Abstract
Purpose
Soluble vascular adhesion protein-1 (sVAP-1) and adropin are 2 biomarkers of endothelial dysfunction. The main purpose of this study was to evaluate the levels of sVAP-1 in patients with moderate and severe obstructive sleep apnea (OSA) compared to healthy controls, and to further determine the relationship between sVAP-1 and adropin levels, as well as inflammatory biomarkers and sleep parameters.
Methods
In this study, we included 50 male patients with OSA (25 moderate and 25 severe) and 20 age- and sex-matched control subjects. Patients with OSA underwent polysomnography. All subjects underwent fasting peripheral blood sampling for laboratory analysis.
Results
Serum sVAP-1 and inflammatory biomarkers (IL-6, TNF-α, and hsCRP) levels were significantly higher in patients with severe OSA compared to those with moderate OSA and control groups, while plasma levels of adropin showed the opposite trend. Furthermore, sVAP-1 levels had a significant positive correlation with AHI, ODI, TNF-α, IL-6, and hsCRP levels and a significant negative correlation with adropin levels. The receiver operating characteristic analysis showed an Area Under Curve (AUC) of .876 (P < .001) for sVAP-1 levels predicting OSA. Serum sVAP-1 threshold of > 445.5 ng/mL had an 88% sensitivity and 80% specificity for detecting OSA status. Multivariate regression analysis demonstrated that sVAP-1 remained a significant positive predictor of OSA severity.
Conclusions
Increased sVAP-1 levels in OSA patients are significantly correlated with indices of OSA severity, adropin levels, and inflammatory biomarkers, suggesting that sVAP-1 plays a vital role in the pathophysiology of OSA and may become a potential screening tool in the evaluation of OSA severity.
Introduction
Obstructive sleep apnea (OSA) is characterized by partial or complete obstruction of the upper airway during sleep, leading to abnormal ventilation, hypoxemia, and sleep fragmentation. 1 In recent years, more and more evidence has supported a causal link between OSA and cardiovascular disease (CVD).2,3 The pathogenesis of CVD caused by OSA remains largely unknown. One such mechanism refers to vascular endothelial damage, which might be the earliest vascular pathological change in OSA. Vascular endothelial dysfunction (ED) has attracted increasing attention in the field of cardiovascular complications associated with OSA.4,5
Vascular adhesion protein-1 (VAP-1) is a membrane-bound adhesion molecule that is inactive under normal physiological conditions due to being sequestered in the endosome. However, it can rapidly translocate to the cell surface in response to local inflammation, thereby promoting lymphocyte adhesion and migration through the endothelium into inflamed tissue. 6 Unlike other adhesion molecules, VAP-1 can be cleaved by matrix metalloproteases, generating a soluble form of VAP-1 (sVAP-1). sVAP-1 has enzymatic capacity as it can catalyze oxidative deamination of primary amines to aldehydes, thereby releasing hydrogen peroxide and ammonium. This, in turn, amplifies the inflammatory reaction, thus causing damage to the vascular endothelium and leading to arteriosclerosis. 7 Elevation in sVAP-1 levels is observed in many diseases, such as atherosclerosis, diabetes mellitus (DM), obesity, and CVD, and has been widely proposed as an important biomarker of endothelial activity.8-10 Since chronic low-grade inflammation, oxidative stress, and ED are main pathophysiological mechanisms in OSA,4,5 sVAP-1 levels may be increased during OSA development. Therefore, it may become a useful biomarker for evaluating the severity of OSA. To the best of our knowledge, there is no literature regarding the relationship between serum sVAP-1 production and OSA.
Adropin is a novel peptide hormone that is supposed to play a protective role in endothelial function by increasing endothelial nitric oxide (NO) bioavailability through upregulation of endothelial NO synthase (eNOS) expression. 11 Several studies have reported a significant association between adropin and endothelial function in various diseases, including CVD, DM, and OSA. Thus, its ability to regulate endothelial function has been widely studied.12-15 A recent study showed a significant negative correlation between plasma adropin levels and biomarkers of systemic inflammation in patients with OSA. 16 As a result, we suggest that there may be a close association between adropin and sVAP-1.
Therefore, the main objectives of the present study were to determine serum sVAP-1 and adropin levels in a cohort of adult males with moderate and severe OSA compared to controls and to assess the relationship between sVAP-1 and adropin, along with sleep parameters and biomarkers of systemic inflammation.
Methods
Subjects
The present study included male subjects with newly diagnosed OSA who were enrolled at the ENT department of Putuo Hospital, Shanghai University of Traditional Chinese Medicine, between June 2019 and December 2020. OSA diagnosis was based on the polysomnography (PSG) results. The apnea-hypopnea index (AHI) is defined as the number of apnea and hypopnea events that occur per 1 hour of sleep. 17 Therefore, the study participants were classified into 2 groups according to OSA severity as follows: moderate OSA group (AHI: 15–30 events/h, 25 patients) and severe OSA group (AHI: >30 events/h, 25 patients). Exclusion criteria were as follows: (i) patients with central sleep apnea syndrome, DM, severe cardiac disease, and hypertension; known history of severe chronic pulmonary, renal, liver, thyroid, rheumatic, and hematological disorders and active malignant diseases; (ii) diagnosed history of OSA and patients who received medications before the study enrollment; and (iii) female patients.
Non-OSA individuals were enrolled at the physical examination center at Putuo Hospital as controls. The control group consisted of 20 healthy male volunteers matched to those enrolled for OSA based on age and body mass index (BMI). The Snoring, Tiredness, Observed apnea, and high blood Pressure (STOP) questionnaire was used as a screening tool to identify subjects at high risk of developing OSA. Subjects scoring ≥2 points on this questionnaire were excluded from the study. 18 The control group did not undergo a PSG assessment and was subjected to the same exclusion criteria and assessment procedure as the group of patients with moderate and severe OSA.
Anthropometric Measurements
After a detailed medical interview and physical examination, anthropometric measurements were acquired for all subjects included in the study. All subjects wore light clothing when their weight and height were recorded. BMI was calculated as body weight (kg) divided by height squared (m2). Neck circumference was measured at the laryngeal prominence and waist circumference was measured using measuring tape at the midpoint between the 12th rib and the iliac crest. All subjects’ blood pressure was taken at least twice between 8:00 and 9:00 am in a seated position on the right arm.
Polysomnography
All subjects underwent PSG (Polysomnography system, Monet 24, Rembrandt Embla, Reykjavik, Iceland) between 22:00 and 07:00. The monitoring results were automatically recorded using the Alice software and then analyzed by a specialist. Apnea was defined as the absence of ≥90% of airflow for the duration of ≥10 s compared to the baseline level. Hypopnea was defined as a reduction in airflow of ≥30% and lasting for ≥10 s, along with a ≥4% decrease in oxyhemoglobin saturation. AHI was calculated as the total number of apnea and hypopnea events per hour of sleep time. The OSA classifications were as follows: fewer than 5 events per hour, non-OSA; between five and 15 events per hour, mild OSA; between 15 and 30 events per hour, moderate OSA; and more than 30 events per hour, severe OSA.
Blood Sampling and Laboratory Analysis
Fasting blood samples were collected from all subjects, centrifuged immediately, and stored at −80°C until further analysis. All blood samples were analyzed in the same biochemical laboratory and by the same medical biochemistry specialist following standard laboratory procedures. Quantitative evaluation of biomarkers analyzed in the present study was performed with commercially available kits using enzyme-linked immunosorbent assay (ELISA) methods (Human sVAP-1 ELISA kit, Biovendor; hsCRP/IL-6/TNF-α ELISA kit, abcam; Human adropin ELISA kit, Novus.)
Statistical Analysis
All data analyses were performed using SPSS 20.0 Statistics system. Data were expressed as mean±SD. Normality of data distribution was measured using the Kolmogorov–Smirnov test. Significant differences between groups were analyzed using either unpaired Student t tests or ANOVA with post-hoc Tukey Honestly Significant Difference test for continuous variables. Pearson’s correlation analyses were performed to assess the association between sVAP-1 levels and anthropometric measurements, PSG variables, adropin, and inflammatory biomarkers (hsCRP, IL-6, and TNF-α). Multivariate regression analysis was used to determine the independent predictors among the selected variables (serum levels of sVAP-1 and other biomarkers). The regression model was adjusted for age and BMI and inspected for goodness of fit by the Hosmer–Leme show test. In addition, receiver operator curves (ROCs) were calculated to predict the OSA status based on sVAP-1 levels. The statistical significance for all reported data was two-tailed, where P < .05.
Results
Patients’ baseline Characteristics
Patients’ Baseline Characteristics and Plasma Inflammatory Biomarkers.
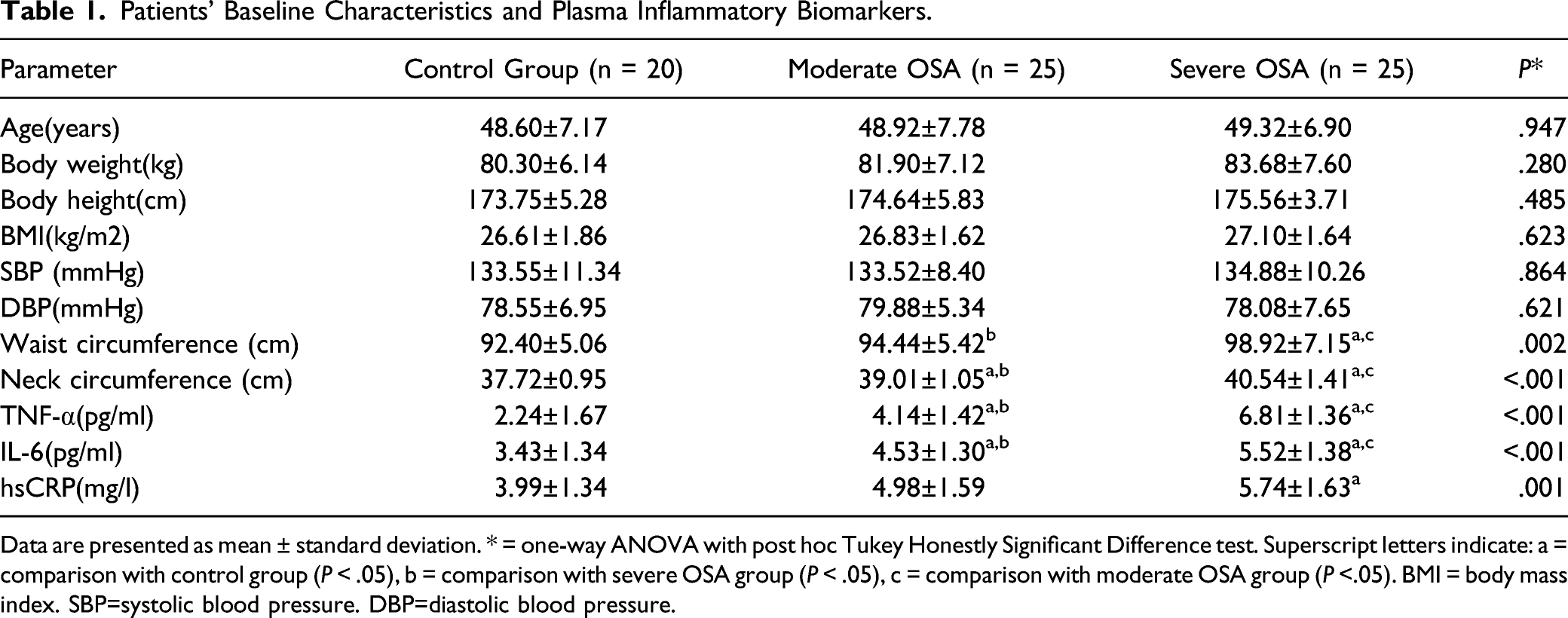
Data are presented as mean ± standard deviation. * = one-way ANOVA with post hoc Tukey Honestly Significant Difference test. Superscript letters indicate: a = comparison with control group (P < .05), b = comparison with severe OSA group (P < .05), c = comparison with moderate OSA group (P <.05). BMI = body mass index. SBP=systolic blood pressure. DBP=diastolic blood pressure.
PSG Data of Moderate and Severe OSA Patients.

Data are presented as mean ± standard deviation. * = Student t test for independent samples. AHI = apnea-hypopnea index. SpO2 = arterial oxygen saturation. ODI = oxygen desaturation index. PSG = polysomnography. TST = total sleep time.
Inflammatory Biomarkers
Inflammatory biomarker levels were significantly higher in patients with severe OSA compared to patients with moderate OSA and controls: IL-6 (5.52 ± 1.38 vs 4.53 ± 1.30 vs 3.43 ± 1.34 pg/mL, P < .001) and TNF-α (6.81 ± 1.36 vs 4.14 ± 1.42 vs 2.24 ± 1.67 pg/mL, P < .001). Plasma hsCRP levels were significantly higher in patients with severe OSA compared to the controls (5.74 ± 1.63 vs 3.99 ± 1.34 mg/L, P < .05). However, there were no significant differences between the other groups (Table 1).
sVAP-1 and Adropin Levels in Patients With OSA and Control Subjects
Serum levels of sVAP-1 were significantly higher in patients with severe OSA compared to those with moderate OSA and controls (816.28 ± 178.58 vs 559.96 ± 184.54 vs 344.45 ± 180.99 ng/mL, P < .001; Figure 1(a)). Plasma adropin levels demonstrated the opposite trend, where patients with severe OSA presented with significantly lower adropin levels compared to those with moderate OSA and controls (3.81 ± 1.12 vs 5.97 ± 1.42 vs 8.27 ± 1.30 ng/mL, P < .001; Figure 1(b)). Averaged biomarkers’ levels between OSA groups and controls(n = 50) (A)sVAP-1, (B) adropin. Data are tested with ANOVA with post-hoc Tukey Honestly Significant Difference test and presented as mean ± standard deviation. * = P < .05 between patients with moderate OSA and controls. † = P < .05 between patients with severe OSA and controls. ‡ = P < .05 between patients with moderate and severe OSA.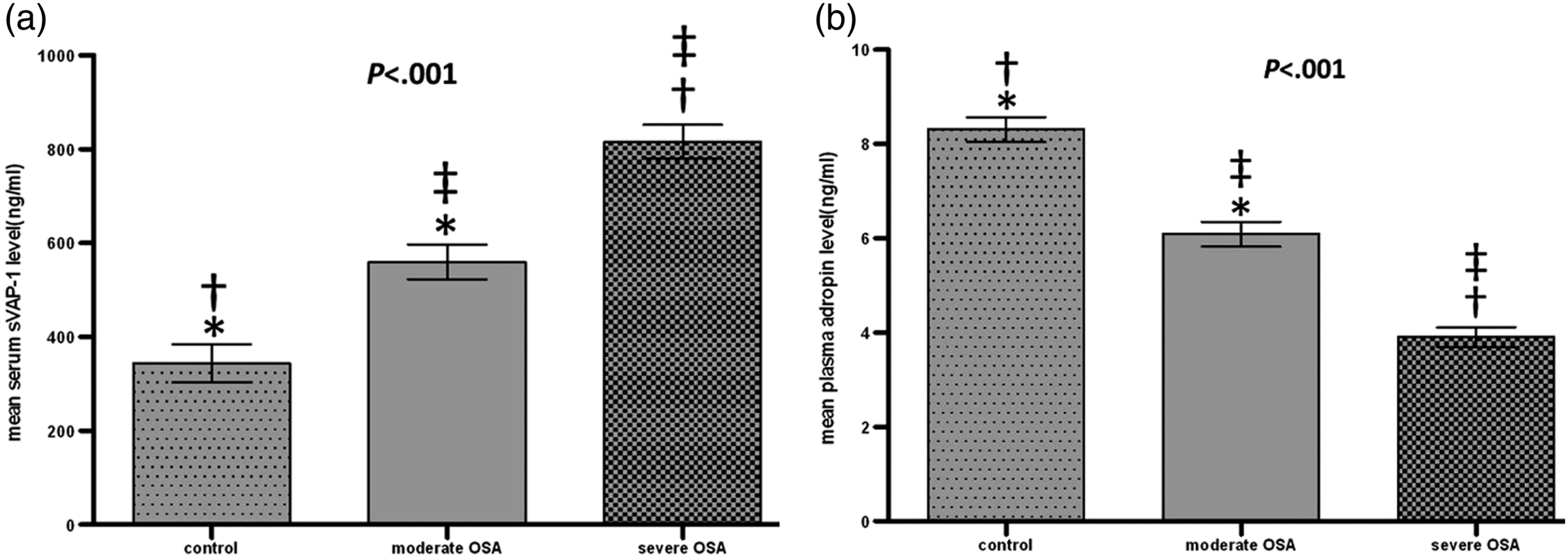
Pearson correlation analysis was performed on 50 patients with OSA to examine the relationship between sVAP-1 levels and OSA severity. Results showed that an increase in AHI was independently associated with an increase in serum sVAP-1 levels (r = .693, P < .001; Figure 2(a)). Similar results were obtained when AHI was replaced by ODI (r = .493, P < .001; Table 3). For the inflammatory biomarkers, serum sVAP-1 levels were significantly positively correlated with plasma levels of TNF-α (r = .753, P < .001), IL-6 (r = .730, P = .013), and hsCRP (r = .476, P < .001; Table 3) and negatively correlated with adropin (r = −.633, P < .001; Figure 2(b)). Adropin was significantly negatively correlated with AHI (r = −.721, P < .001), ODI (r = −.630, P < .001), TNF-α (r = −.759, P < .001), IL-6 (r = −.408, P = .002), and hsCRP (r = −.335, P = .009; Table 3). Correlations in composite of moderate and severe OSA patient groups (n = 50) Correlations between serum sVAP-1 levels and (A) apnea-hypopnea index, (B) adropin in composite of moderate and severe OSA patient groups. Red lines represent Pearson correlation coefficient and green lines represent respective 95% confidence intervals. Correlations Between sVAP-1, Adropin Levels, Inflammatory Biomarkers and Anthropometric Parameters in Patients With Moderate and Severe OSA. * = all P values are 2-tailed, significance level is set at P < .05, r = Pearson correlation coefficient, AHI = apnea-hypopnea index, ODI = oxygen desaturation index, BMI = body mass index, hsCRP = high-sensitivity C-reactive protein, IL-6 =interleukin 6, TNF-α = tumor necrosis factor alpha,sVAP-1= circulating vascular adhesion protein-1.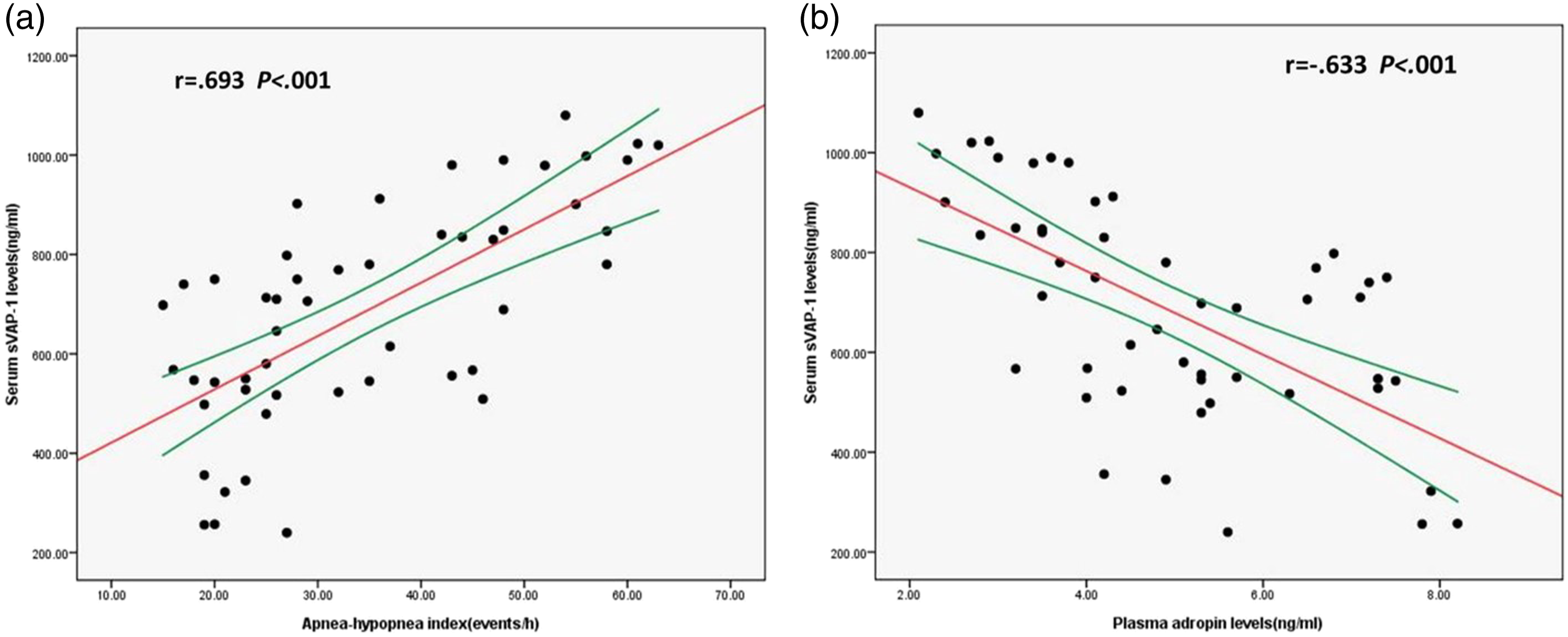
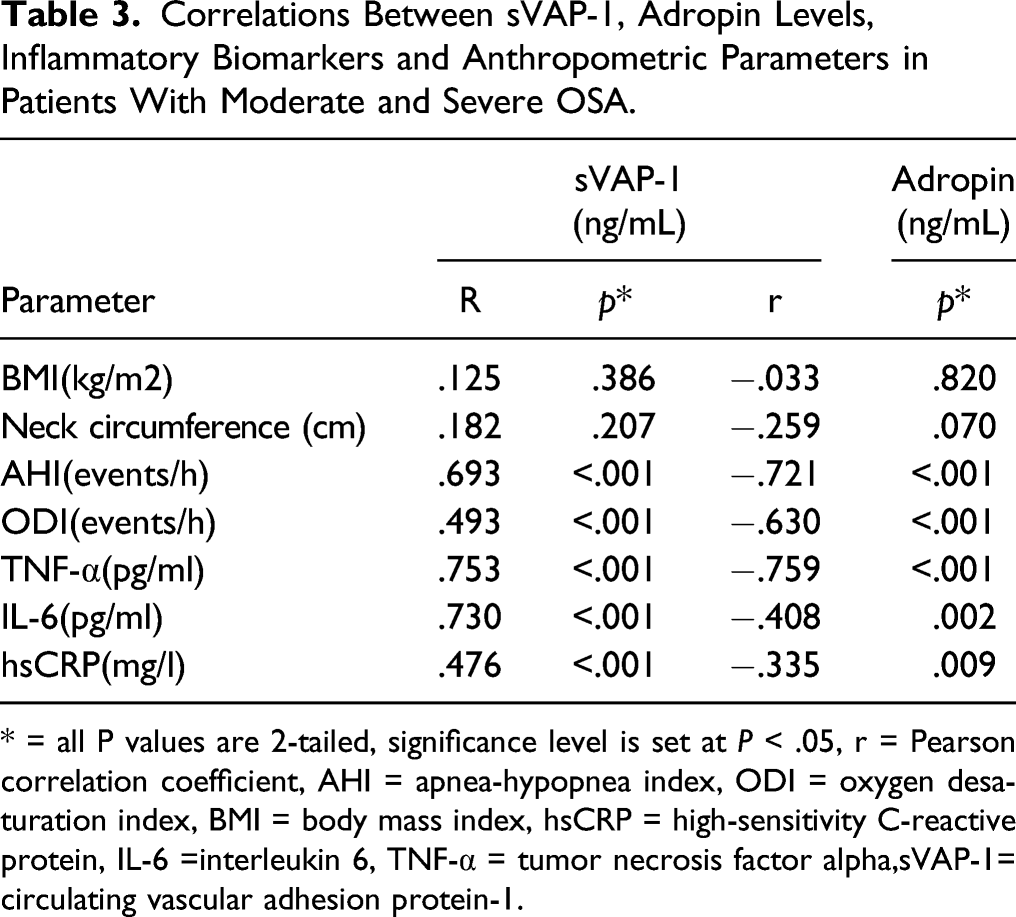
ROC analysis (Figure 3) showed that AUC for serum sVAP-1 levels excluding OSA status was .876 (P < .001, standard error .047, 95% Confidence interval (CI) .784−.968). Serum sVAP-1 cutoff value above 445.5 ng/mL provided 88% sensitivity and 80% specificity for the detection of OSA-positive status. Receiver operating characteristic analysis of sVAP-1 cutoff value in detection of OSA status.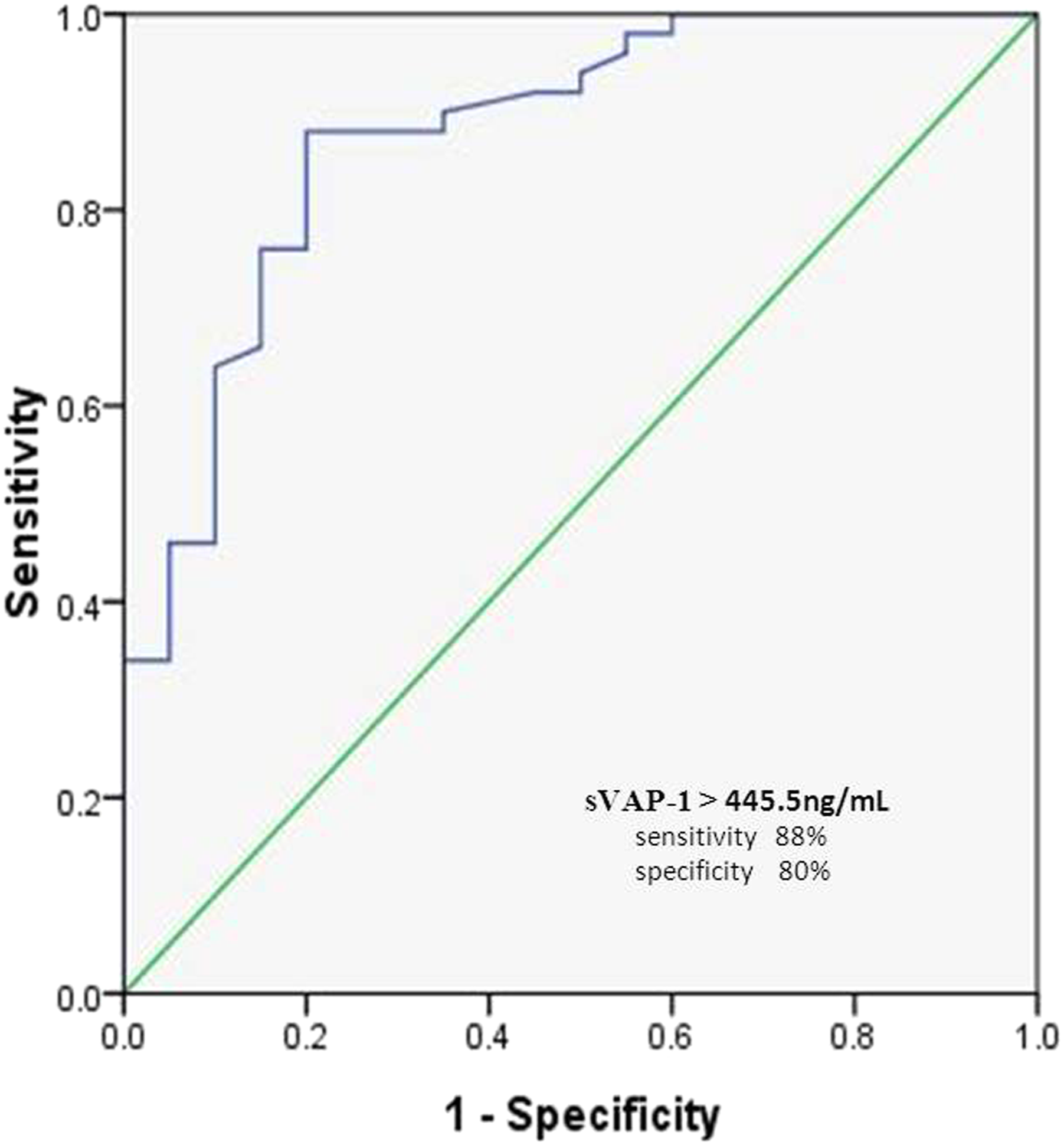
Multivariate Regression Analysis of Biomarkers as Independent Predictors for Moderate and Severe OSA.
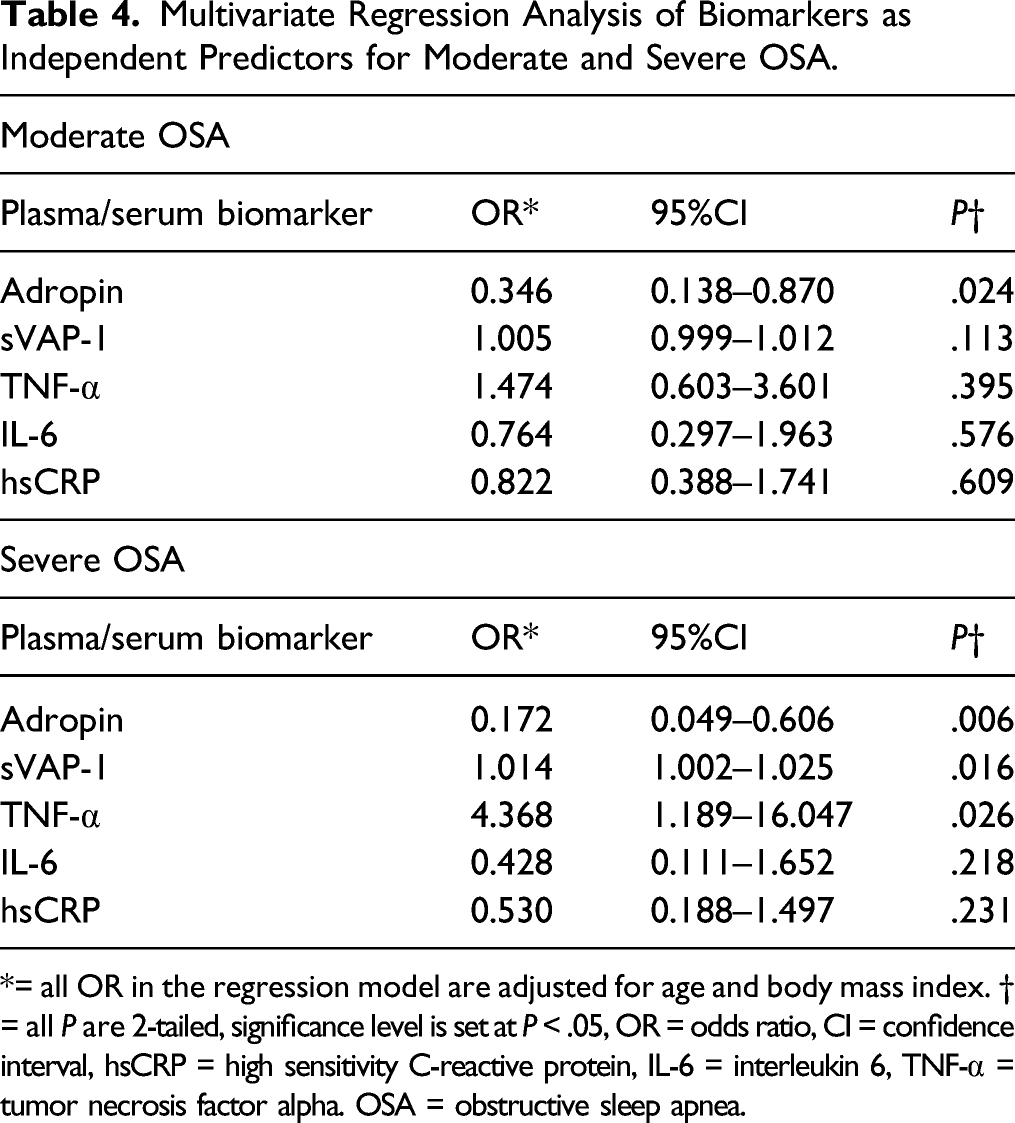
*= all OR in the regression model are adjusted for age and body mass index. † = all P are 2-tailed, significance level is set at P < .05, OR = odds ratio, CI = confidence interval, hsCRP = high sensitivity C-reactive protein, IL-6 = interleukin 6, TNF-α = tumor necrosis factor alpha. OSA = obstructive sleep apnea.
Discussion
In recent years, multiple studies have identified an association between OSA and adverse CVDs, such as hypertension, ischemic heart disease, and stroke.2,3 These chronic diseases are characterized by several pathophysiologic mechanisms, of which ED and inflammation caused by hypoxia must be the most likely ones. Previous researchers have shown that intermittent hypoxia in OSA can cause ED, 19 and that there is a statistically significant correlation between arterial function and hypoxia severity. 20 Compared to healthy controls, patients with OSA have increased plasma levels of endothelin-1, which are closely associated with vascular activity. 21 Other investigators have found an improvement in endothelial function after successful treatment of OSA. 22 It is therefore possible that many factors are involved in the interrelationship between ED and OSA, such as inflammation, oxidative stress, or cellular apoptosis, which are activated during sleep apnea.
In the present study, the relationship between sVAP-1 and disease severity in OSA patients was of particular interest. The study results showed that sVAP-1 was at the highest level in patients with severe OSA, followed by patients with moderate OSA. Serum sVAP-1 levels were significantly correlated with AHI according to PSG parameters, implying that OSA severity affects sVAP-1 expression. sVAP-1 can be secreted by vascular smooth muscle cells, adipocytes, and endothelial cells. In addition, increased sVAP-1 expression has been observed in many inflammatory diseases, including psoriasis, chronic kidney and liver disease, multiple sclerosis, and others.2,3,23 Li et al.24,25 have demonstrated that serum sVAP-1 levels were increased in patients diagnosed with DM and that they could predict mortality and severity of end-stage renal disease. In addition, sVAP-1 has been reported to be involved in proliferative diabetic nephropathy and macrovascular complications, such as increased carotid endothelium layer.24,26 Salmi et al. 27 have demonstrated that sVAP-1 plays an important role in coronary artery disease either through its direct adhesive effect or through its enzymatic activity. In conclusion, sVAP-1 has been shown to be a vascular function-dependent molecule that allows us to hypothesize that it may be a predictive marker of OSA, where ED and inflammation are the main features.
It is well established that intermittent hypoxia is a distinctive feature of OSA and is responsible for upregulation of inflammatory reaction in patients with OSA. Increased levels of sVAP-1 mediate the migration of neutrophils and other adhesive cells to the inflammatory area, after which endothelial cells induce expression of ICAM-1, VCAM-1, IL-8, and others. Kushimoto et al. 28 have found that blocking sVAP-1 expression significantly inhibited inflammatory angiogenesis, suggesting that VAP-1 can accelerate the onset and progression of inflammation. Echoing this, the present findings showed that plasma inflammatory biomarkers, such as hsCRP, IL-6, and, TNF-α, were significantly increased in patients with moderate and severe OSA compared to controls, and that there was a positive correlation between serum sVAP-1 levels and these inflammatory molecules. It is therefore reasonable to assume that increased sVAP-1 levels are most likely responsible for the increased burden of systemic inflammation. Thus, it can be used as an indicator to assess the degree of inflammation in OSA patients.
Data obtained from ROC analysis indicated that sVAP-1 was significant (AUC = .876, P < .001) in the detection of OSA status. Data from multivariate regression analysis showed that plasma sVAP-1 level is an independent predictor of severe OSA. These results suggest that the upward trend in sVAP-1 levels is consistent with the development of OSA severity and it may be used as a valuable biomarker for OSA prediction. Importantly, our model was adjusted for age and BMI, since some authors consider that obesity might be a major confounding factor leading to result misinterpretation. 29 Regression analysis also showed that TNF-α is a significant positive predictor of severe OSA, which is consistent with the findings reported by Li and Zheng, who found significantly higher circulating TNF-α levels in patients with OSA. 30
In the present study, plasma adropin was shown to be an independent negative predictor of severe and moderate OSA based on regression analysis. As demonstrated by Lovrenet et al., adropin is a novel peptide that may play a protective role in endothelial homeostasis. 11 It has also been shown to regulate eNOS activity via the VEGFR2-extracellular signal-regulated kinase pathway, thereby reducing systemic inflammation, leading to an increase in endothelial cell proliferation, migration, and capillary-like tube formation and a decrease in cell permeability and apoptosis. 11 In addition, some studies have shown that adropin can reduce messenger RNA expression levels of TNF-α and IL-6, establishing an inhibitory effect on systemic inflammation. 31 As with OSA, a recent study has shown a significant negative correlation between plasma adropin levels and mediators of systemic inflammation in OSA patients. 16 Circulating adropin concentrations are highly regulated by energy intake and are also involved in cardiovascular function, particularly endothelial function.11,13,15,32 Theoretically, adropin and sVAP-1 may have contrary effects on the course of ED. The present study showed that plasma adropin levels were significantly negatively correlated with sVAP-1, reinforcing the notion that sleep disorders impose a significant ED burden on the affected individuals.
The present correlation analysis also showed a significant inverse relationship between plasma adropin levels and AHI, which is used as an index of OSA severity based on the PSG results. The pathogenesis of ED in OSA remains largely unknown. One such mechanism may involve a dysfunction in eNOS, leading to reduced NO synthesis. 33 NO production and availability in the vascular endothelium is an essential determinant of endothelial function and activity. 34 Mechanistically, adropin’s protective role can be achieved through the interaction of adropin with eNOS, leading to an increased release of NO. A reduction in adropin level in OSA generally reflects the degree of hypoxic burden that can have deleterious effects on eNOS, thus maintaining and further aggravating ED.
The present study has some limitations as it is based on a prospective analysis of a relatively limited number of patients. Furthermore, given the physical differences between men and women, the vascular effects of equally severe hypoxia may be different. Therefore, we attempted to conduct a separate statistical analysis for men and women, which is not described in this report. The present study included only male patients. Therefore, our results cannot be applied to the whole population and further studies are needed to refine our conclusions. Finally, a full-night PSG was not performed in the control group, so it cannot be completely ruled out that some of the healthy volunteers may have had OSA that went undetected. However, low scores (<2) obtained on the STOP questionnaire have a high negative predictive value for OSA status and are a highly sensitive tool for patients with moderate and severe OSA, as confirmed in a large number of validation cohorts and relevant systematic studies.35,36 In addition, another analysis using the full STOP-BANG questionnaire rather than the STOP questionnaire alone was performed to improve sensitivity for moderate and severe OSA. Our analysis showed that none of the 20 subjects were at high risk of OSA. It is therefore reasonable to conclude that volunteer subjects were highly unlikely to have had a clinically significant OSA, despite not having undergone the full nocturnal PSG test.
Conclusion
In conclusion, this is the first study performed in patients with OSA that showed a significant association between sVAP-1 serum levels and PSG sleep parameters, plasma adropin, and circulating inflammatory markers in order to demonstrate the role of sVAP-1 in the pathogenesis of OSA. There is a potential that sVAP-1 may be used as a screening tool for evaluation of OSA severity at a lower cost and with a higher medical compliance compared to the PSG test. Considering the dual function of sVAP-1 both as an enzyme and an adhesive molecule, it may also become a new therapeutic target for medication to prevent vascular damage in OSA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the Ethics Committee of Shanghai University of Traditional Chinese Medicine.
Informed consent
Informed consent was obtained from all individual participants included in the study
Data availability
All the data and material are transparency in this article.
