Abstract
Sclerosing polycystic adenoma (SPA) is a rare salivary gland tumor with about 100 cases reported in the literature. We describe a case of SPA in the parotid gland and review the diagnostic tools used for identifying SPA. A 24-year-old male with a two-year history of right-sided face mass, initially thought to be pleomorphic adenoma of the parotid gland after fine needle aspiration (FNA). Following superficial parotidectomy, histologic features were consistent with SPA. This case illustrates the challenge of pre-operative assessment for SPA. Recent study has suggested SPA is a neoplasm and definitive treatment is surgical excision.
Introduction
Sclerosing polycystic adenoma (SPA) is a rare salivary gland tumor first reported in a 1996 case series 1 and was included in the 4th edition WHO Classification of Head and Neck Tumours. 2 To date, approximately 100 cases have been reported. Sclerosing polycystic adenoma exclusively affects head and neck glandular tissue, most often unilateral parotid glands, followed by submandibular glands. Sclerosing polycystic adenoma has a nearly equal sex distribution (M:F 24:25) and affects all age groups (range 7–84). In this report, we describe a case of SPA in the parotid gland and review the literature with a focus on pertinent diagnostic tools, the classification of SPA as a neoplasm, and its management.
Case
A 24-year-old male presented with a complaint of a mass on the right side of his face. The patient noticed the mass about two years prior. He was not sure if it was growing. He had discomfort when sleeping on the right side, but no other complaints. Examination revealed a hard but mobile parotid mass. Facial nerve examination was normal. Computed tomography scan was obtained and interpreted as “3.3 cm partially necrotic parotid tumor” (Figure 1a). Fine needle aspiration (FNA) was performed and reported as “low-grade neoplasm, consistent with pleomorphic adenoma”. The patient underwent superficial parotidectomy. (a) Axial CT showing right parotid mass, (b) H&E stained specimen (100x) showing irregular proliferation of ducts, (c) H&E stained specimen (400x) with acini displaying enlarged eosinophilic granules, and (d) calponin-stained specimen (100x) showing intact layers of myoepithelial cells.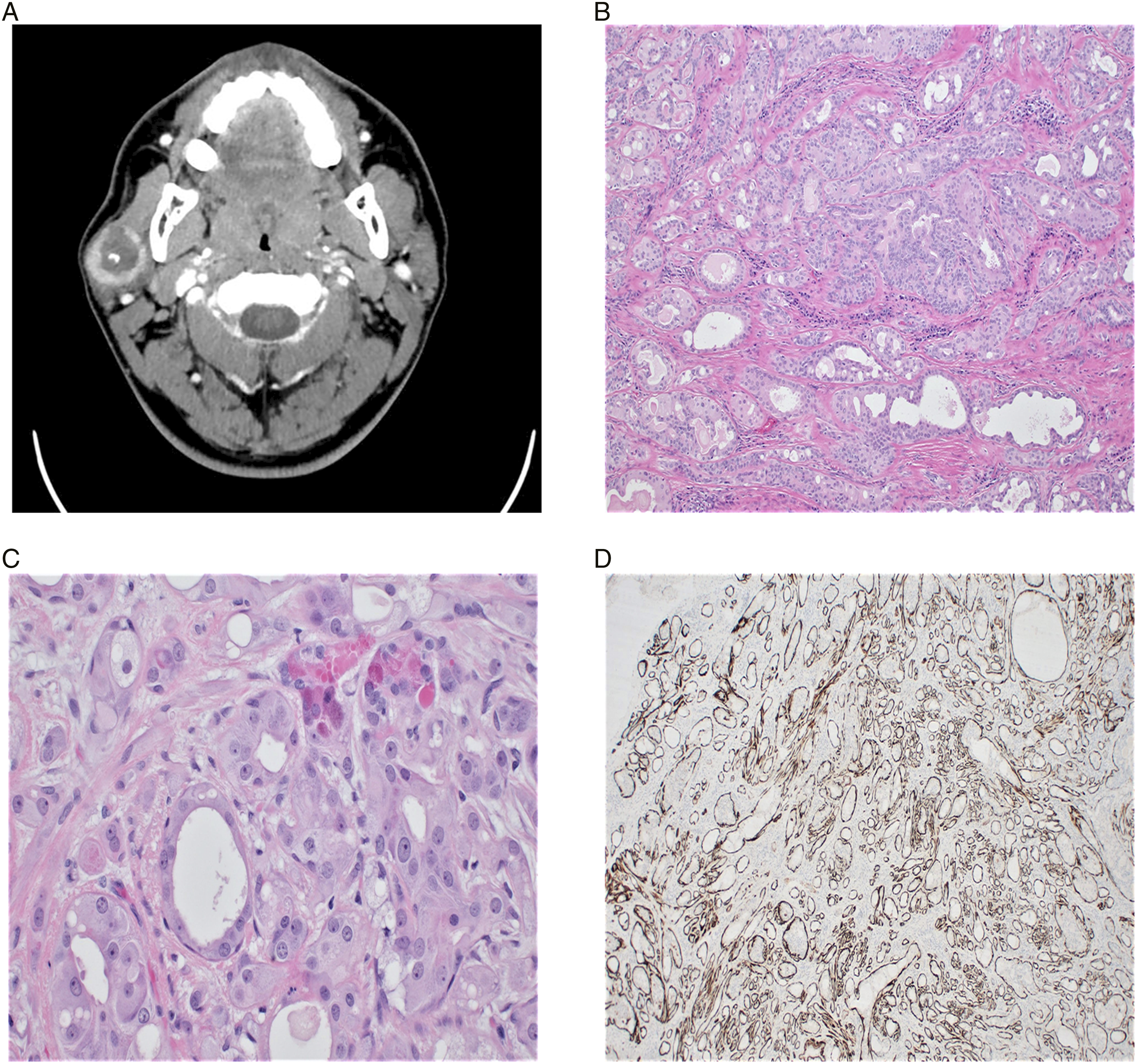
Histology revealed a well-circumscribed lesion (Figure 1b) with irregular ductal and acinar proliferation and associated myoepithelial cells within sclerotic stroma. Ducts showed cystic dilation and apocrine change. Acini contained focal cytoplasmic eosinophilic granules (Figure 1c). The ductal and acinar cells stained positive for anti-cytokeratin (CAM5.2) and epithelial membrane antigen (EMA), with a perimeter of p63 and calponin-positive myoepithelial cells (Figure 1d). Based on these histologic features, a diagnosis of SPA was made.
Discussion
Pre-operative assessment of SPA is challenging. No specific cytopathologic profile has been characterized in SPA. 3 Fine needle aspirations were most commonly misdiagnosed as pleomorphic adenoma or Warthin’s tumor.4-6 This may reflect the relative frequency of these lesions in the parotid, rather than cytopathologic overlap with SPA. Sclerosing polycystic adenoma was misdiagnosed as malignant in several cases.7-10
Histopathologic assessment remains the definitive method of diagnosis. Despite SPA’s rarity, microscopic findings are well described and characteristic. Sclerosing polycystic adenoma is well-circumscribed or encapsulated and shows glandular proliferation and sclerotic stroma. A notable feature is the eosinophilic, even red, cytoplasmic granules seen within acinar cells. Bishop and Thompson point out areas of severe sclerosis can appear invasive; however, an intact layer of compressed myoepithelial cells can be appreciated with careful inspection; thus, confirming SPA is intraductal. 11 These features are similar to sclerosing adenosis of the breast, though SPA lacks the associated malignancy risk.12,13
Sclerosing polycystic adenoma was initially believed to be a reactive, inflammatory process, and, in our review, chronic inflammation or trauma was a recurrent theme, including prior surgery in the area, 14 significant smoking history, 15 and recent radiation treatment. 16 A connection between SPA and EBV infection has also been suggested.17,18
While chronic inflammation and trauma may play a role in its development, recent study suggests SPA is a true neoplasm.19,20 A study of X-inactivation in 6 cases of SPA showed monoclonality at the HUMARA (human androgen receptor) locus. 4 Next-generation sequencing has identified SPA mutations frequently implicated in neoplastic processes, notably loss of function PTEN mutations, 19 leading Bishop and Thompson 11 to conclude that SPA is indeed a neoplasm and should be more appropriately termed SPA rather than adenosis.
Surgical resection is the definitive management for SPA. Previous literature reported recurrence rates as high as 30%; however, this literature review found only 7% of cases were recurrent.1,8,12,20,21 Management with superficial vs total parotidectomy has similar recurrence rates (∼10%) while an excision/enucleation carries about 4 times the risk of recurrence. 22 At least one case of malignant transformation has been described in the case of relapsing disease, which was eventually definitively treated with total parotidectomy. 12
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
