Abstract
Objectives: To compare the efficacy and adverse events of different oral corticosteroids (OCSs) withdrawal methods for chronic rhinosinusitis with nasal polyp after endoscopic sinus surgery (ESS). Methods: This was a randomized prospective study conducted from Oct 2019 to Jan, 2021. 35 patients who underwent ESS were randomly divided into 2 groups. Regular group (n = 18) received 0.4mg/kg/day of methylprednisolone orally for 10 days, tapering group (n = 17) received 0.4mg/kg/day of methylprednisolone orally for 7 days, followed by progressive reduction of 8 mg of methylprednisolone per week for 3 weeks. The visual analogue scale (VAS) score, the Lund-Kennedy endoscopy (LKE) score, and the E score were assessed preoperatively and at half and 1, 2, and 12 months postoperatively. Statistical analyses were performed using SPSS. Results: There was no statistical difference in the baseline characteristics between the 2 groups. The postoperative VAS scores and LKE scores of patients were significantly improved from those preoperatively (P-values < 0.05). There was no statistical difference in the LKE score, E score, and VAS score between the 2 groups both preoperatively and postoperatively (P-values > 0.05). There was no statistical difference in adverse events between the 2 groups (P-values > 0.05). Conclusions: The combination of OCSs and ESS can improve the clinical symptoms of patients and the recovery of nasal mucosa. There was no difference between the 2 drug withdrawal methods in efficacy and adverse events. Drug withdrawal gradually is more complicated therefore, in clinical practice, OCSs withdrawal directly would be the better option for patients.
Keywords
Introduction
Chronic rhinosinusitis (CRS) is a chronic inflammatory disease of paranasal sinuses and nasal mucosa with an incidence of 2%–8% in China.1,2 According to the existence of nasal polyp under nasal endoscope, CRS can be classified into 2 clinical types: CRS with nasal polyp (CRSwNP) and CARS without nasal polyp (CRSsNP). The incidence of CRSwNP accounts for about 20%–33% of CRS. 3 CRSwNP can usually cause worse symptoms such as severe obstruction, olfactory disorders, and continuous headache, and the patients are more likely to relapse. 4 The most commonly used medications for CRSwNP are saline irrigation, topical or systemic corticosteroids, and antibiotics. If the symptoms do not improve after the administration of maximum medications, endoscopic sinus surgery (ESS) is applied for reestablishing nasal sinus mucosa ventilation and mucociliary function. 5 Nasal mucosa is prone to edema, scarring and crusting after ESS. 6 Effective perioperative anti-inflammatory treatment can promote the recovery of the surgical nasal cavity. 4
Corticosteroids (CSs) are widely used in CRSwNP based on their role in suppressing immune activity 7 and regulating nasal mucosal inflammation. 8 Perioperative CSs are associated with improvement in the surgical field, reduction in operative times, and improved postoperative subjective patient outcomes.9,10 CSs nasal sprays are the most commonly used perioperative anti-inflammatory drugs, but nasal sprays can hardly reach the mucosal lesions due to the obstruction of inferior turbinate, middle turbinate, nasal packing, secretion, and crusts on the surface of the surgical cavity. 11 Oral CSs (OCSs) can overcome this issue which has been proved to be effective in the postoperative management for CRSwNP by several studies with regards to mucosal healing. 12 The prolonged duration of OCSs may be beneficial for recovery of surgical mucosa, but the application and withdrawal of OCSs can cause several adverse events. Further research on the usage of postoperative OCSs is necessary to optimize efficacy and adherence while minimizing the potential harm to patients so as to avoiding overprescribing or under prescribing OCSs.
In this study, we compared 2 kinds of usage of OCSs: 0.4mg/kg/day for 10 days without a taper or 0.4mg/kg/day for 7 days followed with a 3-week taper. The purpose of this study is to investigate the difference in efficacy and risk of short-time adverse events between OCSs withdrawal directly and gradually after ESS and provide evidence-based knowledge for daily practice.
Patients and Methods
Patients and study design
This was a randomized prospective study conducted from Oct 2019 to Jan, 2021. Patients from 18–65 years old diagnosed with CRSwNP according to European position paper on rhinosinusitis and nasal polyps 2012 1 who had undergone unsuccessful maximal medical therapy were enrolled in this study and observation of nasal polyp through nasal endoscopy. All patients underwent ESS conducted by one experienced professor at Renmin Hospital of Wuhan University. The exclusion criteria were described as follows: CRSsNP, fungal sinusitis, immunodeficiency, liver or renal deficiency, pregnancy or lactation in women, uncontrolled diabetes, hypertension, gastrointestinal ulceration, the administration of systemic steroids within 3 months before ESS, and concomitant medication with drugs interacting with methylprednisolone. Baseline characteristics of subjects were collected before surgery, including age, sex, Lund-Mackay CT score, and medical history.
35 patients included in this study were randomized into 2 groups. One group (regular group, n=18) received 0.4mg/kg/day of methylprednisolone orally for 10 days. The other group (tapering group, n=17) received 0.4mg/kg/day of methylprednisolone orally for 7 days, followed by progressive reduction of 8 mg of methylprednisolone per week for 3 weeks. Methylprednisolone was taken once daily in the morning since the first day after ESS for both groups. Other medication such as intranasal corticosteroids sprays, antihistamines or antileukotrienes, and antibiotics were similar between the 2 groups. This study was approved by the Ethics Committee of Renmin Hospital of Wuhan University which was performed in accordance with the Helsinki Declaration.
Evaluations of efficacy
Subjective evaluation for the severity of nasal disease symptoms was assessed at preoperatively baseline and at half and 1, 2, and 12 months postoperatively using visual analogue scale (VAS) score. We selected 4 major complaints: nasal obstruction, rhinorrhea, headache, and smell, on the scale of 0–10 points according to symptom severity. For objective evaluation, we used Lund-Kennedy endoscopy (LKE) score to evaluate cavity appearance under the guidance of nasal endoscope preoperatively and at half and 1, 2, and 12 months postoperatively described as follows: for each side: the presence of polyp (0 = absence of polyp, 1 = confined to middle meatus, 2 = beyond middle meatus) and discharge (0 = no discharge, 1 = clear and thin discharge, 2 = clear and thick discharge or purulent and thin discharge, 3 = thick and purulent discharge) and edema, scarring, and crusting (for each: 0 = absent, 1 = mild, 2 = moderate, 3 = severe, scarring and crusting were only applied postoperatively). We used the LKE score as the primary outcome measurement and the 1-year LKE score to calculate the power of this study. E score was also used to evaluate mucosa condition (for each sinus: 0 = normal condition; 1 = sinus only partially observable due to occupation by polyp, and/or discharge; 2 = unobservable due to complete occupation by polyp edematous mucosa, and/or discharge) preoperatively and at half and 1, 2, and 12 months postoperatively. 13
Adverse events
We selected potential adverse events including sleep disorders, edema, digestive tract symptoms, rash/ acne, behavioral/ cognitive changes, weight gaining, pain of limbs, and risk of infection according to the list on the Food and Drug Administration mandated drug label for oral corticosteroids, and collected them in detail at half and 1, 2, and 12 months through questionnaires filled by patients in both groups.
Statistical analysis
Descriptive data are presented as percentages and means ± standard deviations. Comparisons of symptom scores and endoscopy scores between the 2 groups were obtained using T-test. The incidence and percentage of adverse events were compared using χ2 test. Differences were considered statistically significant if P-value was ≤0.05. Statistical analyses were performed using SPSS (version 23.0; IBM Corp., Armonk, NY, USA). Calculation of statistical power was assessed using G*Power (version 3.1; Germany).
Results
Patients baseline
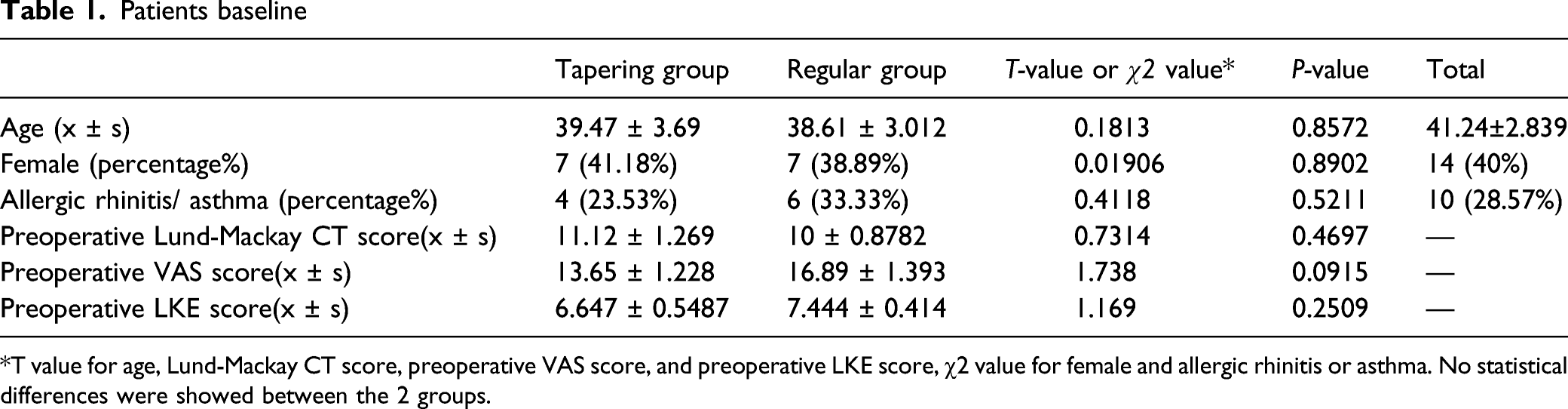
All patients completed the baseline investigation and consented to further follow-ups (Figure 1). Characteristics of all patients were shown in Table 1. The 2 groups were similar in terms of the proportion of female patients and patients who have asthma or allergies. There was no significant difference in preoperative VAS score, Lund-Mackay CT score, LKE score, and age between the 2 groups (all P-values <0.05). Study flow chart Patients baseline T value for age, Lund-Mackay CT score, preoperative VAS score, and preoperative LKE score, χ2 value for female and allergic rhinitis or asthma. No statistical differences were showed between the 2 groups.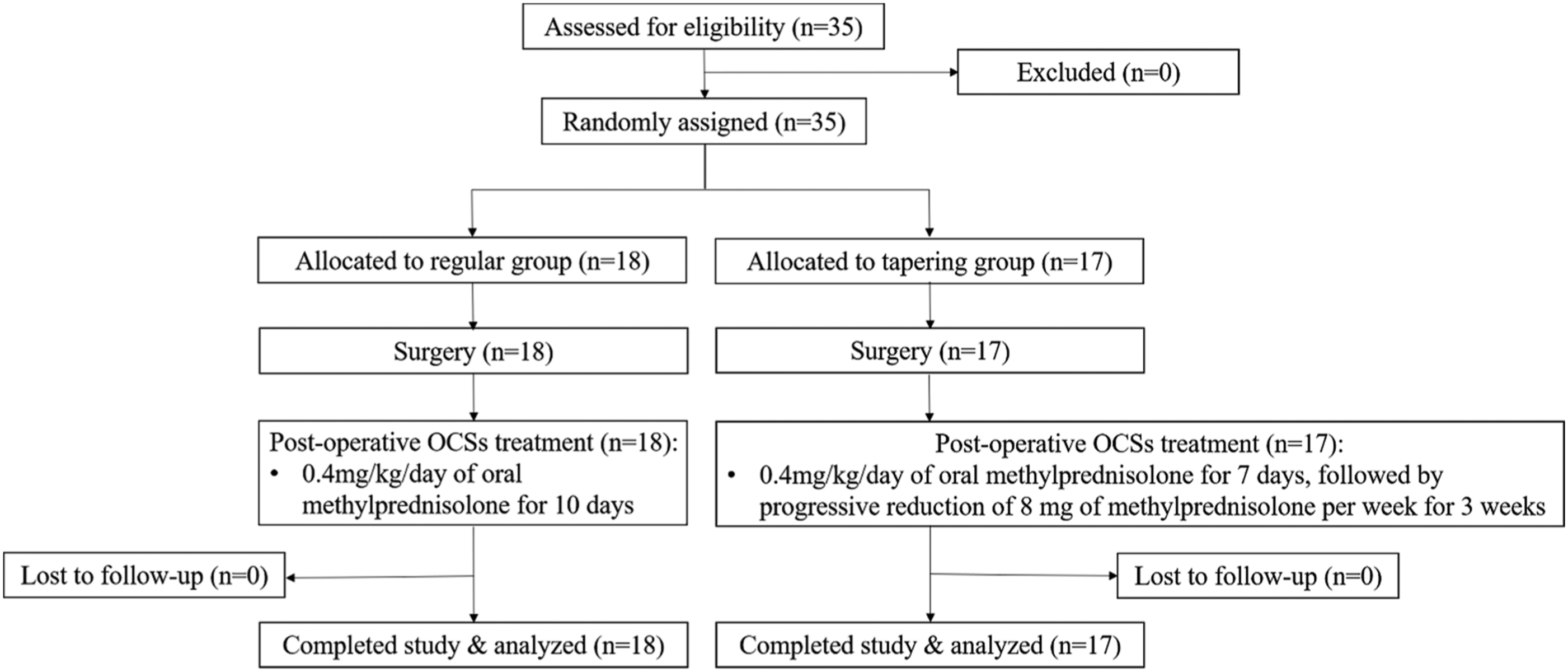
Clinical efficacy
Comparison of preoperative VAS scores and postoperative VAS scores at each time point
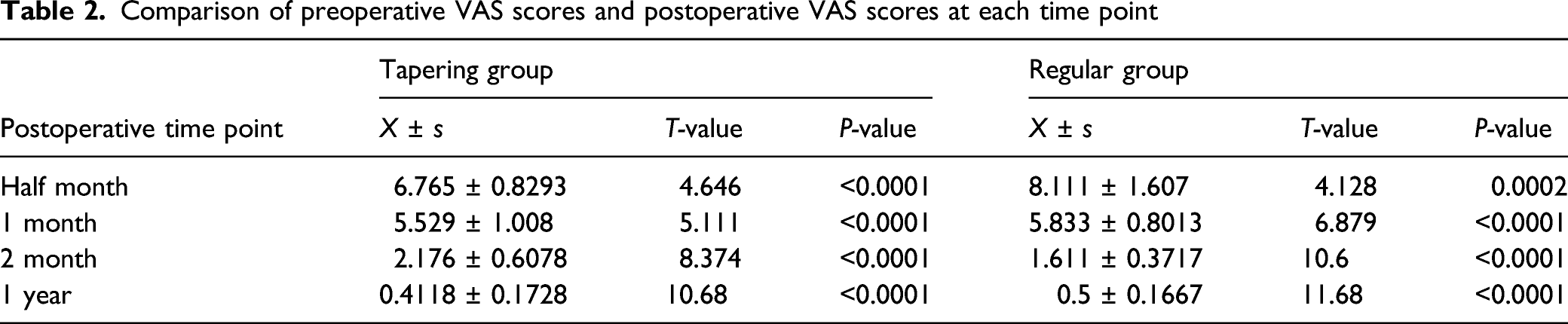
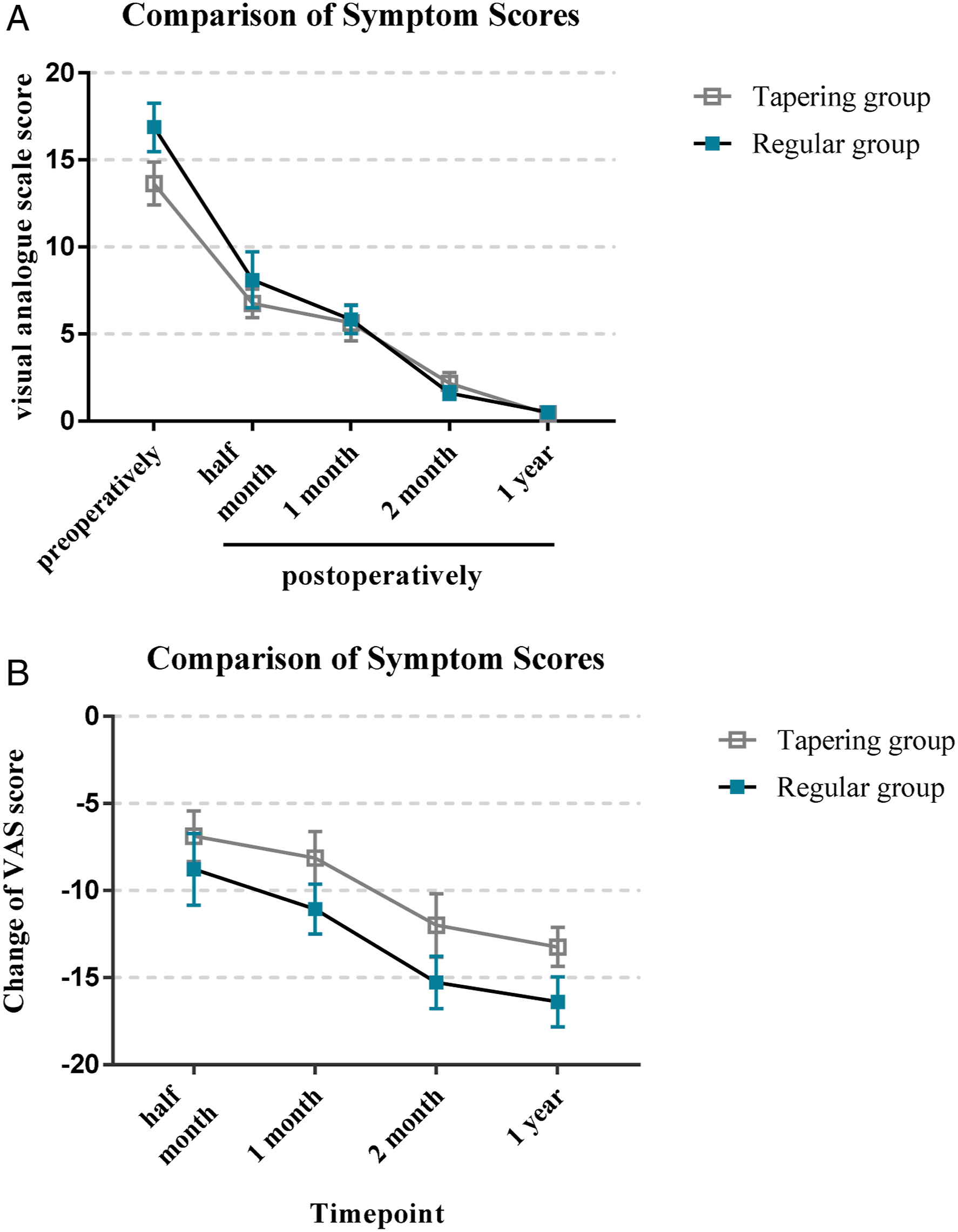
Comparison of symptom scores between the 2 groups before and after surgery. There was no significant difference between the 2 groups in VAS scores at each time point.
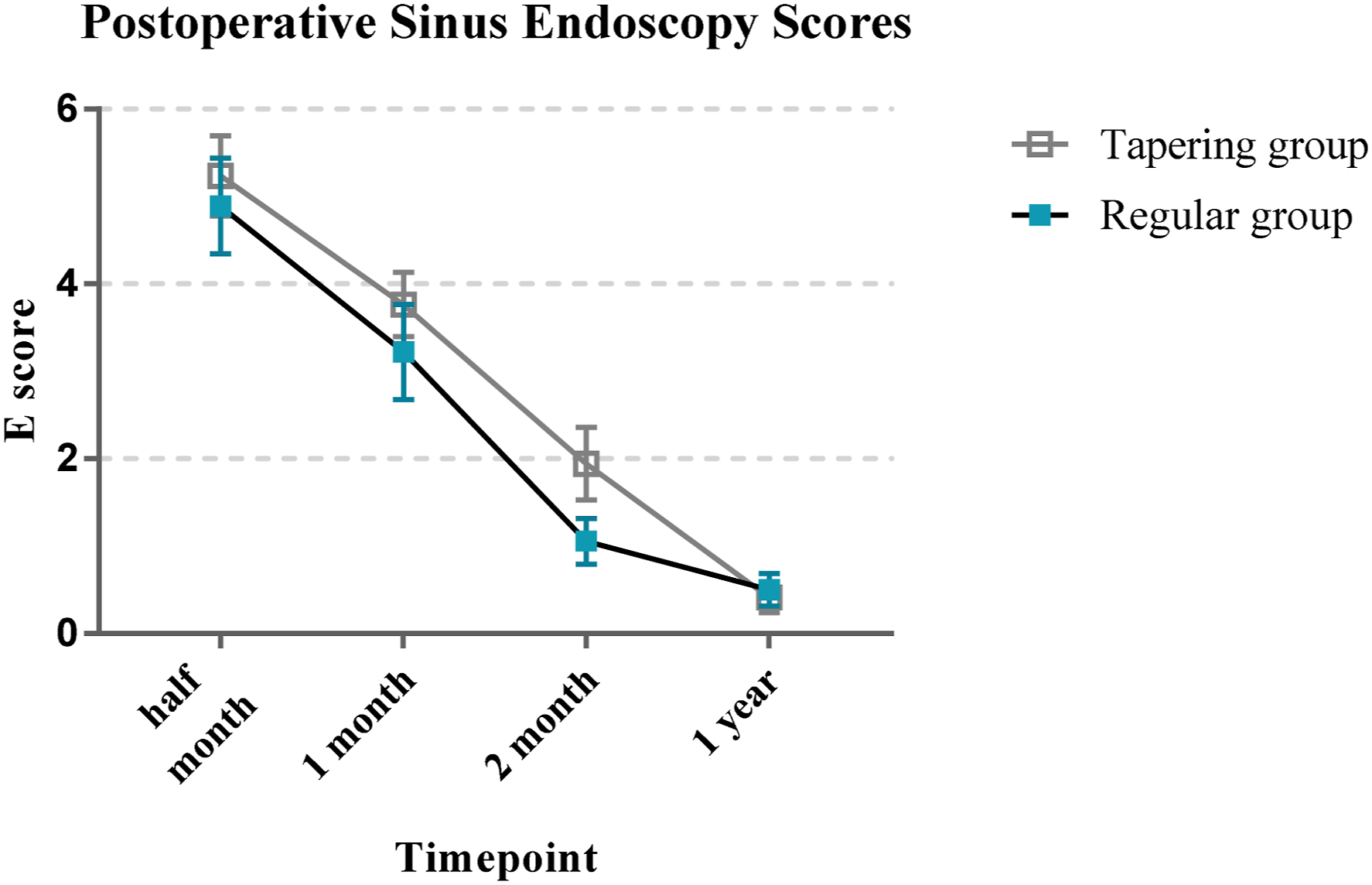
There was also no significant difference in the LKE score and E score between the 2 groups preoperatively and at half and 1, 2, and 12 months postoperatively (all P-values > 0.05, Figures 3-4). Thus, there was no significant difference in the postoperative recovering with the different drug withdrawal methods. The power of this study was 0.52. Comparison of Lund-Kennedy endoscopic score between the 2 groups of patients at each time point after surgery Comparison of E score between the 2 groups of patients at each time point after surgery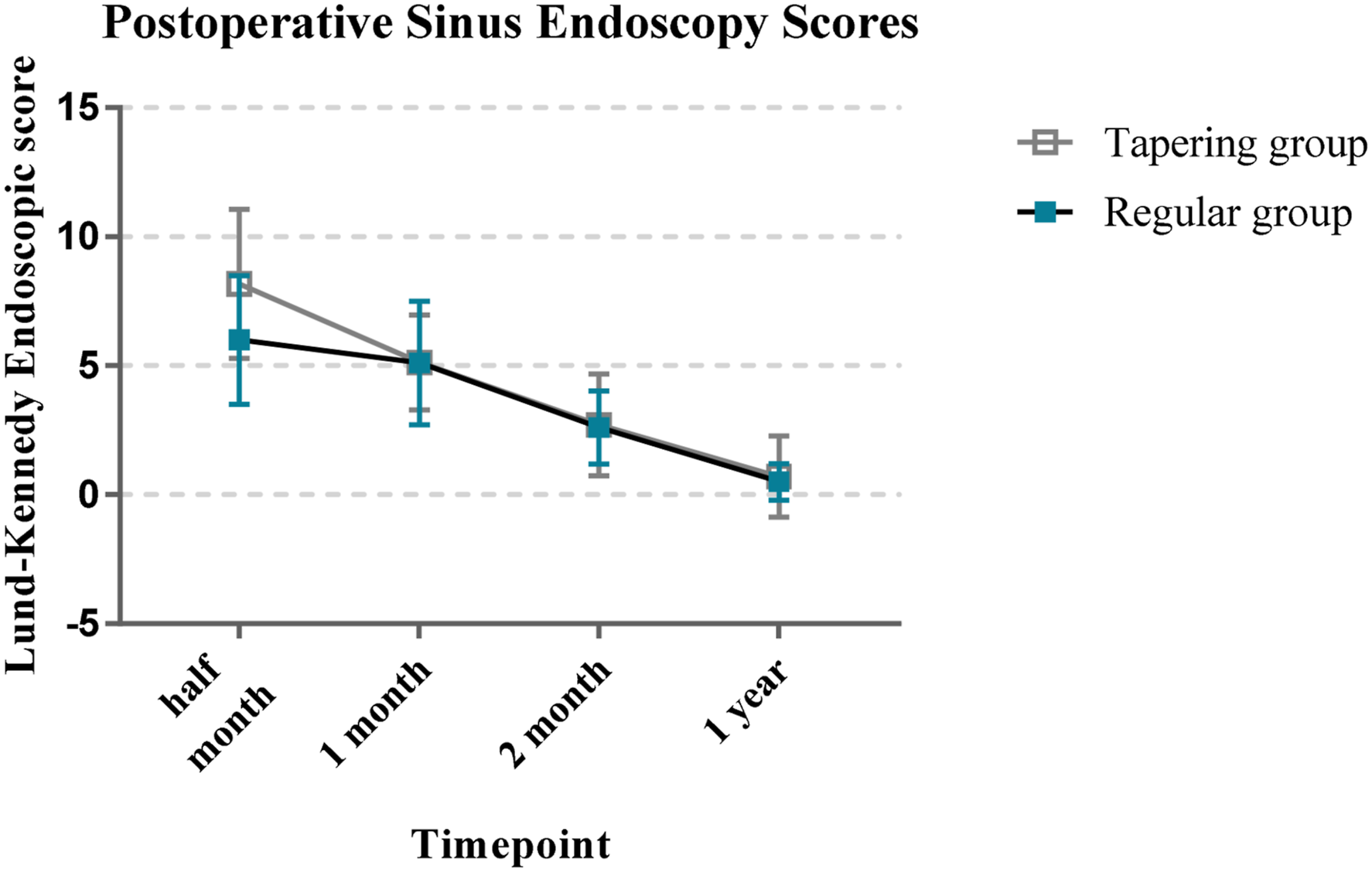
Adverse events
Comparison of each adverse event between the 2 groups
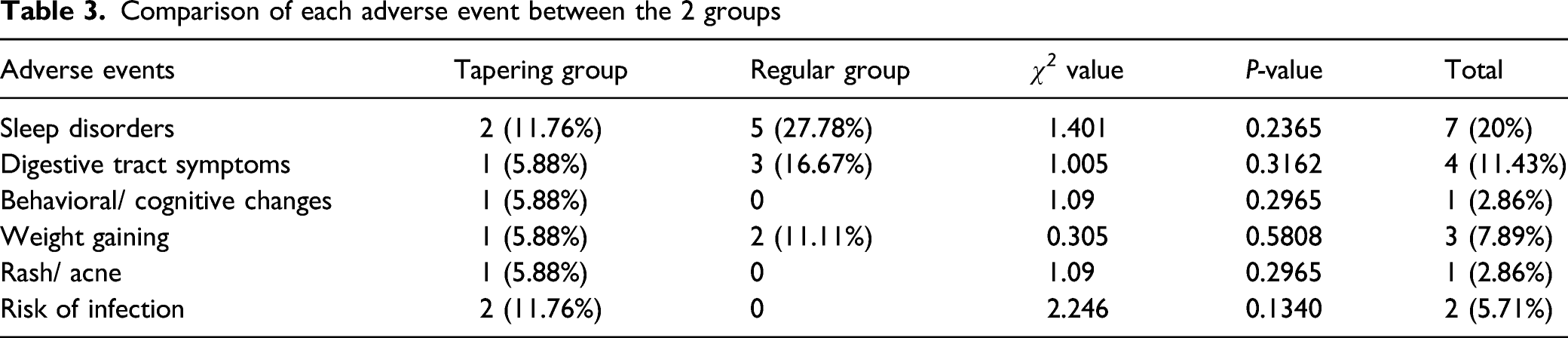
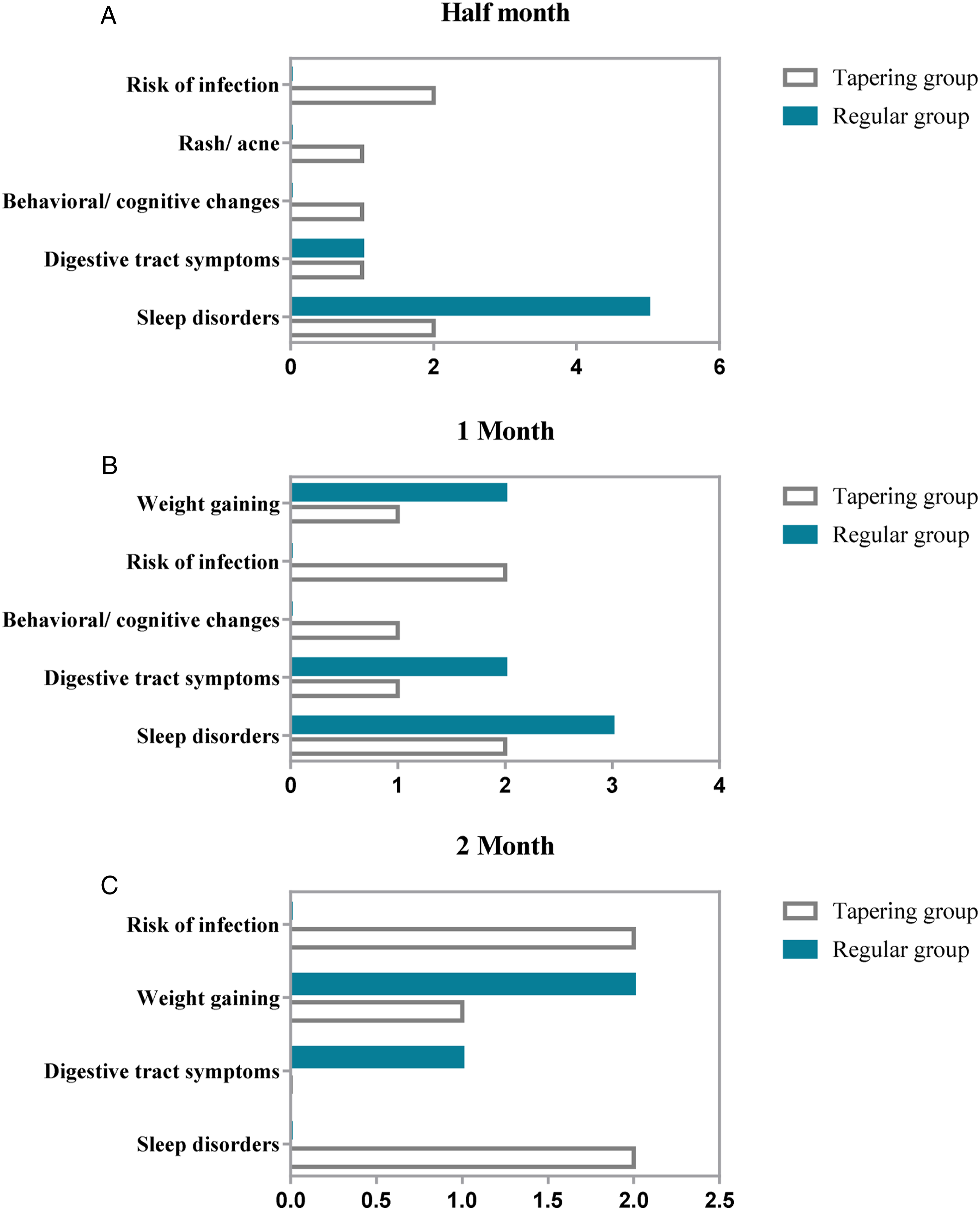
Comparison of adverse events between the 2 groups. There was no difference in each adverse event at each time point between the 2 groups.
Discussion
CSs are recommended to use as a combination with other drugs and surgery to treat CRSwNP within 2–3 weeks. 14 The European position paper on rhinosinusitis and nasal polyps 2020 pointed out that the short-term (7–21 days) use of OCSs was widely applied in CRSwNP, and its perioperative application can reduce intraoperative bleeding and improve surgical vision. 15 Vaidyanathan et al. gave CRSwNP patients 25 mg/d of prednisone orally for 14 days, Epperson et al. gave CRSwNP patients 50 mg/d of prednisone orally for 3 days followed by progressive reduction of 10 mg every 3 days, Martínez-Anton et al. gave CRSwNP patients 30 mg/d of prednisone orally for 4 days followed by progressive reduction of 5 mg every 2 days. All 3 researches showed that the symptoms were significantly improved after treatment, and the polyp size was significantly reduced.16-18 Jorissen et al. gave CRSwNP patients 2 mg/d betamethasone for 7 days after surgery, Shen et al. gave CRSwNP patients 30 mg/d prednisolone for 2 weeks after surgery, Wright et al. gave CRSwNP patients 30mg/d prednisolone for 5 days preoperatively and 9 days postoperatively. These researches showed that endoscopy scores and symptom scores of OCSs group were significantly improved after surgery, and the endoscopy scores of OCSs group improved more significantly compared with the placebo group.12,19,20 It was confirmed that OCSs can effectively improve the edema, scarring, and crusting of the surgical cavity after surgery and promote the recovery of nasal mucosa. However, there was no unified usage of short-term postoperative OCSs, and no comparation between different usages was reported. It was agreed that OCSs tapering should be attempted in all patients with asthma receiving OCSs maintenance treatment by customizing the speed of tapering 21 , but there was no clinical evidence that OCSs taper was beneficial in CRSwNP.
This study was the first to compare the efficacy of different OCSs usages after ESS in CRSwNP. In our study, patients received the same treatment except OCSs and there was no statistical difference in the baseline characteristics between the 2 groups. We selected 4 time points of half and 1, 2, and 12 months post-ESS to observe the short-term and long-term effects of OCSs on postoperative recovery. The results showed that the postoperative VAS score and endoscopy scores of the 2 groups were significantly improved compared with those before surgery, which confirmed the efficacy of the combination of surgery and postoperative short-term OCSs in CRSwNP. But there was no difference in symptom scores and endoscopy scores between the 2 groups at each time point after surgery, indicating that different drug withdrawal methods had similar short-term and long-term effects on the recovery of operative cavity and symptoms after surgery. Although the tapering group was treated for a longer time, there was no significant advantage in symptoms and mucosal recovery.
Although various studies have confirmed the efficacy of OCSs, they are still limited in clinical practice by their adverse events. The common short-term adverse events of OCSs include gastrointestinal reactions and insomnia. 22 Short-term (7–21 days) OCSs is safe in most cases, but ischemic necrosis and fatal varicella zoster have occasionally also been reported. 22 The occurrence of adverse events was positively correlated with the dose and time. 23 Dalal et al. conducted a statistical analysis of 12,697 asthma patients who took OCSs and found that the probability of adverse events increased with the increase of drug dose. 24 Long-term OCSs can lead to hypothalamus–pituitary–adrenal axis suppression, osteoporosis, immunosuppression, etc., 8 which are rarely seen in the short-term OCSs application. Studies have shown that there was no significant change in peripheral blood cortisol and osteocalcin within 1 week of medication in patients with short-term low-dose prednisolone (<10mg/d). 25 Therefore, this study focused on observing short-term adverse events and did not detect peripheral blood indexes, considering that short-term OCSs have little inhibitory effect on the hypothalamus–pituitary–adrenal axis. Suehs et al. reported that gradually reducing the dosage of CSs in asthma can reduce the probability of adverse events. 26 In our study, there were no differences in each short-term adverse event between the 2 groups.
However, there are some limitations in this study. The statistical power of our current study is relatively low (0.52) which may be due to the small sample size, so the number of patients needs to be increased in futher study. Also the follow-up should be carried out at multiple time points after surgery in further study. In addition, corticosteroids levels were not monitored in this study considering that short-term OCSs have little inhibitory effect on the hypothalamus–pituitary–adrenal axis, but it is still necessary to be evaluated to assess adverse events more comprehensively and accurately. Research into the optimal OCSs dose and duration will aid physicians in optimizing efficacy and adherence to therapy while minimizing potential harm to patients from over or under prescribing OCSs.
Conclusion
This study was the first to compare the differences in the improvement of symptoms, the mucosal recovery, and the occurrence of adverse events in CRSwNP patients after ESS with different withdrawal methods of OCSs. The results showed that there was no significant difference in the efficacy and complications of the 2 drug withdrawal methods. Compared with direct drug withdrawal, gradual drug withdrawal is more complicated than direct drug withdrawal, which may lead to poor compliance. Missed medication and misuse may occur in elderly patients, children, and cognitive impaired patients. Therefore, we recommended direct drug withdrawal as the first choice in clinical practice.
Footnotes
Acknowledgments
We thank the teachers of Renmin Hospital of Wuhan University for their help during this research.
Authorship contributions
LSF and RX performed the experiments, analyzed the data, and wrote the manuscript. WZ and HT helped in collecting data. ZZT contributed to design of study and manuscript preparation. YX designed and supervised the study and critically reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the National Natural Science Foundation of China (No. 81770986 and 82071017) and the Fundamental Research Funds for the Central Universities (No. 413000379).
Ethics approval
Ethical approval to report this case was obtained from the Ethics Committee of Renmin Hospital of Wuhan University.
Statement of Human and Animal Rights
All procedures in this study involved in human were conducted in accordance with the Ethics Committee of Renmin Hospital of Wuhan University’s approved protocol (WDRY2021-K084).
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
