Abstract
Objectives
This study aimed to investigate the audiological outcomes and subjective benefits of transcutaneous bone conduction implants (BCIs) in patients with single-sided deafness (SSD).
Methods
This retrospective study was conducted on 11 patients with SSD implantations between 2015 and 2018 at a tertiary center. Pure-tone audiometry, speech reception threshold (SRT), and speech-in-noise (SPIN) tests were performed. Preoperative and postoperative performances were compared. Subjective satisfaction level was assessed using validated questionnaires. A PubMed search was conducted to identify the relevant studies published to date.
Results
All patients demonstrated significant audiological improvements compared with their preoperative condition. The mean SRT improved significantly (p = 0.001) from 109 dB to 23 dB after implantation. The mean SPIN score improved significantly after implantation. The questionnaires showed an overall positive benefit of transcutaneous bone conduction devices (BCDs). A literature search revealed 21 articles, of which 14 reported the use of BCIs in patients with SSD. Our results agree with the published evidence showing the overall benefit of BCI in patients with SSD.
Conclusions
Transcutaneous BCDs could be considered as an alternative treatment option for patients with SSD, it could show good audiological outcomes and high satisfaction levels. Further studies should be conducted on patients with SSD to determine the most appropriate hearing solutions.
Introduction
According to the World Health Organization, hearing defects are the most frequent sensory deficits in humans, affecting more than 250 million individuals worldwide, and on average, 7 in 100 individuals in Saudi Arabia. 1 Single-sided deafness (SSD) or profound unilateral sensorineural hearing loss (SNHL) refers to clinically tested unaidable hearing, defined as severe-to-profound hearing thresholds with poor ability to recognize words.2,3 The etiology of SSD includes pathologies such as cochleovestibular abnormalities, Meniere’s disease, head trauma, enlarged vestibular aqueduct, acoustic neuroma, chronic otitis media, vestibular schwannoma, vascular ischemia, autoimmune disorders, and infections. Often, the onset of hearing loss can occur suddenly, leaving the patient with debilitation. 4
SSD patients face several hearing challenges, including the inability to hear with sufficient loudness, difficulty in precisely localizing the sound source, poor speech intelligibility, and difficulty in hearing well in noisy environments. 2 Functional hearing difficulties associated with SSD affect social and psychological well-being, such as limitations of activities and participation restrictions, including withdrawal from and within situations. 5
SSD patients are treated using different treatment options, from nonsurgical acoustic hearing aids to cochlear implantation under general anesthesia. 6 Cochlear implantation is the gold standard for the treatment of profound SNHL; it is an effective treatment for habilitation and provision of binaural hearing. However, patients with certain conditions such as congenital cochlear nerve hypo/aplasia, acoustic neuroma, and long-duration hearing loss may show lower performance. Patients with SNHL may have different degrees of inner-ear malformations (IEM). In general, patients with unilateral hearing loss have higher rates of IEM compared to those with asymmetric hearing loss. 7 Lin et al. reported a higher percentage of IEM among patients with SSD, which could reach up to 30.9%. 7 Despite this incidence, patients with IEM can still undergo CI and the electrode array could be inserted safely using specific surgical techniques. 8 Furthermore, studies revealed significant benefits and improvements in patients with IEM after CI. 9 Nevertheless, the severity of IEM and cochlear nerve deficiency has an impact on CI outcomes. For instance, patients with a narrowed internal auditory canal showed significantly inferior outcomes with CI.10,11
Bone conduction devices (BCDs) have also been reported as viable treatment options for SSD patients.12,13 Percutaneous bone-anchored hearing aid devices use a surgically implanted abutment to transmit sound by direct conduction through the bone to the inner ear, bypassing the external auditory canal and middle ear, and have been in clinical use for years to successfully treat mixed and conductive hearing loss.However, they have certain limitations owing to the penetration of the abutment into the skin. Some reported complications include failure of osseointegration, chronic infection, trauma, irritation of the skin surrounding the implant, skin flap necrosis, overgrowth of the skin over the device, wound dehiscence, bleeding or hematoma formation, persistent pain, lifelong daily care, and accidental or spontaneous loss of bone-implant. 14
With technological advancements, new transcutaneous BCDs, BonebridgeTM (BB; MED-EL GmbH, Austria), SophonoTM (Sophono Inc., USA), BAHA AttractTM, and the Osia system (Cochlear bone-anchored solutions, Sweden) are now commonly available. Sophono and BAHA Attracts are classified as under-skin-driven BCDs because they transmit vibrations indirectly via the skin, which inevitably leads to less audiological gain. However, Bonebridge is classified as an active transcutaneous direct-drive BCD, which transmits vibrations directly from the transducer implanted under the intact skin to the skull, limiting any loss of audiological gain. 15
We aimed to report the audiological results and subjective assessment of the benefits provided by transcutaneous BCD (Bonebridge) in SSD patients at our center. Furthermore, we conducted a literature search on the use of bone conduction implants (BCIs) in SSD patients until the end of 2020 to compare our results with published evidence to determine whether our results are in line with those of other medical centers.
Patients and Methods
Device description
The investigated device consists of 2 parts (Figure 1): an externally worn audio processor (SAMBA audio processor) and a surgically implanted Bonebridge implant (BCI601), which will be referred to as transcutaneous BCI. A transcutaneous BCI has an internal receiver coil, magnet to hold the audio processor in place over the implant, demodulator to convert the processed digital signal received from the audio processor, and floating mass transducer (FMT) for bone conduction to vibrate the skull. The transition between the demodulator and FMT can be bent in any direction (±90°) as required to fit the patient’s anatomy. In cases where the bone is not sufficiently thick to submerge the FMT, BCI lifts are used to elevate the FMT to avoid exposing the dura underneath. Parts of the investigated transcutaneous bone conduction implant.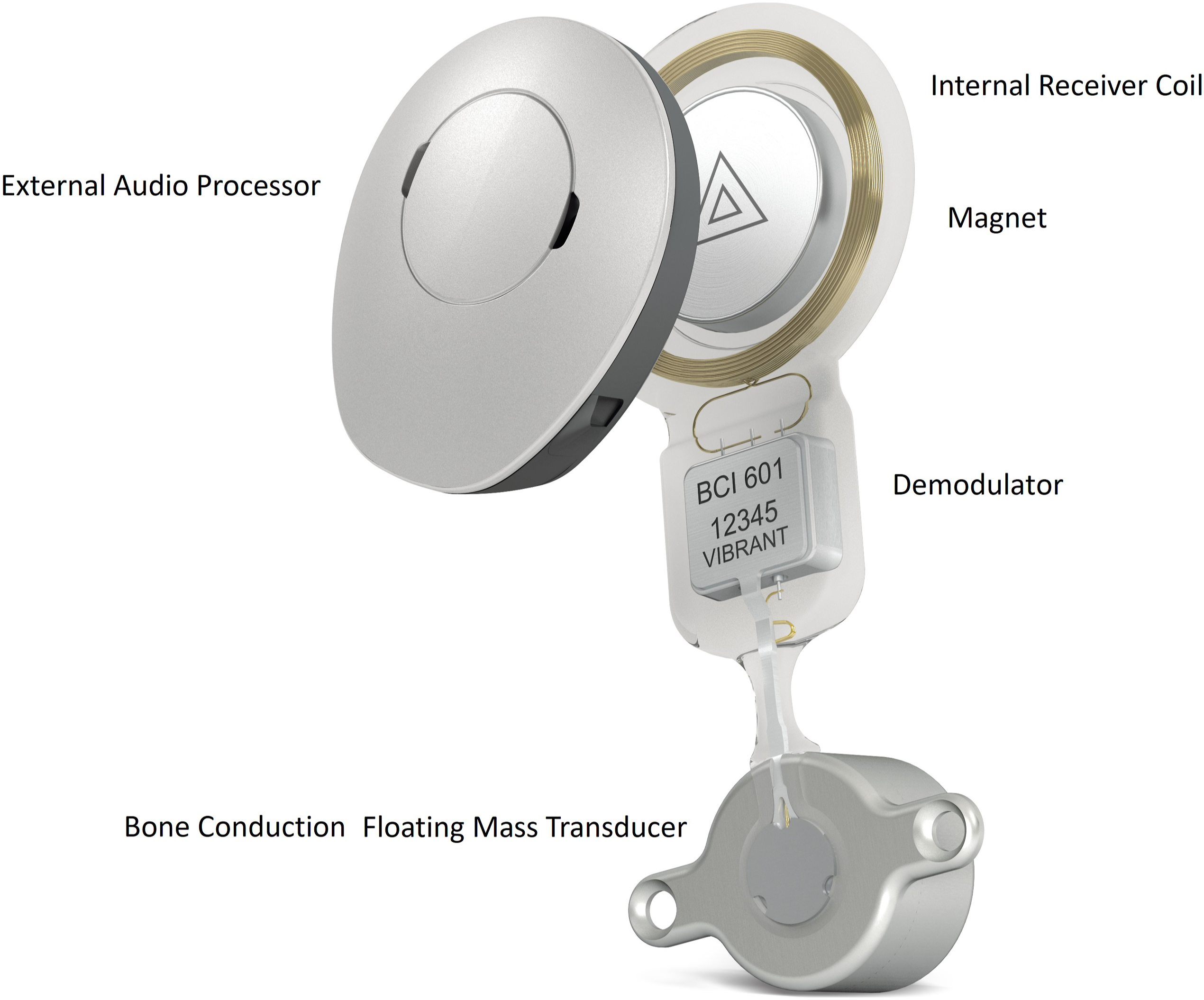
Patients
After obtaining ethical approval from the institutional review board (IRB# 20-36), we retrospectively collected the data for this study. The inclusion criteria were all SSD patients, with any hearing etiology of hearing loss, who underwent transcutaneous BCI surgery at our center between 2015 and 2018, and had a minimum of 2 years of follow-up after implantation. We did not exclude any SSD patients with a transcutaneous BCI. Demographic data, including age at implantation, sex, etiology, and follow-up duration, were collected for the included patients. Furthermore, relevant intraoperative and postoperative data, including complications, have also been reported.
Audiological and Speech Tests
During routine clinical practice, unaided pure-tone air and bone conduction thresholds were measured preoperatively and postoperatively in an anechoic chamber with calibrated audiometers. Preoperative and postoperative pure-tone audiometry (PTA) followed the procedure recommended by the British Society of Audiology. For the preoperative unaided measurements, pure tones with frequencies of 0.25–8 kHz were presented through inserted earphones and a bone vibrator.
The pure-tone average (PTA4) was calculated by averaging 4 main thresholds (0.5, 1, 2, and 4 kHz). The same frequencies were presented and tested postoperatively in a sound field using a speaker positioned at 90° azimuth. The speech reception threshold (SRT) and speech in noise (SPIN) in the narrow band were tested under 2 different settings (Figure 2). In this setup, the SIN scores indicate a hearing level of at least 50% correct speech recognition. Aided and unaided speech in noise tests were measured using a monosyllabic Arabic word list developed by Ashoor et al.
15
Setup for measuring the speech in noise (SPIN).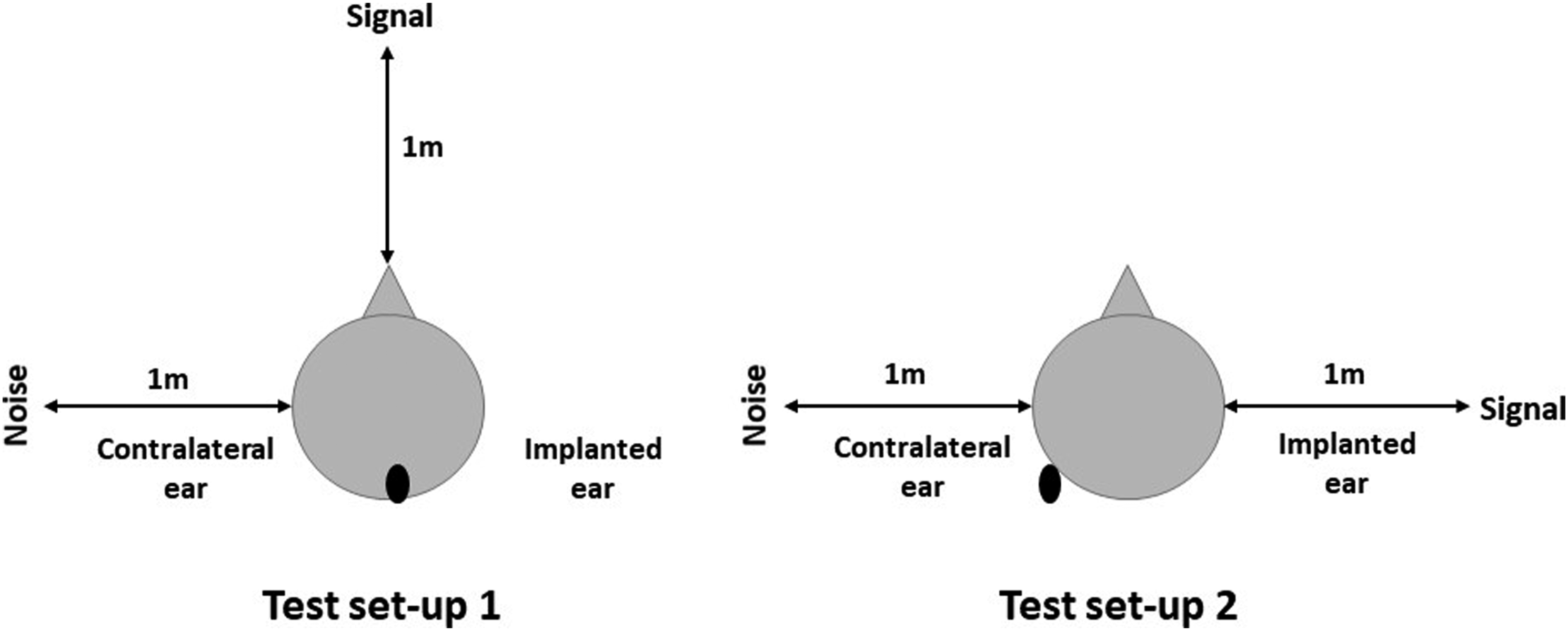
Subjective Assessment
The subjective perceptions of the users were determined using translated and validated versions of the Abbreviated Profile of Hearing Aid Benefit (APHAB), Hearing Handicap Inventory (HHI), and Bern Benefit of Quality of Life in Single-Sided Deafness (BBSS). The language of the questionnaire was each patient’s native language, either Arabic or English.
The APHAB is a collection of questions that evaluate the effort needed for ease of communication, speech understanding in background noise, speech understanding in moderately reverberating rooms, and negative reactions to environmental sounds. 16 This questionnaire was scored by assigning a score to each answer and calculating the global score of each subscale as the mean of the scores for all items. For each subscale, the benefit was calculated by subtracting the preoperative and postoperative global scores. In general, a lower score indicates an improvement. The HHI is a collection of questions that evaluates the social and emotional effects of hearing loss.17,18 The total score of this tool ranges from 0 to 100, where 0 indicates that no handicap is perceived, and 100 indicates that a significant handicap is perceived. The BBSS is a collection of questions designed specifically for SSD patients. 19 Patients in the preoperative condition were asked to grade their hearing on a scale of 0–5 in 10 different situations, with 0 being the worst and 5 being the best.
Furthermore
Statistical Analysis
All statistical analyses were performed using GraphPad Prism TM version 8.4.0 (GraphPad Software, La Jolla, CA, USA). The mean, standard deviation, and range were used to describe the participants’ characteristics. To compare preoperative and postoperative data, the normality of the data was checked. A parametric paired t-test was performed to test the significance between groups for data with a normal distribution, and the Wilcoxon non-parametric test was used to test the others. Statistical significance was set at P < 0.05.
Results
Patients
Patient Demographics.
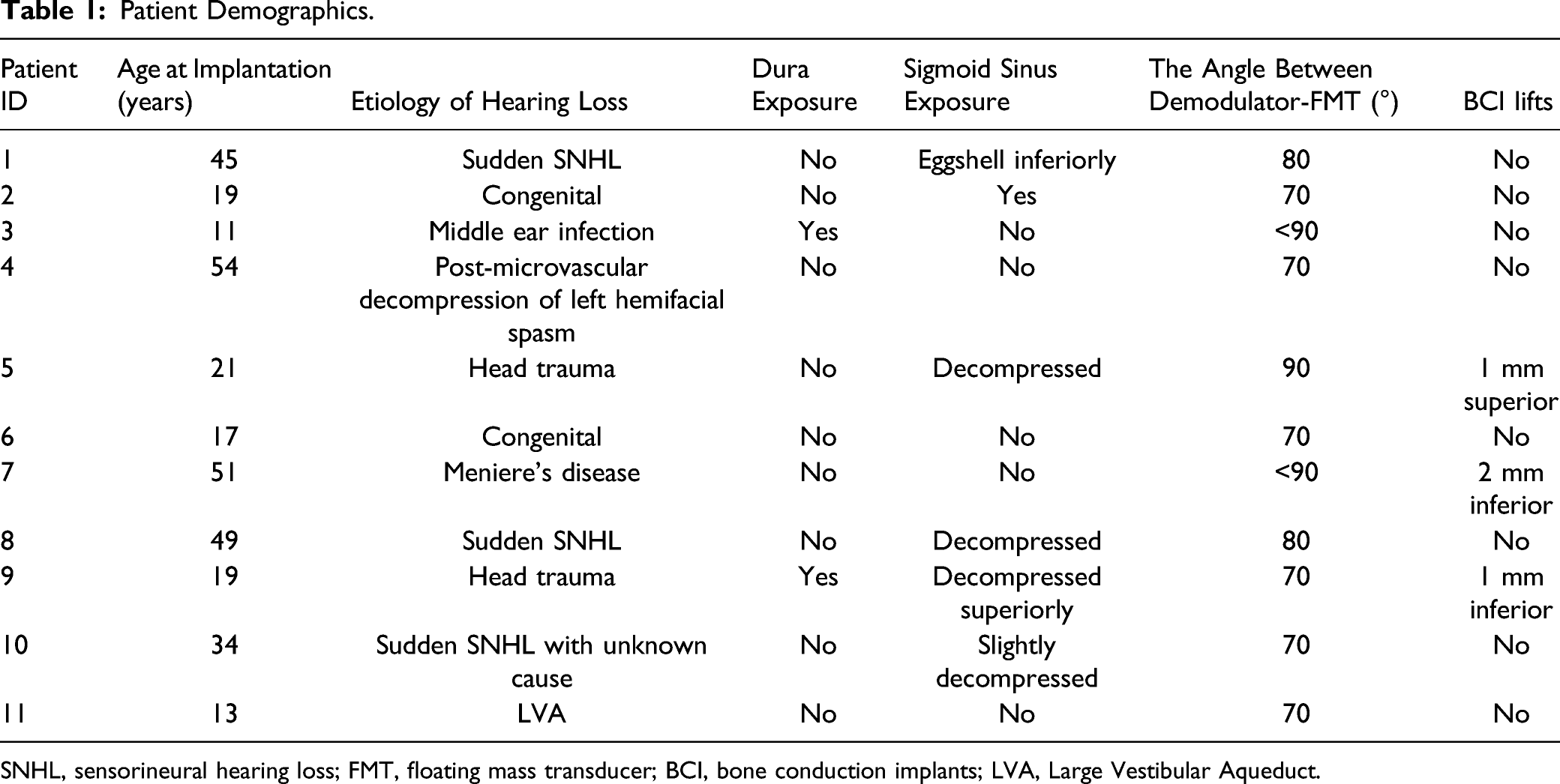
SNHL, sensorineural hearing loss; FMT, floating mass transducer; BCI, bone conduction implants; LVA, Large Vestibular Aqueduct.
Pure-Tone Audiometry
The hearing thresholds measured at the four frequencies in the deaf ear, the contralateral normal hearing ear, and deaf ear post-implantation with transcutaneous BCI are shown in Figure 3A. Patients’ hearing thresholds improved significantly across all four frequencies when tested in the implanted ear between pre- (111 dB HL) and post-implantation (23 dB HL) (p = 0.001). Compared to the contralateral normal ear (12.5 dB HL), the hearing threshold of post-implantation hearing with transcutaneous BCI (23 dB HL) was significantly higher (p = 0.001) (Figure 3A). Pure-tone average (A) and speech reception threshold (B) comparison between deaf ear, a contralateral normal hearing ear, and deaf ear with transcutaneous bone conduction device (BCD) post-implantation.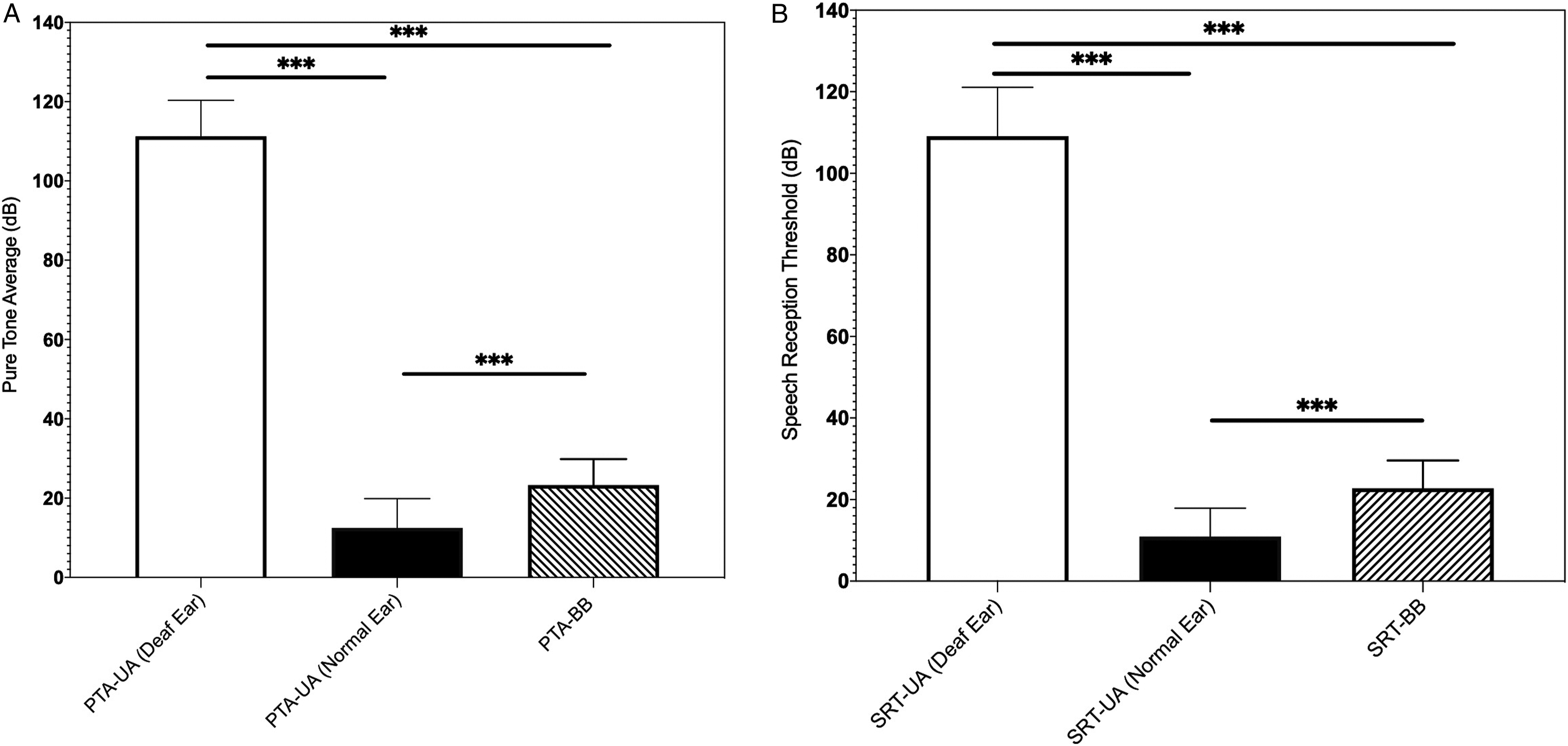
Speech Audiometry
SRT improved significantly (p = 0.001) in the implanted ear postoperatively (23 dB HL) compared with the preoperative condition (109 dB HL). However, SRT of the contralateral normal hearing ear (11 dB HL) was significantly better (p = 0.0009) than the postoperative SRT with transcutaneous BCI (23 dB HL) (Figure 3B).
Speech in Noise
Speech in Noise (SPIN) Test Pre- Versus Post-Operatively in two Different Test Setups.

SNR, signal-to-noise ratio.
Questionnaires
Figure 4A shows a comparison of the APHAB subscale scores before and after surgery. Users perceived that the Ease of Communication using transcutaneous BCI improved significantly (p = 0.0009) compared to the preoperative condition (12.62 ± 9.80 vs. 53.95 ± 27.46), that is, less effort was required. The patients also perceived that their hearing in background noise improved after implantation (44.48 ± 12.58 vs. 38.58 ± 13.47). However, this difference was not statistically significant (P = 0.505). The reverberation (RV) of transcutaneous BCI users improved using the BB compared to the preoperative condition (41.58 ± 12.71 vs. 49.80 ± 17.09%), although not significantly (p = 0.154). Furthermore, there was a trend toward non-significant improvement (p = 0.719) in transcutaneous BCI users’ perception of aversiveness by transcutaneous BCI users' post-implantation compared to pre-implantation (26.76 ± 23.43 vs. 30.42 ± 25.80). Subjective rating of listening situations is determined using 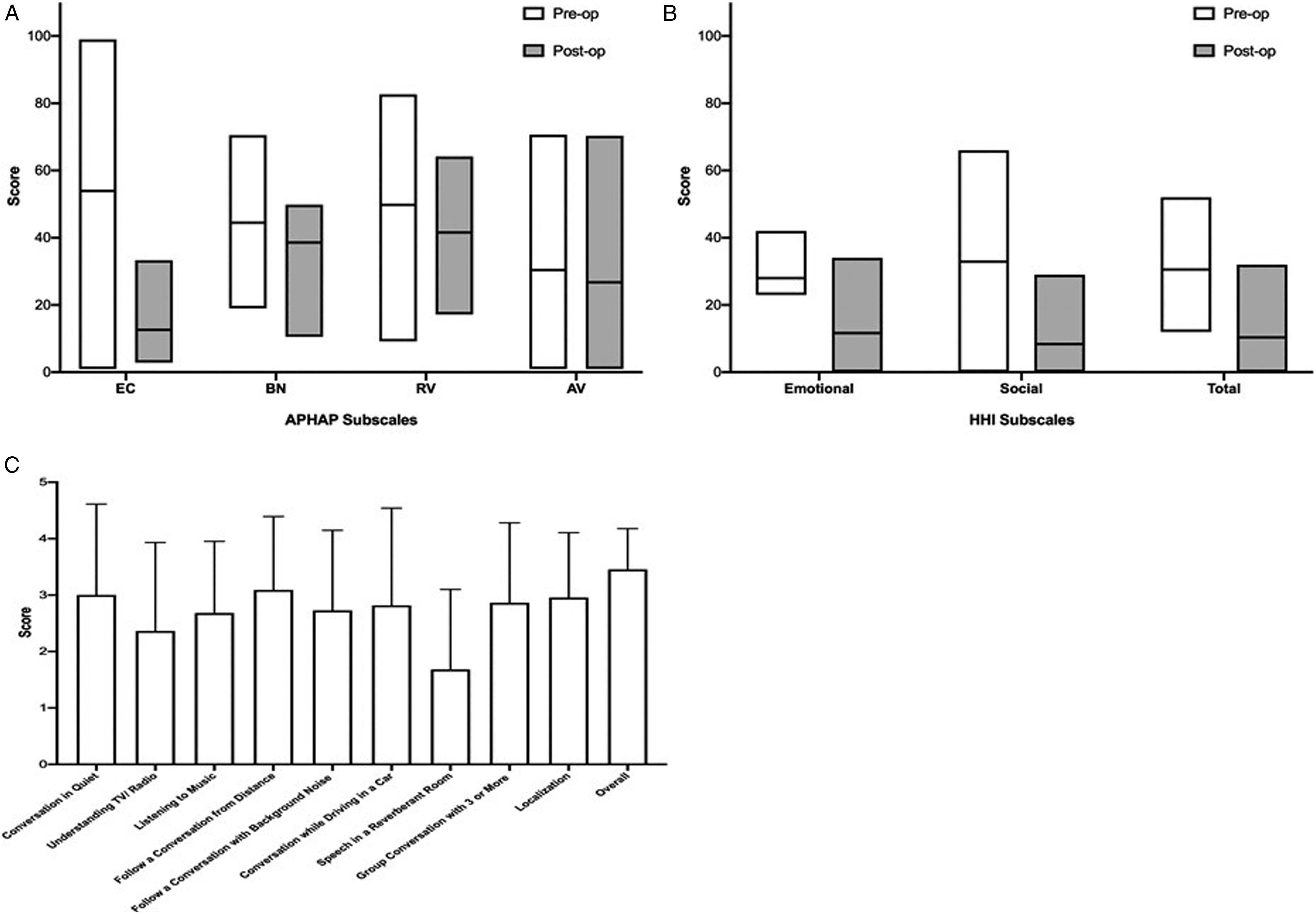
The mean total HHI score improved significantly (p = 0.001) in the postoperative condition compared to the preoperative condition (10.4 ± 8.9 vs. 30.5 ± 10.12). Furthermore, both social and emotional categories of HHI were significantly improved after implantation (p = 0.004 and p = 0.002, respectively). The average HHI results are shown in Figure 4B. The average results for the 10 BBSS questions are presented in Figure 4C. The patients reported an average benefit of using the device of 3.45 compared to the preoperative condition (5indicates the greatest possible benefit).
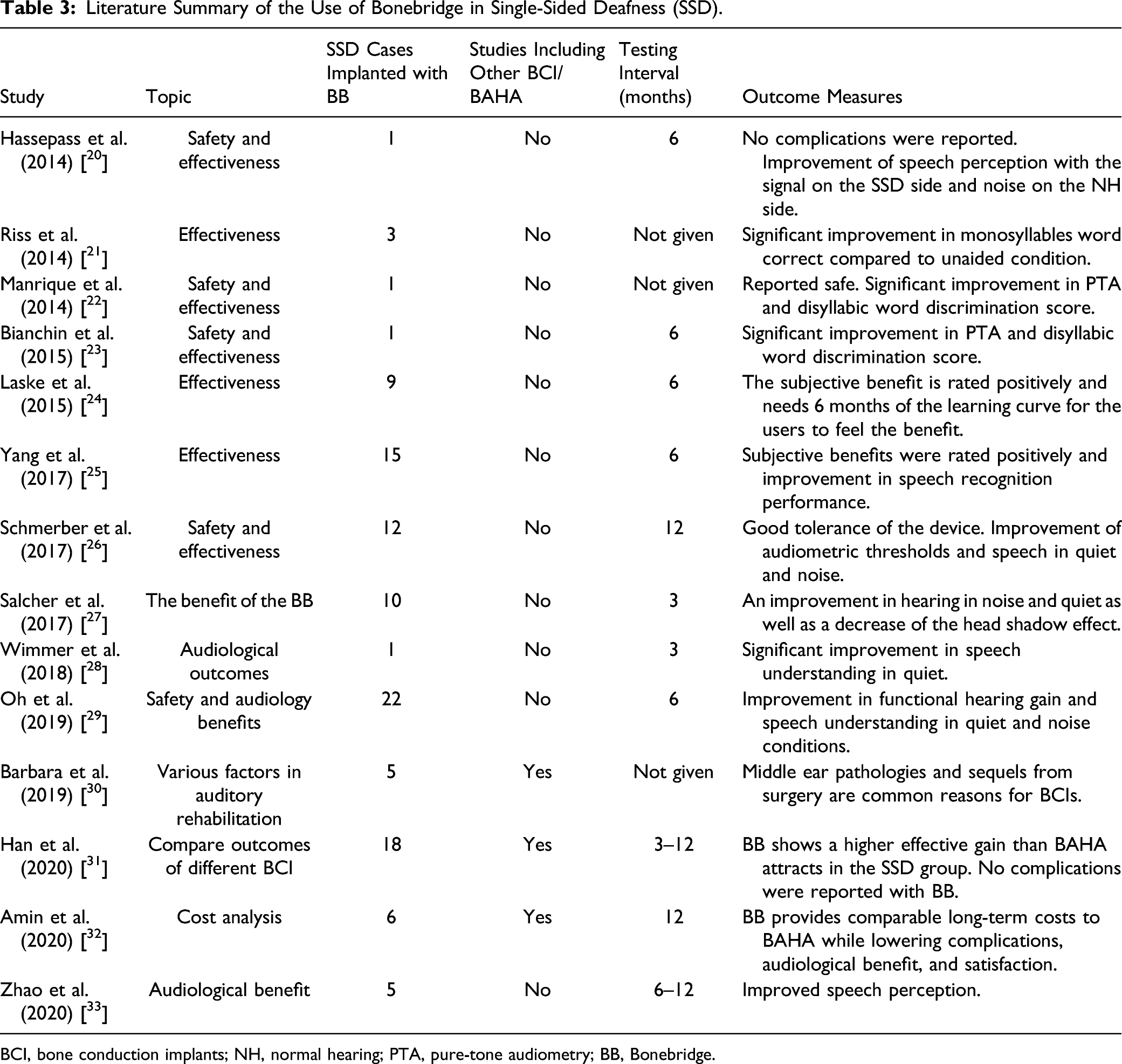
The literature review search yielded 21 articles published until the end of 2020. After exclusion of articles that did not meet the search criteria by including BB as a treatment for SSD, 14 articles were included in the review.20-33 These studies aimed to determine the safety and audiological benefits of transcutaneous BCI. A study reported the cost-benefit relationship of transcutaneous BCI and compared it with a BAHA device. 32
Literature Summary of the Use of Bonebridge in Single-Sided Deafness (SSD).
BCI, bone conduction implants; NH, normal hearing; PTA, pure-tone audiometry; BB, Bonebridge.
Discussion
This retrospective study demonstrated the clinical and subjective benefits of transcutaneous BCI in SSD patients. The objective benefits were evaluated using different audiometric measurements, and 3 different questionnaires were used to assess subjective benefits. In 2 different hearing situations, with the signal coming from the front and noise coming from the contralateral side (S0NCL) and the signal coming from the implanted side and noise from the contralateral side (SBBNCL), the SPIN of the patients for correctly repeating at least 50% of the words correct in background noise was significantly better in the postoperative condition. SBBNCL is the head shadow situation, and the maximum benefit was measured in this situation with a significant improvement of
The placement of the bone conduction FMT plays an important role in the audiological outcomes. In our study and for all patients, bone conduction FMT was implanted in the mastoid at the sino-dural angle, which is the most common position of such percutaneous systems in line with the surgical approach of Salcher et al. 27 ; therefore, a similar result is not surprising. Laske et al. 24 implanted the bone conduction FMT in a surgical position that is remarkably similar to the position reported in our study and Salcher et al. 27 However, the audiological test conducted was different because the noise was presented from the front and showed less benefit (−1.65 dB SNR) than that reported in our study (−2.3 dB SNR), in which the sound signal was presented from the front and noise on the contralateral side.
In the subjective assessment of the experience of transcutaneous BCI users with the device, patients felt a significant improvement in the ease of communication on the APHAB, with a score of 41.3% (p < 0.001). Background noise, reverberation, and aversiveness tended to improve by 5.9% (p = 0.37), 8.22% (p = 0.27), and 3.6% (p = 0.72), respectively. Improvement in the category “ease of communication” is generally much more pronounced than perceived hearing improvement in other categories, in line with the result of Salcher et al. 27
The subjective HHI rating showed an overall lower effect on hearing loss after BB implantation in terms of both emotional and social effects. In total, the preoperative condition had an effect of 30.5%, which decreased to 10.4 after BB implantation (p < 0.005), indicating an improvement after the transcutaneous BCI operation.
All patients experienced a subjective benefit in all hearing conditions assessed using the BBSS. This assessment shows the added benefit of transcutaneous BCI compared to the preoperative condition. On average, all 11 patients scored 3.45, which is higher than the score reported by Salcher et al. (2.8). 27 One patient reported no benefit and answered zero for some questions, whereas all remaining patients responded with positive values only. The patient who gave the lowest score on the BBSS also showed the lowest scores on the SPIN.
Although our retrospective findings were obtained from 11 participants without direct comparison between percutaneous and transcutaneous BCD, our findings demonstrate that comparable results for transcutaneous BCI in SSD patients can be obtained, as reported in earlier studies with percutaneous bone-anchored devices. Our findings on safety and audiological performance agree with those of previous reports on transcutaneous BCI in SSD patients (Table 3). The testing interval in our study was at least 2 years, which is much longer than that reported in the literature (3–12 months). The highly decreased postoperative complication rates due to intact skin above the implant are a significant advantage of BB over percutaneous BCIs and need to be highlighted. Audiologically, transcutaneous BCI is a better alternative to percutaneous devices when the bone thickness allows transcutaneous BCI implantation.
CI was approved by the Food and Drug Administration in 2019 for use in SSD. 2 It showed significant audiological and speech improvements. However, CI may not be the appropriate treatment for cases with aplasia either in the cochlear nerve or in the cochlea itself. The CI showed significant benefits and improved outcomes in cases with IEM. 9 Nevertheless, patients with a narrowed internal auditory canal showed significantly lower outcomes.10,11 In such patients and those who have long-duration of hearing loss and show positive satisfaction with the soft bands, BCI could be a viable treatment option to improve their quality of life. BCI can provide those patients with better language and music perception, the ability to use the devices without rehabilitation shortly after the surgery, and less cost compared to CI. This study has some limitations, such as the small number of participants, and the diversity in their etiology of hearing loss.
Conclusion
Transcutaneous BCI at the sino-dural angle is an alternative in patients with SSD. In this study, the benefits of transcutaneous BCI in SSD patients were comparable to those in earlier reports. An improvement in the speech in noise, as well as a decrease in the head shadow effect, can be expected in patients with the transcutaneous BCI. The general satisfaction of SSD patients with transcutaneous BCI is high, and postsurgical complications are limited. Proper counseling, sufficient trial time before surgery, and realistic expectations are helpful.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
