Abstract
Epstein-Barr virus-positive anaplastic plasmacytoma in the sinonasal tract is uncommon. Herein, we present the case of a 40-year-old male patient with a right sinonasal tract filled with a soft tissue mass, as shown on CT. Apart from frequent nosebleeds, he did not report any specific sinonasal symptoms, such as nasal obstruction, discharge, or loss of smell. The patient underwent functional endoscopic sinus surgery under general anesthesia. The diagnosis of Epstein-Barr virus-positive anaplastic plasmacytoma was confirmed by lesion biopsy, subsequent immunohistochemical staining, and in situ hybridization.
Keywords
Introduction
Solitary plasmacytoma (SP) is a subtype of plasma cell neoplasm, defined by the sole mass of clonal plasma cells, no or minimal bone marrow plasmacytosis, and no symptoms other than those associated with the primary lesion. 1 Extramedullary (extraosseous) plasmacytoma (EMP) (in soft tissues) and solitary bone plasmacytoma are the two most common presentations of SP. 2 EMP may involve any site or organ, but it most frequently occurs in the head and neck area, including the sinuses, nasopharynx, and oropharynx. 3 Epstein-Barr virus (EBV)–related plasmacytoma has rarely been reported. Plasmacytomas rarely express anaplastic features and there are no guidelines for treatment. We searched the PubMed, Web of Science, and EMBASE databases and found no more than five English articles on anaplastic plasmacytoma in the sinonasal tract. In this study, we describe a case of Epstein-Barr virus (EBV)–positive anaplastic plasmacytoma of the right sinonasal tract, provide the main clinical and pathological features, and comprehensively review the limited literature on sinonasal anaplastic plasmacytomas.
Case presentation
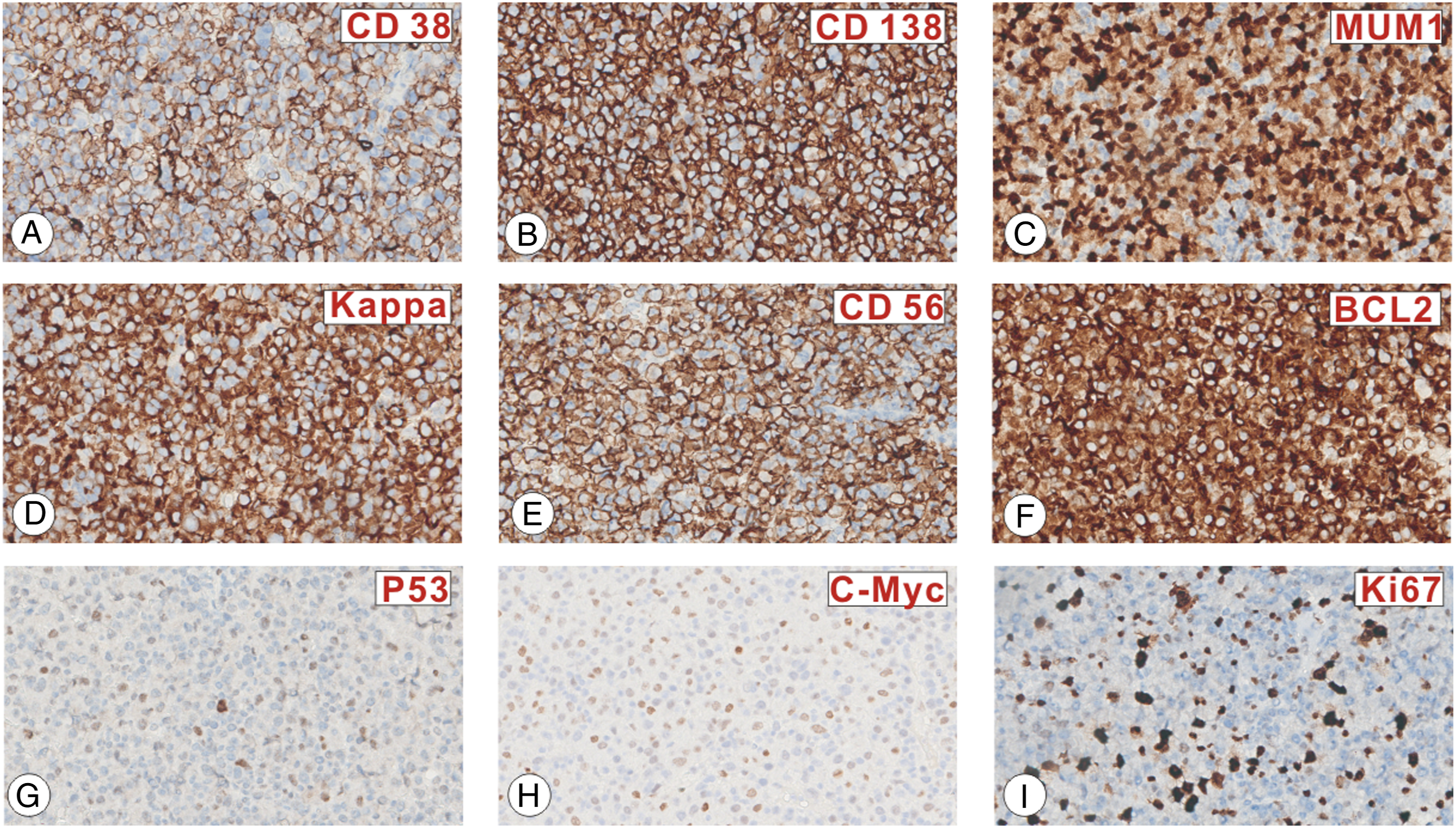
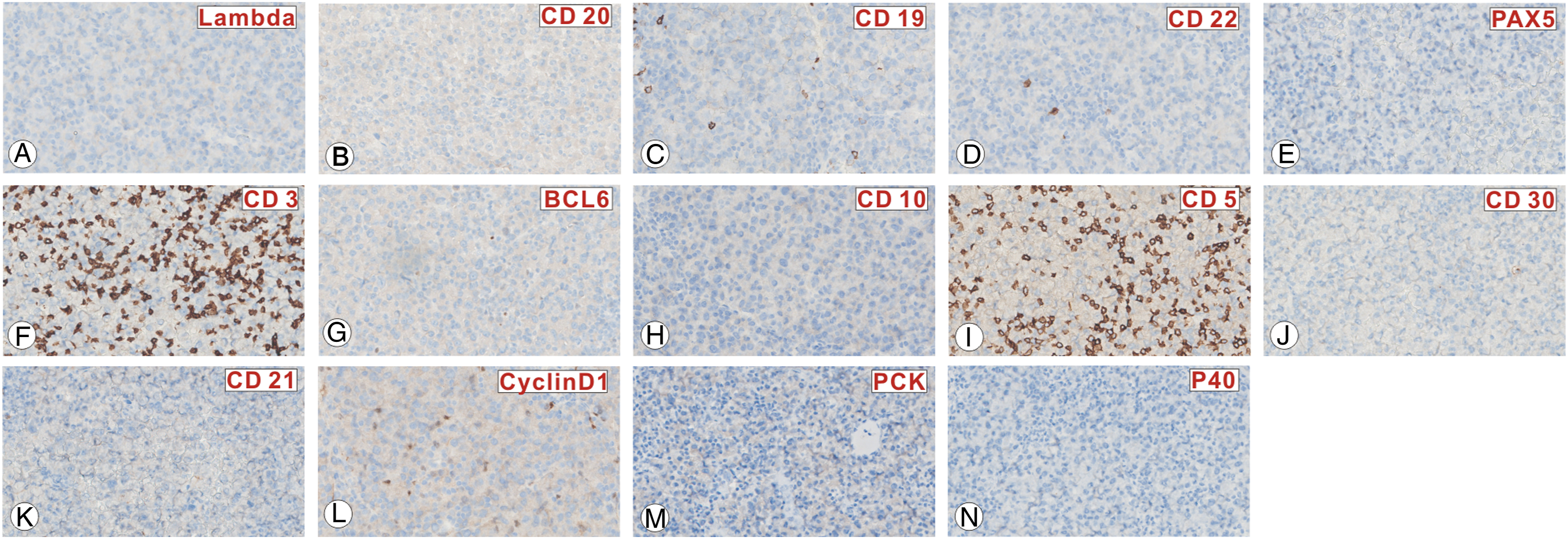
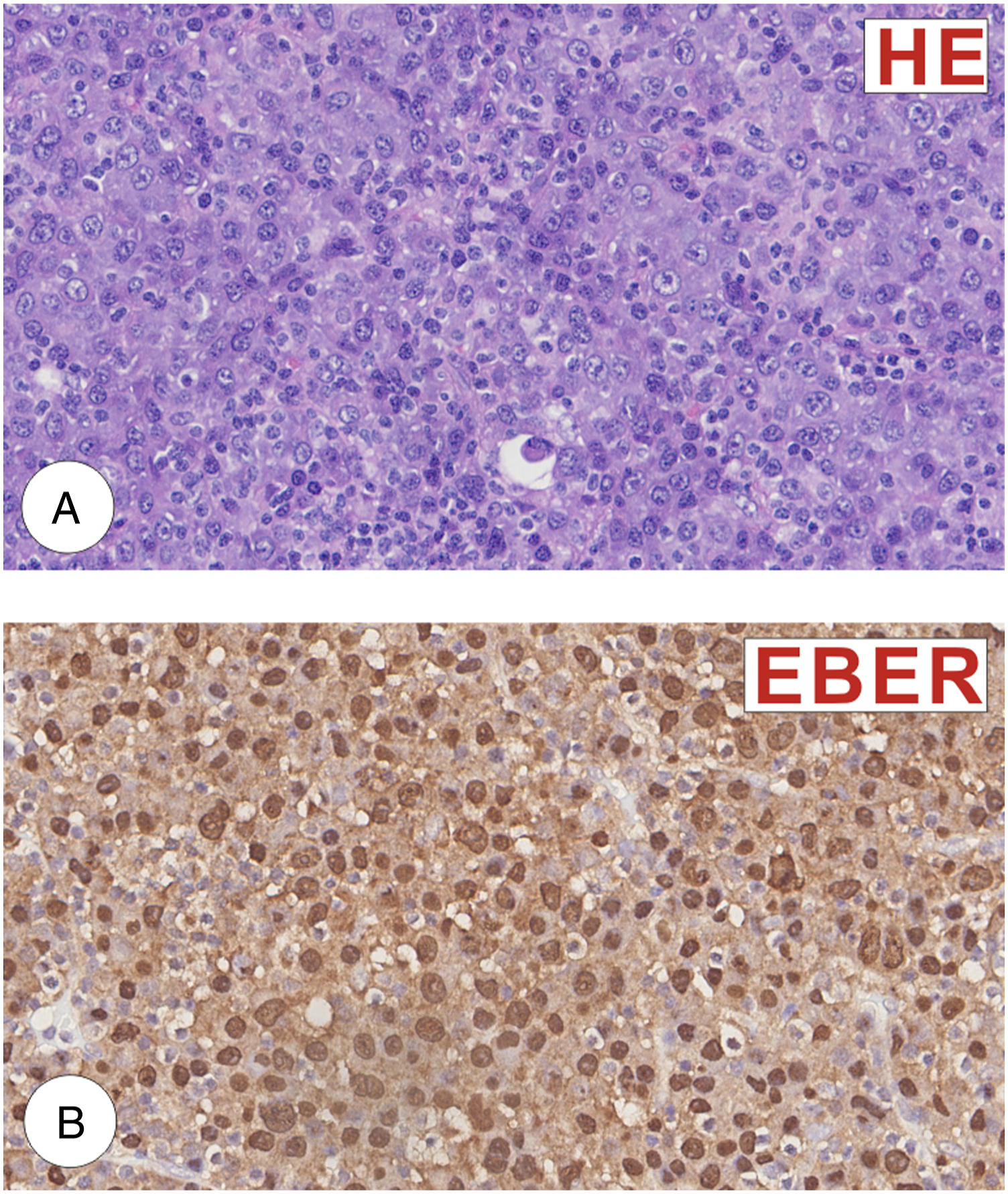
A 40-year-old man complained of frequent nosebleeds for ten days. He did not report any specific sinonasal symptoms such as nasal obstruction or discharge and loss of smell. Nasal bleeding could be stopped with local pressure. He underwent a computed tomography (CT) examination at a local hospital which indicated that the right sinonasal tract, extending to the right lamina papyracea of the ethmoid bone, causing bone erosion, was filled with a soft tissue mass. He was then directed to the otolaryngology department at Wuhan Union Hospital and underwent nasal endoscopic and magnetic resonance imaging (MRI) examinations. Nasal endoscopy revealed a dark brown lesion in the right middle meatus, while MRI showed a diffuse mass infiltrating the right sinonasal tract extending to the right lamina papyracea of the ethmoid bone. On T1-weighted imaging, the lesion was hypointense, while on T2, it was heterogeneous to hypointense, becoming enhanced upon contrast administration. The patient underwent rigid functional endoscopic sinus surgery with a biopsy. During the operation, the lesion was seen extending throughout the lamina papyracea of the ethmoid bone with bone erosion anteroinferior to the middle turbinate, filling the right nasal cavity (Figure 1). Histopathological analysis revealed abnormal, hyperchromatic, plasmacytic cell proliferation with numerous mitoses. Immunohistochemical staining revealed that the lesions were focal or partially positive for CD38, CD138, MUM1, CD56, Kappa, Bcl2, P53 (20%+), c-Myc (50%+), and Ki-67 (30%) (Figure 2) and negative for CD3, PAX5, Bcl6, CD20, CD19, CD21, CD22, PCK, P40, Lambda, and CyclinD1(Figure 3). In situ hybridization revealed that the lesions were EBV-encoded RNA (EBER) positive. These biopsy results support the diagnosis of EBV-positive anaplastic plasmacytoma (Figure 4). After surgery, whole-body PET/CT revealed localized disease spreading from the right sinonasal tract to the ethmoid bone’s right lamina papyracea. He was directed to the oncology department, and was recommended to undergo a four-week radiotherapy course. CT scan and MRI of the paranasal sinuses. A–D, Coronal scan, E–H, axial scan. A and E showed a soft tissue mass filling the right sinonasal tract, extending to the right lamina papyracea of the ethmoid bone and causing bone erosion. B–D and F–J showed a diffuse mass infiltrating the right sinonasal tract extending to the right lamina papyracea of the ethmoid bone; on the T1-weighted image, the lesion was hypointense; it was heterogenous to hypointense on T2-weighted and enhanced upon contrast administration. The tumor cells are positive for CD38 (A), CD138 (B), MUM1 (C), Kappa (D), CD56 (E), Bcl2 (F), P53 (20%+) (G), c-Myc (50%+) (H), and Ki-67 (30%) (I). The tumor cells are negative for Lambda (A), CD20 (B), CD19 (C), CD22 (D), PAX5 (E), CD3 (F), Bcl6 (G), CD10 (H), CD 5(I), CD30 (J), CD21 (K), CyclinD1 (L), PCK (M), and P40 (N). (A) Histopathologic image (H&E: 400x) of the left nasal tissue mass shows abnormal, hyperchromatic, plasmacytic cellular proliferation with abundant mitosis. (B) The tumor cells are EBER-positive on in situ hybridization.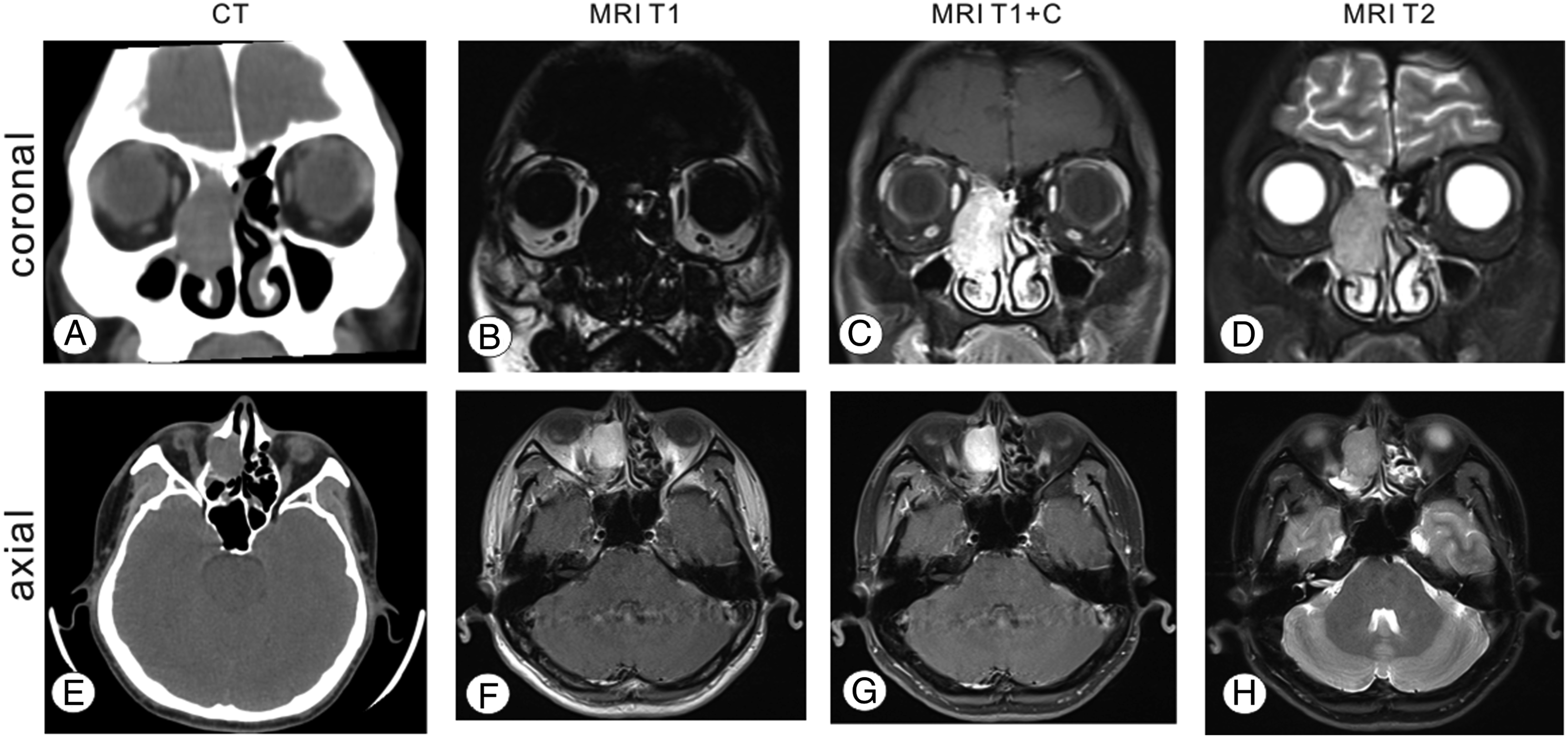
Discussion
SP is very uncommon. Its cumulative incidence is only 0.15/100.000, while EMP is rare, accounting for only 3–4% of all SP. 4 Head and neck EMP mainly affects older adults with a mean age of 55 years old and has a slight male predominance. 5 In a retrospective review of 22 patients, the five-year OS was 84.2%, and the EMP’s five-year multiple myeloma (MM)–free survival rate was 75.9%. 6 There were no specific clinical symptom imaging results for EMP. Frequent epistaxis and nasal obstruction are the two main complaints associated with EMP. This lesion appears as a well-defined or infiltrative soft tissue tumor with variable enhancement on CT. Most EMP on T1 are iso/hypointense, whereas those on T2 are iso/hyperintense with considerable contrast enhancement. 7 The assessment of cytoplasmic immunoglobulins is of crucial importance in diagnosis. Plasmablastic, extracavitary primary effusion, and ALK-positive large B-cell lymphomas are all differential diagnoses for anaplastic plasmacytomas. They have a similar high-grade morphology but exhibit varying plasmacytoid morphologic characteristics, and all express the plasma cell markers, MUM1, CD38, and CD138, while lacking the pan-B-cell markers, PAX5, CD19, and CD20, and show variable CD30, CD45, CD56, CD79a, and EMA expression. 8 Plasmacytomas are defined by their monomorphic infiltrate against reticular stroma, spherical nuclei, chromatin “cartwheel” arrangement, and abundant cytoplasm. 9 EMP with anaplastic features is an uncommon type of plasma cell malignancy diagnosed by histopathological analysis. Anaplastic plasmacytomas are often of high histological grade and morphology, comprising large atypical cells with eccentric nuclei, abundant cytoplasm, and excessive mitosis. 10
Most anaplastic plasmacytoma cases (60–75%) were EBER-positive on FISH. The presentation of ESP with anaplastic features and EBV-positivity, as in our patient, is poorly understood due to its rarity. 11 The role of EBV in the pathogenesis of plasmacytoma remains unclear; many researchers believe that plasmablastic lymphoma (PBL) may occur after EBV infection as a high-grade transformation from preexisting plasmacytoma. 12 A rare immunocompetent of a 74-year-old case was reported by Ambrosio et al., first diagnosed with preexisting EBV-positive, MYC-negative plasmacytoma that transformed to PBL. Immunohistochemistry revealed many relapses with increasing proportions of EBER and MYC-positive cells. MYC expression was also found to play a role in the transformation to PBL, and reduced MYC expression is associated with a less aggressive clinical course and decreased transition risk to PBL. 13 In our case, immunohistochemistry reported a 30% Ki-67 proliferative index and 50%+ MYC expression, suggesting a high transformation risk to PBL. Our patient thus required an aggressive treatment protocol and close follow-up.
In conclusion, we report an EBV-positive anaplastic plasmacytoma in the sinonasal tract, which is distinct from PBL, despite the close resemblance. Comprehensive medical and immunohistochemical examinations should support accurate diagnosis, which may have prognostic and therapeutic consequences.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (81771005) and the Research Grant of Union Hospital, Tongji Medical College, HUST (F016.02004.21003.126).
