of lymph node metastasis on overall survival (OS) and disease-specific survival (DSS) were analyzed using Cox regression analysis. The differences in OS and DSS between groups were compared using the Kaplan–Meier method. P values of less than 0.05 were considered statistically significant. To control potential confounding factors, propensity score matching (PSM) was conducted to examine the treatment differences after accounting for sex, age, race, T stage, N stage, and M stage. All analyses were performed using SPSS 25.0 and R-upset 1.4.0.
Results
Associations between clinical characteristics and LN metastasis in patients with SNNM.
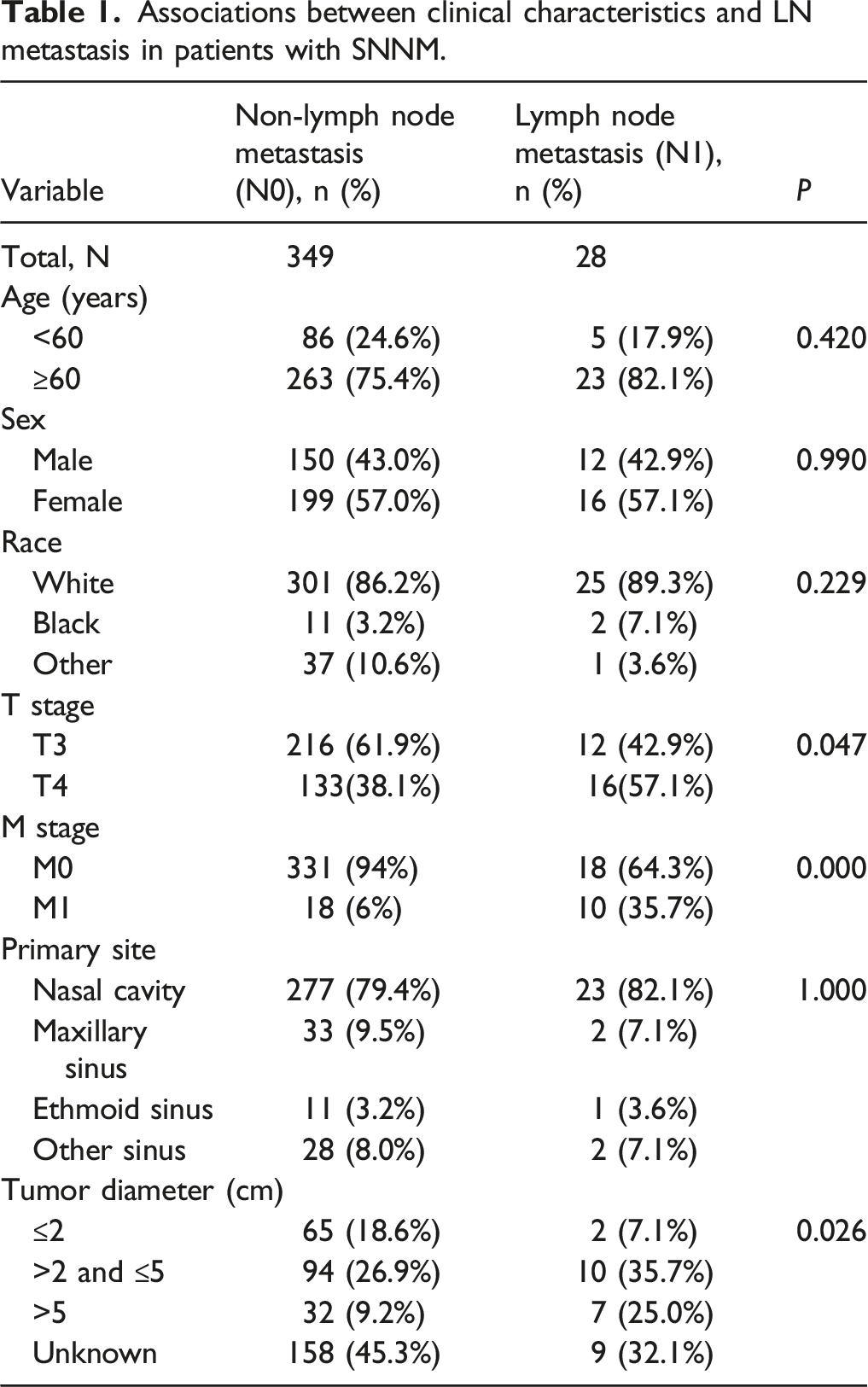
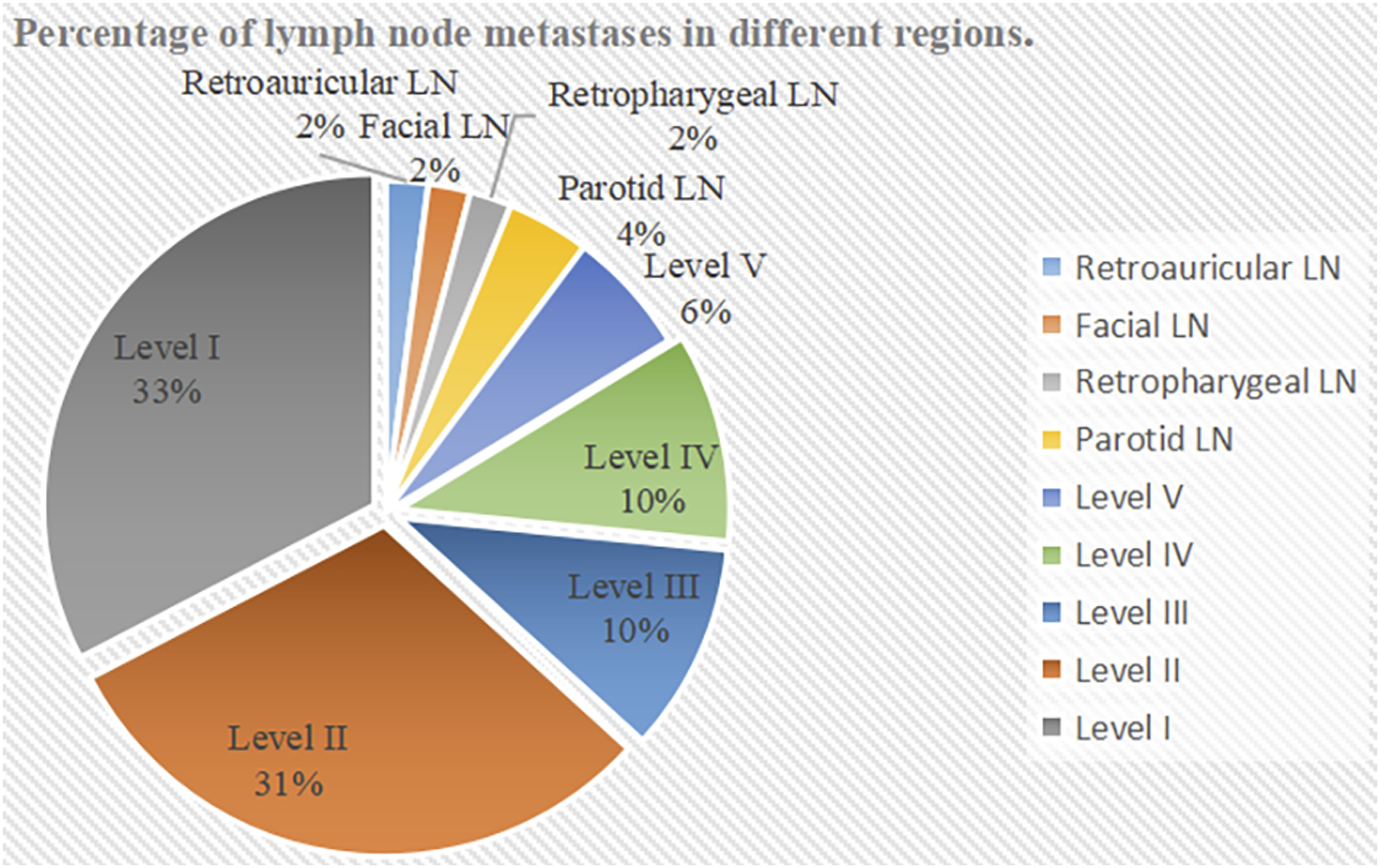
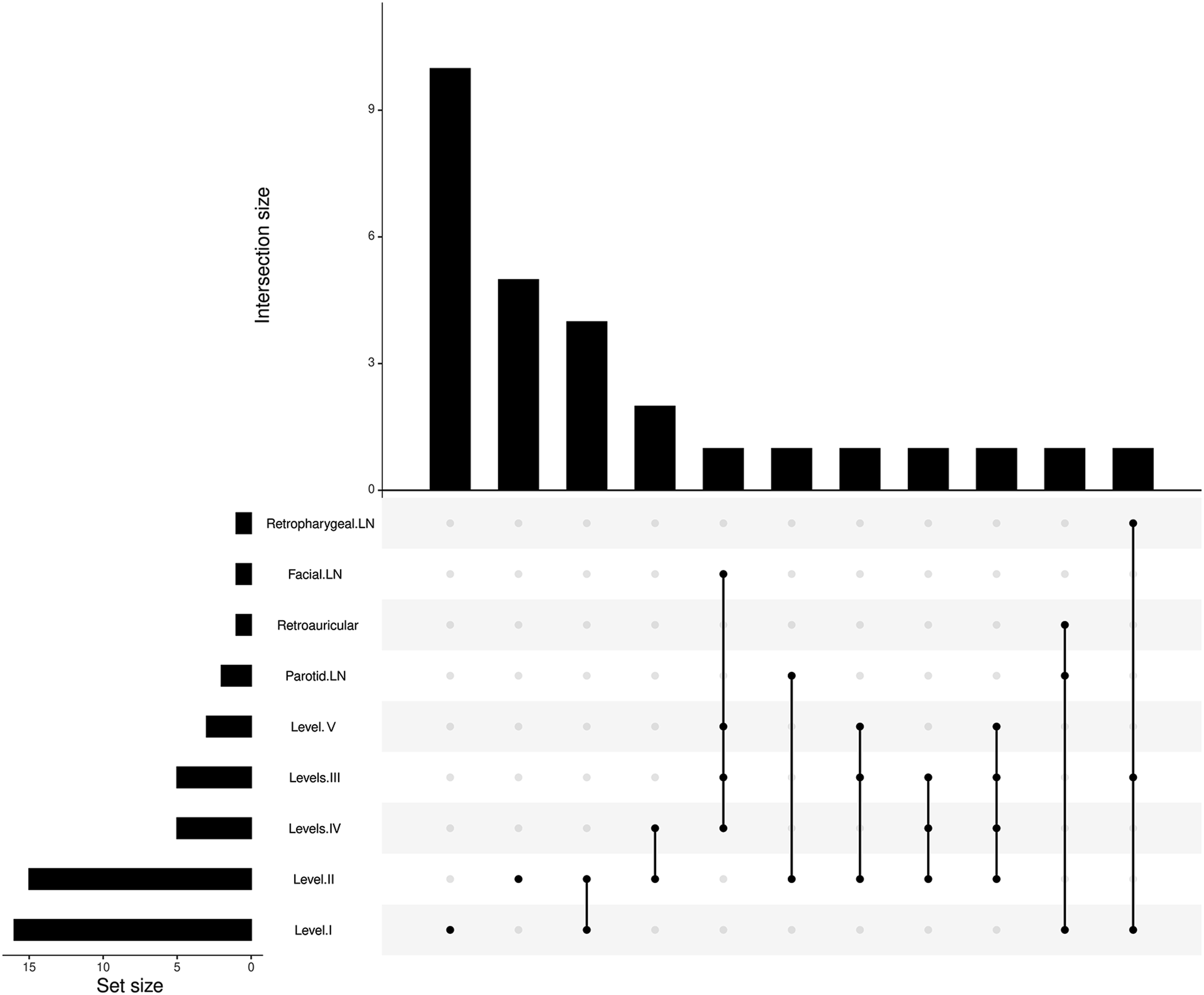
The rate of LN metastasis among the SNMM cases was 7.6% (28/377). SNMM most commonly metastasized to lymph nodes of level I (57.1%) and level II (53.6%), followed by levels III (17.9%) and IV (17.9%). Metastasis to other lymph nodes in the head and neck such as those in the level Ⅴ, parotid, facial, retropharyngeal and retroauricular regions was rare. The metastasis rates and percentage of lymph node metastases in different regions are shown in Figures 1 and 2. Association of lymph nodes in different regions are also shown in Figure 3. Metastasis rates of lymph nodes in different regions. Percentage of lymph node metastases in different regions. Association of lymph nodes in different regions.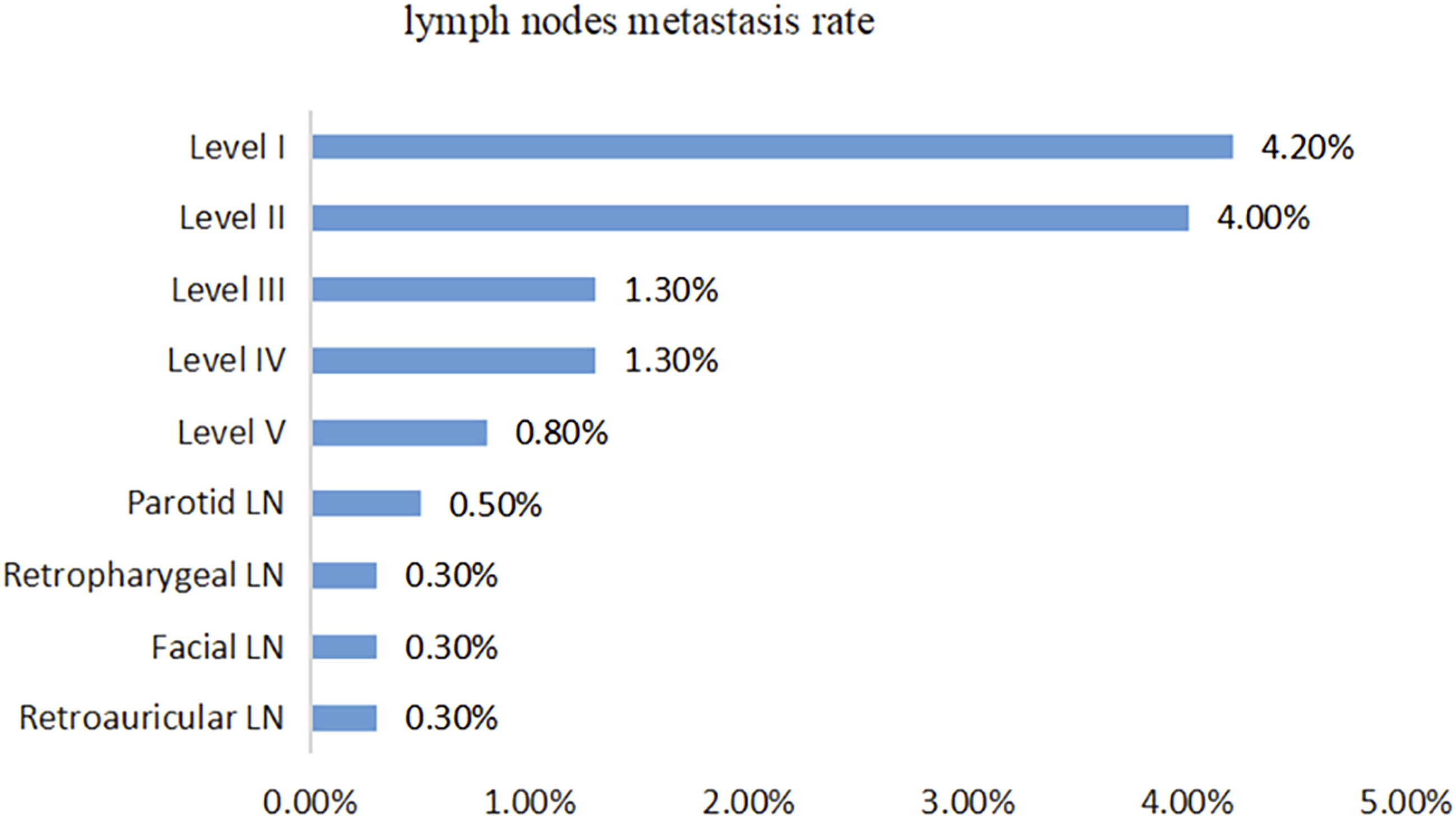
The 5-year OS and DSS rates for this cohort were 33.1% and 37%, respectively. The 5-year OS rates of SNMM patients without and with lymph node metastasis were 35.2% and 5.3%, respectively, while the 5-year DSS rates of patients with N0 and N1 were 39.6% and 5.3%, respectively (Figures 4A and 4B). (A) Analysis of OS rates in SNMM patients without (N0) and with (N1) lymph node metastasis. (B) Analysis of DSS rates in SNMM patients without (N0) and with (N1) lymph node metastasis.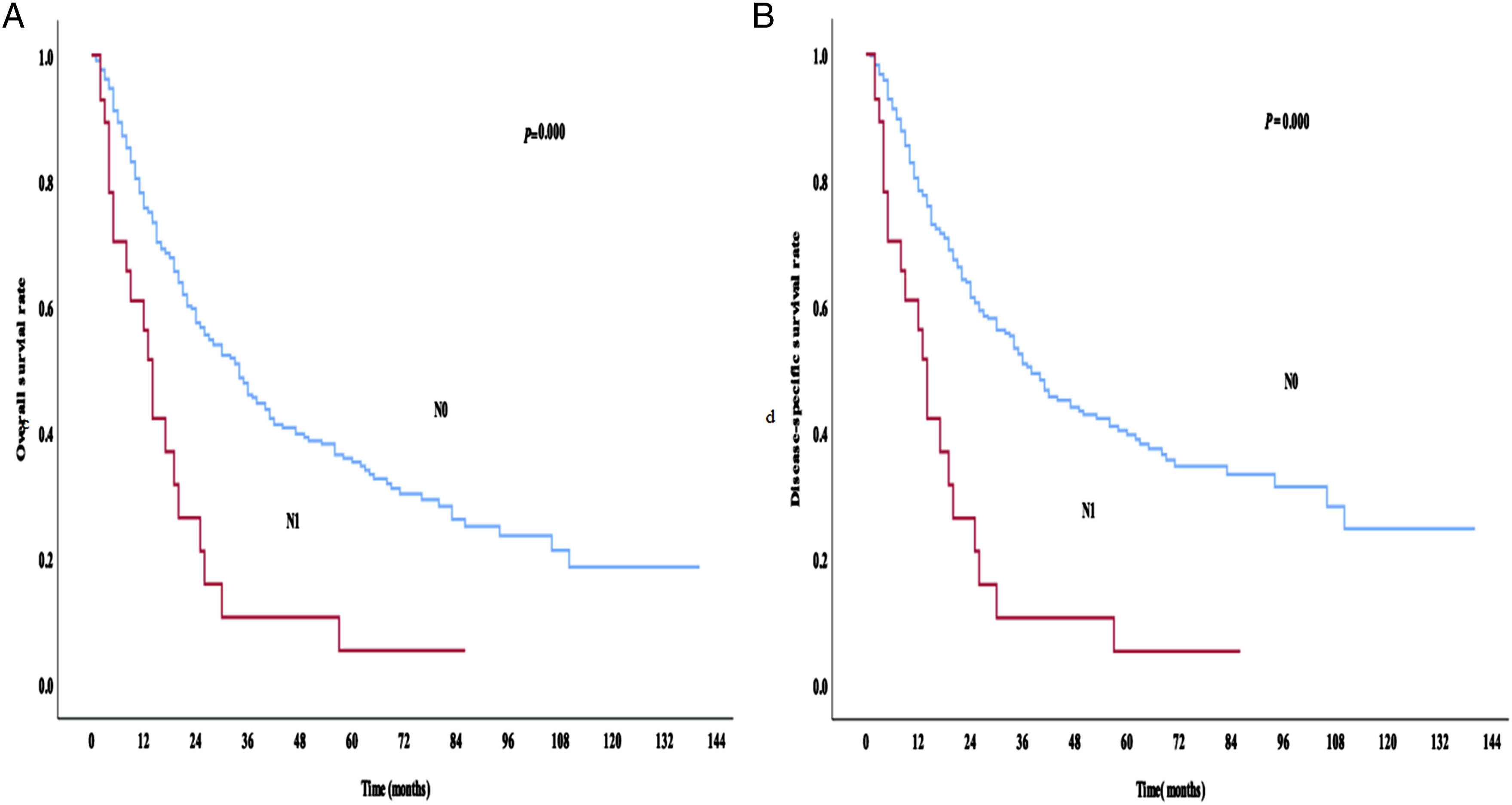
In this cohort, 104 (27.6%) patients underwent surgery, 220 (58.4%) patients were treated with surgery followed by radiotherapy, and 53 (14.1%) remained untreated. The 5-year OS and DSS rates in the untreated group were 16.9% and 18.5%, respectively, and the 5-year OS and DSS rates in the surgery group were 36.1% and 42.6%, respectively. The 5-year OS and DSS rates in patients with surgery plus radiotherapy were 35.2% and 38.2%, respectively. No significant differences of survival rates were detected between the treated subgroups (P = 0.953), while significant difference was observed between the treated and untreated groups (P = 0.000; Figures 5A and 5B). No statistical difference of OS or DSS was observed between the subgroups of N0 and N1 patients from different treatment groups (P = 0.718 for OS, P = 0.914 for DSS). (A) OS rates of SNMM patients who received different treatment methods. (B) DSS rates of SNMM patients who received different treatment methods.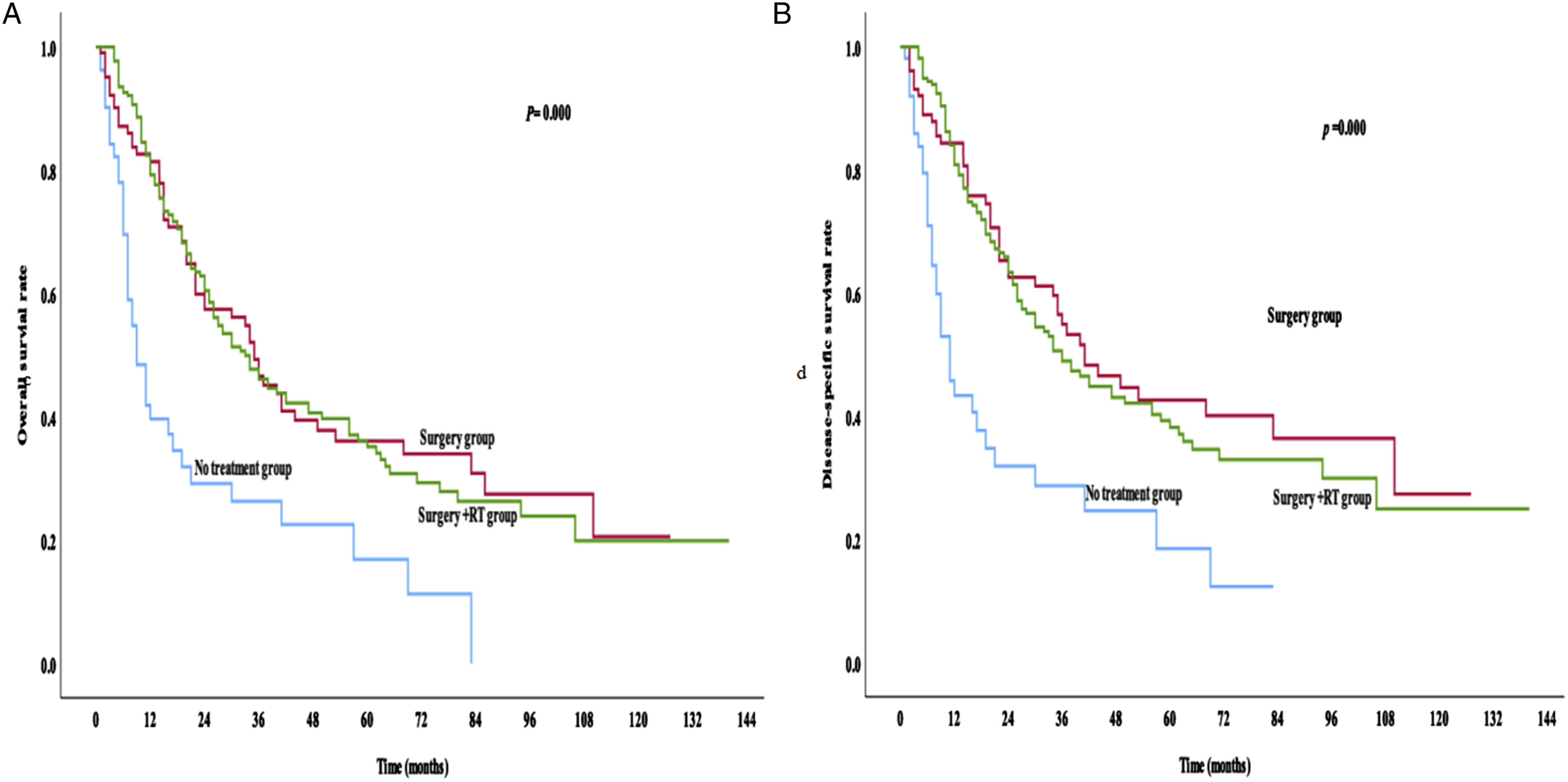
Univariate analysis of influencing factors on prognosis.
Multivariate analysis of independent factors affecting OS and DSS in SNMM patients.
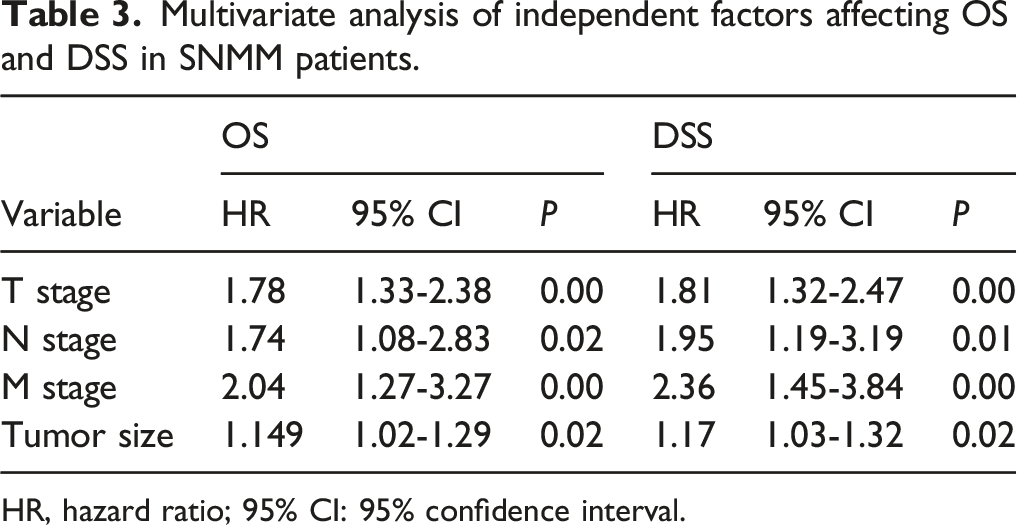
HR, hazard ratio; 95% CI: 95% confidence interval.
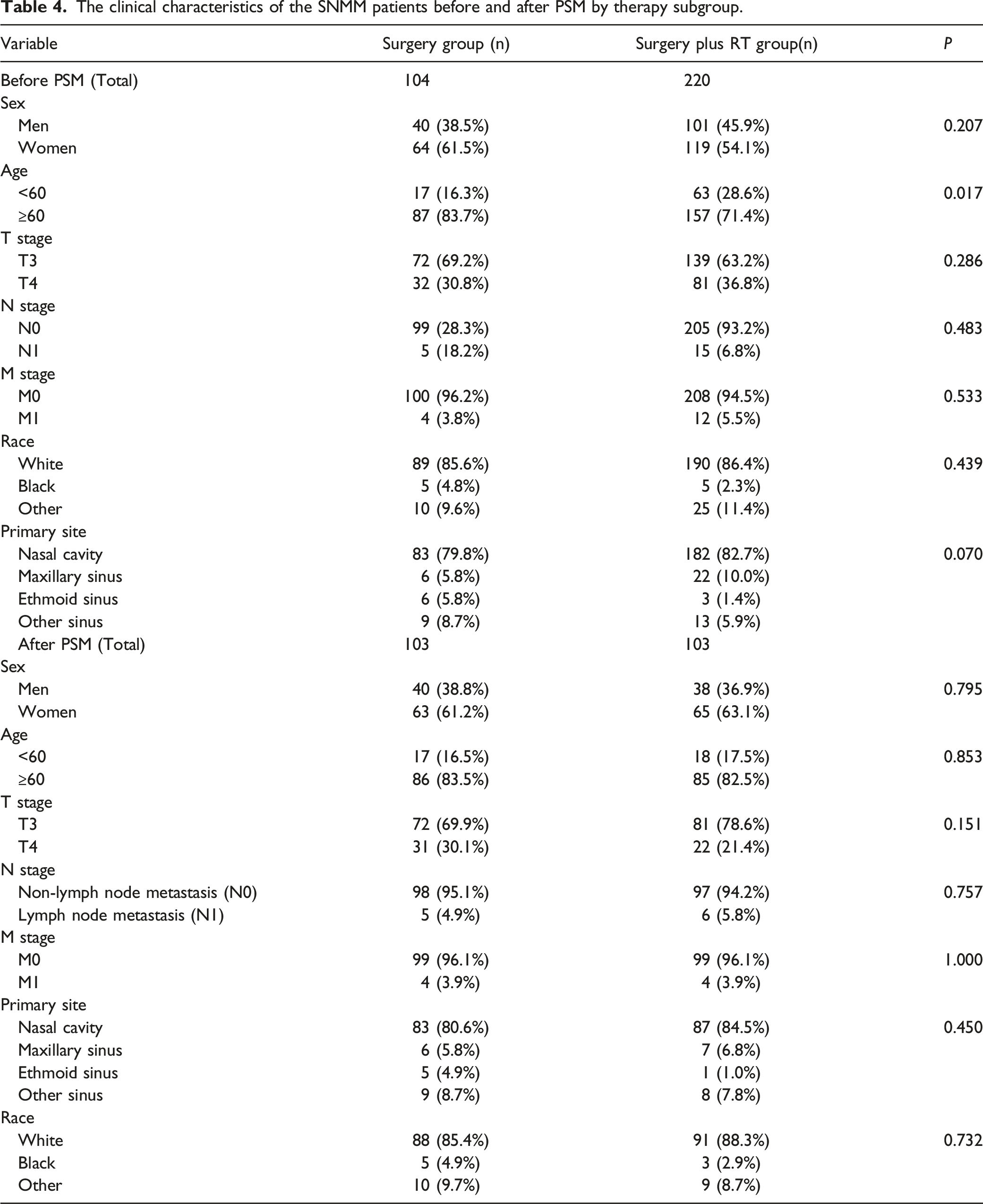
The clinical characteristics of the SNMM patients before and after PSM by therapy subgroup.
Discussion
Previous literatures have reported the LN metastasis rates of 3.5–20% in patients with SNMM at initial diagnosis.1,2,7,11 Similarly, we reported a LN metastasis rate of 7.6% (28/377) in our cohort. Although the lymph node metastasis rate in SNMM at initial diagnosis was relatively low, approximately 20–40% of patients developed regional recurrence that complicated their disease course.2,13-15 In this cohort, lymph node metastasis was associated with distant metastasis (P = 0.000), indicating that occult metastasis may occur in patients with lymph node metastasis and then further develop into distant metastasis.
The LN metastatic patterns of SNMM remains unclear. Previous studies revealed that level I and level II nodes were the most common sites of lymph node metastasis of SNMM.8,13 Consistently, in the present study, level I (57.1%) and level II (53.6%) nodes were also the most common sites of metastasis, followed by levels III (17.9%) and IV (17.9%). Metastases to other sites in the head and neck such as the level Ⅴ, parotid, facial, retropharyngeal, and retroauricular nodal were rare. The pattern of nodal metastasis was similar to that of squamous cell carcinoma, 16 indicating level I and level II lymph nodes may be the sentinel lymph nodes, with metastasis occurring first in level I and level II and then spreading to other sites of the head and neck.
Whether lymph node metastasis affects survival among SNMM patients remains controversial. Lund et al. reported that lymph node metastasis did not have an impact on either local control or OS. 17 However, Low et al. found that nodal metastasis at presentation was an adverse prognostic factor for OS and DSS in SNMM patients, 8 which concurs with our findings. The discrepancy among these studies may be due to statistical bias caused by the small sample cohort. Overall, lymph node metastasis was associated with distant metastasis, indicating that nodal metastasis may promote distant metastasis, resulting in poor survival outcomes.
Distinct differences of the OS and DSS rates were observed between SNMM patients without and with lymph node metastasis (5-year OS rates without and with lymph node metastasis were 35.2% and 5.3%, respectively, while the 5-year DSS rates without and with lymph node metastasis were 39.6% and 5.3%, respectively). Although nodal metastasis was found to be an important prognostic factor for survival, the associations between clinical factors and nodal metastasis in SNMM patients has remained largely unexplored and controversial. Amit et al presented that all the clinical and pathological characteristics, including pT stage, were not correlated with nodal metastasis. 13 However, tumor size and T stage were correlated with nodal metastasis in our cohort, this difference may be due to the relatively small sample cohort and single-center selection bias in the former study. The lymph node metastasis rate (3.1%, 9.6%, and 17.9%) increased as the tumor diameter (≤2 cm, >2 but ≤5 cm, and >5 cm, respectively), and extension enlarged in this study, suggesting that a larger tumor burden could promote lymph node metastasis, Tumor diameter could be a factor of assessing the probability of nodal metastasis and prognosis from another perspective.
According to previous studies, the prognosis of patients with SNMM is poor.7-9 A systematic review showed 5-year OS rates have ranged from 20 to 60%, and the mean 5-year OS and DSS rates were 27% and 26%. 18 These rates are consistent with those observed in the present study (33.1% and 37.0%, respectively). Gal et al demonstrated T stage, N stage, and M stage were independent factors affecting the prognosis of SNMM patients, age was also significantly associated with survival. 1 Though we found age was not associated with the survival, the 5-year OS was better in patients younger than 60 years (36%) than patients older than 60 years (32%). Excepting the T, N, and M stages, we also found that tumor size was independently and significantly related to the prognosis of SNMM patients. Advanced stage was associated with poor survival, whereas race and sex were not associated with the survival.
The role of radiotherapy in the management of SNMM has remained unclear, with some authors considering that postoperative radiotherapy may improve local control19-21 and OS. 12 However, other researchers have noted that SNMM is radio-resistant, and thus, postoperative radiotherapy could not improve survival.1,10,11 To the best of our knowledge, most of the studies were not analyzed by PSM, which was used to control potential confounding factors caused by selection bias. In the present study, treatment method was shown to be an important prognostic factor for SNMM patients, compared with the lack of treatment group, as patients treated with surgery or surgery plus radiotherapy showed better survival. However, no survival benefit was observed in the surgery plus radiotherapy group of patients with or without lymph node metastasis compared to the surgery alone group before and after PSM analysis. But it was still possible that these surgery plus radiotherapy group patients may have positive margins after surgical resection, suggesting the use of radiotherapy should be undertaken cautiously after comprehensive consideration.
Due to the rarity of SNMM, most of the studies on this disease were case reports and single-center retrospective studies. The use of SEER data allowed us to specifically focus on SNMM, and the SEER database includes the most comprehensive data source of cancer epidemiology, survival and end results, covering approximately 34.6% of the United States cancer cases. 22 However, it does not provide information of radiotherapy modes and surgery procedures, which may confound our analyses.
This study demonstrated that level I and level II lymph nodes may be the sentinel nodes for SNMM metastasis, with metastasis possibly first occurring in these lymph nodes and then spreading to other sites in the head and neck. Advanced T stage and increasing tumor size could promote LN metastasis. LN metastasis may promote distant metastasis and remains an important prognostic factor for patients with SNMM. Finally, postoperative radiotherapy did not improve survival of patients with or without lymph node metastasis.
Footnotes
Author contributions
WD and LX wrote, conceived the manuscript, and confirm the authenticity of the data. JL, ZL, TH, and XC carried out the literature and study research and generated the figures. TZ and TL revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundings of Science and Technology Projects in Guangzhou (2060206), the National Science Foundation of China (81972896 and 81302368), the Project of State Key Laboratory, State Key Laboratory of oncology in South China (HN2019-09), and the Guangdong Province Natural Science Foundation (2019A1515010288 and 2015A030313058).
