Abstract
Malignant sinonasal mucosal melanoma is a rare and aggressive neoplasm. As it shows nonspecific clinical symptoms, magnetic resonance imaging (MRI) is the most effective diagnostic tool. Before a mass is histopathologically confirmed, MRI is regarded as the optimal differential diagnostic procedure, especially in patients who present with a unilateral sinonasal cavity mass. However, we recently treated a patient who showed atypical MRI findings, resulting in a preoperative misdiagnosis of a vascular tumor. By pathologic examination, the patient was confirmed to have a malignant mucosal melanoma that originated from the nasal septum. This case demonstrates that unilateral nasal cavity masses that present with nonspecific symptoms, such as epistaxis, should be evaluated for possible malignant neoplasms, even if the mass has benign-looking characteristics in preoperative endoscopic findings and MRI images.
Introduction
Malignant mucosal melanomas within the sinonasal cavity are rare and present with aggressive features. Their prevalence is reported to be <4% of all head and neck cancers, 55% of all head and neck melanomas, and 0.3% to 2% of all melanomas.1,2 Moreover, the 5-year survival is reportedly under 35%. 3 Malignant sinonasal mucosal melanomas arise from mucosa melanocytes, and the primary sites are nasal septum, nasal lateral wall, and the inferior nasal concha.1,2 As symptoms are nonspecific, magnetic resonance imaging (MRI) with a contrast agent is regarded as the prime technique for diagnosis, because, characteristically, melanin has strongly hyperintense and hypointense signal intensity for muscle in nonenhanced T1-weighted and T2-weighted images. 4 Here, we present a case of nasal septal malignant mucosal melanoma, which was preoperatively misdiagnosed as a vascular tumor due to endoscopic findings and MRI images.
Case Report
A male in his 70s with no medical history presented with intermittent bleeding from the left nasal cavity and nasal obstruction. The symptoms had started approximately 3 months prior and progressively worsened. He had no history of anticoagulant medication use and was a nonsmoker. Nasal endoscopy showed a red-purple colored mass that filled his left nasal cavity (Figure 1A). Images of his paranasal sinus captured by MRI showed a 2.6 × 2.3 cm mass with low signal intensity on T1- and a heterogeneous intermediate to high signal intensity on T2-weighted images (Figure 1B). The mass showed a diffusion restriction pattern. Based on these findings, the mass was suspected to be a vascular tumor, such as a lobular capillary hemangioma. The patient underwent endoscopic sinus surgery, and the mass was found to be attached to the anterosuperior mucosa of the nasal septum. However, the mass was not attached to the inferior turbinate or posterior nasal septum. The mass and the anterosuperior septal mucosa were removed. Postoperatively, a histopathologic examination revealed that the mass consisted of malignant epithelioid cells and contained prominent eosinophilic nuclei in epithelial cells with irregular contours. Immunohistochemical staining of the mass was positive for melan-A and S-100 (Figure 2). As a result of these findings, the mass was confirmed as malignant mucosal melanoma. Neck computed tomography and positive emission tomography scans showed no definite lymph node or distant metastasis. Therefore, the patient was diagnosed as stage I by the Ballantyne classification and as T3N0M0 by American Joint Committee on Cancer staging. 1 Chemoradiotherapy was performed postoperatively to eradicate any residual melanoma cells that may have remained in the surgical margins. After treatment, the patient maintained a “no evidence of disease” status for 7 months.
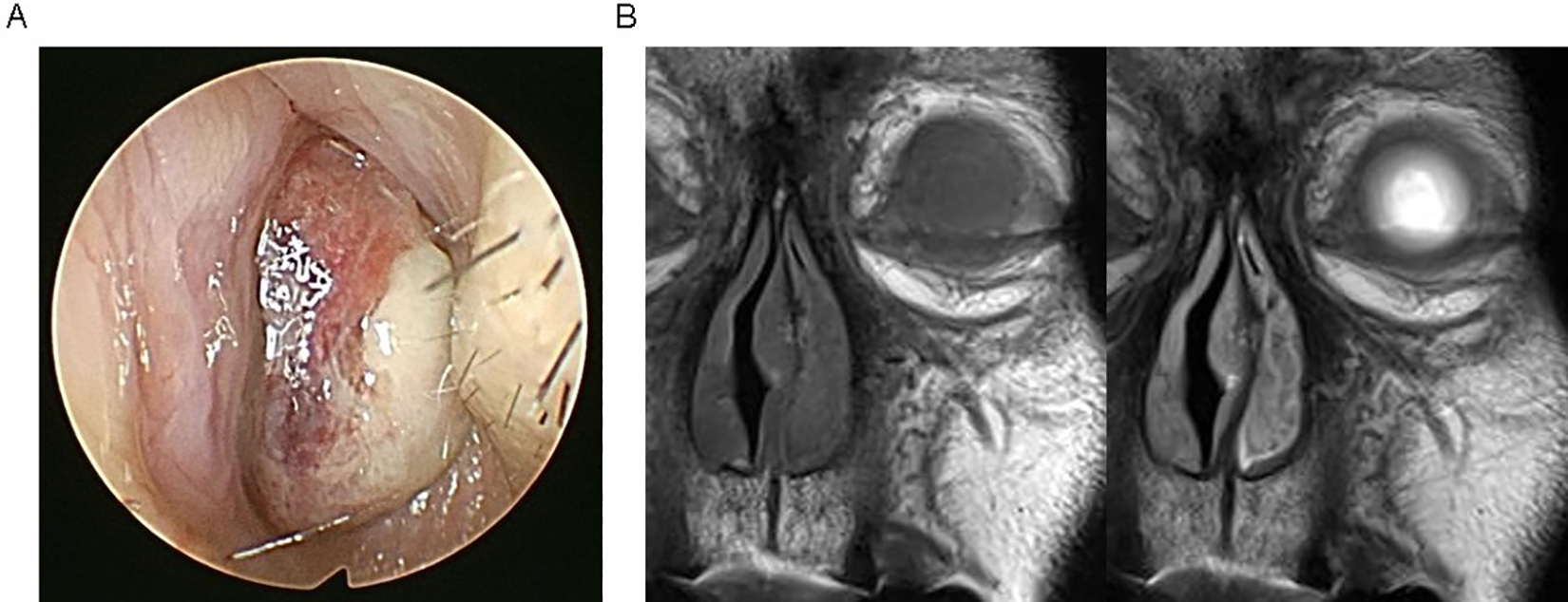
A, Endoscopic finding of the left nasal cavity. B, Contrast-enhanced magnetic resonance imaging (MRI) coronal view (left, T1-weighted; right, T2-weighted image).
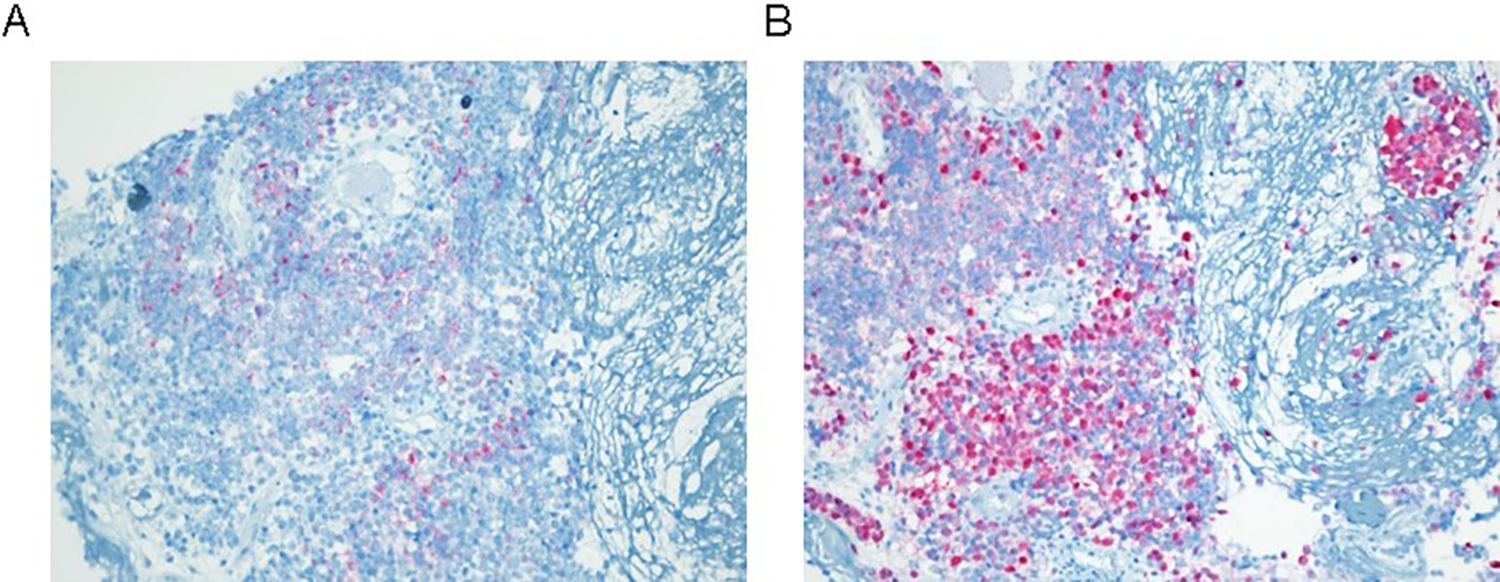
Results of immunohistochemical staining of biopsy specimen. A, melan-A, (B) S-100; original magnification ×100.
Discussion
Malignant mucosa melanomas are very rare cancers that account for just 1% of all cases of melanomas. 2 The most common sites are the head and neck, and the incidence is slightly, but not significantly, higher in women than men. 5 They are usually found in people younger than the age of 65. Sun radiation, cigarette smoking, and formaldehyde exposure have been suggested to be risk factors for sinonasal mucosal melanoma. 6 In most cases, MRI is useful for melanoma diagnosis and typically shows a high signal intensity on T1- and a low signal intensity signal on T2-weighted images. 7 This is likely because melanomas have a high melanin content or hemorrhage, which is typically correlated with hyper signal intensity and hypo signal intensity on T1- and T2-weighted images, respectively. Magnetic resonance imaging with diffusion-weighted imaging and dynamic contrast enhancement imaging can also provide useful information for diagnosing sinonasal mucosal melanomas. 2 In typical cases, MRI with a contrast agent is regarded as the best technique for the diagnosis of a unilateral sinonasal cavity mass because it can typically distinguish between solid lesions and inflammatory mucosal lesions. Also, specific characteristics of MRI images enable the differential diagnosis of various solid lesions. Vascular tumors show intermediate T1 and T2 signal intensities with multiple flow voids from feeding vessels. Inverted papillomas have a “cerebriform” appearance on T2- and/or contrast-enhanced T1-weighted MRI images. 7 Therefore, MRI findings can help distinguish melanoma from other types of tumors. However, in our case, we identified low signal intensity on T1- and high signal intensity on T2-weighted MRI images, which most likely indicated a vascular tumor. Malignant tumors need more attention during preoperative evaluations due to the possibility of lymph node or distant metastasis and require wide resections, which include the tumors and any primary attached sites. Therefore, it is important to remember that, while malignant mucosal melanomas commonly show typical characteristics features in MRI images, they might also show atypical signal intensity. Furthermore, a previous study found that MRI signal intensity is associated with malignant mucosal melanoma prognosis and that atypical signal intensity can be associated with poor outcome. 8
Surgery is regarded as the first-line treatment for malignant mucosal melanomas. 2 Since malignant mucosal melanomas are composed of cells with a high post-irradiation regenerative capacity, radiation is not an effective management tool for malignant mucosal melanoma. 2 Radiotherapy is suggested in cases of residual tumor cells in the surgical margins or local recurrence. Chemotherapy is applied for palliative treatment or in metastatic cases. 2 Recently, immunotherapies, such as anti-PD1/PDL-1 agents, have been suggested; however, specific data are lacking. In spite of these new therapeutic trials, the prognosis of malignant mucosal melanoma in the sinonasal cavity remains very poor. Five-year overall survival is less than 40%, and the survival duration does not exceed 28 months.9,10 In about half of cases, local recurrence occurs, with metastasis commonly observed in the lungs, liver, bone, and (less often) brain. 7 Advanced age and larger tumor size have been suggested as common clinical characteristics associated with poor prognosis. 11 Therefore, early diagnosis and proper management at the time of the first surgery may be the most important factor in managing malignant mucosal melanoma.
Conclusion
Magnetic resonance imaging plays a crucial role in the diagnosis and characterization of unilateral sinonasal masses. Furthermore, MRI is the gold standard technique for the diagnosis and local–regional staging of sinonasal malignant mucosal melanoma. However, such as in the case presented here, it is important to be aware that sinonasal malignant mucosal melanomas can show atypical images in MRI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
