Abstract
Substance abuse is a rare but known cause of sensorineural hearing loss (SNHL). We report a case of acute SNHL in a 28-year-old man following an overdose of methamphetamine and incidental ingestion of fentanyl. On initial encounter, he had moderate-to-severe hearing loss in the right ear and severe-to-profound hearing loss in the left ear in addition to acute kidney injury, liver failure, and lactic acidosis. The patient was treated with a two-week course of high-dose steroids and expressed a subjective improvement in hearing. This case highlights the importance of auditory testing following a drug overdose and is one of the only documented cases of hearing loss following methamphetamine use in recent years. There is a paucity of literature regarding the mechanism causing acute SNHL secondary to methamphetamines. Proposed etiologies include neurotransmitter depletion or reduced cochlear blood flow as possible causes of ototoxicity.
Introduction
Methamphetamine is a sympathomimetic amine with psychostimulant properties. It is one of the most used illicit drugs with an estimated 4.7 million Americans reported to have knowingly tried it at least once. 1 Methamphetamine enters well-perfused organs rapidly, including the brain, liver and kidney increasing release and blocking reuptake of monoamine neurotransmitters such as dopamine, norepinephrine and serotonin. Health consequences may occur including myocardial infarction, malignant hypertension, hemorrhagic stroke, and psychosis.2–4 Fentanyl, a synthetic opioid, has been increasingly laced in stimulants such as methamphetamine and others which drastically increases the risk of overdose. 5 While sensorineural hearing loss (SNHL) has been reported as a sequela of illicit drug use, there is sparse literature describing its occurrence with amphetamine or fentanyl overdose. We present a patient with SNHL and concomitant myocardial injury, acute kidney injury with rhabdomyolysis, and liver failure following an overdose of methamphetamine and fentanyl.
Case Report
The patient was a 28-year-old male who presented to the ED after being found unconscious by his mother in his car. A glass pipe and unmarked canister were in the car. Subsequent urine toxicology was positive for methamphetamines and fentanyl and the patient endorses a history of methamphetamine and cocaine use, but denies knowledge of fentanyl use. The patient was arousable but lethargic in the emergency department and immediately complained of hearing loss including all sounds except faint beeping from machines in the room. Communication was limited to writing. He admitted using substances in the past including methamphetamine and cocaine, but he denied memory of the overdosing event and details of the incident. He denied otalgia, otorrhea, tinnitus, vertigo, and any past otologic surgeries or hearing problems. On exam, bedside Weber and Rinne tests on bilateral ears with a 512-Hertz tuning fork were inconclusive as the patient expressed only feeling the vibration and could not hear the tone. Lab results revealed myocardial injury (troponin 5.67 ng/mL and ST depressions on EKG), acute kidney injury with rhabdomyolysis (creatinine 5.33 mg/dL, creatinine phosphokinase 14,683 U/L), liver failure (INR 2.2, total bilirubin 1.4 mg/dL, ALT/AST 8,046/9,145 IU/L), and lactic acidosis (1.8 mmol/L). He began treatment for SNHL with Prednisone PO 60 mg for 14 days with a 6-day taper. Although hyperbaric oxygen treatment was recommended, the patient’s insurance did not cover the procedure, and he declined. An audiogram taken three days following the inciting event showed the right ear with moderate-to-severe hearing loss (56% speech discrimination) and the left ear with severe-to-profound hearing loss (48% speech discrimination). Results at ten days after prednisone was started showed relatively flat, bilateral, SNHL at 80 dB in the right ear and 65 dB in the left ear with unchanged discrimination
1
and a normal type A tympanogram (Figure 1). Bilateral sensorineural hearing loss and normal tympanogram.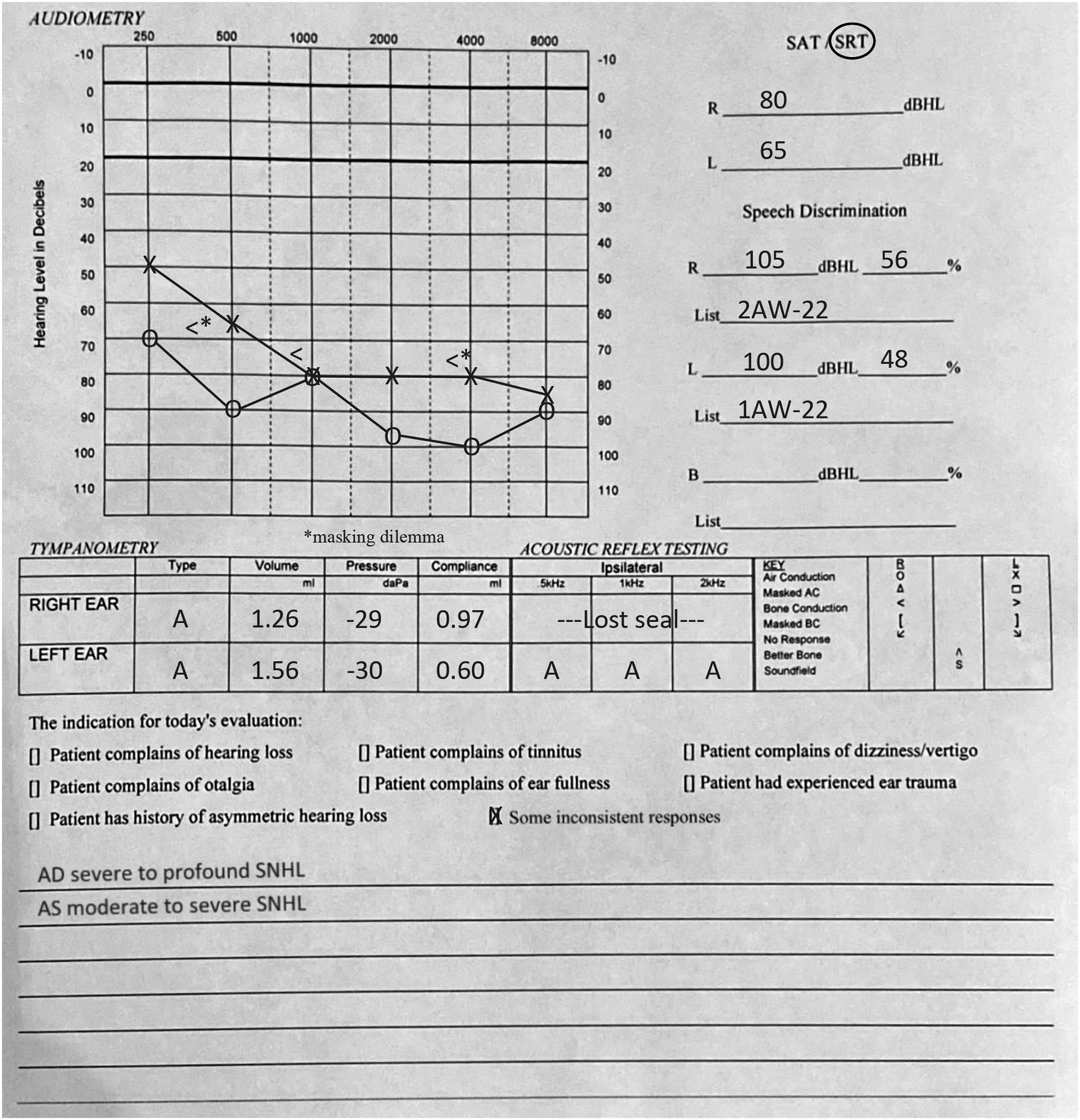
Discussion
Many pharmacologic agents, including antibiotics, heavy metals, and recreational drugs, have been identified as ototoxic. Although evidence exists that establishes the mechanism by which stimulants damage the ear, few data have been presented for amphetamine-induced SNHL. Iqbal reported seven inpatient heavy amphetamine users in Saudi Arabia who developed bilateral hearing loss. 6 Hearing loss was recovered in every case within four to ten days following cessation of chronic use. The author hypothesizes damage to the neurons and depletion of neurotransmitters to be the mechanism of ototoxicity, but evidence establishing the mechanism of amphetamine-induced SNHL does not exist.
Mechanisms of SNHL have been proposed for cocaine, another widely used recreational stimulant, that may provide clues for other stimulants. Shivapuja et al. investigated the mechanism of chronic and acute, high-dose cocaine on ear physiology in rats. Chronic cocaine use reduced cochlear catecholamine innervation, while acute cocaine use reduced inner ear cochlear blood flow. 7 Ischemic change of the temporal lobe is one of the leading theories for amphetamine-induced SNHL given its identification in several other recreational drug overdoses including cocaine. 8
Although our patient’s hearing loss occurred immediately following a drug overdose, our ability to pinpoint the exact cause is limited due to polysubstance abuse and the complex nature of the patient’s presentation. There is no knowledge of the frequency and amount of drug use by the patient, how much methamphetamine or fentanyl was ingested, the timing of the intoxication or whether other substances were consumed that are not included in the urine toxicology panel. Future research to understand the pathophysiology of amphetamine ototoxicity would be beneficial to guide evaluation and treatment.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
