Abstract
Introduction
Malignant salivary gland tumors are a rare and heterogeneous group of head and neck cancer with a relatively low incidence in the general population, accounting for <1% of all malignant tumors. 1 There is a multitude of histological classifications of salivary gland tumors, which are classified following the latest World Health Organization (WHO) classification of head and neck cancer. The varieties of which make subtype of salivary gland tumors significantly rare. Mucoepidermoid carcinoma (MEC), representing approximately 1/3 of salivary gland tumors and accounting for 70% of major salivary gland malignancies, is the commonest malignancy of the salivary gland. 2
The research shows that the prognostic factors of MEC included age, tumor stage, histological grade, tumor margin status, lymph node involvement, and adjuvant therapy, in which staging and grade may have the most significant effect on prognosis.3,4 Histologically, MEC is typically composed of a mixture of three cell populations including mucous, intermediate, and epidermoid. According to the ratio of epidermoid cells to mucous cells, mucoepidermoid carcinoma (MEC) was sub-classified into low, intermediate, and high grades. Generally, low-grade MEC has an excellent prognosis that was reported over 97% at 5 years survival ratio. Despite the multidisciplinary therapeutic strategies, the prognosis of high-grade MEC is still dismal and the 5-year overall survival rate of only 22.5%. 5
High-grade mucoepidermoid carcinoma typically occurs in the major salivary glands (e.g., the parotid gland) and is associated with advanced T and N stages. 6 So far, the American Joint Committee on Cancer (AJCC) Tumor, nodes, and metastases-staging (TNM) system, an authorized tool for prognostic prediction of MEC patients, is widely used in clinical practice. However, the AJCC staging system is short of certain pathological characteristics and treatment information, and the use of AJCC staging system to predict the prognosis of patients has specific limitations.
Therefore, exploring more predictors and constructing a multivariable survival prediction model based on personalized characteristics was necessary so as to provide clinical decision support and comprehensive, individualized prognosis assessment in a convenient manner. Nomograms are visual description of predictive statistical models for individual patients, and they have been widely applied to predict an individual patient survival, such as gastric cancer and esophageal cancer. Some scholars have established a survival prediction model for parotid gland mucoepidermoid carcinoma. 7 However, the clinicopathologic characteristics, treatment strategy, and outcome of low-grade pMEC are different from high-grade pMEC. Therefore, it is necessary to further explore the prognostic factors and personalized treatment for patients with high-grade parotid mucoepidermoid carcinoma.
In this study, 519 patients diagnosed with high-grade mucoepidermoid carcinoma of the parotid gland between 2004 and 2015 were evaluated retrospectively using SEER*Stat software based on the SEER database established by the National Cancer Institute in 1973. We explored the prognostic risk factors associated with prognosis and constructed a nomogram, in order to provide a basis for prognostic evaluation of patients with high-grade mucoepidermoid carcinoma of the parotid gland.
Patients and methods
Data sources and research factors
As Figure 1 shows, the patient records were obtained from the SEER database through the version 8.3.9 of the SEER*Stat software: Incidence-SEER 18 Regs Custom Data (with additional treatment fields), Nov 2018 Sub (1975–2016 varying)—Linked To County Attributes—Total U.S., 1969–2017 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2019, based on the November 2018 submission. Flow chart for cases screening.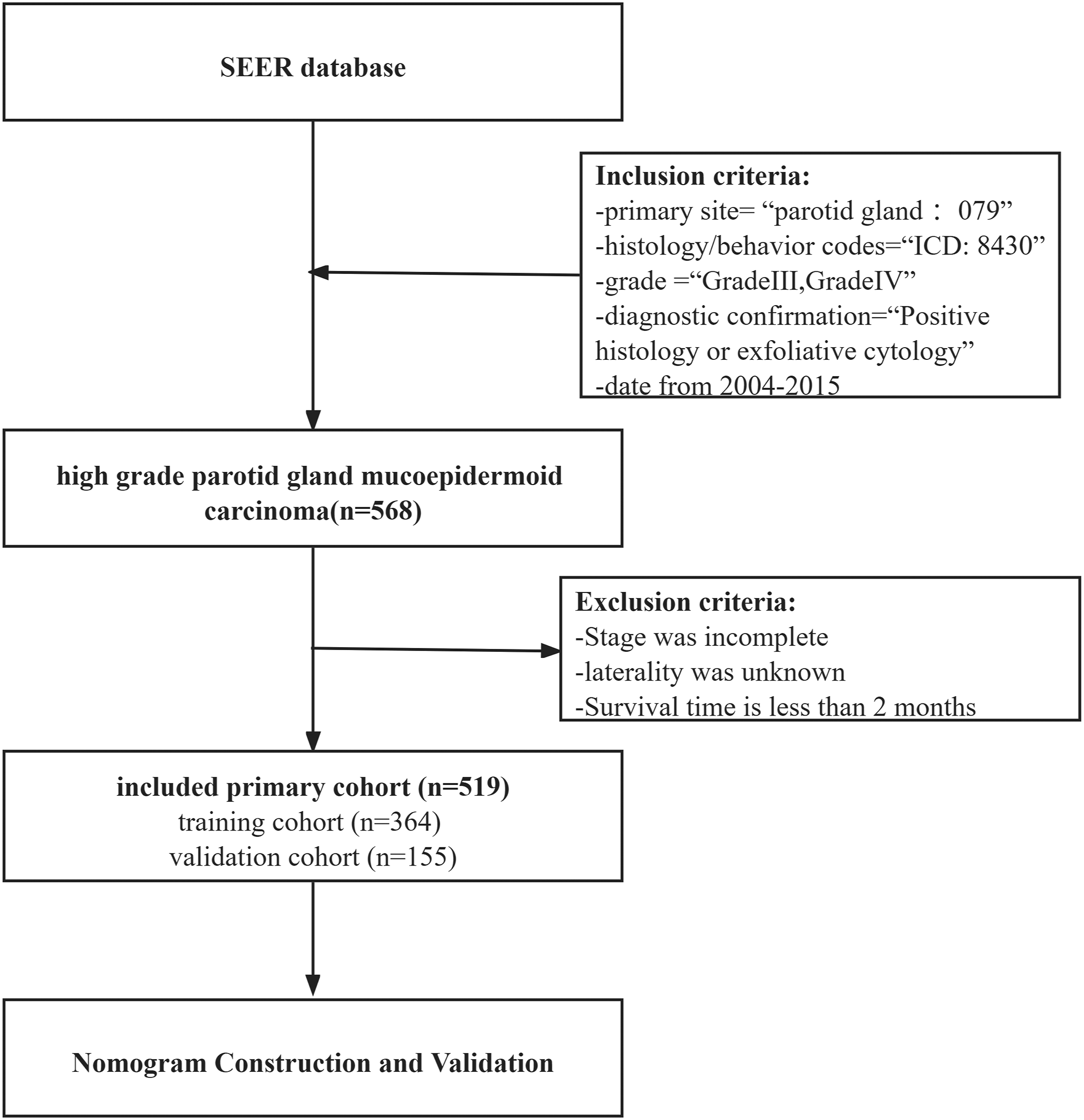
We screened cases from the SEER database by choosing the primary tumor location of salivary gland tumor using the variables ‘‘primary site’’ (parotid gland = 079), ‘‘histology/behavior codes’’ (International Classification of Diseases code (ICDO) = 8430), ‘‘diagnostic confirmation’’ (positive histology or exfoliative cytology), and the date from 2004–2015. Additionally, the following ‘‘grade’’ (Grade III and Grade IV) were selected. We selected several survival-related variables including general characteristics (age, sex, and marriage status), tumor characteristics (laterality, grade, stage, regional lymph nodes status, and extraparenchymal lesions), and treatment information (lymph node dissection status, surgery status, radiotherapy status, and chemotherapy status).
The exclusion criteria are as follows: 1. Stage was incomplete. 2. Laterality was unknown. 3. Survival time is less than 2 months.
All patient data are collected from the SEER database with public nature; thus, the patient consent was not required.
Statistical analysis
The endpoint for this study was overall survival, defined as time from diagnosis to death or to the end of the study period that calculated from diagnosis to death or to the end of the study period. The initial data were randomly divided in a 7:3 ratio into a training cohort (N = 364) and a validation cohort (N = 155) using the R software (“caret” package). Statistical significance via log-rank test was used to compare training cohort and a validation cohort. A log-rank test was used to estimate statistical significance between the cohorts. Clinical data were statistically analyzed employing SPSS 26.0 statistical software. The counting data were expressed as N (percentage %). The univariate Cox regression models were performed to identify potential independent prognostic indicators. Subsequently, the multivariate Cox regression analysis was applied to determine the independent factors associated with prognosis. Univariate and multivariate Cox proportional hazards regression models were performed to identify independent prognostic indicators. P-values < 0·05 represented that the difference is statistically significant.
Nomogram construction and validation
The R programming environment with the rms package was used to build a visual nomogram that predicted the survival of high-grade pMEC patients at 3 and 5 years, incorporating all independent factors associated with prognosis. The area under the receiver operating characteristic (ROC) curve (AUC) was performed to assess the discrimination ability of 3- and 5-year survival of the prognostic nomogram. Calibration curves were drawn to assess the consistency between the predicted and actual probabilities. Moreover, decision curve analysis (DCA) was implemented to investigate the clinical usefulness of the nomogram.
Results
General characteristics
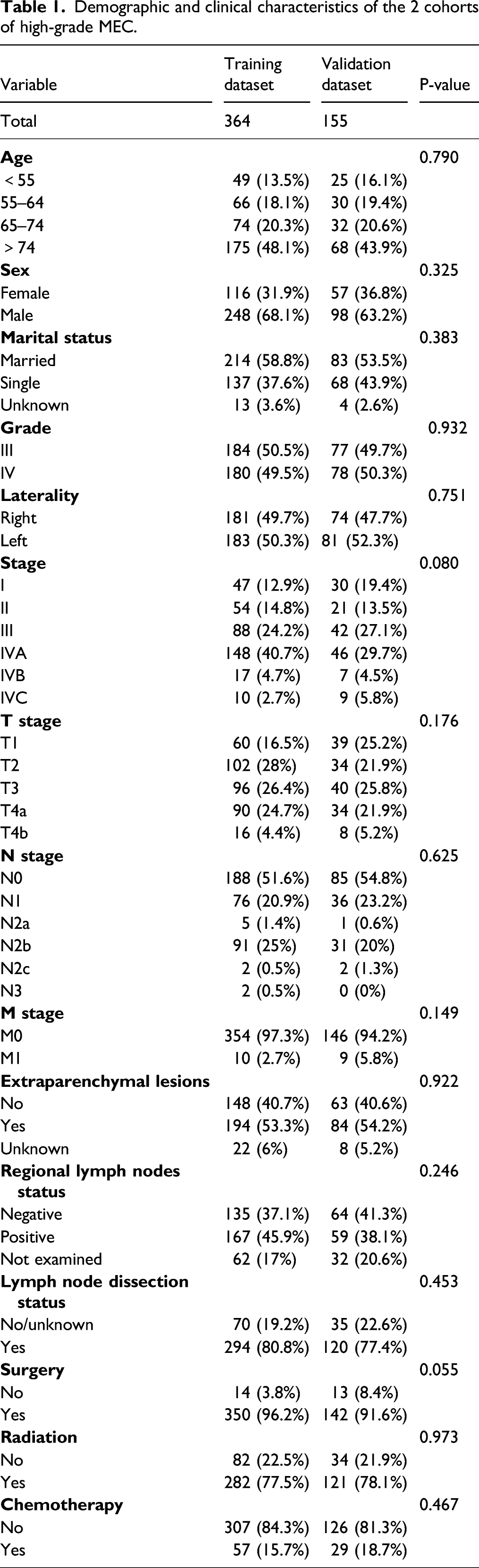
Demographic and clinical characteristics of the 2 cohorts of high-grade MEC.
Explore the independent factors affecting patient survival time
Univariate and multivariate analyses on prognostic factors for overall survival in high-grade MEC patients.
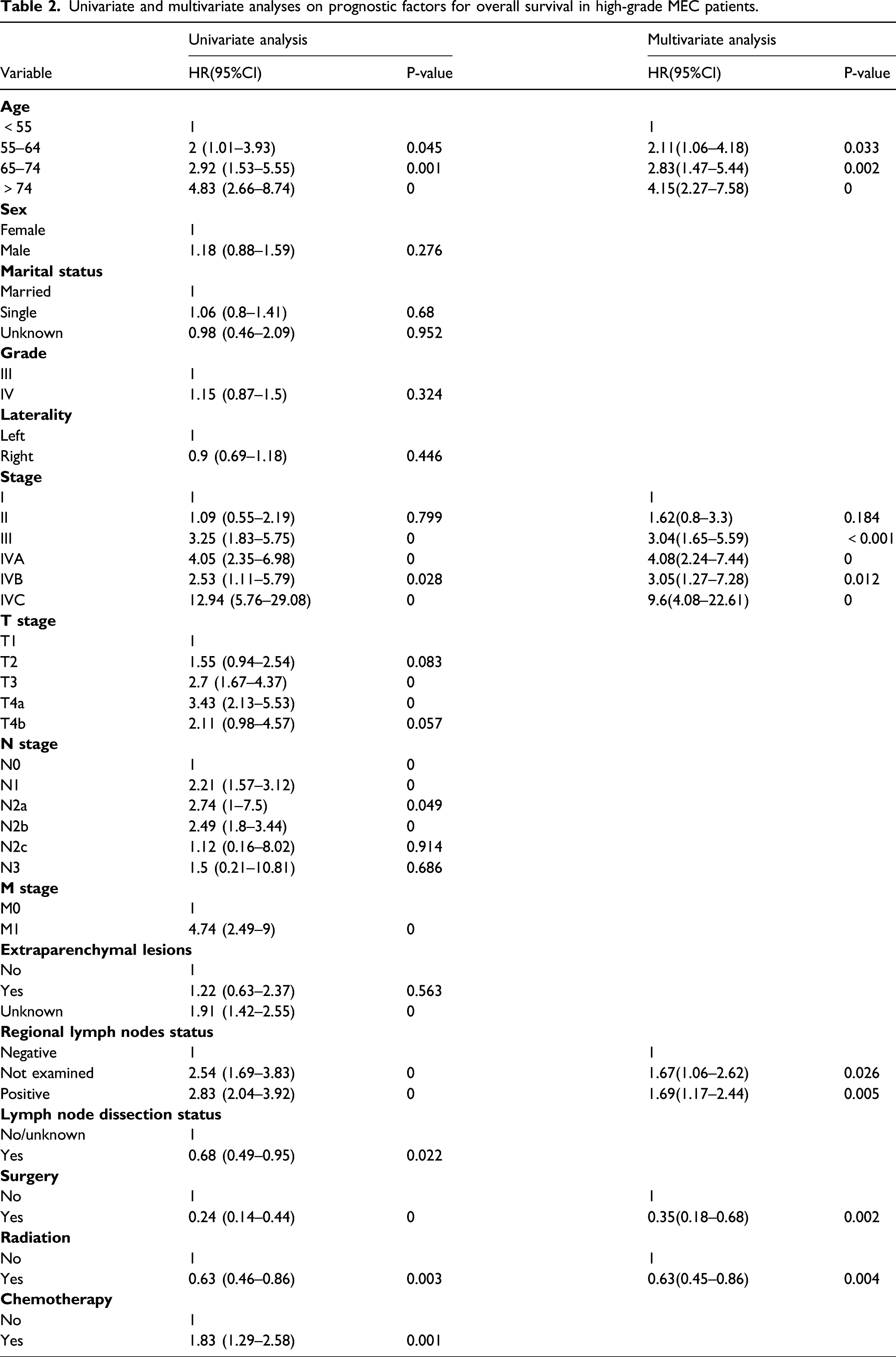
Multivariate Cox stepwise regression analysis was performed on the results of univariate regression analysis, and screen out 5 independent prognosis factors related to OS, including aged 55–64 years (HR = 2.11, p =0.032 versus aged younger than 55 years), aged 65–74 years (HR = 2.83, p = 0.002 versus aged younger than 55 years), aged older than 74 years (HR=4.15, p < 0.001 versus aged younger than 55 years), AJCC stage II disease (in comparison with stage I, HR = 1.62, p = 0.183 ), AJCC stage III disease (in comparison with stage I, HR = 3.04, p < 0.001), AJCC stage IVA disease (in comparison with stage I, HR=4.08, p < 0.001), AJCC stage IVB disease (in comparison with stage I, HR = 3.05, p = 0.012), AJCC stage IVC disease (in comparison with stage I, HR = 9.6, p < 0.001), regional lymph nodes were not examined(HR = 1.67, p = 0.026 versus regional lymph nodes negative),and the regional lymph node test results are positive (HR = 1.69, p =0.005 versus regional lymph nodes negative),surgery (in comparison with no/unknown, HR =0.35, p = 0.002),radiotherapy (in comparison with no/unknown, HR =0.63, p =0.004) (Table 2). We found that older age, advanced AJCC stage, and positive regional lymph nodes were independent risk factors for OS. In addition, treatment with radiotherapy and surgery were significant independent protective factors for OS.
Nomogram construction and validation
Based on the multivariate analysis, we established nomogram prognostic evaluation model to predict the 3‐ and 5‐year OS of high-grade pMEC patients (Figure 2). On the nomogram plot, we scored each independent factor and then summed each factor score. Lastly, we vertically project the total points to the bottom scales of the nomogram and obtain the 3- and 5-year survival rate. The nomogram revealed that stage had the highest effect to prognosis, followed immediately by age, surgery, regional lymph nodes status, and radiotherapy. Nomogram predicting 3- and 5-years OS for high-grade MEC.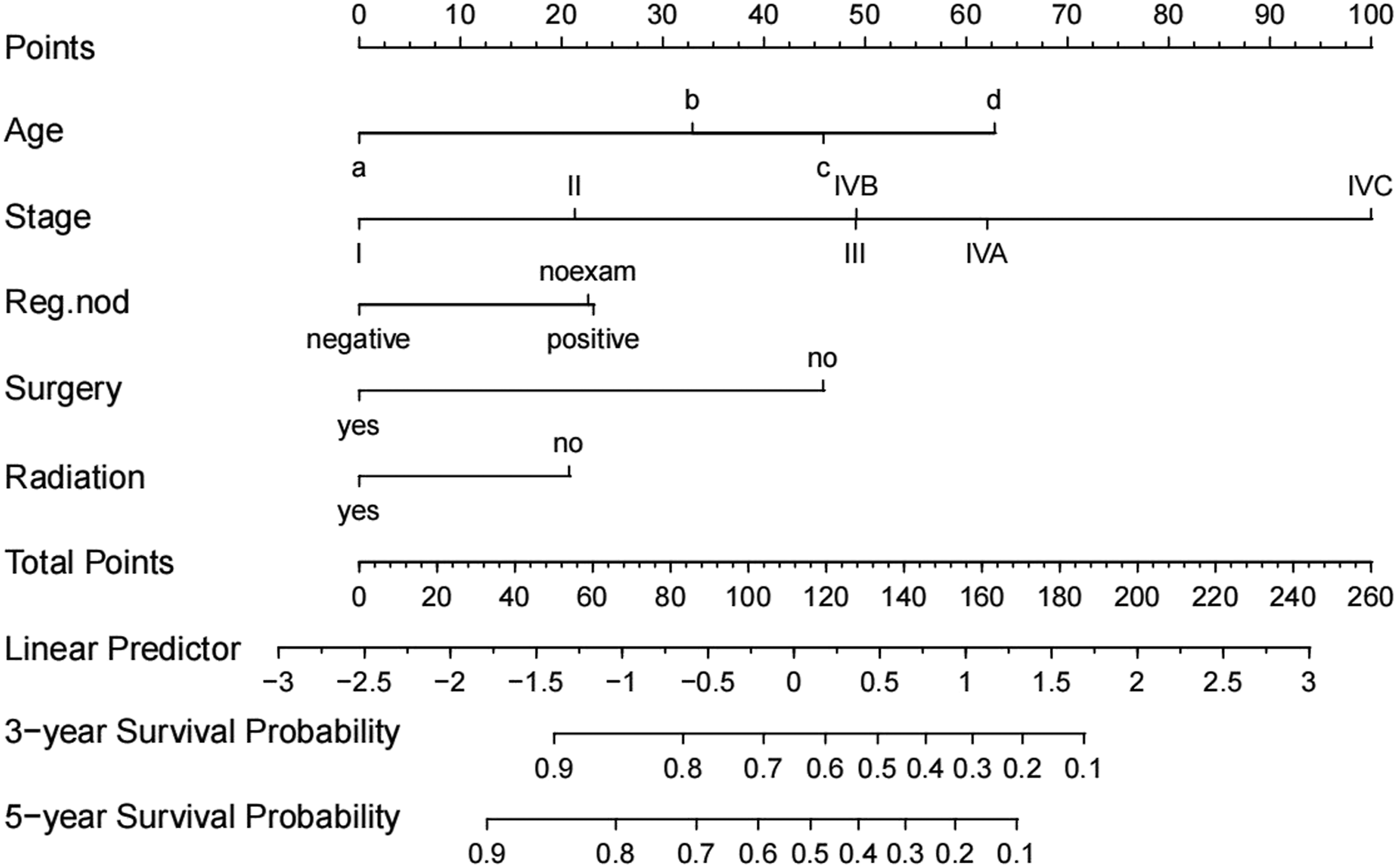
We plotted 3- and 5-year ROC curves based on the relationship between the true positive rate (TPR, sensitivity) against the false positive rate (FPR, 1-specificity), and the area under the ROC curve (AUC) was calculated (Figure 3 A B). In the training cohort, the 3- and 5-year AUCs were 0.779 and 0.793, respectively, and 0.803 and 0.805 in the validation cohort. The ROC curves demonstrated good predicting power of our nomograms. We performed 1000 bootstrap re-sampling analyses on the data and obtained calibration plots of the prediction model. As Figure 3 C D shows that the calibration plots for 3-year OS probabilities for the model demonstrated good agreement between the actual value and the predicted value in the training and validation dataset. Calibration plot analysis on 5-year OS probabilities for the model demonstrated a small over-estimation of mortality risk. Furthermore, we plotted decision curve analysis (DCA) curves to assess the clinical utility by quantifying the net benefits of the nomogram (Figure 3 E F). As DCA curves shown that utilize the nomogram to predict the 3- and 5-year OS probabilities present a better clinical net benefit than the treat-all-patients and treat-none schemes. The ROC curves, calibration curves, and DCA curves for high-grade MEC. A: The AUC for 3- and 5-years OS probability of the training cohort for high-grade MEC. B: The AUC for 3- and 5-years OS probability of the validation cohort for high-grade MEC. C: Calibration curves for 3-years OS probability in high-grade MEC. D: Calibration curves for 5-years OS probability in high-grade MEC. E: DCA of the training cohort for high-grade MEC. F. DCA of the validation cohort for high-grade MEC.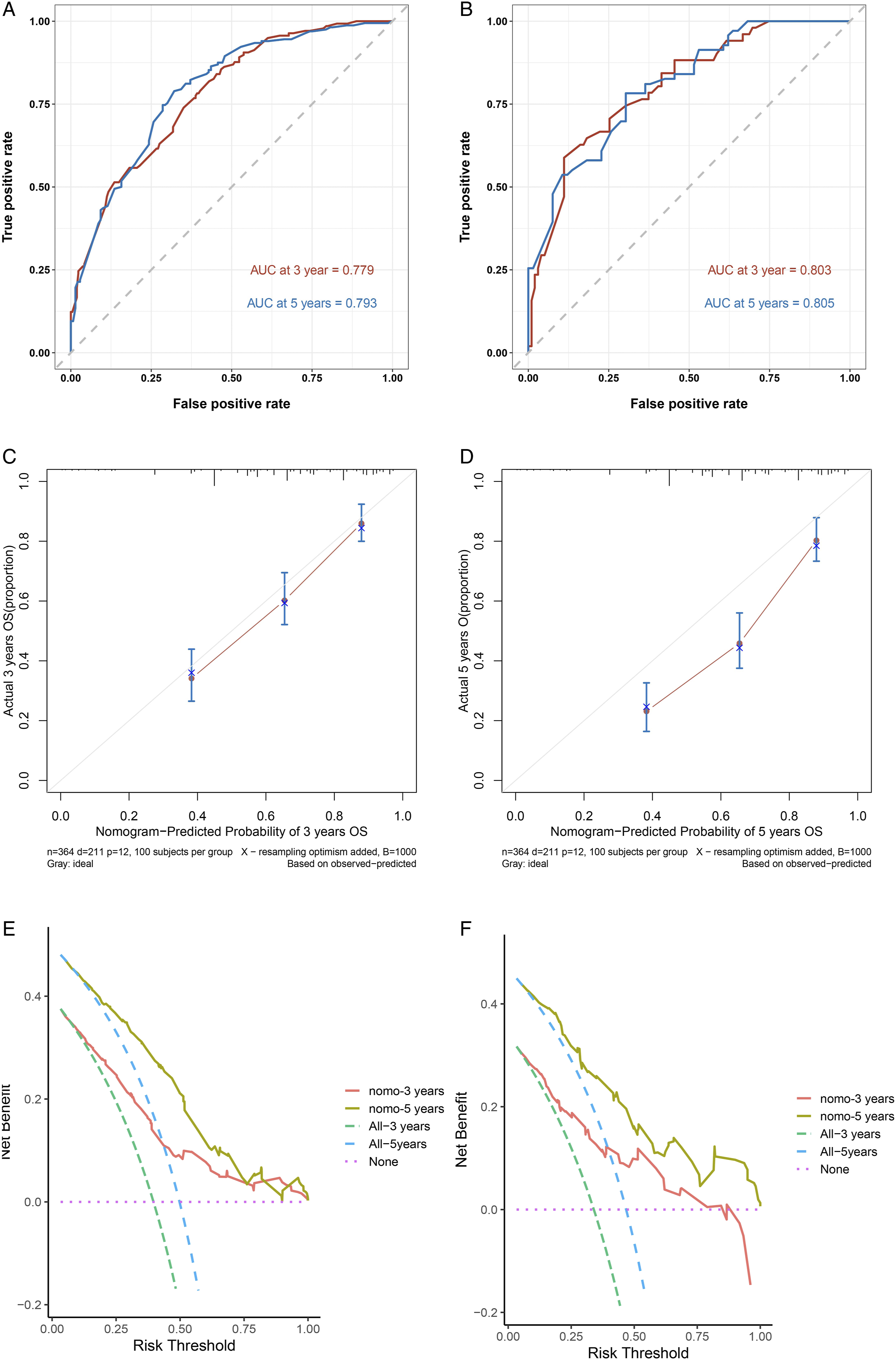
Discussion
The most prevalent malignancies of the salivary glands are mucoepidermoid carcinoma (MEC), with incidence of MEC as 2.3/1,000,000 people per year. 8 MEC constitutes approximately 1/3 of all salivary gland malignancies, and it can arise in either the parotid gland or minor glands, with about 70% of cases arising in the parotid gland. 9
The prognosis of MEC patients was significantly correlated with the histologic grade. The histological grading of MEC was divided into four groups. Grades III (poorly differentiated) and IV (undifferentiated) are classified as the high-grade group with the worst prognosis. The presented study enrolled 519 eligible high-grade pMEC patients derived from the public database SEER. We conducted a multivariate analysis and the result showed that older age, advanced AJCC stage, and positive lymph nodes were independent risk factors for OS. In addition, we found that treatment with radiotherapy and surgery were significant independent protective factors for OS. In the present study, the 3-year and 5-year overall survival rates were 62.1% and 51.1%, respectively. By integrating multiple significant prognostic factors, we constructed a nomogram that offers a comprehensive and intuitive to predict 3- and 5-year overall survival of the high-grade pMEC patients. Age at diagnosis, AJCC stage, and histologic grade were found to significantly affect the survival probability of MEC. 10 Typically, MEC is predominantly consisted of mucous, intermediate, and epidermoid cells. According to the ratio of epidermoid cells against mucous cells, MEC was sub-classified into low, intermediate, and high grades. The high-grade MEC is characterized by a predominance of epidermoid cells,whereas low-grade MEC usually contains a high proportion of mucous cells. Identifying histologic tumor grade is essential, as histologic grade has prognostic implications and guides treatment. 11 The overall survival of patients with high-grade MEC (grade III and grade IV) has a worse prognosis than patients with low-grade MEC (grade I and grade II). Studies have reported that the disease-free survival at 5 years was 97% for low-grade MEC, while high-grade MEC has a 5-year disease-free survival rate of 22.5% only. 5
The clinicopathologic characteristics, treatment strategy, and outcome observed substantial variance between the low-grade MEC and the high-grade MEC. A retrospective analysis revealed that high-grade MEC is associated with male sex, elderly group, larger size, and had a greater tendency to local invasion, distant, and lymph node metastases. 12 The data presented in this paper demonstrate high-grade pMEC often occurs in older males, as the age increased, the adverse outcomes increased gradually. Especially, when aged older than 74 years, the incidence of adverse events was significantly increased.
There were reports in the literature that reveal that the majority of patients (83%) had no lymph node metastases. 13 As the histologic tumor grade increases, the risk of lymph node metastases also significantly increases. 12 Data in this study showed that positive regional lymph nodes were found in 43.5% of patients, and positive regional lymph nodes were an independent risk factor for prognosis.
A previous study of the National Cancer Database (NCDB) shows that patients with high-grade MEC have higher staging. 6 Despite aggressive strategy of treatment undertaken, advanced stage is still considered as the most important independent risk factor for local failure. 14 Our study found that the majority of patients have stage III (24.2%, 27.1%) or stage IV (48.1%, 40%) disease and the risk for death was significantly increased in stage IVC disease (in comparison with stage I, HR =9.6, p < 0.001).
In this study, surgery and radiotherapy were independent protective factors affecting the overall survival, with surgery performed on 98.3% of patients and 77.6% received radiotherapy. For all grades of MEC, surgery treatment remains the primary and curative therapy for patients with clinically resectable tumors. When the histopathological features are a low-grade MEC without advanced T stage, regional lymph node and surgical margins positive, lymphovascular and perineural invasion, and excellent long-term disease control may be achieved using surgery alone.4,15 There are studies which show that in early-stage MEC the postoperative 5-year survival rate was 96 %. 16 Typically, surgical resection with or without lymphadenectomy is treatment for high-grade MEC. If lymph nodes are involved, adjuvant radiotherapy is followed. Some studies suggest that adjuvant radiotherapy may not improve the overall survival. There may be some bias in these studies that not all groups correct for. 17
The staging and treatment modalities depend heavily on histology. Ferrell et al 18 reported that postoperative radiotherapy improved OS compared to surgery alone, in high-grade MEC. A retrospective study has found clear survival benefits in patients who underwent postoperative radiotherapy. 19 Other studies also demonstrated that adjuvant radiation provides survival benefit for high-risk patients. Postoperative radiotherapy was superior to surgery alone in long-term local control rate among the patients with high risk factors after surgery, including stage T3-4, close and incomplete resection, bone invasion, or perineural invasion. 20 Another study also demonstrated that radiotherapy can significantly increase the local control rate and enhance survival of patients with advanced stage and regional lymph node positive, especially for patients with positive resection margin postoperatively; postoperative radiotherapy has significant benefit in local control rate. 21 Thus, when patients are diagnosed as high-grade MEC or with high-risk factors, postoperative radiotherapy should be considered.
The efficacy of chemotherapy is limited and was used as a palliative treatment. In recurrent or metastatic MEC, cisplatin-based chemotherapy, including single agent cisplatin or combination with other chemotherapeutic therapy, has certain efficacy. In the MEC cohort with single-agent paclitaxel, three of those fourteen patients responded. 22 In the present study, the overall survival rate was decreased in patients who received chemotherapy. A review based on NCDB showed that CRT did not benefit patients even if they had high risk factors. 23 This may be connected with decreased survival in patients who have undergone chemotherapy, in which chemotherapy increased systemic toxicity or has a poor clinical efficacy. 18
Inevitably, our study has its limitations. Firstly, this study was designed using retrospective data derived from the SEER database, which presents with inherent biases. Secondly, the SEER database does not provide date that may have an impact on patient survival, such as radiotherapy dose, chemotherapy drugs, molecular markers, smoking and drinking history, nutritional status, and other factors. In the future, the focus of our research is to incorporate more prognostic factors and perform external validation, making the models more reliable.
Conclusions
We explored the predictors of high-grade pMEC patients and found that older age, advanced AJCC stage, positive regional lymph nodes, radiotherapy, and surgery resection were independent predictors for OS. Then, we constructed a multivariable survival prediction model to predict 3- and 5-year OS rates based on personalized characteristics which was necessary so as to provide clinical decision support and comprehensive, individualized prognosis assessment in a convenient manner.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
