Abstract
Objectives
The anterolateral thigh (ALT) flap is a versatile workhorse flap for the reconstruction of head and neck defects; however, variations in the location of perforators are a significant concern. This prospective study proposes a rapid, effective, and convenient method for mapping the location of sizeable perforators on the curved thigh skin surface using the visual coordinate system in computed tomographic angiography (CTA) images.
Methods
Fourteen patients suffering from defects after head and neck carcinoma resection underwent CTA prior to ALT flap reconstruction at the Department of Otolaryngology-Head and Neck Surgery, Beijing Friendship Hospital, Capital Medical University, from January 2018 to August 2021. Using sterile gauze swab developing lines pasted on the patients’ thighs, we established a coordinate system visualized both in CTA images and the thighs. The perforator information was then compared between the CTA and intraoperative findings.
Results
Twenty-one perforators were detected on CTA in the designed flaps, which were also confirmed during the operation with a visual pulse. The course and source of the perforators were consistent with the intraoperative findings. The distances of perforator coordinate points between CTA and intraoperative findings were <5 mm. Another five perforators (mostly without visual pulse) found intraoperatively could not be identified on preoperative CTA. Most flaps survived in their entirety, and one flap suffered a small area of necrosis due to perforator compression.
Conclusion
This study demonstrated the efficacy of a novel perforator mapping method for anterolateral thigh flaps.
Keywords
Introduction
Reconstruction of complex soft tissue defects in the head and neck region is challenging due to the area’s complex anatomy. Numerous studies have proposed different flap types to repair these complex defects.1–4 Perforator flaps have gained popularity over the last decade for reconstructive surgery. First introduced by Song et al. in 1984, the anterolateral thigh (ALT) flap has become known as a versatile workhorse flap for restoring multiple soft tissue defects. The ALT flap can be modified to include muscle, subcutaneous tissue, nerve, or fascia. The ability to fabricate individualized flaps using various perforators enables the use of ALT flaps for cases with complicated composite resections.5–7 However, despite well-described anatomy, the limiting factor of variable vasculature makes the unpredictability of ALT perforators a challenge even for experienced surgeons.8–11 Therefore, accurate preoperative determination of the location of reliable perforators is important for safely planning perforator flaps. Preoperative imaging is one means to predict individual vascular anatomy and maximize flap vascularity and operative success. At present, five main tools can be used to preoperatively assess perforators: hand-held Doppler (HHD), color duplex sonography (CDS), digital subtraction angiography (DSA), computed tomography angiography (CTA), and magnetic resonance angiography (MRA).10–15 Recently, CTA was reported to be successfully used in the preoperative assessment of ALT perforator flaps, with the perforators located by virtual reference lines connecting the anterior superior iliac spine (ASIS) and patella in preoperative CTA images.10–12,14 Nevertheless, it is still a challenge to effectively position the perforators shown on the CTA onto the patient’s thigh. To address this issue, we used sterile gauze swab developing lines pasted on the patient’s thighs to establish a visual coordinate system in the CTA images that could effectively locate perforators on the patient’s thighs before surgery. Here, we propose a rapid, effective, and convenient method to mark perforator locations on the skin surface preoperatively.
Patients and methods
Patients’ data and the details of perforators detected by computed tomographic angiography.
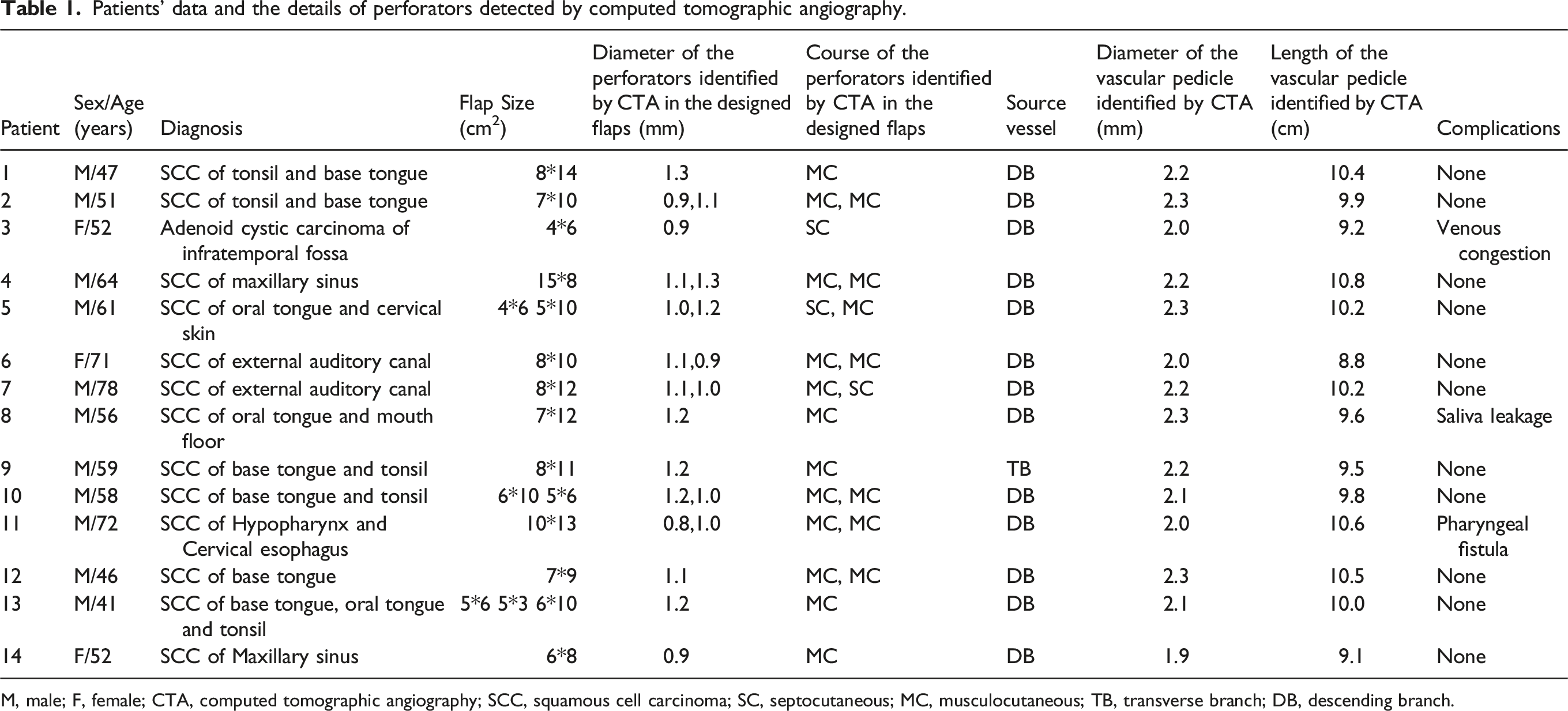
M, male; F, female; CTA, computed tomographic angiography; SCC, squamous cell carcinoma; SC, septocutaneous; MC, musculocutaneous; TB, transverse branch; DB, descending branch.
With the patient in the supine position, we drew a line from the ASIS to the superior lateral border of the patella on both thighs. The line was defined as the x-axis; the midpoint of this line was defined as the origin, and the y-axis line was constructed vertically at the midpoint of the x-axis. Next, we pasted sterile gauze swab developing lines on the x-axis and y-axis to establish a coordinate system.
CTA imaging protocol
The patients were examined using a 160 mm detector, spectral CT (Revolution CT; GE Healthcare, Waukesha, WI, USA). CTA images were obtained using fast kV-switching Gemstone spectral imaging (GSI) with the following parameters: rotation speed = 0.8 s, automatic mA, pitch = 0.516: 1, and reconstruction algorithm ASIR-V = 30%.
Scans were obtained in an antegrade direction from a point superior to the ASIS to just below the patella. The contrast medium (Iohexol, 350 mgI/mL) was injected at 0.8 mL/kg with a flow rate of 4 mL/s. Subsequently, 60 mL of saline was injected with a flow rate of 4 mL/s. After scanning, the data were reconstructed at 50-keV level and 0.625-mm thickness image and transmitted to a GE ADW 4.7 workstation (Advantage workstation; GE Healthcare, Chicago, IL) for revascularization using GSI (Gemstone Spectral Imaging) viewer software.
Image interpretation
The images were preoperatively reviewed and discussed by radiologists and plastic surgeons. The subcutaneous vasculature of the anterolateral thigh was obtained by curved projection reformation (CPR) and the volume rendering technique (VRT). We traced the perforators up to the source vessel, and only the perforators originating from the same source vessel were recorded. Next, we marked the points where the perforators pierced the deep fascia on the axial images, vertically projected the points to a tangent line perpendicular to the curved skin surface of the thigh, recorded the points on the visual coordinate system, and finally marked the skin (Figure 1). This method made the CTA data more meaningful for the surgeon by converting three-dimensional (3D) data to two-dimensional (2D) data. The perforator diameter (at the point of origin), course, origin, vascular pedicle diameter (at the point of origin), and length (from the origin to the point of the dominant perforator piercing the deep fascia) were also recorded (Table 1). The process of perforator mapping of ALT flaps using CTA and visual coordinate system in patient 13. (A) Establishing visual coordinate system by pasting sterile gauze swab developing lines on the thigh, superior lateral border of the patella (solid arrow), anterior superior iliac spine (hollow arrow). (B) Curved projection reformation image showing the subcutaneous vasculature of the anterolateral thigh, subcutaneous perforator (solid arrow), descending branch of the lateral circumflex femoral artery (hollow arrow). (C) Volume rendering technique reconstruction of skeletal, muscular and major vascular structures of the lower extremities, highlighting the descending branch of the LCFA and cutaneous perforator(hollow arrow),visual coordinate system(solid arrow). (D) Axis image of CTA, recording the point of cutaneous perforator piercing the deep fascia on the curve skin surface, the point of cutaneous perforator piercing the deep fascia(solid arrow), projection point on the curve skin surface (hollow arrow). (E) Volume rendering technique showing the cutaneous perforator (hollow arrow) and the source vessel (solid arrow). (F) Marking the data on the coordinate system of the thigh, origin (solid arrow), projection point of cutaneous perforator piercing the deep fascia (hollow arrow).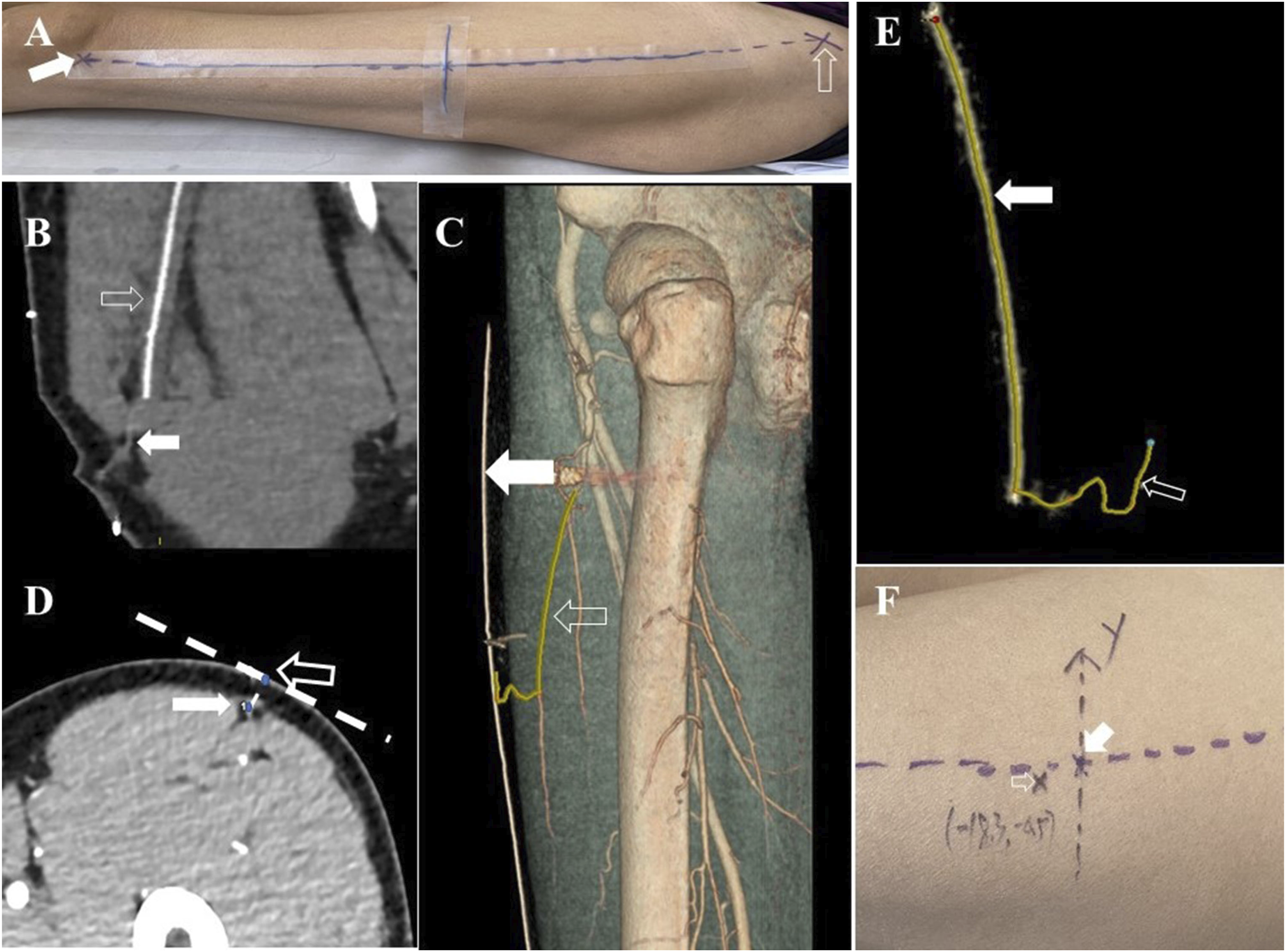
Surgical process
The decision to use the left or right thigh as the flap donor was determined by correlating the radiologic findings with the preoperative evaluation. Ultimately, we chose the thigh with the larger diameter and higher number of perforators. The form and dimension of the flap templates were designed by measuring the defects. Generally, the flap was designed to be centered at the preoperative marker of a sizeable perforator with the shortest intramuscular course. The medial incision of the flap was made down to the fascia above the rectus femoris muscle. Subfascial dissection continued laterally until the perforators piercing the deep fascia were identified. All the perforators encountered were preserved with the dissection proceeding to identify their source arteries. For musculocutaneous perforators, intramuscular dissection was undertaken, and approximately 0.5 cm of the muscle around the vessel was preserved to protect it. Only the perforators originating from the same vessel (mostly the descending branch of the LCFA) were ultimately reserved. After the lateral incision was made, the vascular pedicle was elevated from the distal to the proximal end, and the vascular branches to the muscle were ligated. Various amounts of vastus lateralis muscle were included in the flaps when the muscle was needed. Generally, the vascular pedicle was disconnected at its origin (Figure 2). Finally, the flaps were transferred to their recipient sites, microscopic anastomosis was performed, and the donor sites underwent primary closure. Information about the perforators (location, course, and origin) obtained from CTA was compared with the intraoperative findings in the region of the designed flaps. The diameter and length of the vessels were not measured during the operation to avoid prolonged surgical time and surgical injury. Surgical process of elevating anterolateral thigh flaps in patient 13. (A) Designing ALT flaps according to the head and neck defects, origin (small black arrow), projection point of the cutaneous perforator piercing the deep fascia (small white arrow), flap used for the reconstruction of lateral pharyngeal wall (large black arrow),flap used for the reconstruction of base tongue, flap used for the reconstruction of oral tongue (hollow white arrow). (B) Confirming the cutaneous perforator percing the deep fascia (white arrow). (C) Islanding the anterolateral thigh flaps, origin (hollow arrow), actual point of the cutaneous perforator piercing the deep fascia(solid arrow). (D) Elevating the anterolateral thigh flaps.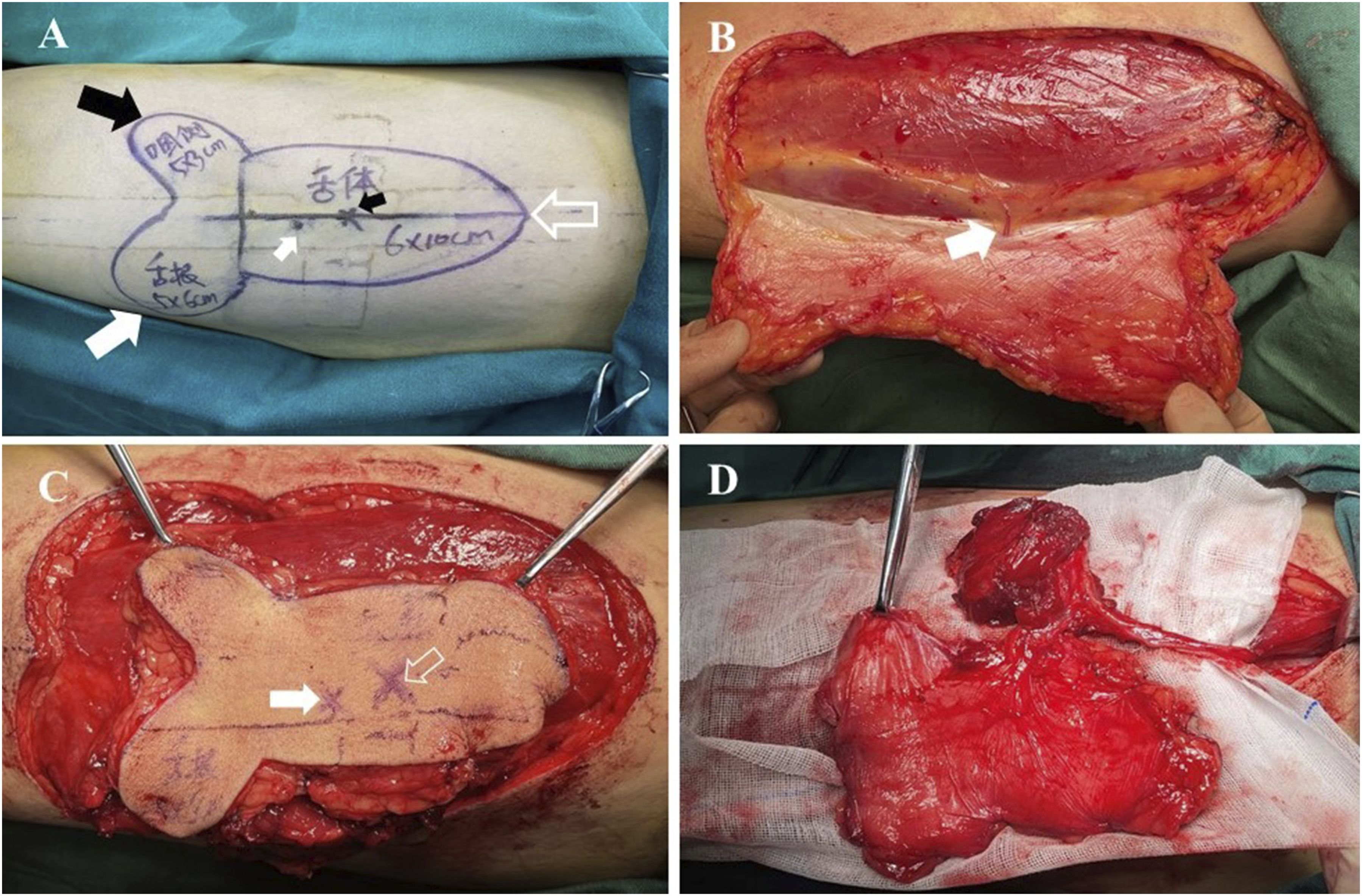
Statistical analysis
The data were reported as the mean ± standard deviation (SD). Coordinate data of the perforators were compared between CTA and intraoperative findings. No statistical analysis was performed due to the relatively small sample size. Considering the accuracy requirements of the actual operation and the presence of measurement errors, the distances of perforator coordinate points between CTA and intraoperative findings were defined at <5 mm as effective mapping without the necessity to revise the operative strategy.
Results
For the 14 patients, 21 perforators were detected on CTA in the designed flap. The course and source of these perforators were consistent with the intraoperative findings. Of the perforators, 85.7% (18/21) were musculocutaneous, and 14.3% (3/21) were septocutaneous. Furthermore, 92.9% (13/14) of source vessels were the descending branch of the LCFA, and only one was the transverse branch of the LCFA. The diameters of the perforators and the pedicle identified by CTA in the designed flaps were 1.07 ± 0.14 and 2.15 ± 0.13 mm, respectively. The length of the vascular pedicle was 9.9 ± 0.60 cm.
Perforators’ data compared between CTA and intraoperative findings in the designed flaps.
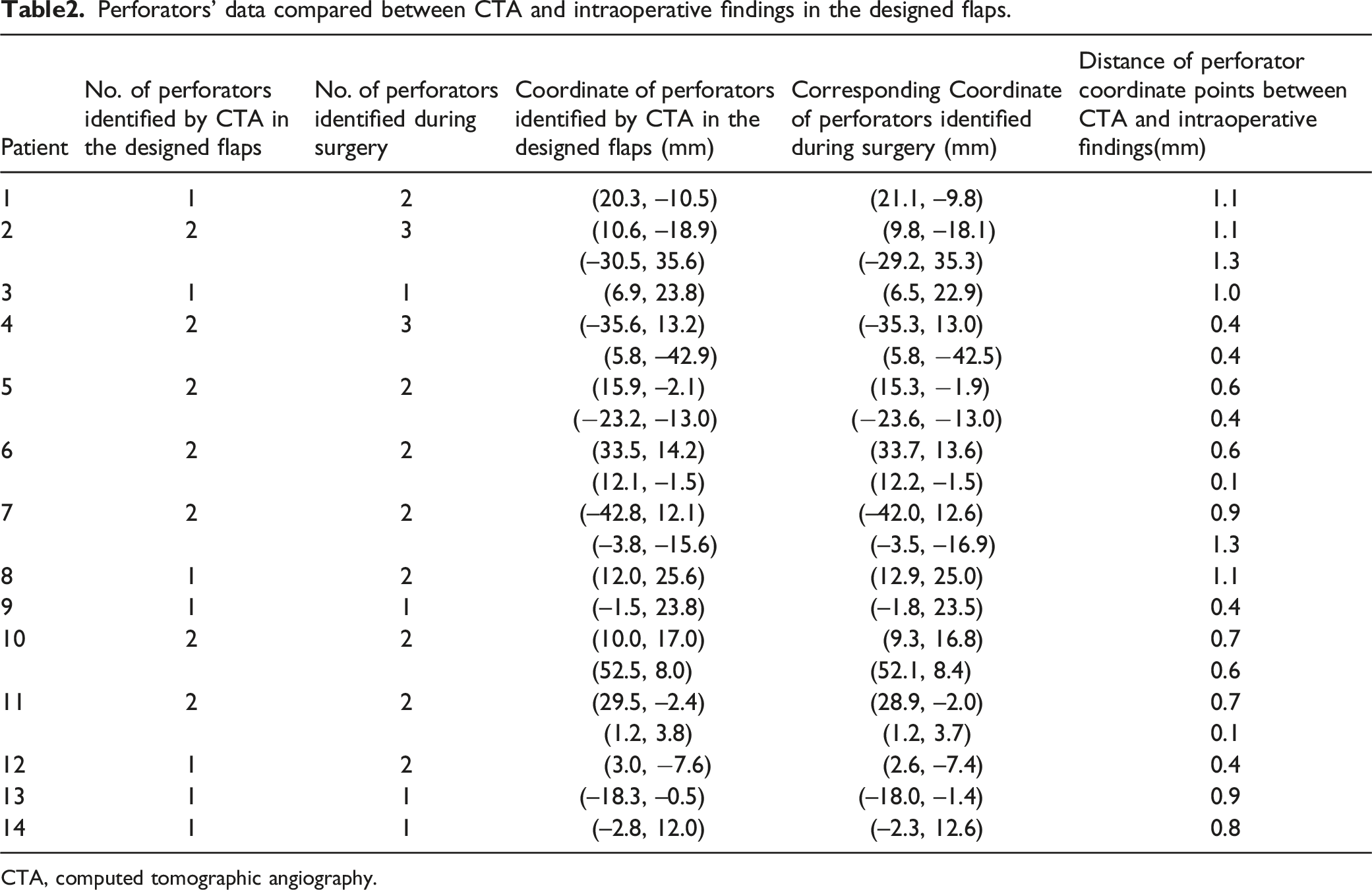
CTA, computed tomographic angiography.
Complications included saliva leakage in one patient, pharyngeal fistula in one patient due to the infection of dead space, and venous congestion in one patient due to perforator compression, which resulted in a small area of flap necrosis (0.5 × 1 cm). All other flaps survived in their entirety. There was no donor site morbidity. Table 1 lists additional outcomes.
Discussion
The ALT flap is a recently popularized flap with applications in the reconstruction of head and neck defects. Its advantages include providing a large skin paddle, fascia, muscle, and a long vascular pedicle with good caliber, facilitating vessel anastomosis. There is also minimal donor site morbidity and the opportunity for a two-team combined approach, decreasing the surgery and anesthesia time. Its main disadvantage is variable vasculature.5-7
Previous studies have focused on statistical data of cutaneous perforator location, the success of gaining ALT flaps based on cadaver dissection, and surgical experience.5-7,16,17 It has been postulated that cutaneous perforators are located at the midpoint of the reference line drawn between the ASIS and superolateral border of the patella or in the inferolateral quadrant of a circle with a radius of 3 cm drawn from the midpoint as the center. However, preoperative planning should be based on individualized data rather than statistics to increase the probability of achieving optimal outcomes with minimal loading for the plastic surgeon.
Of the main tools currently available to preoperatively assess cutaneous perforators, MRA and DSA are not yet in routine practice due to image resolution limitations and availability. Though noninvasive and inexpensive, HHD and CDS can be time-consuming and inaccurate due to their being too sensitive with low specificity. More importantly, they cannot reproduce 2D or 3D images of complete vascular anatomy that can be used by the surgeon during flap design or flap elevation. The advantage that CTA offers is providing an image with accurate visual details of the caliber and course of the vessels and their relationships with other anatomic structures.10-14 However, there are still issues to be addressed.
Firstly, perforators are better visualized when surrounded by subcutaneous fat than by more radiopaque muscle, skin, or fascia. The reality is that most patients with head and neck cancer have experienced significant weight loss, which increases the difficulty of mapping the perforators. 18 On the other hand, as the thighs have a highly curved surface, manipulating 3D data requires multiple oblique planes and makes viewing the course of perforators more difficult. Secondly, the ability to accurately translate 3D images of cutaneous perforators into 2D images on the highly curved surface of the thigh is more meaningful for plastic surgeons. Most prior studies located the perforators through a virtual reference line on CTA images,10,11,18,19 which was obviously less precise during the actual operation.
To address the above issues, using statistical data and gauze swab developing lines pasted on the patient's thighs, we established a coordinate system that was visualized both in the CTA images and thighs. The x-axis roughly followed the septum between the rectus femoris and vastus lateralis. The developing lines were easy to obtain, very soft, and fit tightly onto the curved skin surface of the thighs, without many artifacts in the CTA images.
Identifying the subcutaneous course of perforators is much easier than the intramuscular course. The reconstruction of the subcutaneous vasculature of the thigh was first obtained by CPR and VRT. Then we traced the subcutaneous course up to the source vessel, focusing on the cutaneous perforators emerging beneath the origin of the coordinate system and originating from the descending branch of the LFCA. In our cohort, 92.9% (13/14) of source vessels were the descending branch of the LFCA. Twenty-one perforators were detected on CTA in the designed flap, which were also confirmed during the operation with a visible pulse. Notably, another five perforators found intraoperatively could not be identified by CTA. However, as most of these perforators were not sizeable and without a visible pulse, it was not necessary to revise the operative strategy. The diameter of perforators identified by CTA was 1.07 ± 0.14 mm. It is not easy to determine the threshold of the diameter of the vessels visualized by CTA. It could be claimed that sizeable perforators are identifiable by CTA in most cases.
As ATL flaps are usually harvested beneath the deep fascia, the location where the perforators pierce the deep fascia is very important. We vertically projected this on the curved skin surface of the thigh in the CTA axial images, recorded it in the visual coordinate system, and finally marked it on the patient’s thigh. All 21 perforators identified by CTA were also confirmed during the operation in our study. The distance of perforator coordinate points between CTA and intraoperative findings was <5 mm in all the cases. We believe that this mapping technique provided sufficient effective perforator information during the actual operation without the necessity to revise the operative strategy. It should be noted that the rotation of the lower extremity may cause displacement of the skin surface from the deep tissue, leading to more errors in locating perforators. Keeping patients in the same position while pasting the developing lines and performing CTA and the operation could help minimize displacement of the detected perforators.
We also recorded the diameter and length of the vascular pedicle in the CTA images preoperatively to confirm whether the vascular pedicle was suitable for vessel anastomosis. However, we did not compare the data during the operation to avoid an unnecessarily prolonged surgery time and surgical injury. In our cohort, the diameter of the vascular pedicle identified by CTA was 2.15 ± 0.13 mm, with a length of 9.9 ± 0.60 cm, which was sufficient for free flap vessel anastomosis in the reconstruction of head and neck defects.
The significant disadvantages of CTA are exposure to ionizing radiation, using a potentially nephrotoxic contrast medium, and imaging small perforators with weak signal intensities. It is important to realize that anatomical knowledge is the cornerstone of a successful flap harvest. The deficiency of this study is that the sample size was relatively small, which was not conducive to effective statistical analyses.
Conclusion
Our findings demonstrated that this fast and simple mapping technique could achieve sufficient effective perforator information of the ALT flaps, supporting the preoperative selection of a sizable perforator with the shortest intramuscular course for the reconstruction of head and neck defects.
Footnotes
Declaration of conflicting interests
The author(s) declare no potential competing interests concerning the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Key Science and Technology Program of Beijing Municipal Commission of Education [grant number KZ202110025035], Research and Development Project of Scientific Research Instruments and Equipment of Chinese Academy of Sciences-major instruments project [grant number YJKYYQ20180039] and Digestive Medical Coordinated Development Center of Beijing Municipal Administration of Hospitals [grant number XXZ0604].
Ethical approval
The study protocol was approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University (2022-P2-149-01). All the patients provided written informed consent forms.
Data availability
All data, models, and code generated or used during the study, can be available upon reasonable request from the corresponding author.
