Abstract
Histiocytic sarcoma (HS) is an aggressive and rare hematological malignancy. Its treatment has not been established, and most patients die within 2 years of diagnosis. Resection can provide a favorable prognosis for solitary lesions. We present the case of an 80-year-old Japanese man with HS. He presented a history of a slow-growing painless mass in the lower part of his right jaw. Ultrasonography showed a swollen lymph node in the vicinity of the right submandibular gland. Contrast-enhanced computed tomography revealed a heterogeneous, low-contrast mass on the right of the neck. Magnetic resonance imaging revealed a heterogeneously enhanced mass in gadolinium-enhanced T1-weighted images. The fine needle biopsy showed spindle-shaped cells and HS was suspected. Fluorodeoxyglucose positron emission tomography revealed uptake by the tumor alone. The patient underwent right upper neck dissection and resection of the submandibular salivary glands. No postoperative adjuvant treatment was administered, but 2-year survival was achieved. Histopathological examination showed proliferation of large, pleomorphic atypical cells without differentiation into lymphocytes, which proved their differentiation into histiocytes. A bone marrow biopsy showed no evidence of monocytic leukemia. Thus, a diagnosis of HS was made. With local treatment alone, our patient achieved long-term survival, maintaining his quality of life.
Introduction
Histiocytic sarcoma (HS) is a rare neoplasm characterized by malignant proliferation of cells with morphological and immunophenotypic features similar to those of mature tissue histiocytes.1,2 It accounts for less than 0.5% of all malignancies of the hematopoietic system. 1 Approximately one third of cases occur in the lymph nodes, about one third in the skin, and about one third in several other extranodal sites, most commonly the intestinal tract. 1 HS usually demonstrates aggressive behavior, and treatment options are limited, resulting in a poor prognosis. 1 In this paper, we report a case of primary HS originating from a lymph node in the submandibular region that was treated with surgical resection and had a good prognosis for more than 2 years.
Case report
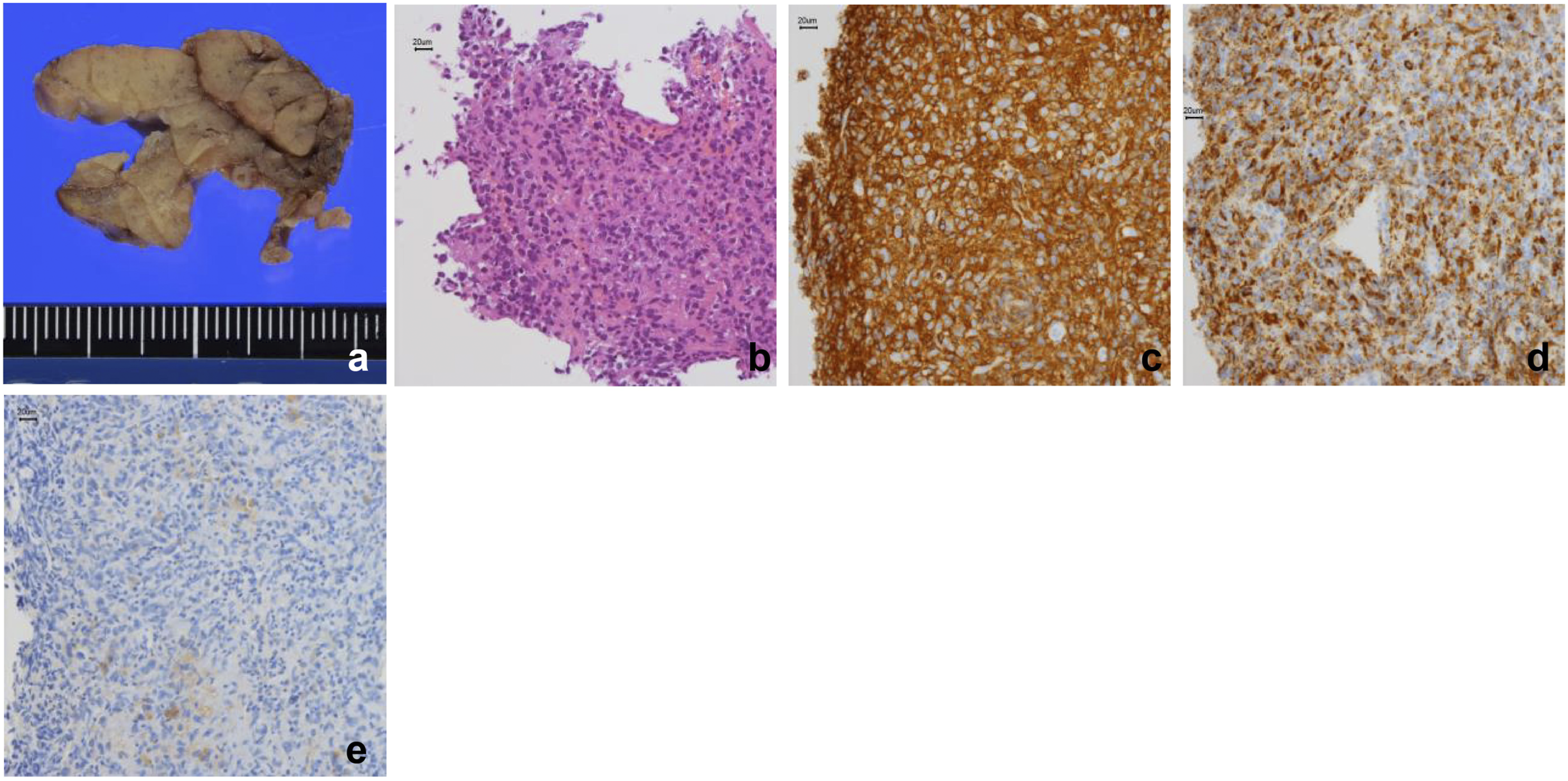
An 80-year-old Japanese man came to our hospital with a slow-growing painless mass in his upper neck area and was admitted to our department. Computed tomography (CT) revealed a well-circumscribed solid mass exhibiting a slightly heterogeneous enhancement measuring 3.8 × 3.5 × 2.6 cm on the outer side of the right submandibular gland. Magnetic resonance imaging (MRI) of the axial section revealed a hypointense mass on T1-weighted images (Figure 1C) and T2-weighted images (WIs) (Figure 1D). The mass was heterogeneously enhanced by gadolinium in the axial (Figure 1E) and coronal (Figure 1F) sections of the MRI. Fluorodeoxyglucose positron emission tomography (PET)/CT revealed tumor uptake and no uptake in any other lymph nodes or bone marrow (Figure 1G and H). Fine needle aspiration cytology (FNAC) was performed and suggested a nonepithelial malignant lesion. The results of the core needle biopsy (CNB) suggested HS due to the histological characteristics of the spindle-shaped cells and their immunostaining patterns. The patient was treated with dissection of the right upper neck and excision of the submandibular gland. The marginal mandibular branches of the facial and hypoglossal nerves were preserved. The resected specimen was serially sectioned, revealing a homogeneous, slightly lobulated, well-circumscribed nodule measuring 3.8 × 3.8 × 2.6 cm without extracapsular extension (Figure 2A). Pathologically, tumor cell growth was observed against the background of normal lymph nodes and the tumor consisted of large oval or pleomorphic atypical cells with distinct nucleoli (Figure 2B). The surgical margins were found to be negative for malignancy. Malignant lesions were not observed in the other resected neck lymph nodes. Tumor cells were positive for CD45 and CD68 and focally positive for the S-100 protein (Figure 2C-E). Immunoreactivity for epithelial cell membrane antigen, pancytokeratin, CD3, CD20, CD30, CD34, or ALK was not observed. Patients with acute monocytic leukemia were excluded due to the absence of abnormal peripheral blood and bone marrow aspiration samples. Based on these results, primary HS was confirmed in the neck lymph node. The patient refused any additional treatment. No evidence of recurrence or metastasis was observed 2 years after surgery. Image findings of the tumor. Contrast-enhanced computed tomography image showing a well-circumscribed, solid, heterogeneous low-contrast mass measuring 3.8 cm × 2.6 cm in the right neck (A, B). MRI image showing a mass with low signal intensity on T1-weighted images (C) and T2-weighted images (D). Gadolinium contrast-enhanced MRI image revealing a strongly heterogeneous contrast-enhanced mass (E, F). A positron emission tomography scan image revealing strong accumulation, consistent with a mass in the right neck (G). No accumulation is seen at other locations, including the bone marrow and neck lymph nodes (H). MRI, Magnetic resonance imaging. Histologic findings of the tumor. The resected specimen measuring 3.8 × 3.8 × 2.6 cm, with no extracapsular extension (A). Pathologically, the tumor is composed of large oval or pleomorphic cells with well-defined nucleoli (B). Immunohistologically, the tumor cells are CD45- and CD68-positive and focally positive for S-100 protein (C-E).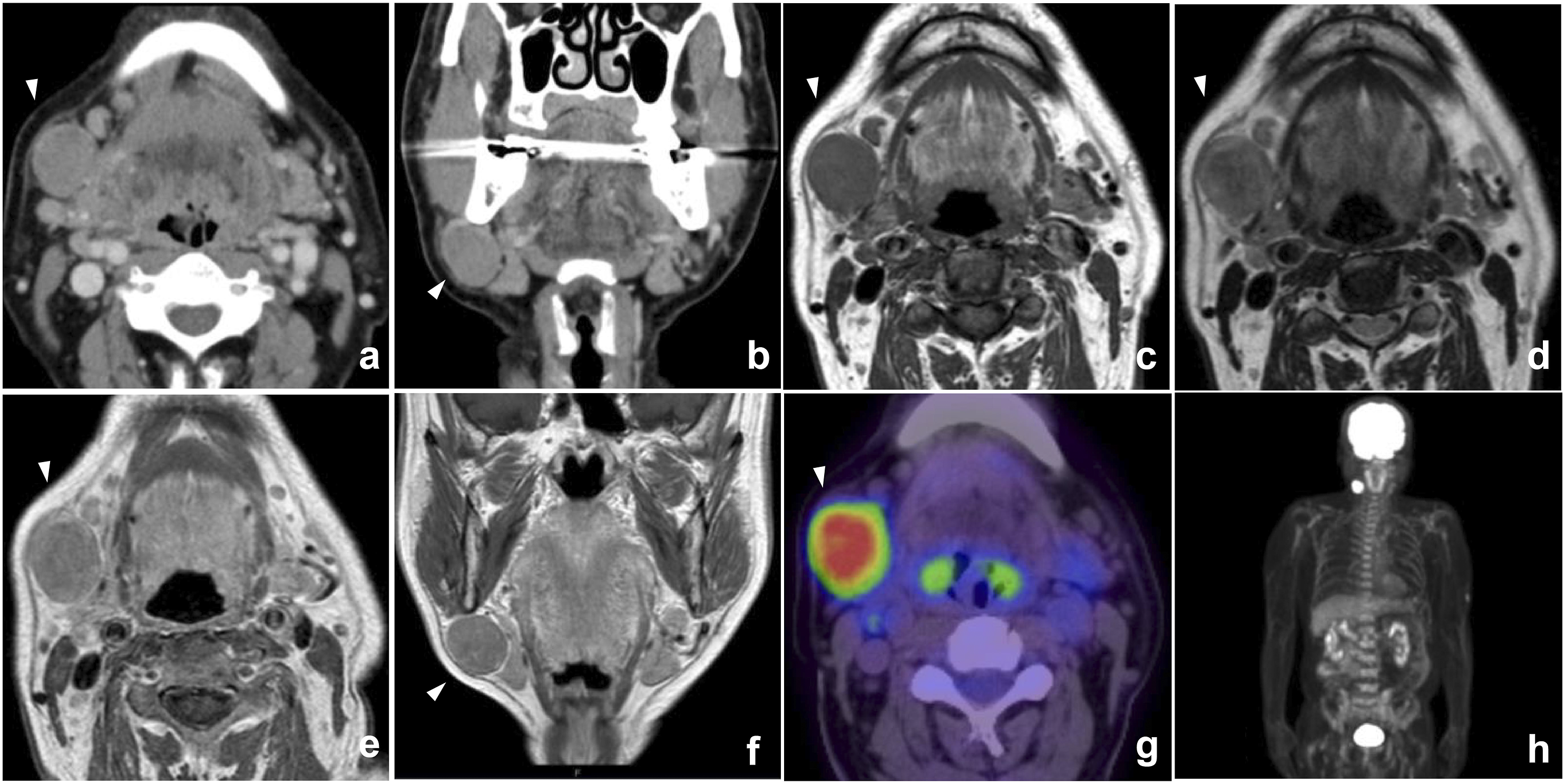
Discussion
Clinical features of primary histiocytic sarcoma cases in the head and neck region.
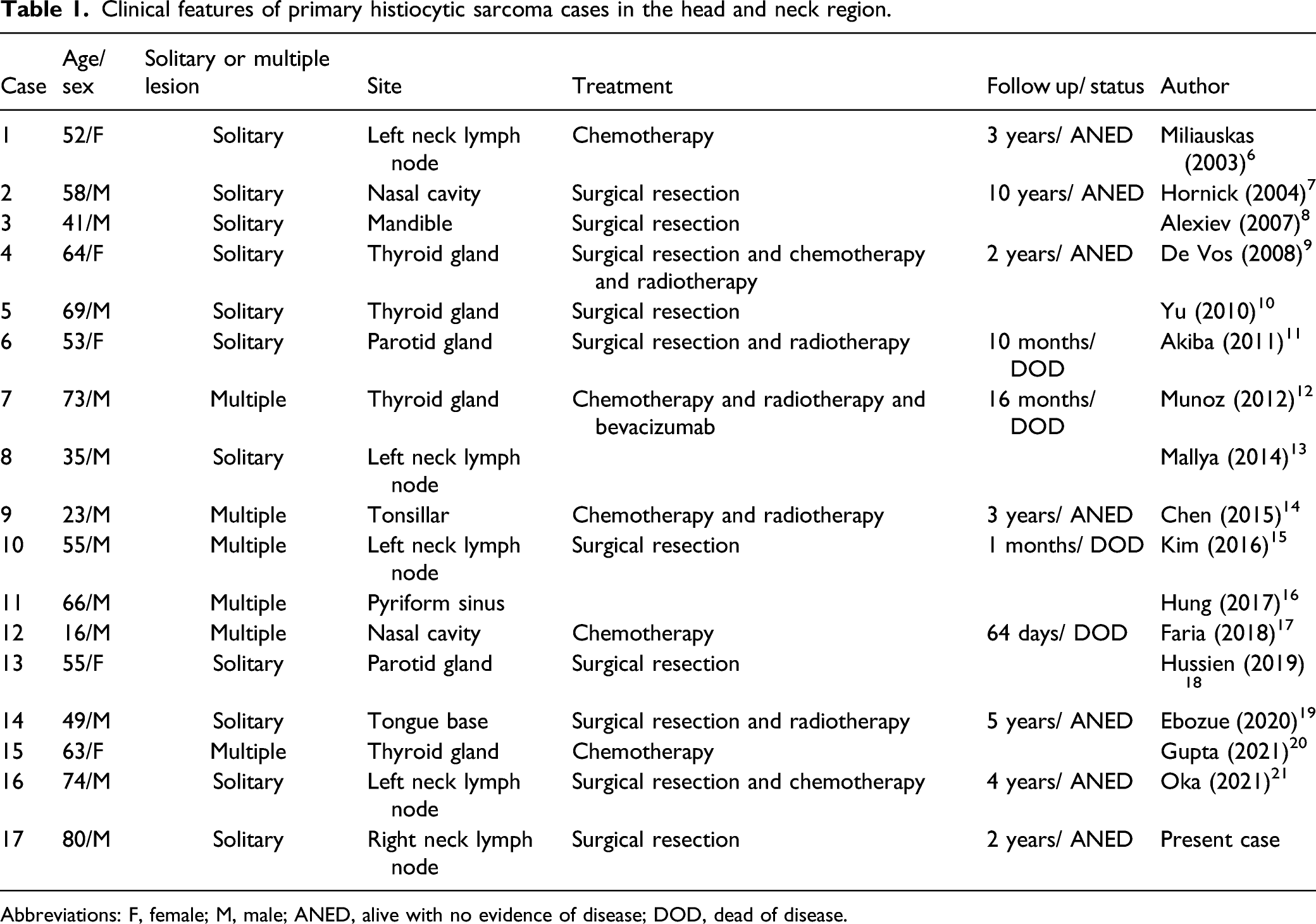
Abbreviations: F, female; M, male; ANED, alive with no evidence of disease; DOD, dead of disease.
Histopathologically, they show morphological and immunohistochemical similarities with mature histiocytes. Tumor cells are large, usually round to oval in shape, but may be spindle-shaped. 1 The cytoplasm is rich and acidophilic. The nuclei are pleomorphic, with well-defined nucleoli, and may be multinucleated. Necrotic and hemophagocytic images can be obtained.1,16Immunohistochemically, HS expresses CD45, CD68, CD4, and lysozymes, which are the main markers of histiocytic differentiation. 23 In our case, the expression of CD68 suggested that the tumor was of histiocytic origin. However, CD68 has also been found to be expressed in other carcinomas, including melanoma and dendritic cell tumors. A specific marker for histiocytes has been sought and Nguyen et al. showed that CD163, a hemoglobin scavenger receptor, may be more specific to histiocytes. 24 However, it should be emphasized that none of these markers is specific for histiocyte differentiation and that other markers must be excluded. CD3 and CD20, which are positive in normal T and B cells, are typically absent. 1 The S-100 protein may be positive, but is generally only partially positive, 1 and immunohistological negativity for CD20 and CD30 is associated with diffuse large B-cell lymphoma (DLBCL). Immunohistological negativity for CD20 and CD30 helps differentiate diffuse large B-cell lymphoma (DLBCL) and anaplastic large cell lymphoma (ALCL). 20 ALK is also useful for differentiating ALCL. 1 Facchetti et al. reported the expression of programmed death ligand 1 (PD-L1) in HS. 25 The WHO classification of HS requires a bone marrow biopsy to exclude acute monocytic leukemia forming masses. 2 However, in practice, monocytic leukemia rarely presents as an extramedullary mass. Furthermore, although morphologically similar, HS can be differentiated immunohistochemically using the negative myeloid markers CD33 and CD34.
Genetically, the absence of detectable clonal rearrangements of the IGH or TCR genes was previously required for diagnosis. However, Feldman et al. found that patients with follicular lymphoma and associated HS shared these genetic mutations, 4 and Chen et al. reported a certain probability of rearrangement of the clonal IGH gene, even in solitary cases. 26 Due to its rarity, the genetic characteristics of HS have not been fully clarified; however, with the advent of next-generation sequencing, this is now being worked on. The BRAFV600E mutation has been reported in many cases, indicating that activation of the RAS/RAF pathway may be involved in malignant transformation.25,27 Hung et al. reported a KMT2D mutation, suggesting the involvement of chromatin dysregulation. 16 However, due to the rarity of this tumor, its genetic cause has not been fully elucidated, and further studies are needed.
HS is often diagnosed secondary to lymphoma or in a clinically advanced state, with limited response to chemotherapy and high mortality.1,3 Takahashi et al. reported that many patients progressed aggressively and died in 2 years. 1 On the other hand, in cases involving the head and neck region, some patients survived for more than 2 years after surgery ± chemotherapy ± radiotherapy (Table 1). A study using the National Cancer Database (NCDB) examined 330 cases of HS and reported that overall survival (OS) improved in HS patients treated with local therapy (surgery alone or with radiation therapy) compared to systemic therapy. 3 Dalia et al. also reported that adjuvant radiotherapy after surgical treatment improved OS. 23 Based on these results, it is optimal to perform surgical treatment and adjuvant radiation therapy as much as possible, especially if local treatment is possible. Although there are no clear indications for neck dissection, it should be performed in accordance with other malignant tumors in the head and neck region. Chemotherapy can be used in cases of systemic symptoms or inoperable cases, but no treatment regimen has been agreed upon.3,25 As mentioned above, the expression of PD-L1 in HS has been reported and may attract attention as a therapeutic target in the near future.
Ongoing research is needed to fully understand the clinicopathological and genotypic characteristics of this rare tumor and to further elucidate its etiology. A treatment regimen should be established as soon as possible. Fortunately, our patient has been free of recurrence for 2 years and we will continue to closely monitor him.
Conclusions
In this paper, we report a case of primary histiocytic sarcoma of a neck lymph node. The lesion was solitary and surgical resection of the primary lesion and neck dissection of the affected side were performed. No further treatment was performed after surgery, and the patient has remained disease-free for 2 years now.
Footnotes
Author contributions
AK, YI, RS, TW and JH performed the surgery and provided bedside care. AK and NB drafted the manuscript. HN, HK, and KS performed the pathological investigations. YH conceived the study design. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ Note
This material has not been published elsewhere, either in whole or in part. All authors have been personally and actively involved in the substantive work of preparing the manuscript and are jointly and individually responsible for its content.
Ethical approval
All procedures performed on patient tumor samples in this study were carried out in accordance with the ethical standards of the Institute Ethics Committee and the Declaration of Helsinki of 1964 and its subsequent amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from the patient and his family for the publication of clinical details and images.
