Abstract
Importance:
Necrotizing fasciitis is a relatively uncommon and potentially life-threatening soft tissue infection, with morbidity and mortality approaching 25% to 35%, even with optimal treatment. The challenge of diagnosis for necrotizing soft tissue infections (NSTIs) is their rarity, with the incidence of approximately 1000 cases annually in the United States. Given the rapid progression of disease and its similar presentation to more benign processes, early and definitive diagnosis is imperative.
Findings:
Signs and symptoms of NSTIs in the early stages are virtually indistinguishable from those seen with abscesses and cellulitis, making definitive diagnosis difficult. The clinical presentation will depend on the pathogen and its virulence factors which ultimately determine the area and depth of invasion into tissue. There are multiple laboratory value scoring systems that have been developed to support the diagnosis of an NSTI. The scoring system with the highest positive (92%) and negative (96%) predictive value is the laboratory risk indicator for necrotizing fasciitis (LRINEC). The score is determined by 6 serologic markers: C-reactive protein (CRP), total white blood cell (WBC) count, hemoglobin, sodium, creatinine, and glucose. A score ≥ 6 is a relatively specific indicator of necrotizing fasciitis (specificity 83.8%), but a score <6 is not sensitive (59.2%) enough to rule out necrotizing fasciitis. In terms of imaging, computed tomography (CT) imaging, while more sensitive (80%) than plain radiography in detecting abnormalities, is just as nonspecific. Computed tomography imaging of NSTIs demonstrates fascial thickening (with potential fat stranding), edema, subcutaneous gas, and abscess formation. Magnetic resonance imaging (MRI) has demonstrated sensitivity of 100% and specificity of 86%, though MRI may not show early cases of fascial involvement of necrotizing fasciitis.
Conclusions and Relevance:
Necrotizing soft tissue infections are rapidly progressive and potentially fatal infections that require a high index of clinical suspicion to promptly diagnose and aggressive surgical debridement of affected tissue in order to ensure optimal outcomes.
Prompt surgical and infectious disease consultation is necessary for the treatment and management of these patients. While imaging is useful for further characterization, it should not delay surgical consultation. Necrotizing soft tissue infection remains a clinical diagnosis, although plain radiography, CT imaging, and ultrasound can provide useful clues. In general, the management of these patients should include rapid diagnosis, using a combination of clinical suspicion, laboratory data (LRINEC score), and imaging (MRI being the recommended imaging modality), prompt infectious disease and surgical consultation, surgical debridement, and delayed reconstruction. Laboratory findings that can more strongly suggest a diagnosis of NSTI include elevated CRP, elevated WBC, low hemoglobin, decreased sodium, and increased creatinine. Imaging findings include fascial thickening (with potential fat stranding), edema, subcutaneous gas, and abscess formation. Broad-spectrum antibiotics should be started in all cases of suspected NSTI. Surgical debridement, however, remains the lynchpin for treatment of cervical necrotizing fasciitis.
Keywords
Introduction
Necrotizing fasciitis is a relatively uncommon and potentially life-threatening soft tissue infection, with morbidity and mortality approaching 25% to 35%, even with optimal treatment. While necrotizing fasciitis can affect the dermis and epidermis, most cases involve the deeper layers of adipose tissue, fascia, and muscle, making it difficult to differentiate from other more benign skin pathologies. The degree of tissue destruction depends on the pathogen involved and its virulence factors. 1
Given the rapid progression of disease and its similar presentation to more benign processes, early and definitive diagnosis is imperative. Signs of toxicity are invariably disproportionate to skin manifestations and physical signs, requiring a high index of suspicion on the physician’s part to accurately diagnose necrotizing fasciitis. Only 15% to 34% of patients with necrotizing fasciitis have an accurate admitting diagnosis. 1 Infection may follow trauma, however small of a scrape. One of the most important interventions is early surgical debridement. 2
Case Report
Patient History
A 29-year-old woman presented to our hospital after being treated at an outside hospital 6 days ago with oral antibiotics for a neck infection. The patient presented with fever, chills, and significant neck pain. A computed tomography (CT) scan (Figure 1) was performed that revealed a deep neck abscess, subcutaneous emphysema, and thickening of the platysma worrisome for necrotizing fasciitis. The patient was brought to the operating room where she underwent initial drainage of the neck abscess, debridement of the muscle, and excision of the skin and platysma. The patient was admitted to the intensive care unit where she was managed by the Critical Care Team, Thoracic Surgery, as well as the head and neck service.
The patient underwent a modified Blair incision with elevation of the subplatysmal flaps, revealing necrosis of the platysma extending to the subdermal subcutaneous fat and an abscess of the sternocleidomastoid muscle with necrotic involvement of the left mastoid tip extending to the tail of the left parotid gland. These areas were debrided, irrigated, and covered with plain gauze with bacitracin saline solution. Cultures of this patient’s tissue eventually grew group A streptococci. The patient recovered well over the course of several months with aggressive wound care.
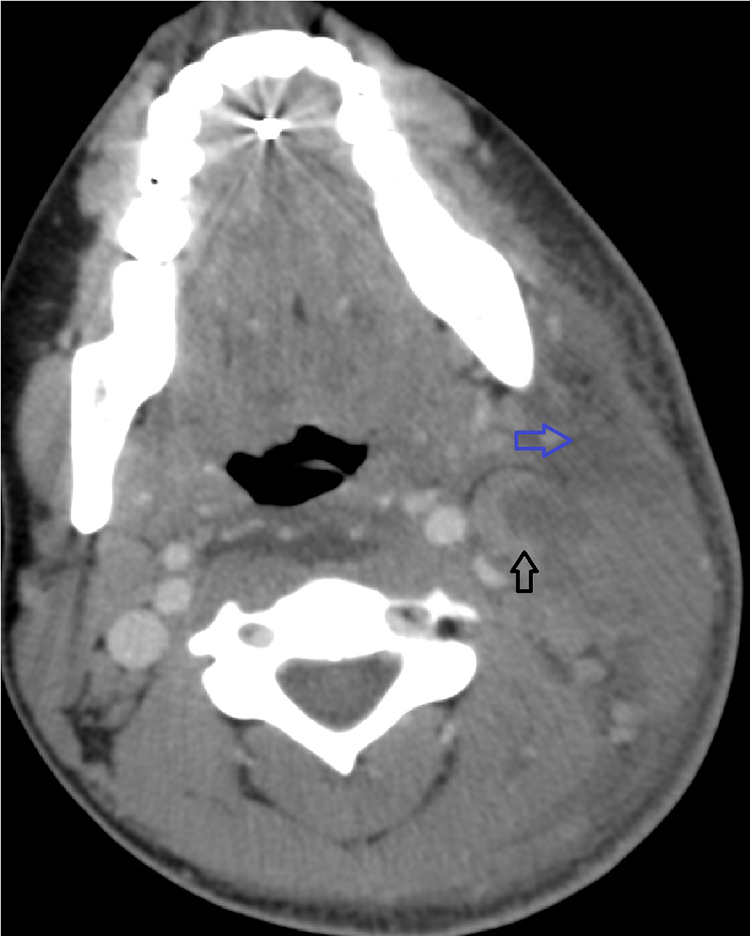
Contrast-enhanced CT scan through the neck demonstrating regions of hypoattenuation with soft tissue stranding and obliteration of fat pads (blue arrow). An enlarged lymph node with necrotic center is also noted (black arrow). CT indicates computed tomography.
Discussion
Epidemiology
The challenge of diagnosis for necrotizing soft tissue infections (NSTIs) is their rarity. There are approximately 1000 cases annually in the United States. Although NSTIs are more common in obese individuals and the immunocompromised, they can also occur spontaneously in young, otherwise healthy individuals without predisposing risk factors. Immunocompromised patients, such as those with active corticosteroid usage, malignancy, undergoing chemotherapy or radiation therapy, a diagnosis of HIV or AIDS, or organ transplant with long-term immunosuppression, may not exhibit symptoms as early or as severely. 3 In general, immunocompromised patients tend to present with lower systolic blood pressures, glucose levels, and white blood cell (WBC) counts. Because their presentation is apparently less acute, they generally require a greater degree of illness to warrant transfer, admission to a surgical service, and are significantly less likely to undergo surgical debridement, resulting in a mortality rate twice as high as the general population. 4
Pathophysiology
Necrotizing fasciitis can be classified into 3 categories, with type 1 infections representing polymicrobial infections, type 2 involving Group A Streptococcus, typically streptococcus pyogenes, and type 3 representing gram negative marine organisms, most notably vibrio vulnificans. 5
Although necrotizing fasciitis typically affects the perineum and trunk, our case reflects its ability to affect the head and neck. Most cases of necrotizing fasciitis occur due to trauma to skin that then seeds bacteria. Infection begins in the deep tissue, where bacteria can rapidly proliferate within viable tissue. Fibrous attachments between subcutaneous tissues and fascia limit spread to areas like the hands, feet, and scalp, but because the trunk and limbs do not have these attachments, there is a tendency for infection to spread. 1 Edema can also occur secondary to venous and lymphatic spread. This spread can also cause thrombosis of blood vessels that leads to tissue ischemia and possible gangrene of subcutaneous fat and the dermis. 6 Once the fascia breaks down, infection of the muscle leads to myositis, as occurred in our patient. Although rare, subcutaneous emphysema, known colloquially as gas gangrene, can also occur in the necrotizing tissue and is often caused by Clostridium species.
Clinical Presentation and Diagnosis
Signs and symptoms of NSTIs in the early stages are virtually indistinguishable from those seen with abscesses and cellulitis, making definitive diagnosis difficult. In addition to the cardinal signs of infection which include swelling, erythema, warmth, and fever, NSTIs present with a constellation of additional findings that can help a physician in distinguishing it from relatively benign superficial skin infections. Necrotizing soft tissue infections are characterized by severe pain, extending beyond the obviously involved areas and disproportionate to examination findings. Later in the disease process, the clinical signs become more identifiable. In the minority of cases (7%-44%), findings include bullae, ecchymoses, skin and muscle necrosis, gas in tissue identified on radiography or as crepitus on physical examination, or cutaneous anesthesia secondary to dermal necrosis. Any of these signs should prompt immediate surgical exploration. 4
While clostridial NSTIs are classically associated with subcutaneous gas formation and crepitus on examination, this finding is not always present. Moreover, clostridial species are often difficult to grow and isolate in culture, making blood cultures a poorly sensitive test for ruling them out. Once patients manifest signs of shock, mortality exceeds 50%. 7 An important distinction to be made between clostridium infection and most other causes of NSTIs is that the onset of symptoms and progression to florid necrosis and sepsis occurs within hours of inoculation rather than 2 to 3 days before symptom onset.
One of the most important factors in diagnosing an NSTI is the time course of disease, as established by a thorough history. In contrast to abscesses or cellulitis, which progress over the course of days or weeks, NSTIs progress extremely rapidly, on the order of hours. To support the diagnosis, there are several helpful modalities, including laboratory evaluation, imaging evaluation, and operative exploration.
Laboratory risk indicator for necrotizing fasciitis score
There are multiple laboratory value scoring systems that have been developed to support the diagnosis of an NSTI. The scoring system with the highest positive (92%) and negative (96%) predictive value is the laboratory risk indicator for necrotizing fasciitis (LRINEC), developed by Wong and colleagues. 8 The LRINEC score was devised from a comparison of 89 patients with an NSTI and 314 patients with severe cellulitis or abscess, or both. 4 The scoring system scores patients on various laboratory values including C-reactive protein, WBC count, hemoglobin level, serum sodium level, serum creatinine level, and serum glucose level at hospital admission. For an overview of the scoring system in table form, please refer to Table 1.
Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) Scoring System.
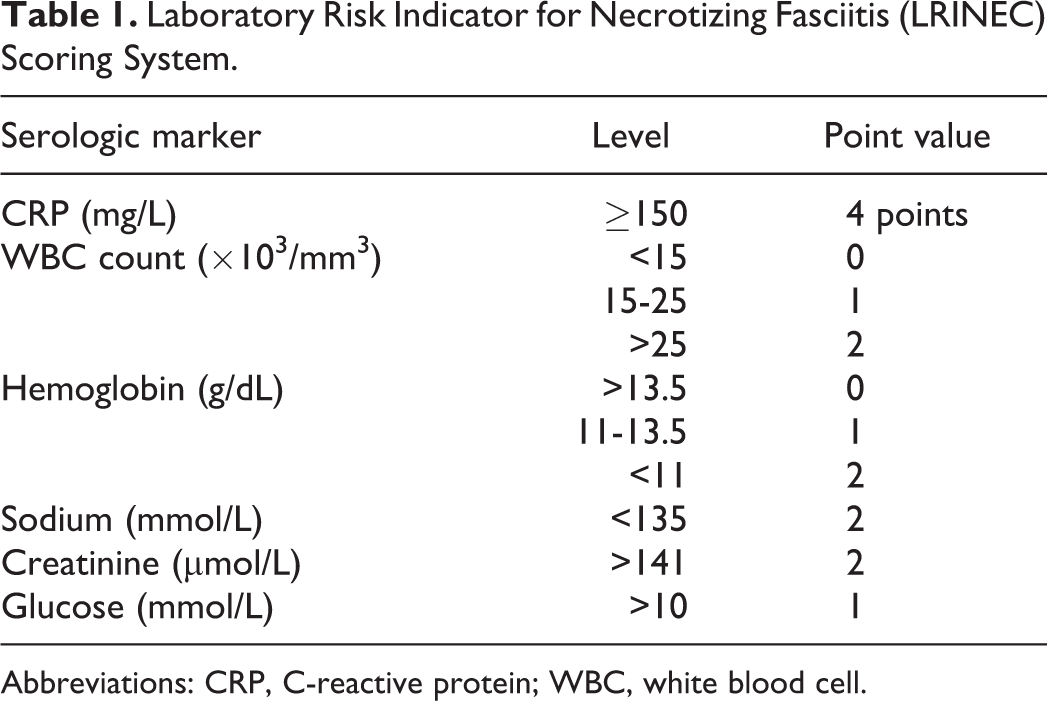
Abbreviations: CRP, C-reactive protein; WBC, white blood cell.
The score is determined by 6 serologic markers: C-reactive protein (CRP), total white blood cell (WBC) count, hemoglobin, sodium, creatinine, and glucose. A score ≥6 is a relatively specific indicator of necrotizing fasciitis (specificity 83.8%), but a score <6 is not sensitive (59.2%) enough to rule out necrotizing fasciitis.
Although imaging can sometimes be useful in ascertaining the depth of infection or in determining the presence or absence of gas, it is generally agreed that the potential benefit of imaging is outweighed by the time required to obtain it. If imaging is obtained, however, the below findings may be of use to detect cases of NSTI.
Computed tomography and magnetic resonance imaging findings
Computed tomography imaging, while more sensitive (80%) than plain radiography in detecting abnormalities, is just as nonspecific. 9 Computed tomography imaging of NSTIs demonstrates fascial thickening (with potential fat stranding), edema, subcutaneous gas, and abscess formation as seen on Figure 2. Magnetic resonance imaging (MRI) has demonstrated sensitivity of 100% and specificity of 86%, though MRI may not show early cases of fascial involvement of necrotizing fasciitis.10,11 Magnetic resonance imaging (MRI) has been shown to be more sensitive than either CT or plain radiography in detecting NSTIs but is less specific, owing to overlapping imaging findings of tissue enhancement on T2-weighted imaging, frequently seen following trauma or inflammatory processes. On T1 weighted images, there is often high signal intensity with loss of muscle texture. On T2 weighted images, there is subcutaneous and intramuscular edema in a reticulated pattern as well as fluid collections in the fascia. 12 Ultrasound lacks both sensitivity and specificity for detecting NSTIs but remains a sensitive modality for ruling out abscesses. 13
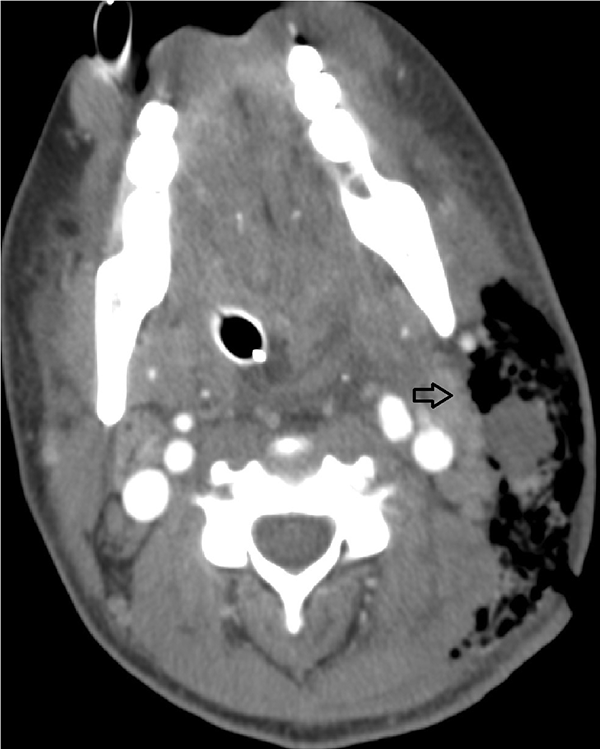
Axial CTs show the diffuse cellulitis and myositis, which are more localized on the left with obliteration of the fascial planes. There are localized gas collections seen in within the soft tissues (black arrow). CT indicates computed tomography.
The definitive standard for diagnosing NSTIs is operative exploration when a high index of clinical suspicion is present. Upon visualization of the fascial layers, a surgeon would be able to assess the degree of involvement of fascia, surrounding adipose, and muscle tissue.
Management and Treatment
Empiric therapy should include coverage for gram-positive, gram-negative, and anaerobes, such as vancomycin or linezolid plus piperacillin-tazobactam.7,14 Consideration for possible infection with methicillin-resistant Staphylococcus aureus (MRSA) should be given and appropriate MRSA treatment should be initiated if suspected.
The lynchpin for treatment of necrotizing fasciitis is surgical debridement. The room’s temperature should be kept warm enough to prevent hypothermia. Dressings must be kept to prevent tissues from drying out but to provide easy access in case complications such as secondary infections occur. It is important to note the exact characteristics such as size, appearance, and any other notable features to stage the wound and as a baseline for further considerations.
Therapy with hyperbaric oxygen is a commonly cited adjunct to wound debridement that involves delivering 2 to 3 atms of oxygen in a high-pressure chamber. This level of pressure results in arterial oxygen tension as high as 2000 mm Hg and tissue oxygen tension of 300 mm Hg. 15 For reference, 100% oxygen at normal atmospheric pressure only achieves arterial oxygen tension of 300 mm Hg and tissue oxygen tension of 75 mm Hg. Hyper-saturation of tissue with oxygen in cases of NSTI has been shown to result in less edema, enhanced granulation tissue formation, improved leukocyte-mediated oxidative burst, inhibition of bacterial toxin production and release, and synergistic efficacy with concurrent antibiotic administration.
Covering and reconstruction of the wound also plays a role in the outcome of the patient, as immediate tissue coverage protects against superinfection, as well as fluid and electrolyte loss, allowing for the patient to mobilize early while reducing pain. Traditional reconstructive options include primary closure, split-thickness skin grafts, full-thickness skin grafts, delayed primary closure, healing by second intention, tissue expansion, and pedicled or free flaps. 4
Complications and Prognosis
Complications from treating necrotizing fasciitis are numerous. The wound may become secondarily infected requiring further excision and debridement which increases the already apparent risks, such as hypotension, hypothermia, and large wounds subject to infection. It is possible to use mafenide or Dakin solution soaks, pulse lavage, and negative pressure wound therapy. These all serve to help remove debris and bacteria from the wound. 4
It is important for patients to begin using the affected body parts as soon as possible, as early mobilization can improve outcomes. The 3 main types of pain that patients experience are background pain, or pain that is almost always present of equal consistency throughout, breakthrough pain, which is an acute worsening of pain that may be a result of therapy, and procedural pain, typically seen when further debridement must be done or dressings must be changed. As in most cases of pain management, long-acting narcotics may be used for consistent pain, nonsteroidal anti-inflammatory drug for breakthrough pain, and opioids may be used for procedural pain, which is often described as burning sensation. 4 In small randomized clinical studies, gabapentin has demonstrated opioid-sparing effects, reduction of acute pain, and reduction in paresthetic sensation. 16
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
