Abstract
Purpose
The purpose of the present study was to investigate the effects of COVID-19 on audiovestibular system with Transiently Evoked Distortion Otoacoustic Emissions (TOAE), Distortion Product Otoacoustic Emissions (DPOAE), video head impulse test (vHIT) and caloric test.
Methods
Audiovestibular findings of 24 patients with moderate/severe COVID-19 and 24 healthy controls were compared using pure tone audiometry, tympanometry, TOAE, DPOAE, caloric test, and vHIT.
Results
On audiometry, the pure tone averages of the COVID-19 patients were higher than the controls (P = .038). The TEOAE amplitudes at 4000 and 5000 Hz (P = .006 and P < .01), and DPOAE amplitudes at 3000, 6000, and 8000 Hz (P < .001, P = .003 and P < .001) were significantly lower in COVID-19 patients compared to the controls. On vestibular tests, there was no significant difference between the caloric test results of the patients and the controls (P > .05). On vHIT testing, amplitudes of right semicircular canal was found to be significantly lower in COVID-19 group compared to the control group (P = .008).
Conclusion
COVID-19 may affect inner ear functions causing a subtle damage in the outer hair cells and lateral semicircular canals. It must be kept in mind that COVID-19 may cause cochleovestibular problems.
Introduction
Viral infections may result in different forms and severities of hearing loss. Hearing loss may be due to the direct effect of the virus on the inner ear structures, or due to an indirect effect through immune-mediated mechanisms. 1 -3
SARS-CoV-2 infection or COVID-19 has caused a pandemic, and its exact symptoms, diagnosis and treatment are not clear even after 2 years. 4 It has been reported that COVID-19 may cause vertigo and hearing loss, 5,6 however, the mechanisms are not clear. It was suggested that arterial and venous thrombosis as well as microvascular occlusion might be associated with the pathogenesis of the disease. 7,8 Since the microcirculation of cochlea is vulnerable to thrombotic events, cochleovestibular symptoms may also be associated with thrombovascular events.
This study aimed to investigate the effects of COVID-19 on cochleovestibular system by using audiovestibular tests.
Materials and methods
This prospective case-control study was approved by the local ethical committee of our tertiary medical center (No: E1-20-718/718). A total of 24 patients with mild/moderate COVID-19 who admitted between July 1, 2020 and August 30, 2020, and 24 healthy age- and sex-matched controls were included in the study. The ages of the participants ranged between 18 and 65 years.
There were 10 female and 14 male COVID-19 patients with a mean age of 45.7 ± 6.7 years. The control group consisted of 14 female and 10 male individuals with a mean age of 48.9 ± 6.9 years. None of the patients had any history of neuro-otologic disorders, ototoxicity, or chronic systemic conditions including diabetes mellitus and autoimmune disorders.
The patients were hospitalized, and none of them were intubated in the intensive care unit. All patients received favipiravir and anticoagulants as the standard of care. Favipiravir was administered p.o. 1600 mg/day on the first day, then 600 mg b.i.d. for 4 days, and .1 mg/day enoxaparin was injected subcutaneously.
The patients and the controls were evaluated with audiovestibular tests. The tests were performed 2 weeks after the positive SARS-CoV-2 PCR test results, after the patients had become negative.
Audiovestibuler testing
The tests included pure tone thresholds (GN Otometrics, Madsen, Astera-2, Denmark), tympanometry (GN Otometrics, Madsen New Zodiac, Denmark), acoustic reflexes, transient-evoked otoacoustic emissions (TEOAE) and distortion product otoacoustic emissions (DPOAE) (GN Otometrics, Madsen Capella Cochlear Emissions Analyzer, Denmark). The vestibular tests were performed with videonystagmography (GN Otometrics, ICS Charter 200, Denmark), video head impulse test (v-HIT) (Interacoustics, Denmark), and air caloric test.
Statistical analysis
The analysis of the data was performed with IBM SPSS Statistics version 25.0 software (IBM Corporation, Armonk, NY, USA). Shapiro-Wilk test was used to investigate whether the normal distribution assumption was met. Categorical data were expressed as numbers (n) and percentages (%) while quantitative data were presented as mean ± standard deviation (SD) and medians (25th–75th percentiles). The mean differences between the groups were compared with Student’s t test, and the Mann–Whitney U test was applied for the comparisons of not-normally-distributed variables. Categorical data were analyzed with continuity corrected χ2 or Fisher’s exact tests, when appropriate.
A P value less than .05 was considered as statistically significant. However, the Bonferroni correction was performed for each possible multiple comparison, in order to control any Type I error.
Results
There was no significant difference between COVID-19 patients and the controls for their ages or genders (P > .05). All patients and controls had Type A tympanometries and normal acoustic reflexes.
The pure tone averages were 12.2 dBHL in COVID-19 patients and 13.1 dBHL in the controls. The pure tone averages of the patients were significantly higher than the controls (P = .04). The difference was due to the higher pure tone averages of the COVID-19 patients at 4 kHz (P = .002). Otherwise, there was no significant difference between the frequency-specific pure tones of the patients and the controls (P > .05) (Figure 1). Pure tone audiometry results of COVID-19 patients and the controls.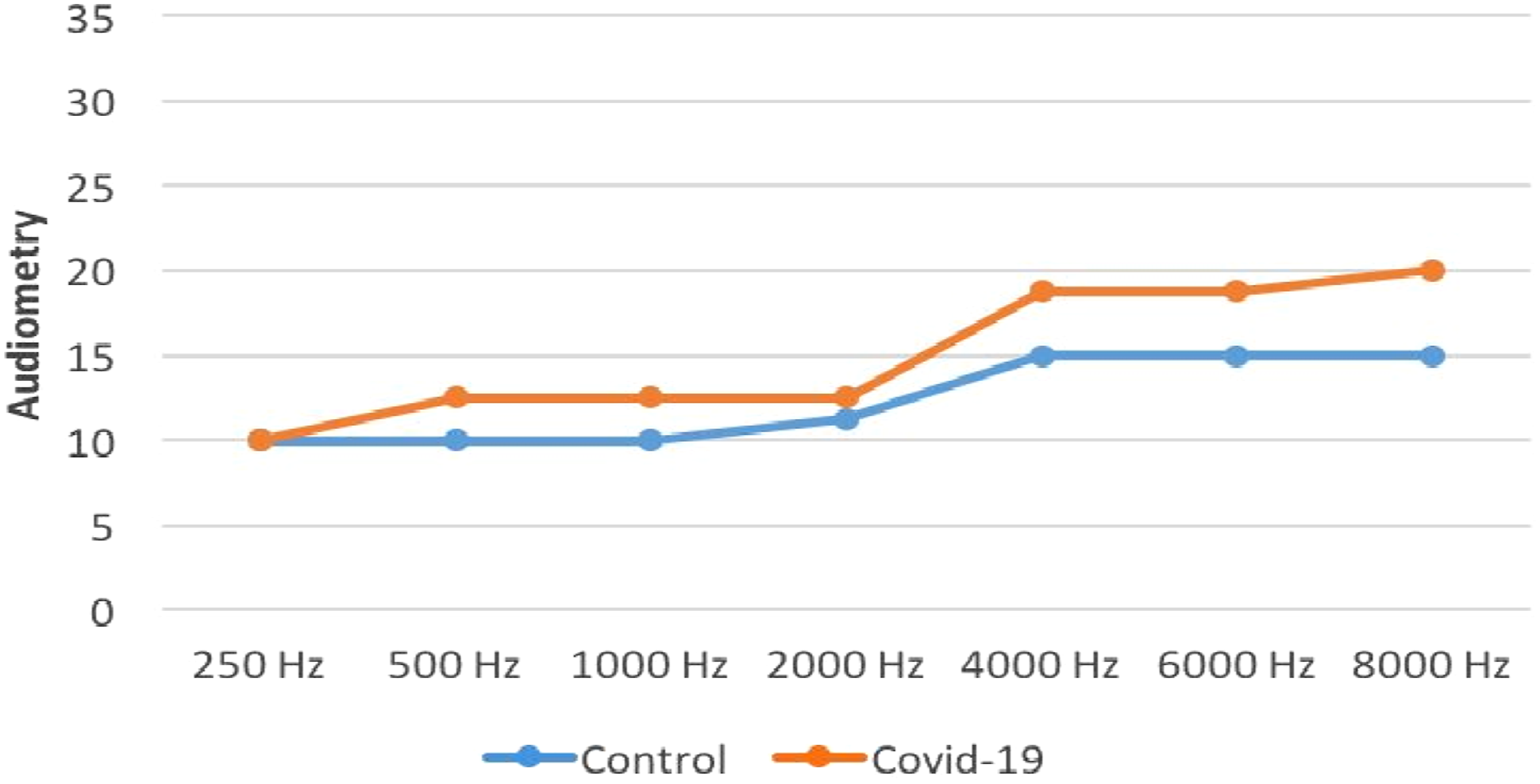
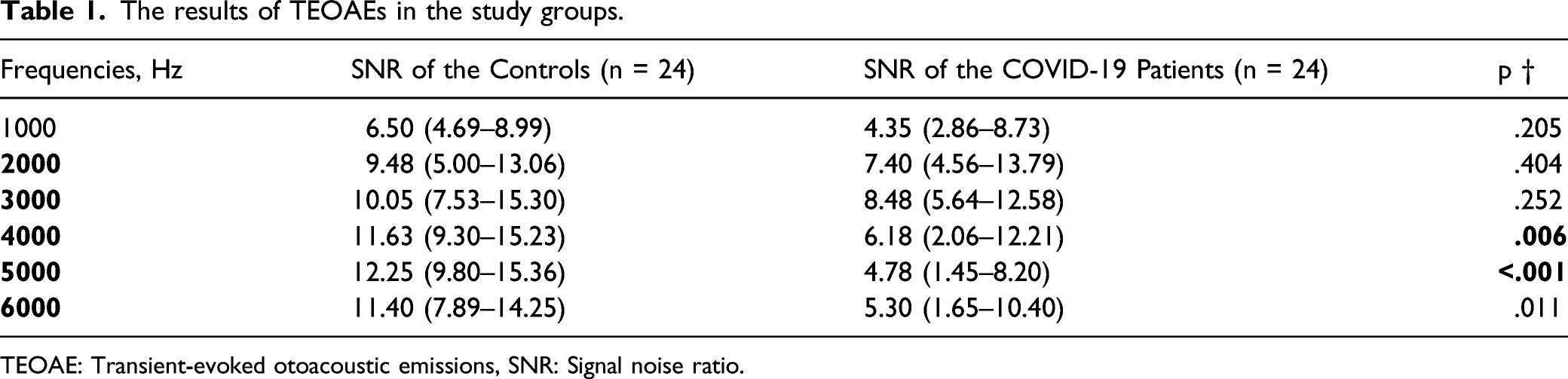
The amplitudes of TEOAEs were significantly lower in the patients compared to the controls at 4 kHz and 5 kHz (P < .01). The TEOAE amplitudes of the patients and the controls were similar (P > .05) except for the amplitudes at 4 kHz and 5 kHz (Figure 2 and Table 1). The results of TEOAEs in COVID-19 patients and the controls. TEOAE: Transient-evoked otoacoustic emissions. The results of TEOAEs in the study groups. TEOAE: Transient-evoked otoacoustic emissions, SNR: Signal noise ratio.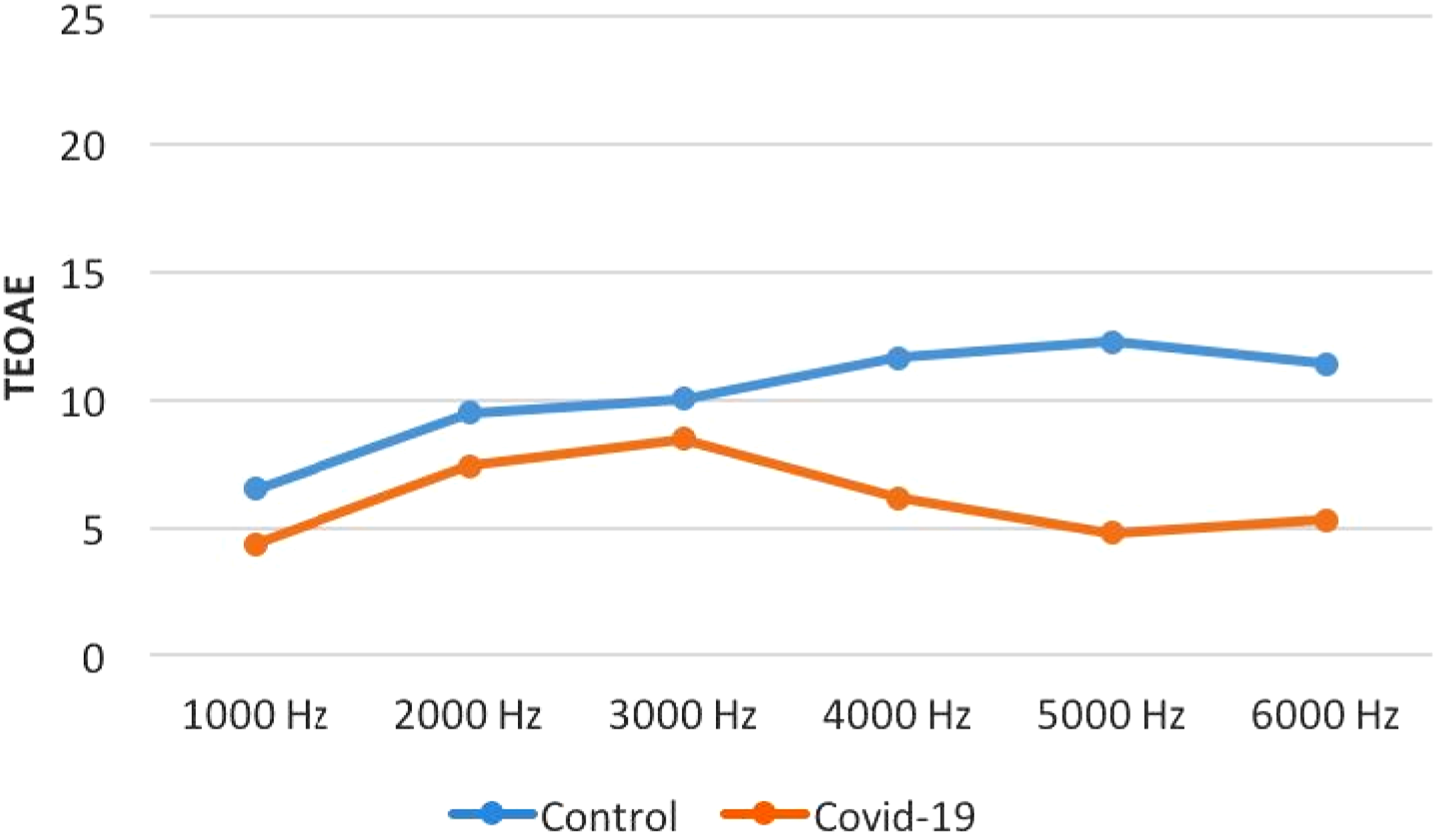
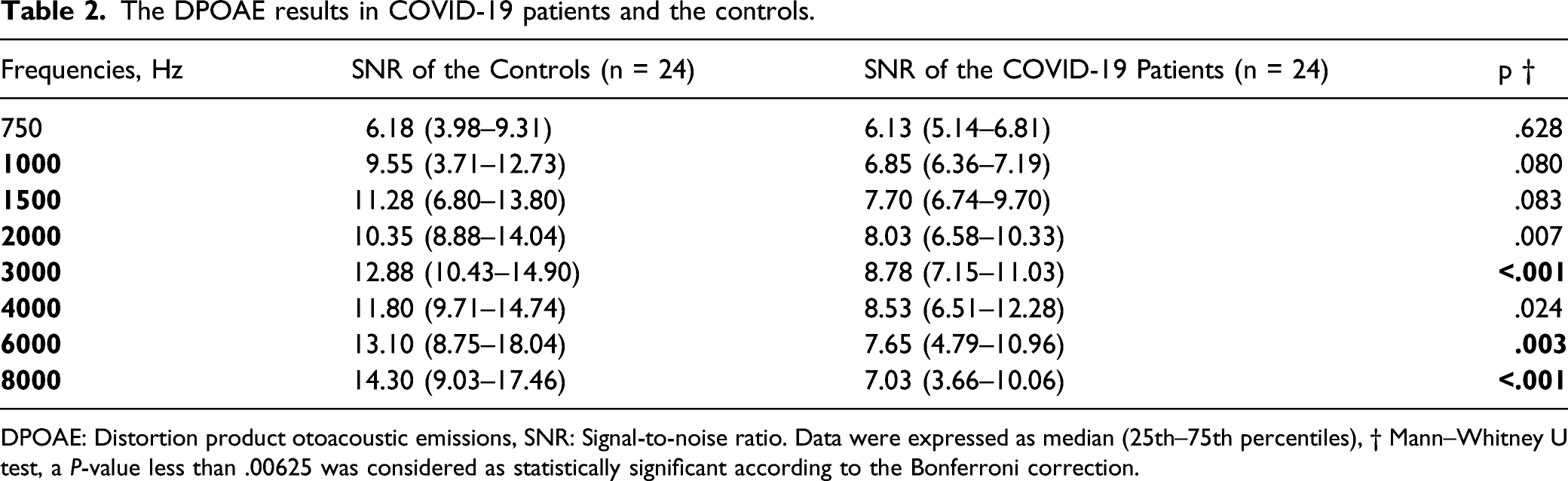
There was no significant difference between the patients and the controls regarding the DPOAE amplitudes (P > .05) except for the amplitudes at 3 kHz, 6 kHz, and 8 kHz. The DPOAE amplitudes were significantly lower in the COVID-19 patients compared to the controls at 3 kHz, 6 kHz, and 8 kHz (P < .01) (Figure 3 and Table 2). The DPOAE results in COVID-19 patients and the controls. DPOAE: Distortion product otoacoustic emissions. The DPOAE results in COVID-19 patients and the controls. DPOAE: Distortion product otoacoustic emissions, SNR: Signal-to-noise ratio. Data were expressed as median (25th–75th percentiles), † Mann–Whitney U test, a P-value less than .00625 was considered as statistically significant according to the Bonferroni correction.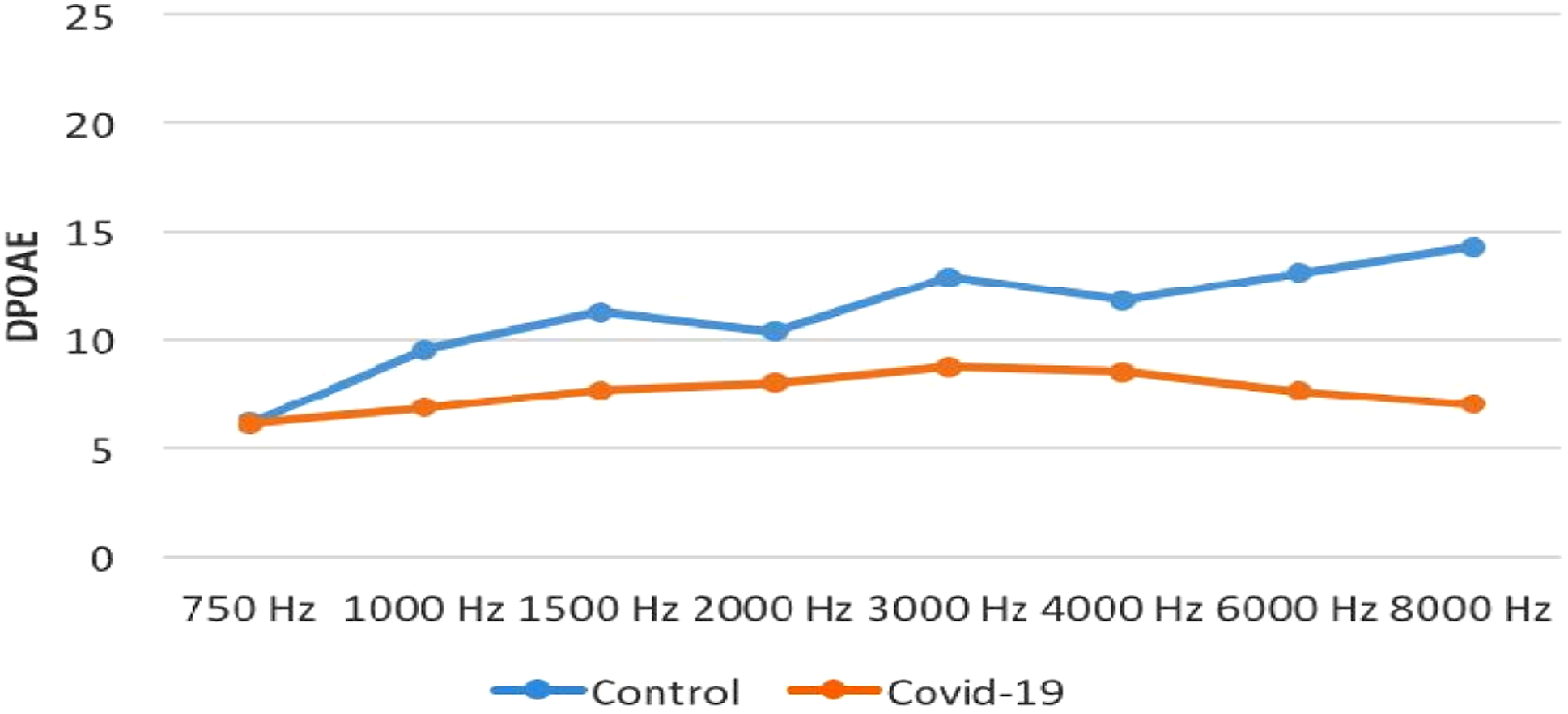
v-HIT results in the COVID-19 patients and the controls.
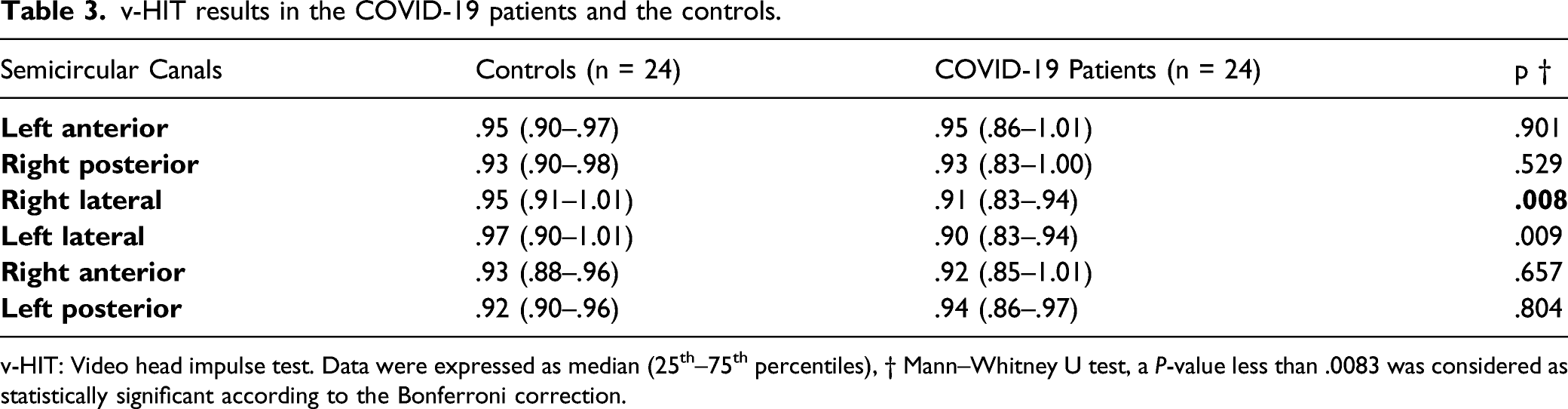
v-HIT: Video head impulse test. Data were expressed as median (25th–75th percentiles), † Mann–Whitney U test, a P-value less than .0083 was considered as statistically significant according to the Bonferroni correction.
Discussion
COVID-19 may lead to multi-organ failure due possibly to inflammatory reactions or coagulopathy disorders. 8 It is also known that some viral infections may cause labyrinthitis and vestibular neuritis. 9 The inner ear damage may also be related to immune-mediated mechanisms. 1 -3 There are limited number of reports regarding the effect of COVID-19 on audiovestibular system. Our study may help illuminating the effect of COVID-19 on inner ear functions.
The audiovestibular effects of COVID-19 have been reported. 6,10,11 Its effect on the cochlea was reviewed by Fancello et al. 10 Beukes et al. 11 reviewed the audiological symptoms of COVID-19, and reported the tinnitus rate as 8%. Mustafa et al. 6 reported elevated pure tone averages and decreased TEOAE amplitudes. The results of our study are comparable with the results of previous studies.
Unlike the studies mentioned above, Dror et al. 12 compared asymptomatic COVID-19 patients with a healthy control group using TEOAE, DPOAE, and ABR, and did not find any significant differences between the groups. However, their study group consisted of 12 cases who had asymptomatic SARS-CoV-2 infection. It may be supposed that different results may be obtained due to the inclusion of different patient groups into the study, and few number of patients may also affect the results.
Normal tympanometry results suggest that the middle ear is spared in COVID-19. That is, COVID-19 does not increase the probability of otitis media with effusion. Likewise, a normal functioning acoustic reflex arc suggests the integrity among the cochlear nerve, facial nerve, and their central connections in the brainstem.
We found significant differences between COVID-19 patients and controls for the TEOAE and DPOAE amplitudes. TEOAE shows the status of outer hair cells in a wide frequency range. DPOAEs may be helpful with regard to determination of subtle problems within the cochlea. In our study, the DPOAE amplitudes showed a tendency to decrease in the high frequencies. According to these findings, it is possible to say that COVID-19 may lead to outer hair cell damage mainly situated in the basal turn of the cochlea, and audiological problems associated with the disease are related to cochlea, rather than central auditory connections.
Dizziness was reported as the most common neurological symptom in the study of Mao et al, 13 indicating the neuroinvasive potential of SARS-CoV-2. Baig et al 14 suggested that the virus invades neural tissues via the angiotensin converting enzyme-2 receptors on the capillary endothelium. In addition to that, it was reported that viral invasion, hypoxia, hypercoagulopathy and immune-mediated reactions may play role in neuroinvasion. 15 A review published in 2021 reported the prevalence of hearing loss, tinnitus and rotational vertigo as 6%, 14.8% and 7.2%, respectively. 16 Our study supports those findings.
On vestibular testing, no significant difference was found between the patients and controls regarding caloric test results, however decreased gain was found in 1 of the lateral semicircular canals on v-HIT. This was an interesting finding, because the caloric test helps in comprehending the activity of lateral semicircular canals. Thus, the findings of caloric test and v-HIT were not in agreement. However, it is also possible that the balance problems encountered in COVID-19 may be due to subtle changes in the semicircular canals or due to involvement of other parts of the vestibular apparatus.
Little et al. 17 have argued that agents like chloroquine, hydroxychloroquine, azithromycin, lopinavir, ritonavir, interferon, ribavirin and ivermectin which are used in COVID-19 treatment might have caused cochleovestibular symptoms. Therefore, cochleovestibular side effects of the medications should be kept in mind.
Our study has some limitations. Subjective vestibular assessment based on questionnaires was not carried out. We did so, since we wanted to evaluate the audiovestibular system with objective tests, because subjective evaluations may be misleading. The majority of COVID-19 patients complain of balance problems, as observed in the clinical practice. This subjective balance problem may be attributed to systemic effects of the viral disease rather than the audiovestibular effects of COVID-19. The patients who were included in this study had clinical data and audiograms in the database of our hospital. Therefore, the impact of a prior insult on the results could be eliminated.
Conclusion
COVID-19 may affect inner ear functions in the form of a subtle damage to the outer hair cells or lateral semicircular canals. The mild high-frequency sensorineural hearing loss encountered may remain unrecognized unless an audiogram is performed.
Footnotes
Author contribution
All authors declare that they all meet the author criteria.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Ethical approval was taken from the Ministry of Health and the local ethical committee. (protocol: E1-20-718).
Consent about participate and publication
All patients were informed about the study and their informed consents were obtained.
Data availability
Data is available on request from corresponding author.
