Abstract
Objective
Surgical resection is standard treatment for pleomorphic adenoma (PA) of the parotid gland. A small number (2–5%) of these tumors recur. Recurrence usually necessitates reoperation, which is technically challenging and puts the facial nerve (FN) at risk. The aim of this study is to characterize the recurrent parotid PA population and compare outcomes after surgery for singly recurrent and multiply recurrent tumors.
Methods
This study was a retrospective chart review of patients at a single tertiary care academic medical center who underwent operations for recurrent PA of the parotid gland between 2007 and 2020. Demographic data, details of surgical interventions, pre- and postoperative FN function, and recurrence rates were studied. These factors were compared between patients with singly and multiply recurrent tumors.
Results
Thirty-eight patients met criteria: 4 patients presented for primary PA and subsequently recurred, 26 with a first recurrence, 7 with a second recurrence, and 1 with a fourth recurrence. Multiply recurrent PAs were more likely to require at least partial nerve sacrifice at the time of reoperation (P = 0.0092). Significantly worse long-term FN outcomes were seen following surgery for multiply recurrent PA (P = 0.008). There was no significant difference between the rate of re-recurrence following first revision surgery vs second-fourth revision surgery. Time to reoperation was significantly shorter between the first and second revision surgery than between the primary surgery and first revision (P = 0.0017).
Conclusion
Surgery for recurrent PA incurs high risk to the FN, and this risk appears to increase in the setting of multiple recurrences.
Introduction
Eighty-five percent of parotid tumors are benign.1-3 Of those, 65% are pleomorphic adenomas (PA), making them the most common neoplasms of the parotid gland. 4 PA typically presents during the fourth to fifth decade of life and has a strong female predominance.5,6 Surgical resection with facial nerve (FN) preservation has been established as the standard of care. 7 If left untreated, there is a 1.5–13.8% chance of malignant transformation into carcinoma ex-pleomorphic adenoma.7-10 Following resection, a small number (2–5%) of PAs recur, and recurrent PA can be a complex problem to manage. 7 A major cause of primary recurrence is iatrogenic tumor puncture or spillage following violation of the tumor capsule during resection.11-13 However, this is often underreported and poorly documented. Other risk factors for recurrence include advanced age, location in the deep parotid lobe, increased tumor size, and tumor adherence to the FN. 11
Recurrent tumors often require reoperation, which can be technically challenging and carries a high risk of FN injury. Risk factors for postoperative FN morbidity include surgery type and duration of intraoperative FN exposure.14,15 Larger tumor size is associated with greater postoperative FN dysfunction, likely attributable to the need for more extensive nerve dissection and mobilization.
16
Temporary FN weakness or paralysis is a relatively common finding in the immediate postoperative period, occurring in 10–30% of cases and most commonly involving the marginal mandibular branch.
17
Permanent unilateral facial paralysis is much less common, occurring in less than 1% of patients.16,17 The risk for permanent FN damage increases with successive surgeries. Complicating factors in surgery for recurrence include the need to dissect a scarred tumor bed and a predominance toward multifocal disease (Figure 1).14,16,17 Axial (A) and coronal (B) T2-weighted magnetic resonance images with fat suppression demonstrating a multifocal recurrent pleomorphic adenoma (white arrows) of the left parotid gland.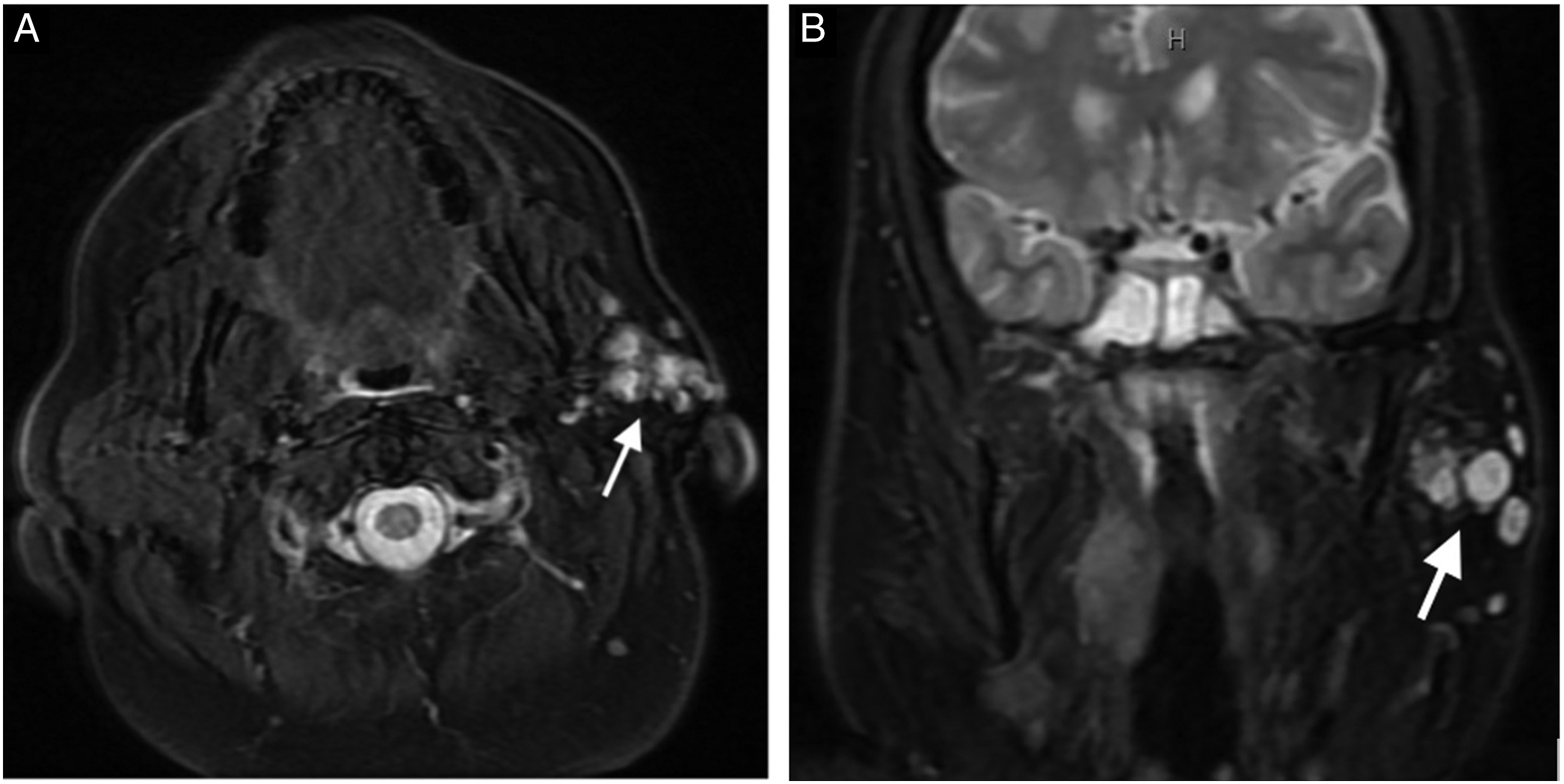
There is a lack of consensus in available literature regarding the ideal strategy for nerve-preserving management of recurrent PA. A superficial parotidectomy involves FN dissection and removal of parotid tissue lateral to the nerve. 17 A total parotidectomy involves a complete FN dissection with removal of tissue lateral and medial to the nerve. Both carry risk of FN damage. Damage may occur through stretching, thermal injury, or transection of the main trunk or any of its distal branches. 13
Intraoperative facial nerve monitoring is employed by some surgeons and has been shown to decrease the incidence of postoperative facial nerve weakness in retrospective studies. 18 This is suggested to be particularly beneficial in patients with large tumors and in recurrent disease.19,20 However, others propose that intraoperative facial nerve monitoring may result in less careful dissection due to overreliance, which may paradoxically lead to increased facial nerve injury. 21
Radiation therapy (RT) plays a controversial role in treatment of recurrent pleomorphic adenoma. RT is often considered for adjuvant treatment in cases where complete PA excision is not possible, in cases involving multiple recurrences, and with multifocal disease. 22 While some studies have found no significant advantage in tumor control rate following postoperative RT, there is some evidence that it may provide better local control than surgery alone.23-26 Many remain reluctant to employ RT due to its side effect profile and the risk of RT-induced malignancy.26,27 Additionally, there is a paucity of prospective data comparing the use of adjuvant RT to surgery alone, and thus there is a lack of consensus as to whether RT should be routinely used in the treatment of PA. 22
The purpose of this study was to characterize the recurrent PA population at a single tertiary referral center between 2007 and 2020 and to determine whether postoperative outcomes differed following surgery for a first recurrence vs surgery after multiple recurrences.
Methods
An ICD-9/10 search of the EMR was conducted for patients with a diagnosis of “benign neoplasm of a major salivary gland.” The results were refined using a keyword search for “pleomorphic adenoma” to identify all patients who received a diagnosis of PA between 2007 and 2020. Those with poor case documentation, primarily extra-parotid tumors, or surgical pathology inconsistent with PA were excluded.
Data regarding the patient’s FN function at presentation, as well as tumor histopathology, size, and location, were collected. Intervals between surgery and recurrence, the number of recurrences, and the postoperative complications were noted. Data regarding radiotherapy, when used, were obtained. Demographic factors such as age, race, and sex were recorded. This study was deemed exempt by the Loyola University Chicago Institutional Review Board.
Statistical analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA). Data were analyzed using chi-squared and student t-tests with a significance threshold set to P < .05.
Results
Five hundred and four patients were identified as having been diagnosed with PA. After applying our exclusion criteria, we were left with 38 patients who underwent surgery for recurrent PA of the parotid gland between 2000 and 2020 (Figure 2). No patients were lost to follow-up. Flowchart of data collection (ICD = international classification of diseases; CUI = controlled unclassified information; PA = pleomorphic adenoma; PPS = parapharyngeal space; XRT = external beam radiation therapy).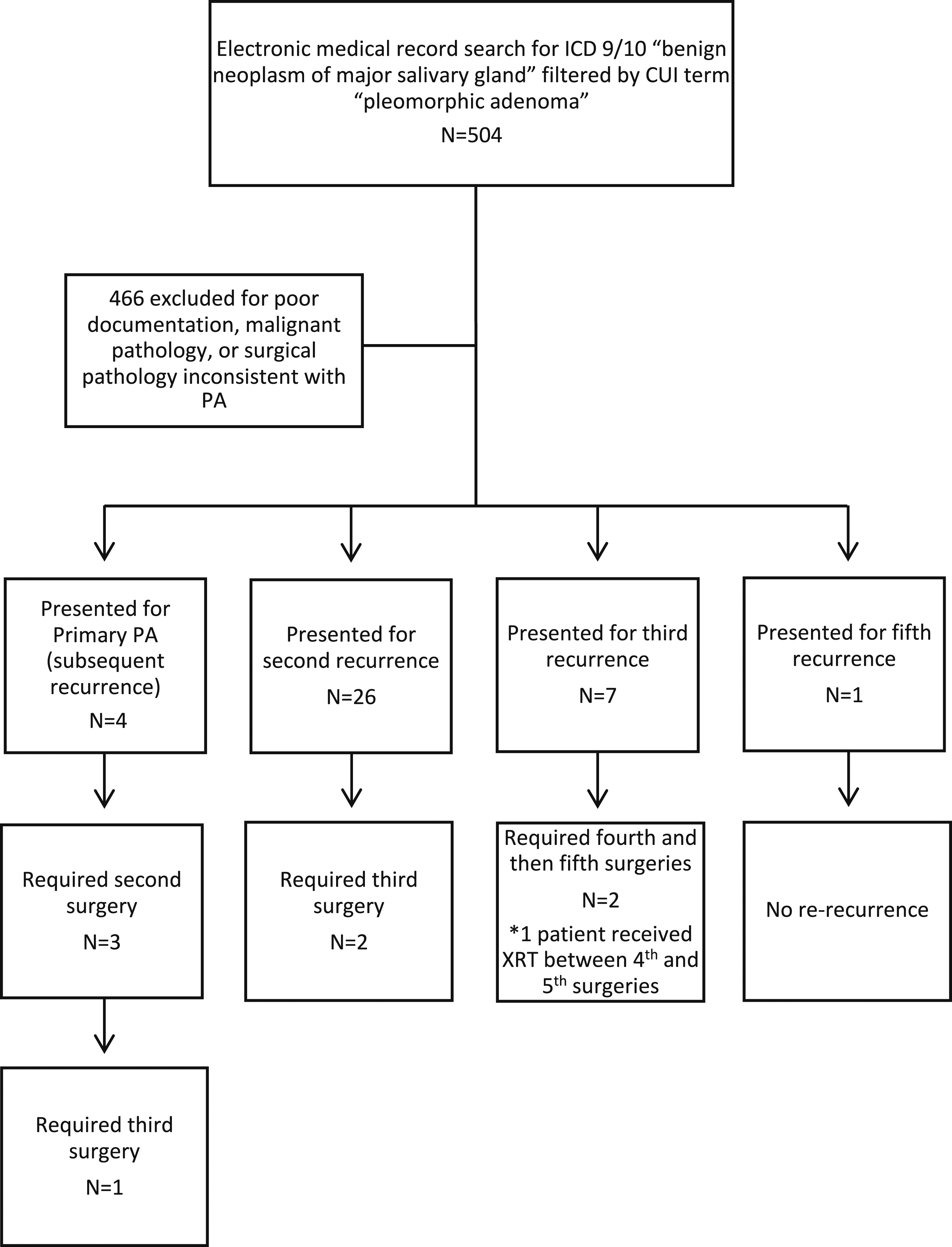
Twenty-six of 38 (68%) patients were female. The age at the time of the first surgery ranged from 15 to 66 years, with an average age of 36.5 years. Twenty-six (68.4%) presented with a first recurrence, 7 (18.4%) with a second recurrence, and 1 (2.6%) with a fourth recurrence. Twenty-three (60.5%) of the patients who presented with recurrence had a prior surgery at an outside hospital. Four (10.5%) patients first presented for primary PA, underwent surgery, and subsequently recurred. Nineteen (50%) tumors were right sided. Thirty-one (81.6%) had House-Brackmann (HB) grade I FN function on presentation, 2 (5.3%) had HB grade II, and 1 (2.6%) had HB grade VI. Five (13.2%) had Frey syndrome documented during their initial clinic visit.
Description of procedures performed.
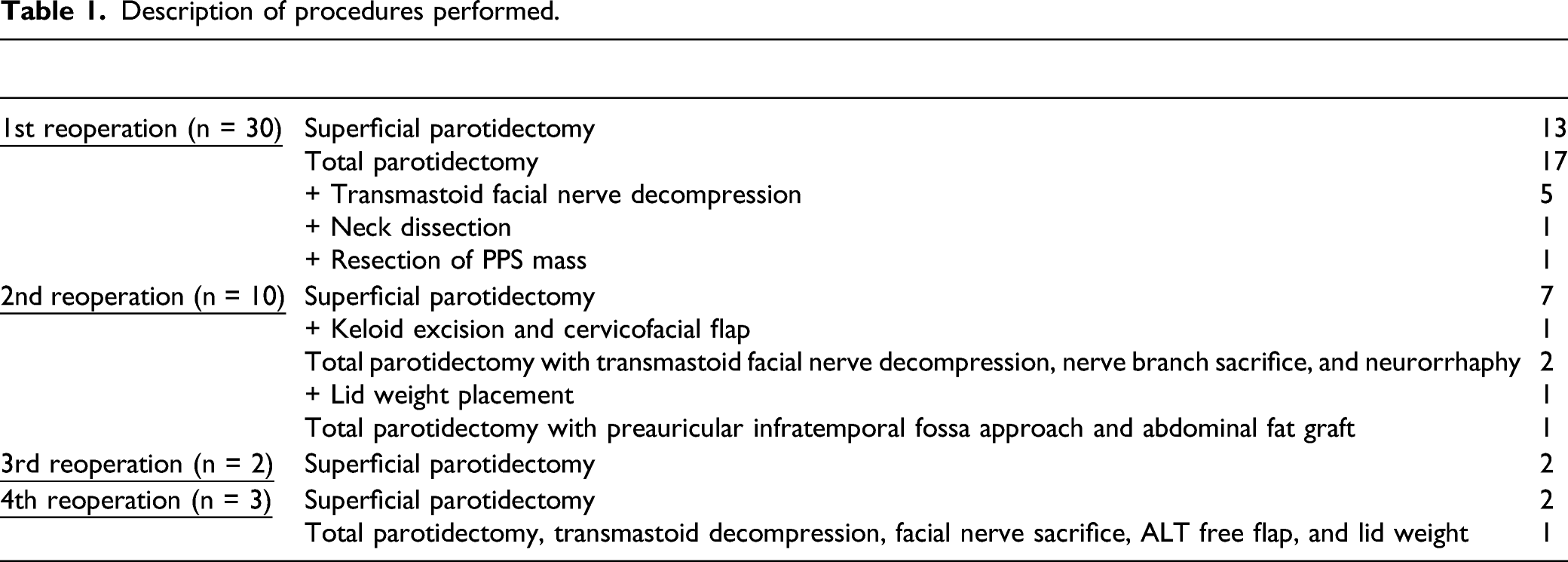
When comparing surgeries for first recurrence (FR) and those for multiple recurrences (MR), surgeries for MR were more likely to require at least partial nerve sacrifice at the time of reoperation (P = .009). Surgeries for MR had significantly worse FN outcomes at long-term follow-up (>60 days postoperatively) than did surgeries for FR (P = .008) (Figure 3). When comparing the FR and MR groups, there was no significant difference in FN function documented at preoperative or immediate (<30 days) postoperative clinic visits. Whether an operation was for FR or MR showed no significant correlation with surgical margin status or likelihood of finding multifocal disease. Average preoperative vs postoperative facial nerve function as graded on the House-Brackmann scale. Error bars denote standard deviation.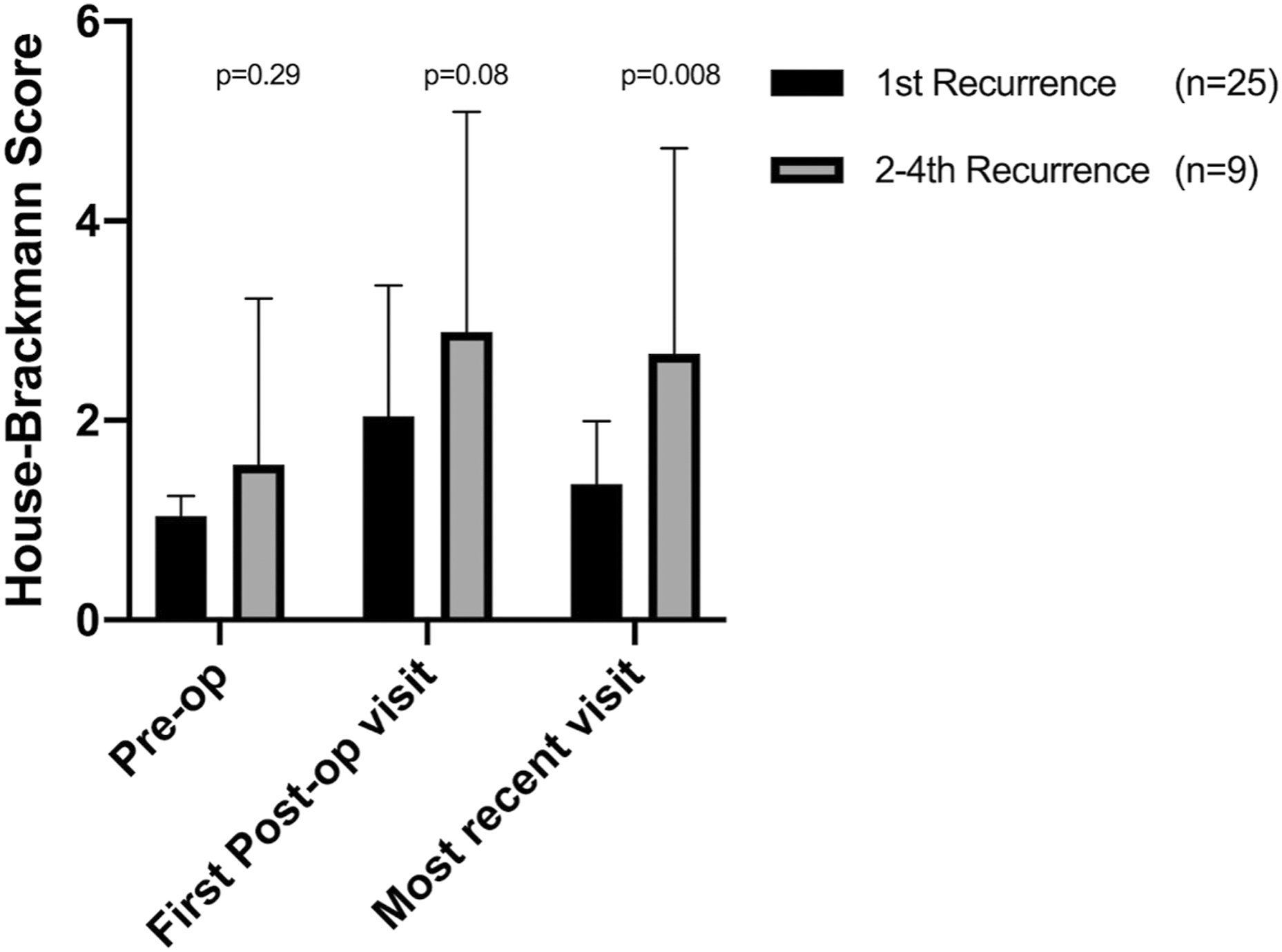
When comparing re-recurrence rates following surgeries for FR and MR, there was no significant difference. However, the time to reoperation was significantly longer between the primary and first revision surgeries than between subsequent revision surgeries (P = .002).
Discussion
Consistent with previous reports, our patients were treated for their primary PAs at an average age of 36.5.5,6 Our re-recurrence rates following first reoperation and those following further reoperations fall within the broad ranges reported in existing literature (11–50% and 20–60%, respectively).5,6
Interestingly, the time to the first reoperation was significantly longer than the time between subsequent reoperations. This may indicate that tumors recur more quickly after the first reoperation than after a primary resection. If so, this would underscore the need for close and careful monitoring following resection of a recurrent PA. It is also possible that subsequent recurrences are treated sooner because patients are more likely to have frequent visits with their physician following reoperation, which would allow re-recurrences to be identified earlier. In contrast with existing literature, when comparing outcomes after surgery for singly recurrent and multiply recurrent tumors in our group, there was no significant difference in the incidence of positive margins on pathology or in the likelihood of finding multicentric disease. 28
On initial presentation, most of our patients had normal FN exams documented. Following surgery for both singly and multiply recurrent tumors, there was a significant decrease in ipsilateral HB score as documented preoperatively and at office visits occurring within the first month. While FN function tended to gradually improve over time, on average, HB scores did not return to baseline at follow-up. As discussed, the risk of temporary and permanent FN injury during these surgeries is well documented. 13 The use of intraoperative FN monitoring showed no significant difference in postoperative FN function. This finding may suggest that intraoperative FN monitoring is less useful in recurrent cases as successive surgeries may predispose the FN to a greater risk of injury. We found no significant difference in FN function between patients with singly and multiply recurrent PAs at the preoperative and immediate postoperative visits. However, there was a significant difference in persisting FN dysfunction between patients with singly and multiply recurrent PAs at long-term follow-up. This may indicate that repeated exposure and dissection of the FN carries greater risk of damage of a more permanent nature.
This study is not without limitations. Data were collected via retrospective chart review. This precluded random sampling. A longitudinal, prospective collection of data would have allowed for a more accurate analysis and understanding of causal relationships but would be difficult given the rarity of this condition. There was likely an element of selection bias given that many patients were lost to follow-up, limiting the study population. Additionally, several variables that could have influenced the interpretation of our data were omitted from a number of patient charts.
Future studies are warranted to clarify specific mechanisms by which PAs recur as this may help guide treatment strategies for primary and recurrent disease. Furthermore, the heterogeneous presentation of recurrent PA makes it important to characterize prognostic factors for postoperative FN function. This may allow otolaryngologists to better tailor their treatment approach to each patient.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
