Abstract
Ganglioneuroblastoma is a rare peripheral neuroblastic tumor located anywhere in the sympathetic nervous system but rarely in the retropharyngeal space. Diagnosis can often be difficult based on imaging alone. We describe one case of a child presenting with snoring. The lesion was located in the rare retropharyngeal space, and its histology finally revealed ganglioneuroblastoma. Therefore, clinicians should be aware of pediatric patients with respiratory symptoms indicating cervical ganglioneuroblastoma. To make a definite diagnosis as soon as possible, a core needle biopsy or even immunohistochemistry may need to be performed before surgery.
Introduction
Peripheral neuroblastic tumors (pNTs) are the most common extracranial solid tumors in childhood 1 and are most commonly seen in children aged 0–4 years old. 2 According to the International Neuroblastoma Pathology Classification (INPC), pNTs include neuroblastoma (NB), ganglioneuroblastoma intermixed (GNBi), ganglioneuroblastoma nodular (GNBn) and ganglioneuroma (GN), and GNBn can be divided into a favorable subset (FS) and an unfavorable subset (US).3,4 Ganglioneuroblastoma (GNB) is a rare pNT that can be located in any part of the sympathetic nervous system: adrenal glands (47%), abdomen/retroperitoneum (24%), thorax (15%), pelvis (3%), neck (3%), and other primary tumor sites(8%). 5 In the literature of the past few decades, there have been 8 reports describing 15 cases of primary cervical GNB, 5 of which are located in the retropharyngeal space.6-13 In this report, we present the case of an 8-year-old girl with a cervical GNB located in the retropharyngeal space.
Case report
An 8-year-old girl was admitted to the hospital with snoring and paroxysmal nocturnal dyspnea for more than one year. She had other accompanying symptoms, such as the sensation of a pharyngeal foreign bod and dysphagia without Horner’s syndrome. Her family and personal pathological history were insignificant. Physical examination showed that the mucosa of the posterior wall of the oropharynx bulged with a smooth surface and clear boundary, up to the soft palate plane and down to the epiglottic apex plane. No positive lymph nodes were found by cervical palpation. Contrast-enhanced magnetic resonance imaging (MRI) scans showed a giant mass located in the posterior wall of the pharynx, approximately 3.7 cm × 1.9 cm × 4.6 cm in size, with smooth margins (Figure 1A). The lesion could also be seen on contrast-enhanced computed tomography (CT) scans, which showed that the cervical lymph nodes were not invaded and the surrounding bones were not damaged (Figure 1B). Tumor cells could be found by fine-needle aspiration (FNA), which tended to be granulosa cell myoblastoma. According to the growth morphology, manifestations, FNA results, and imaging characteristics, it was preliminarily considered retropharyngeal granular cell myoblastoma. Preoperative magnetic resonance imaging (MRI) scans showed an uneven mass in the retropharyngeal space, which narrowed the oropharyngeal cavity(A). Preoperative computed tomographic (CT) scans showed a large mass with uniform density in the posterior wall of the oropharynx. The adjacent blood vessels were not invaded, and the surrounding bone structure was normal (B).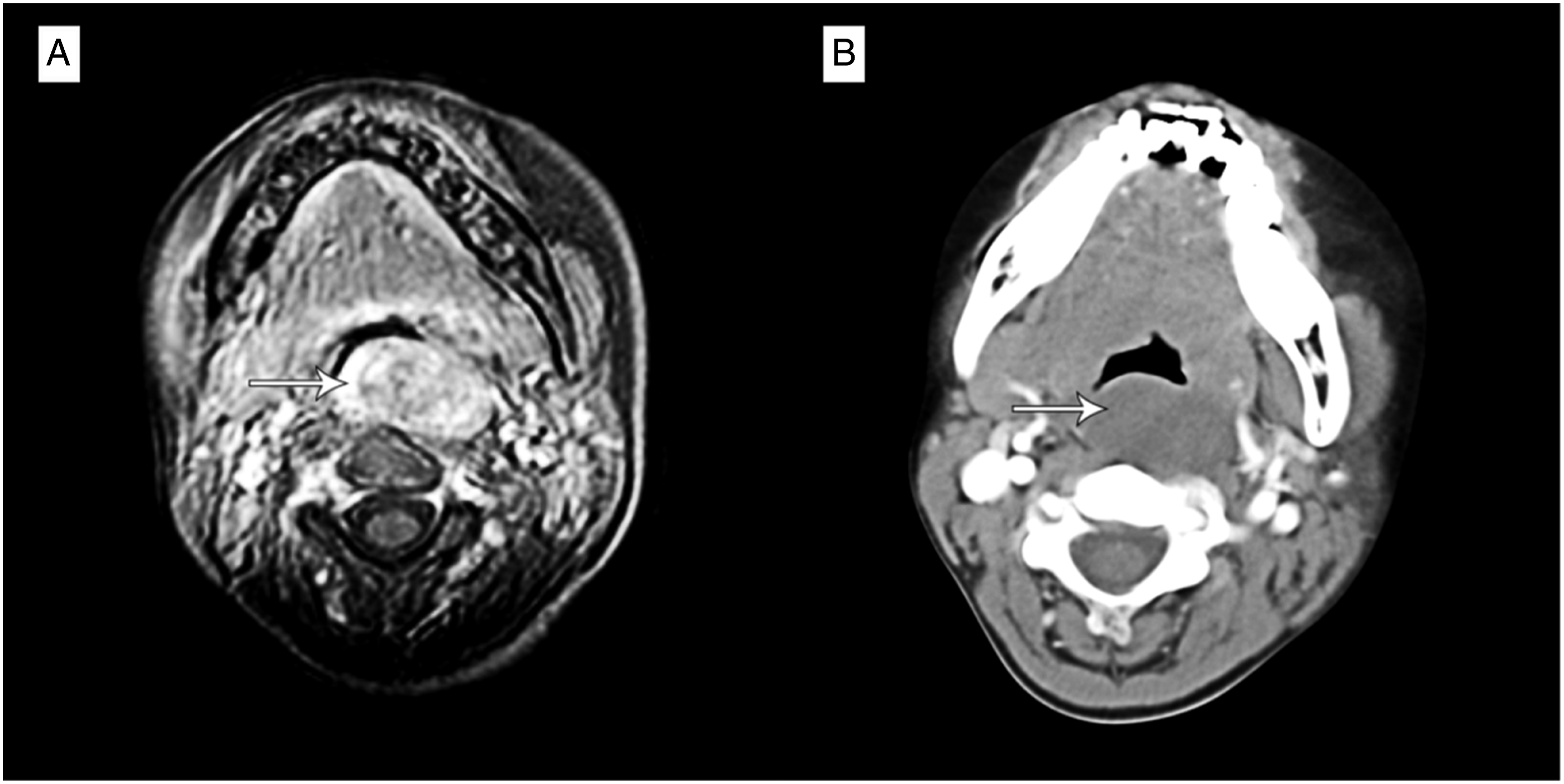
To minimize damage, the tumor was removed by transoral surgery. Two encapsulated, solid masses were found in the retropharyngeal space. Among them, the larger one was approximately 4.0 cm in diameter, which was oval-shaped, and the smaller one was approximately 2.0 cm in diameter, which was lobulated. The cut surface was grayish-white and was easy to separate from the surrounding tissue. Postoperative histopathology showed that the mass was rich in Schwann cells, and the cancer nest in the mass was formed by a mixture of small neuroblasts and large mature ganglion cells (Figure 2A). Further immunohistochemical results showed that the expression of synaptophysin (Syn), neurofilament (NF), and chromogranin A (CgA) was positive (Figure 2B–D). After the operation, the patient no longer had snoring or paroxysmal nocturnal dyspnea, and there were no postoperative complications. Five courses of chemotherapy were given in the oncology department after the operation. In the following 4 years, the neuron-specific enolase test and neck MRI examination were performed every year. After 4 years of follow-up, the patient was in good condition and no recurrence was found. (A) Histopathological findings (H&E staining): The tumor was rich in Schwann cells (blue arrow), and the cancer nest was formed by a mixture of small neuroblasts (white arrow) and large mature ganglion cells (black arrow) (original magnification × 200). Positive staining for Syn (B), neurofilament (C), and chromogranin A (D) (original magnification × 200).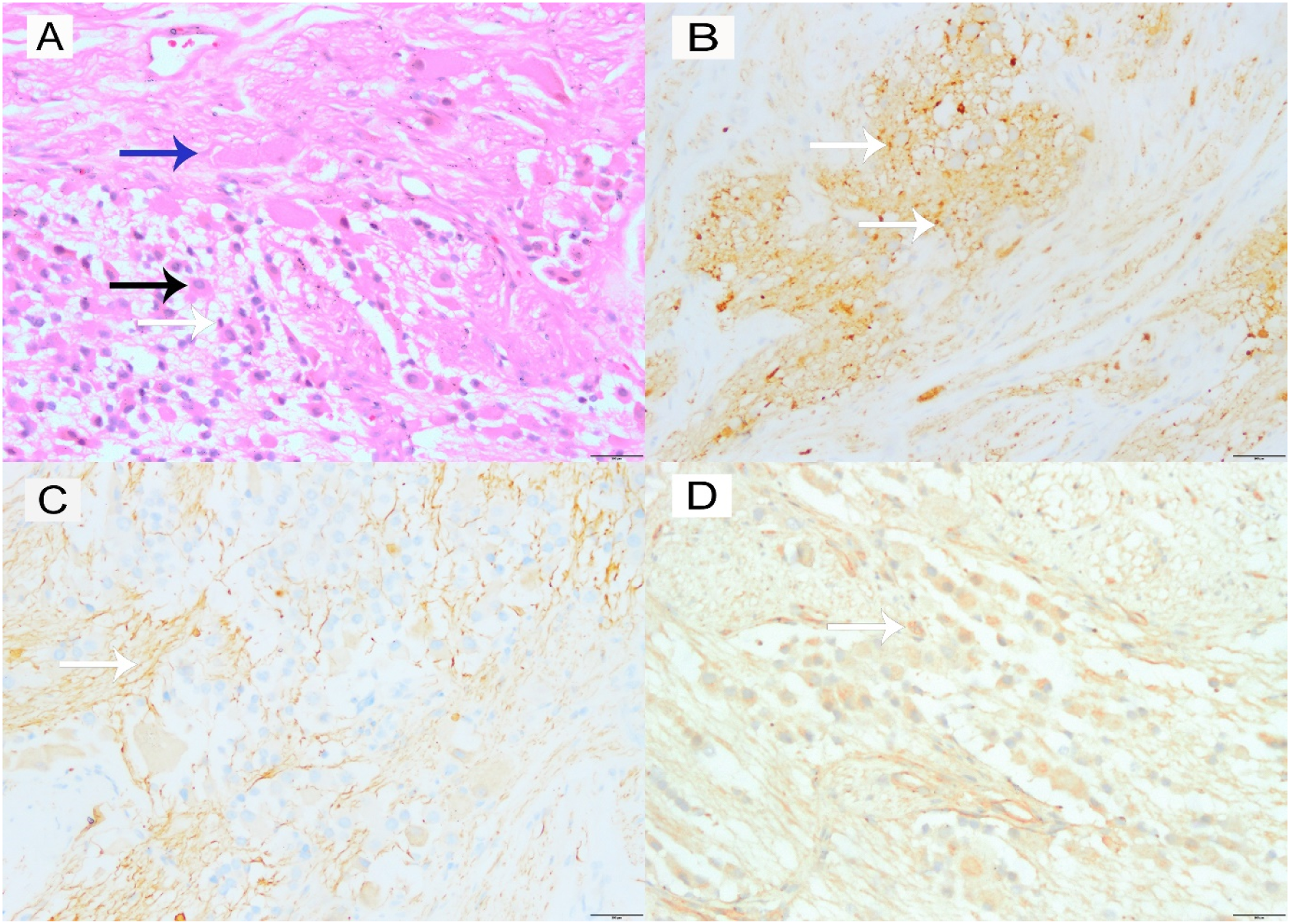
Disscussion
Cervical masses are common in the pediatric population and most of them are benign such as inflammatory or congenital masses, more than 10% of which are malignant. 10 Approximately 5% of pNTs occur in the cervical region, 14 GNB is a malignant tumor, although it is more benign than NB. 15 GNB is commonly seen in early childhood, especially in children aged 0–4, and occurs slightly more frequently in boys than girls (ratio 1.2:1). 2
An accurate diagnosis of cervical GNB involves not only the observation of preoperative clinical symptoms, but also imaging, histopathologic examination, and immunohistochemical staining. 11 The clinical symptoms of cervical GNB include a neck mass, cranial nerve neuropathies such as Horner’s syndrome, dysphagia, or respiratory symptoms such as snoring and even dyspnea during sleep.11,16,17 Our case showed respiratory symptoms and dysphagia, but no Horner’s syndrome. However, due to the rarity of this disease, she was misdiagnosed with granulosa myoblastoma in the retropharyngeal space before the operation. The final diagnosis still depended on histological examination. 18
Imaging examinations play an important role in diagnosis, CT and MRI can generally be used to visualize masses, and Moukheiber et al. 8 reported that calcifications were found in the tumor in 50% of cases of GNB. However, according to the study of Lonergan et al, 19 compared with CT, MRI is superior to CT in determining the tumor origin and regional invasion of GNB because of its excellent tissue identification and multiplanar imaging. In MRI, GNB is typically heterogenous, and variably enhancing, and it appears as a low signal intensity in T1-weighted scanning and high signal intensity in T2-weighted scanning. In our case, CT scans showed a large mass with clear boundary irregular low density and no noticeable enhancement or calcification. At the same time, MRI scans a showed smooth boundary and the lesion had hypo‐intense signal on T1-weighted images with a heterogenous hyperintense signal on T2-weighted images. Neither CT nor MRI scans showed tumor invasion of the surrounding structures.
Despite the above disease characteristics, accurate preoperative diagnosis of GNB is still challenging due to the lack of standard clinical presentation and imaging characteristics. Therefore, it is not surprising that misdiagnosis often occurs. 11 Histopathological examination and immunohistochemical staining are required to make a definite diagnosis. According to previous research, core needle biopsy (CNB) offers the advantage of preserving tissue architecture with increased tissue yield. 20 It has a higher sensitivity and specificity than FNA in cervical masses. 21 If we perform CNB on cervical masses not diagnosed by FNA before surgery and perform immunohistochemistry when necessary, fewer GNBs may be misdiagnosed. For histopathological examination, GNB consists of both mature and intermediate-form ganglion cells and neuroblasts. For immunohistochemical staining, NF, Syn, CgA, S-100, cluster of differentiation 30 (CD30), and CD15 were generally positive in GNB. 22 In our case, histopathology showed neuroblasts, mature ganglion cells, and many Schwann cells. Meanwhile, immunohistology showed Syn (+), NF (+), and CgA (+), consistent with GNB. The tumor was classified as Stage 1 according to the International Neuroblastoma Staging System. 23
The treatment for GNB includes surgery, chemotherapy, radiotherapy, and biological therapy. However, treatment choice depends on tumor stage, patient age, and biological prognostic factors. 24 Luo et al. 25 reported that low-risk patients were often managed with surgical resection or observation alone; intermediate patients received chemotherapy and the number of cycles depends on their response and surgical resection of the primary tumor; high-risk patients remained controversial, but it was concluded that ≥90% resection of the primary tumor was both feasible and safe for most high-risk patients. Several surgical approaches include the transcervical, cervical-parotid, mandibular split, transcervical-transmastoid, infratemporal fossa, transoral, and minimally invasive approaches. 26 The surgical strategy is determined by location, size, and pathology. 27 In our case, the patient’s tumor was located in the retropharyngeal space of the oropharynx and laryngopharynx, and the patient had no limitation of mouth opening. To minimize the damage and take care of the beauty, we chose the transoral approach to remove the tumor altogether, and there were no postoperative complications. Considering the borderline malignancy of the tumor, further chemotherapy was performed in the oncology department.
The prognosis of GNB primarily depends on patients’ age and disease stage. In general, the prognosis of patients under one year of age is significantly better than that of older children with the same tumor stage. 28 In univariate analysis, large tumors, high neuron-specific enolase (NSE) and lactate dehydrogenase (LDH) levels, positive lymph nodes, macroscopic residue, and N-Myc gene amplification have adverse effects on the results. 29 Disease recurrence mainly occurs in the first 2 years after surgery. 30 Approximately 1% of neuroblastoma metastasizes through blood vessels or the lymphatic system, and the common metastatic sites are the liver, lung, bone, and bone marrow. 31 Therefore, patients should be examined every 3 months in the first and second years and then every 6 months. 11 In our case, the prognosis was favorable based on complete tumor resection, and further chemotherapy was performed. There was no recurrence or metastasis for 4 years after surgery. However, she needed a longer follow-up to ensure that there is no recurrence or metastasis. The frequency of follow-up could be reduced.
Conclusion
Here, we have presented a case of an 8-year-old female with cervical GNB, one of the few GNBs located in the retropharyngeal space. Our report suggests that clinicians should be alert to pediatric patients with respiratory symptoms, which may indicate cervical GNB. It is important to make the preoperative diagnosis as confirmed as possible for choosing treatment for GNB. When FNA cannot make a definite diagnosis, we might need to perform a core needle biopsy or even immunohistochemistry to confirm the diagnosis further.
Footnotes
Author’s Note
The patient’s permission was obtained.
Declaration of Conflict Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
