Abstract
Otolaryngologic manifestations of infection with Blastomyces species are extremely rare and restricted geographically to recognized endemic regions. Here, we describe a case of laryngeal blastomycosis that presented as slowly progressive dysphonia. While a preliminary diagnosis was made using routine histopathology, a species identification of Blastomyces dermatitidis was made using polymerase chain reaction amplification and rapid genotyping without the need for fungal culture. All symptoms resolved following 1 month of antifungal therapy. Rapid molecular differentiation of B dermatitidis from Blastomyces gilchristii provides important insights into pathogenesis given recent recognition of differences in clinical spectra.
Introduction
Blastomycosis, caused by 6 distinct species in the Blastomyces genus,1-3 is a potentially serious fungal infection of humans and other mammals. The disease is reported sporadically worldwide but is of particular concern in areas of endemicity including the Great Lakes region and Mississippi River Valley. 4
Blastomyces species are dimorphic fungi that have the unique ability to undergo morphologic changes in response to environmental conditions. 5 Following inhalation of infectious conidia from the soil by a susceptible mammalian host, the fungus converts to a pathogenic yeast with a characteristic broad-based bud form. Blastomyces species are protean in the spectrum of clinical disease produced, varying from asymptomatic infection to fulminant respiratory failure with secondary dissemination to skin, bone, and other organs possible.6,7 Extrapulmonary disease manifestations are believed to spread from a primary and often unrecognized pulmonary infection with very rare exceptions.8,9 Among the 6 described Blastomyces species, Blastomyces dermatitidis and Blastomyces gilchristii have been most studied with B gilchristii being primarily associated with acute pulmonary disease and B dermatitidis being associated more with disseminated disease. 7
Otolaryngologic manifestations of blastomycosis are extremely rare with descriptions being limited to case reports and short reviews.10-12 Laryngeal blastomycosis can be particularly hard to diagnose, especially in the absence of pulmonary symptoms, as both gross and microscopic lesions can superficially resemble neoplastic processes.13,14 Symptoms are often limited to dysphonia but can also include cough, throat pain, and dysphagia. Here, we describe a case of isolated laryngeal blastomycosis that remained minimally progressive despite delayed diagnosis.
Case Report
A 66-year-old male with elevated body mass index, history of hypertension, hyperlipidemia, type II diabetes mellitus, hypothyroidism, chronic back pain, and liver disease but without a smoking history presented to an otolaryngology clinic with a 9-month history of dysphonia worsening over the prior 3 months. He was determined to have a right vocal cord lesion and was placed on vocal rest for 2 weeks with scheduled follow-up. Three weeks later, he underwent fiberoptic laryngoscopy. The health care provider noted dry mucous membranes of the nose, throat and larynx, and distinct thickening of both vocal cords with no lesion being identified. It was noted that the patient was prescribed several medications for chronic back pain that may produce side effects including mouth and throat dryness, potentially leading to irritation and lesion formation. Cancer was not suspected and the patient was urged to make dietary and lifestyle modifications, including treatment for acid reflux disease.
The patient presented again 9 months later with a complaint of persistent hoarseness, and another fiberoptic laryngoscopy was performed. An excess of granulation tissue was noted with commissure thickening that was suspicious of reflux disease. Due to retirement of the treating provider, the patient was referred into our health care system and appointed 3 months later. Microlaryngoscopy was subsequently scheduled with biopsy and pathologic examination that revealed hyperkeratotic and reactive squamous mucosa with microabscesses and mild squamous atypia. A prednisone taper was prescribed for control of inflammation based upon these findings. At 1 month follow-up postsurgery, the patient had not improved on prednisone and was placed on a 10-day course of oral cephalexin. One month following completion of the antimicrobic, a chest radiograph was obtained during preoperative clearance for a second planned biopsy. The imaging was determined to be normal. Two weeks later, a second biopsy was performed and budding yeast characteristic of Blastomyces species were identified on histopathologic examination (Figure 1). Based upon these findings, a review of the initial biopsy did reveal a single yeast form being discernible within one of the microabscesses, demonstrating the slow growth rate and pronounced tissue reaction to an extremely small organism burden. Fluconazole (200 mg, once daily) was prescribed and initiated 11 days after the second biopsy rather than itraconazole due to potential drug interaction with the patient’s statin medication. The patient’s dysphonia resolved following 1 month of treatment. A repeat laryngoscopy was performed after 6 months of antifungal treatment and found to be normal. Treatment was stopped, and his disease was considered resolved.
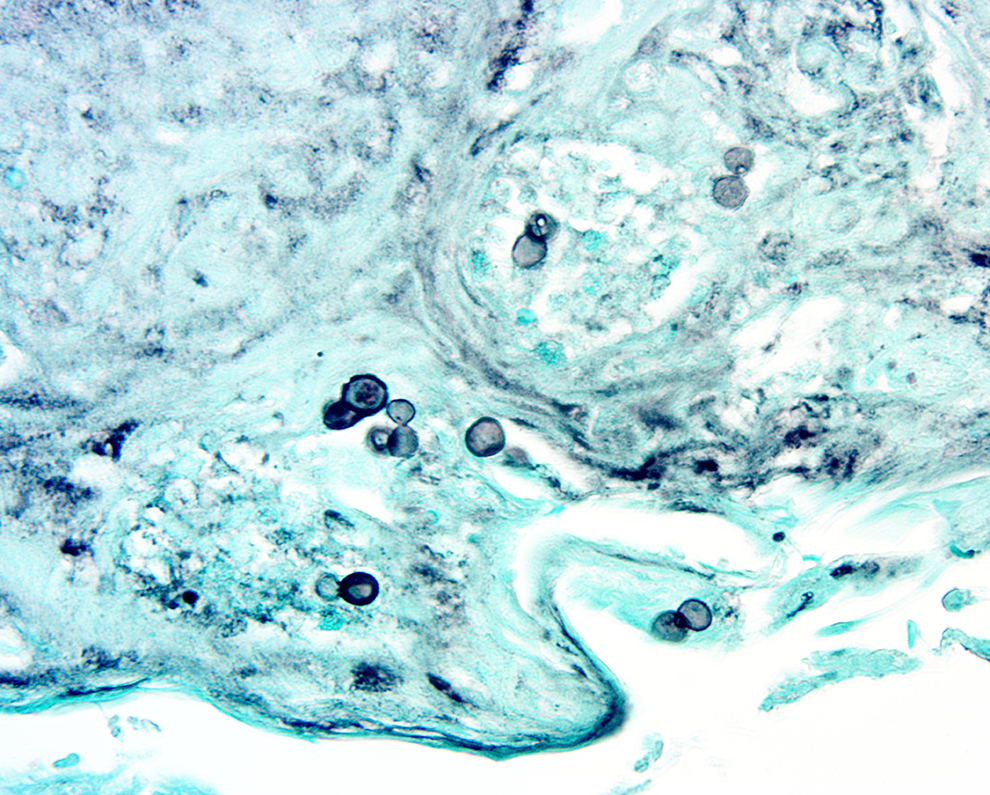
Gomori’s methenamine silver impregnation stain of a vocal cord tissue demonstrating broad-based budding yeast forms typical of a Blastomyces species within foci of acute and chronic inflammation.
The formalin-fixed paraffin-embedded tissue from the second biopsy was subsequently submitted for polymerase chain reaction (PCR) amplification and Blastomyces species genotyping. Four 5-µm-thick sections were cut in duplicate and placed into sterile 1.5-mL sterile microcentrifuge tubes. DNA was extracted using the QIAamp DNA FFPE Tissue Kit (QIAGEN Inc) according to the manufacturer’s recommendations and typed to the species level by use of Sanger sequencing targeting a portion of the inter-transcribed spacer region (ITS2) of rDNA. In addition, the rDNA was also genotyped by use of a Life Technologies Custom TaqMan SNP Assay (data not shown, ThermoFisher Scientific). Species assignment was based on a fixed nucleotide difference between B dermatitidis and B gilchristii at position 19 in both assays with the isolate being confirmed as B dermatitidis. 1
Discussion
Primary, or isolated, laryngeal blastomycosis can be particularly difficult to diagnose due to lack of typical systemic disease presentation and resemblance to laryngeal carcinoma. While such cases are extremely rare, case reports are represented in the literature going back to 1918. 15 Here, we report a case of indolent laryngeal blastomycosis producing dysphonia that was localized to the larynx.
The patient was referred to our health care system with a prolonged history of dysphonia but was without prior documentation or recent history of pulmonary symptoms or respiratory illness. This is consistent with other reports of laryngeal blastomycosis with most being isolated to the larynx as a single lesion. In addition, it is not uncommon in cases of disseminated blastomycosis for initial pulmonary symptoms to be subclinical or self-limiting and resolve spontaneously without medical intervention.16,17 In this case, a chest radiograph was obtained for preoperative clearance 2 weeks prior to diagnosis and was determined to be normal.
The patient continued to complain of hoarseness and throat pain over the 14 months since his initial evaluation, including a 9-month lapse in care. Throughout this time period, he denied dysphagia, shortness of breath, fever, chest pain, or cough and was negative for any skin lesions. We propose several possible explanations for the stable, nonprogressive nature of this infection with pathogen and host immune factors being responsible. Previous studies have established that infections of B dermatitidis are more likely to produce disseminated disease and less likely to present as acute respiratory disease, leading to delayed diagnosis as demonstrated in this case, 7 as compared to the more recently described B gilchristii. Alternatively, this patient did have a significant number of underlying medical conditions which may have limited his ability to mount a robust response to infection. Previous studies have shown that acute pulmonary blastomycosis cases are diagnosed faster than those with limited or no obvious pulmonary involvement. 7 These patients likely benefit from a faster diagnosis in part due to the robustness of their innate immune response. 18 Also, underlying medical conditions can have a masking effect on symptoms of a superimposed disease, as has been shown in studies of sepsis in older adults. 19 Conversely, there is some evidence that prior exposure to Blastomyces species provides protection against future infection. 20 This patient was known to live in an area endemic for blastomycosis, and previous exposure may have provided some level of protective immunity.
At no point during the patient’s evaluation were microbiologic stains, cultures or fungal urinary antigen testing performed given the clinical presentation and rarity of an infectious etiology in this setting. In suspected blastomycosis, these diagnostic modalities, along with use of routine histopathology as in this case, would have been utilized. Fortunately, availability of the FFPE specimen for direct PCR amplification and genotyping was used to confirm the etiologic agent as B dermatitidis.
In summary, we describe a case of isolated and minimally progressive laryngeal blastomycosis producing dysphonia without preexisting evidence of pulmonary involvement. Routine histopathology was used to identify presence of a Blastomyces species, and molecular genotyping was subsequently able to identify the species as B dermatitidis. Infection with Blastomyces species is well known to produce unusual clinical presentations mimicking other clinical syndromes and result in delayed diagnosis and targeted treatment. Importantly, health care providers new to regions where this disease is endemic should be alerted to its presence for consideration in their differential diagnoses. The ability to obtain clinical specimens and perform direct rapid genotyping proved to be especially helpful in this case and can provide timely insight into pathogenesis of strains that produce different clinical spectra. Such information may shed light on important host- and organism-specific factors that could ultimately prove useful in vaccine development strategies.
Footnotes
Authors’ Note
Deepa Bassi, is now with Cancer Treatment Centers of America, WRMC Comprehensive Care and Research Center, Phoenix, AZ, USA.
Acknowledgments
The authors wish to thank Brian Abrahamson and the Histology Laboratory within the Division of Laboratory Medicine, Marshfield Clinic Health System, for their contributions in providing specimens for the molecular genotyping.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Marshfield Clinic Health System, with support from the Marshfield Clinic Research Institute’s Infectious Diseases Distinguished Physician/Scientist Endowment to JKM and the Miller Endowment in Rural and Agricultural Health and Safety Research to TRF.
