Abstract
Objectives
Saccharin test (ST) is a convenient method to assess the efficiency of mucociliary clearance, the primary defense mechanism of the upper airways’ tract. The study objectives are to: (1) substantiate its short- (3 days) and long-term (30 days) repeatability; (2) assess its tolerability; (3) conduct a systematic literature review and to compare our results with the existing evidence.
Methods
Twenty-nine healthy subjects were enrolled in an observational prospective study to perform an ST on three separate visits (at baseline; at follow-up visits at day 3 and at day 30). Transit times were recorded and self-reported nasal and general symptoms noted. A systematic review of the literature was conducted to compare our results with the existing literature.
Results
The mean values (±SD) of ST transit time (STTT) were 7.085 (±2.19), 7.788 (±2.11), and 7.790 (±2.06) minutes at baseline, day 3, and day 30, respectively. Significant linear regression analysis was observed between day 3 and baseline (r = .193; P = .019) and day 30 and baseline (r = .182 P = .024). Significant agreement for the intrasession repeatability was observed with an ICC = .354 (P = .001). Outcomes’ comparisons between baseline vs day 3 (P = .197) and baseline vs day 30 (P = .173) were not statistically significant. ST was well tolerated. Concordance with existing literature’s data and high level of STTT repeatability were confirmed by the qualitative analysis.
Conclusion
STTT reproducibility was good both in the short- and long-term. ST tolerability was very good. Our study data are consistent with the existing literature, indicating ST as a sound methodology for detection of early respiratory health changes and for specific regulatory application in respiratory research.
Keywords
Introduction
Mucociliary clearance (MCC) is part of the innate immunity system, a primary defense mechanism of the respiratory tract; it is designed to provide effective protection against viral and bacterial pathogens, particulate matters, and gaseous material entering the respiratory system.1,2
The MCC system operates by trapping debris and microorganisms in the mucus layer that lines the airway epithelia and clearing them from the respiratory tract; clearance is dependent on effective ciliary beating that propels the mucus blanket toward the larynx for elimination.3,4 Impairment of MCC predisposes to chronic infection of the nose, paranasal sinuses, and respiratory tract. 4
An efficient mucociliary transport relies on coordinated ciliary beating and normal mucus rheology and volume. 5 Dysfunction of the MCC is commonly associated with the impairment of the cilia–mucus interaction, frequently associated with airway inflammatory conditions such as in asthma, allergic rhinitis, chronic rhinosinusitis, and chronic obstructive pulmonary disease (COPD).6–8
Of note, the clearance rate of small particles from the healthy individuals’ nose reflects that of tracheobronchial tree clearance obtained by radioisotopic techniques. 9 Nasal mucociliary clearance transit time (MCCTT) could be measured with the saccharin test (ST), which is non-invasive, well tolerated, and easy to perform. 10 In healthy individuals, MCCTT does not normally exceed 20 minutes. 11 Impaired MCCTT as measured by the saccharin test has been reported in asthma, rhinitis, sinusitis, cystic fibrosis, and bronchiectasis.11–13 Instead, a 30-minute measurement is a commonly accepted threshold for discriminating healthy people from those with dysfunctional MCC.14,15
In the literature, different authors have reported a great variability for the results of STT in the presence of associated comorbidities.11–15 Nasal mucociliary clearance is influenced by two main components, the rheological characteristics of the mucus and the ciliary propulsive properties. Individual factors that can cause a prolonged duration of nasal mucociliary clearance include exposure to tobacco smoke, nasal pathologies, the use of certain drugs (antihistamines, antidepressants, and corticosteroids), and environmental factors (humidity, temperature, and air pollution).14,15
Schuhl et al, in 1995, analyzed the role of changes in nasal mucus rheology in patients with rhinitis and nasal mucociliary clearance. 12 The authors reported a significant difference between allergic patients at 10.27 minutes for allergic rhinitis and 8.8 minutes for healthy subjects (P < .05). Later, Sherly et al the procedural standardization of the saccharin test, stressing the importance of regulative rules such as operating in a controlled temperature and humidity environment, without dust, and avoiding smoking, coughing, sneezing, or eating until the test is completed.
The presence of these important cofactors raises the need for careful standardization of the TS to minimize variability and increase test confidence by improving internal consistency. The literature offers incomplete test descriptions, poor data on its tolerability, and limited information on its repeatability at test–retest.13–16 The study objectives were to: (1) substantiate short- and long-term repeatability of ST transit time (STTT); (2) assess its tolerability; (3) conduct a literature review and to compare our results with the existing evidence.
Methods
Study participants
Healthy subjects (≥18 years old) were recruited from the Hospital staff. They were invited for a screening visit to check their eligibility. The study design is illustrated in Figure 1. Eligibility was determined by the inclusion and exclusion criteria noted below: Flow-chart. Study design. Abbreviations, CEAH: Center of Excellence for the Acceleration of Harm; URTI: upper respiratory tract infection; CRS: chronic rhinosinusitis; COPD: chronic obstructive pulmonary disease; MCTTST, MCCTT-ST: mucociliary clearance transit time by saccharin test.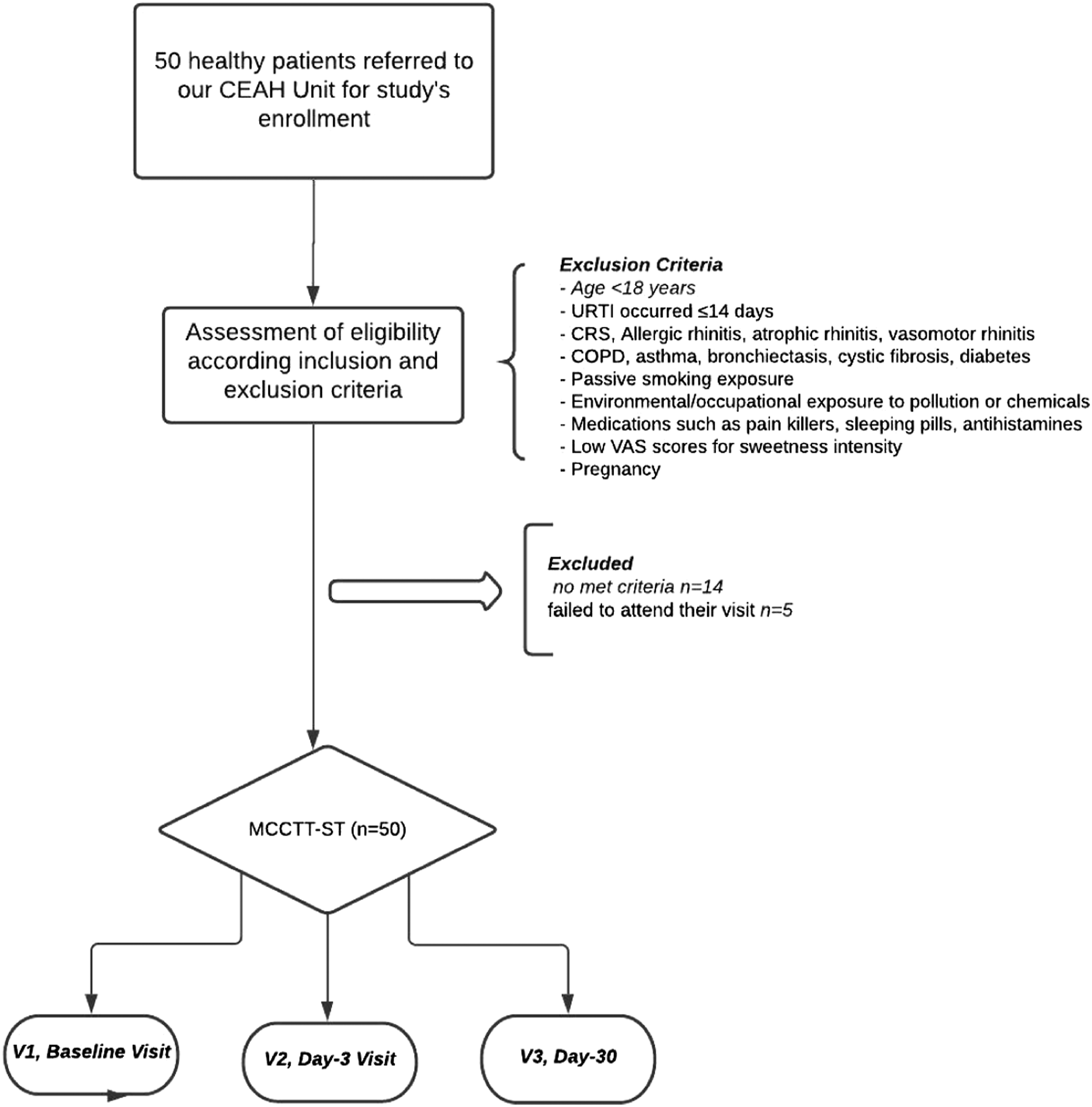
Inclusion criteria:
• > 18 years old • No smoking • Former smoking with at least 6-months abstinence from last cigarette • No vaping • Former vaping (at least 6-months abstinence from last vape)
Exclusion criteria:
• Any conditions potentially impairing cilia–mucus interaction or interfering with MCCTT measurements, including: ⁃ Recent (less than 14 days) history of flu; ⁃ chronic rhinosinusitis, infectious rhinitis, allergic rhinitis, atrophic rhinitis, and vasomotor rhinitis ⁃ any medical disease that may interfere with MCCTT measurements including COPD, asthma, bronchiectasis, cystic fibrosis, diabetes, and deviated nasal septum; ⁃ significant exposure to passive smoking; ⁃ significant environmental/occupational exposure to pollution or chemicals (eg, living in heavily polluted urban or industrial areas), or occupational exposures from employment in chemical or metallurgy industries; ⁃ current use of medications interferes with MCCTT measurements including pain killers, sleeping pills, and antihistamines; poor individual ability to detect sweetness (score below the 25 mm mark on the 0–100 mm VAS for sweetness intensity rating); and ⁃ pregnancy
The local Ethical Review Board “Comitato Etico Catania 1” approved the study and participants gave written informed consent.
Methodological protocol
The study consisted of four morning visits (to avoid the possible influence of circadian rhythms); a screening visit, a baseline visit (V1), a day 3 (±1 day) follow-up visit (V2), and a day 30 (±3 days) follow-up visit (V3) (Figure 2). Subjects were asked to abstain from drinking coffee/caffeinated drinks for at least 4 h prior to each study visit, as these substances can speed up mucociliary function because by stimulating autonomic control.17–19 Study design. Methodological protocol performed.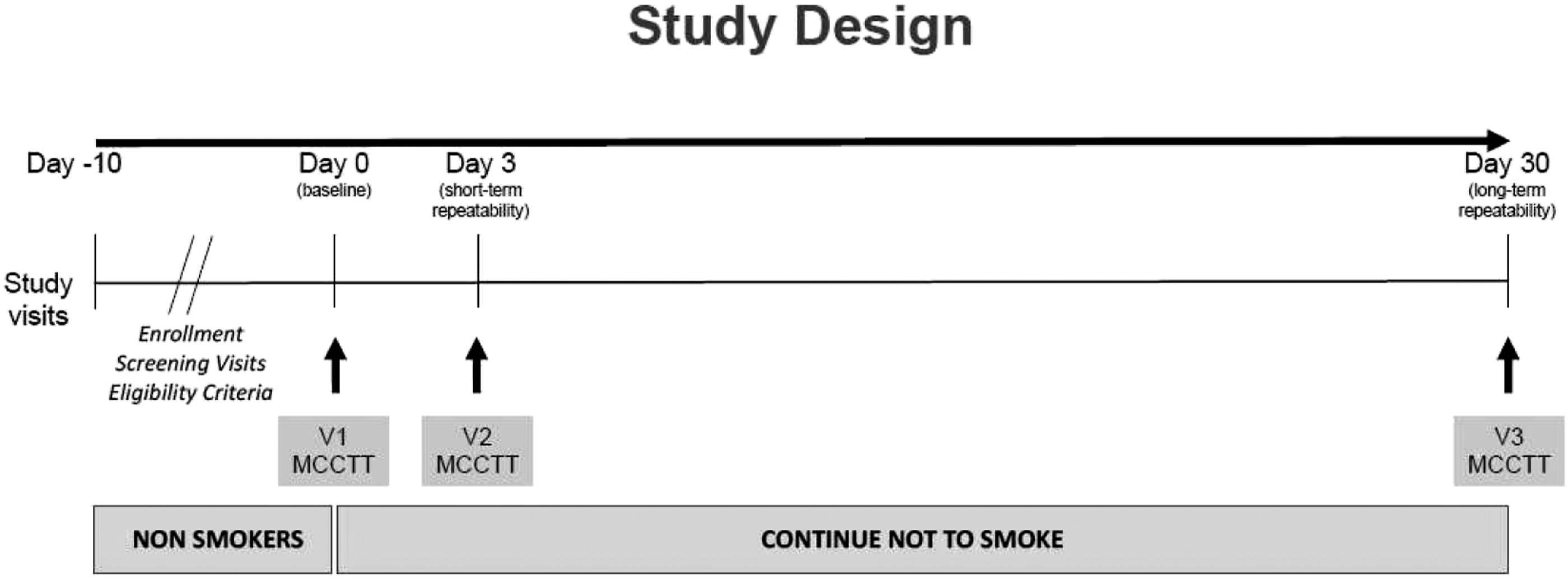
Screening visit
Potential participants attended a screening visit to verify eligibility criteria: socio-demographic data, medical history, medication usage, and smoking/vaping history were noted. Subjects with an exhaled carbon monoxide level >7 parts per million (ppm) were not enrolled in the study, to exclude exposure to cigarette smoke or to environmental sources of carbon monoxide (eg, passive smoking). At screening and prior to enrolment, subjects were tested for their ability to detect sweetness. Perception of sweetness intensity was rated a 0–100 mm VAS. After rinsing the mouth with water and wiping the tongue dry with a paper towel, subjects were instructed to smear a saccharin tablet (Mini-sweeteners; Hermesetas; Switzerland) all around the surface of their tongue. They were asked to rate the intensity of sweetness perception on a 0–100 mm VAS; ratings ranged from “not at all sweet” (at 0 mm) to “extremely sweet” (at 100 mm). Anybody below the 25 mm mark on the VAS was excluded from participation. Eligible subjects were invited to attend the baseline visit.
Baseline visit (visit 1)
Baseline visits were carried out within 10 days of the screening visit. Eligibility criteria were verified once more.
Before beginning the saccharin test, subjects’ nose was rinsed with warm saline (NaCl 0.9% solution). Participants were then invited to acclimatize at controlled environmental conditions (temperature 21–24°C; relative humidity 30–50%) for at least 45 min, during which blood pressure (BP), heart rate (HR), and body mass index (BMI) were measured.
The saccharin test was then carried out (the procedure is detailed in the “Saccharin Test” section below) and baseline transit were times recorded. Self-reported nasal and general symptoms were noted.
Subjects were instructed to avoid taking medications like pain killers or sleeping pills and invited to attend next study visit (V2), to complete the programmed study assessments/procedures.
Day-3 visit (visit 2)
Visit 2 was carried out within 3 (±1) days of the baseline visit. Eligibility criteria were verified once more and subjects were instructed to avoid taking medications like pain killers or sleeping pills within 5 days of the study visit. Saccharin tests were repeated to assess short-term repeatability. BP, HR, and symptoms were noted.
Day-30 visit (visit 3)
The final visit was carried out within 30 (±3) days of the baseline visit. Subjects were instructed to avoid taking medications like pain killers or sleeping pills within 5 days of the study visit. After re-checking eligibility criteria, saccharin tests were repeated to assess long-term repeatability. BP, HR, and symptoms were noted.
Saccharin test method
Room preparation: Saccharin tests were carried out in an air-conditioned examination room. Ambient dehumidification (dehumidifier; Drydigit17; Argo) was programmed 2–3 hours before each study visits to optimize environmental conditions (ie, temperature 21–24°C; relative humidity 40–60%), which were monitored and recorded by a digital thermometer/hygrometer (TP55; ThermoPro) placed on the top of a table in the room.
Instrument’s preparation: Sterile nasal speculum (Hartmann Nasal Speculum; stainless steel. Medium size) and nippers (Malleus Surgical Nippers; stainless steel) were used to position the saccharin tablet (Mini-sweeteners; Hermesetas; Switzerland) inside the nose.
Subjects’ preparation: Nasal washings were carried out by instilling 1.5–2 ml warm saline (NaCl 0.9% solution) into each nostril with a disposable Pasteur pipette. This procedure was repeated twice for each nostril. Subjects were then asked to gently blow the nose to remove any excess of fluids (ie, secretions and saline solution). After nasal washing, subjects were invited to acclimatize for at least 45 min. at controlled environmental conditions.
Saccharin Test: The saccharin test method described here has been modified from that of Andersen et al. 10 Subjects were asked to sit and relax on a comfortable reclining chair. After 45 min acclimatization period, the research investigator (or ENT research nurse) invited the volunteer to slightly raise and tilt the head backward. While illumining a nostril (the one with better nasal breathing as per indication of the subject—the same nostril will be used for all tests providing patency is maintained throughout study visits) with the medical headlight and widening it by using a nasal speculum, the research investigator (or ENT research nurse) identified the small crest that marks the tip of the inferior turbinate. The nipper clasping a saccharin tablet was guided through the speculum. The tablet was gently placed on the medial face of the inferior turbinate, about 1 cm behind its anterior end. The nipper and nasal speculum were withdrawn paying attention not to trigger any sneezing, and a chronometer was started. Subjects were asked to swallow a few times every minute until perceiving the “sweet taste” of saccharin. Self-reported nasal and general symptoms were noted at 3 and 10 min after placing the saccharin tablet in the nose.
Throughout the test, subjects were instructed to avoid to sniff, sneeze, eat, drink, walk, talk, cough, scratch, or blow their nose. If this happens, the test is terminated and subjects re-scheduled.
If at Visit 1 sweet taste is not perceived within 30 minutes, the volunteer is withdrawn from the study. If—at visits 2 and 3—sweetness is not perceived within 30 min, the test is stopped and transit time recorded as “>30 minutes”.
Protocol data extraction and outcomes for a systematic review of the literature
The authors A.M and A.G. analyzed the literature’s data. The study team members solved any disagreements through a discussion. Consequently, the included studies were analyzed to achieve all available data and guarantee eligibility among subjects enrolled. The patient’s features, symptoms, diagnostic procedures, and outcomes were collected. Moreover, we collected the following information: author data, sample size, design of the study and year, statistical analysis, results, and conclusions. The authors of the included studies if required for missing data were contacted using correspondence author’s email or Research Gate (http://www.researchgate.net/).
Electronic database search
We performed a systematic review of the current literature according to the PRISMA checklist for review and meta-analysis 20 and through the PICOS approach. 21
We searched PubMed, Scopus, and Web of Science electronic databases for studies evaluating nasal mucociliary clearance time through saccharin transit test of the literature to date available by two different authors, through MeSH, Entry Terms, and keywords related. The search keywords were the following: “mucociliary clearance,” “nasal mucociliary,” “transit time,” “saccharin test,” “repeatability,” “repeatable”, “reproducibility,” and “reproducible”. We have researched the current literature on this subject. All the titles and abstracts of papers available in the English language were examined by the investigators. The “Related articles” option on the PubMed homepage was also searched via title and abstract search before including them. We used Reference manager softwares (EndNote X7®, Thomson Reuters, Philadelphia, PA for bibliographic management) to collect references and remove duplicates.
The titles and abstracts were reviewed independently by two screeners. Discrepancies were resolved by discussion.
We used the research protocol according to the approved reporting items' quality requirements for systematic review and meta-analysis protocols (PRISMA) declaration. 20 The studies' quality assessment (QUADAS-2) instrument to estimate the included studies' study design features was adopted, and the results of the risk of bias were presented descriptively. 22
Furthermore, we assessed the probable risk of bias in observational studies through the Joanna Briggs Institute Critical Assessment Checklist for Observational Studies. 23
After the title and abstract review, papers were independently reviewed for inclusion with the following criteria. We thus identified and screened full texts for original data, and the related references were retrieved and checked manually to identify other relevant studies.
Eligibility criteria
All studies that met the following criteria were included: 1. Original articles; 2. the article was published in the English language; 3. the studies included nasal mucociliary time evaluation through saccharin transit test; 4. the studies reported detailed information on saccharin transit test repeatability; 5. we excluded from the study case report, editorial, letter to the editor, or review; and 6. studies including animal models were also excluded from the analysis.
Synthesis of results
Due to different laboratory procedures, and various comorbidities identified, the outcomes could influence the results of quantitative analysis. Therefore, we performed a narrative synthesis employing the guidelines of the Synthesis without Meta-analysis reporting items.
Statistical analysis
Linear regression analysis was performed between the measurements obtained at V1 and those obtained at V2 to assess the short-term repeatability of the saccharin test. Similarly, long-term repeatability was evaluated by linear regression analysis between the measurements obtained at V1 and those obtained at V3. Scatter plots of linear regression analyses were generated to visualize repeatability results. Moreover, “Bland and Altman” plots were generated to describe agreement between two quantitative measurements (V1 vs V2 and V3 vs V1). Assessment of the difference from zero of the mean difference between 2 measurements was performed by 1-sample t test. Finally, to evaluate the agreement in the repeatability of the intrasession measurements among the three visits (V1, V2, and V3), the intra-class correlation coefficient (ICC) was computed using a single-measurement, absolute-agreement, two-way mixed-effects model. 24 To identify individual variables that may influence the test results, we carried out multiple regression tests. All analyses were considered significant with a P-value< .05. Analyses of data were performed using R version 3.4.3 (R Core Team, 2017). Furthermore, the random-effects modeling was adopted (standard error estimate = inverse of the sample size) to estimate the summary effect measures by 95% confidence intervals (CI); subsequently, forest plots were generated via the Review Manager Software (REVMAN) version 5.4 (Copenhagen: The Nordic Cochrane Centre: The Cochrane Collaboration). The inconsistency (I2 statistic) was calculated, and the values for low inconsistency = 25%, moderate inconsistency = 50%, and high inconsistency = 75% were established.
Results
Study participants
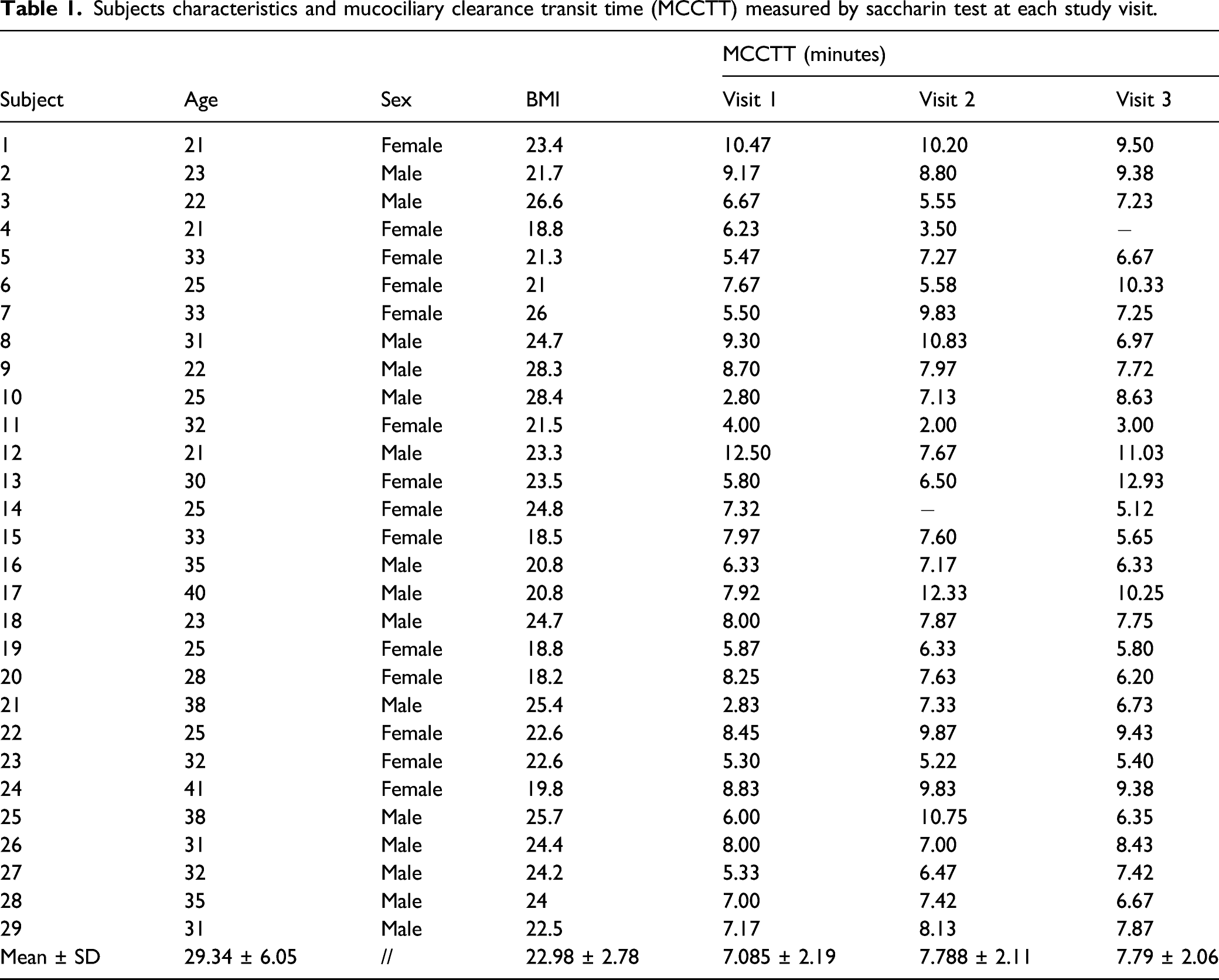
Subjects characteristics and mucociliary clearance transit time (MCCTT) measured by saccharin test at each study visit.
Tolerability of saccharine test
Side effects were infrequently reported in the course of the saccharin test; for example, nasal itch, nasal irritation, and sneezing at 3 min were reported in 20.7%, 16.1%, and 4.6% of the cases (average for V1, V2, and V3 combined), respectively. However, occurrence of side effects was transient and no symptoms were reported at 10 min. Moreover, no significant changes in mean (±SE) resting heart rate and systolic/diastolic blood pressure were observed.
Repeatability of mucociliary clearance transit time by saccharin test
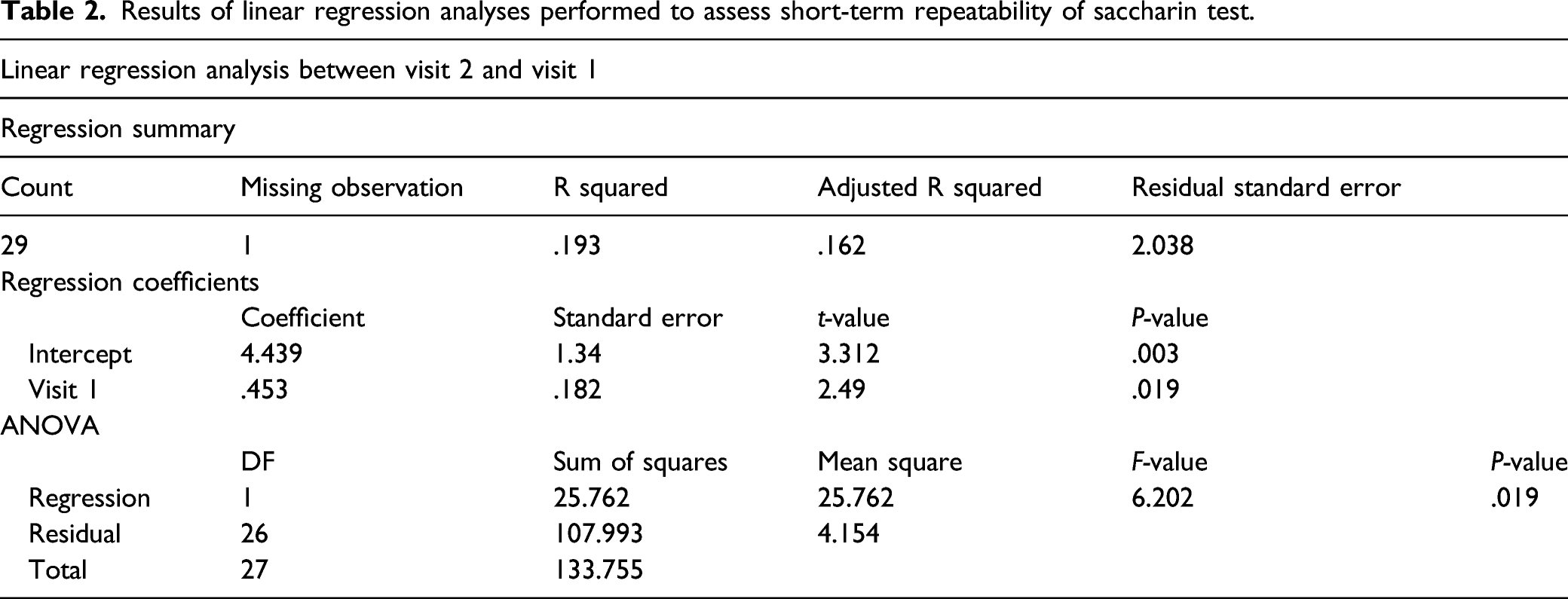
Results of linear regression analyses performed to assess short-term repeatability of saccharin test.
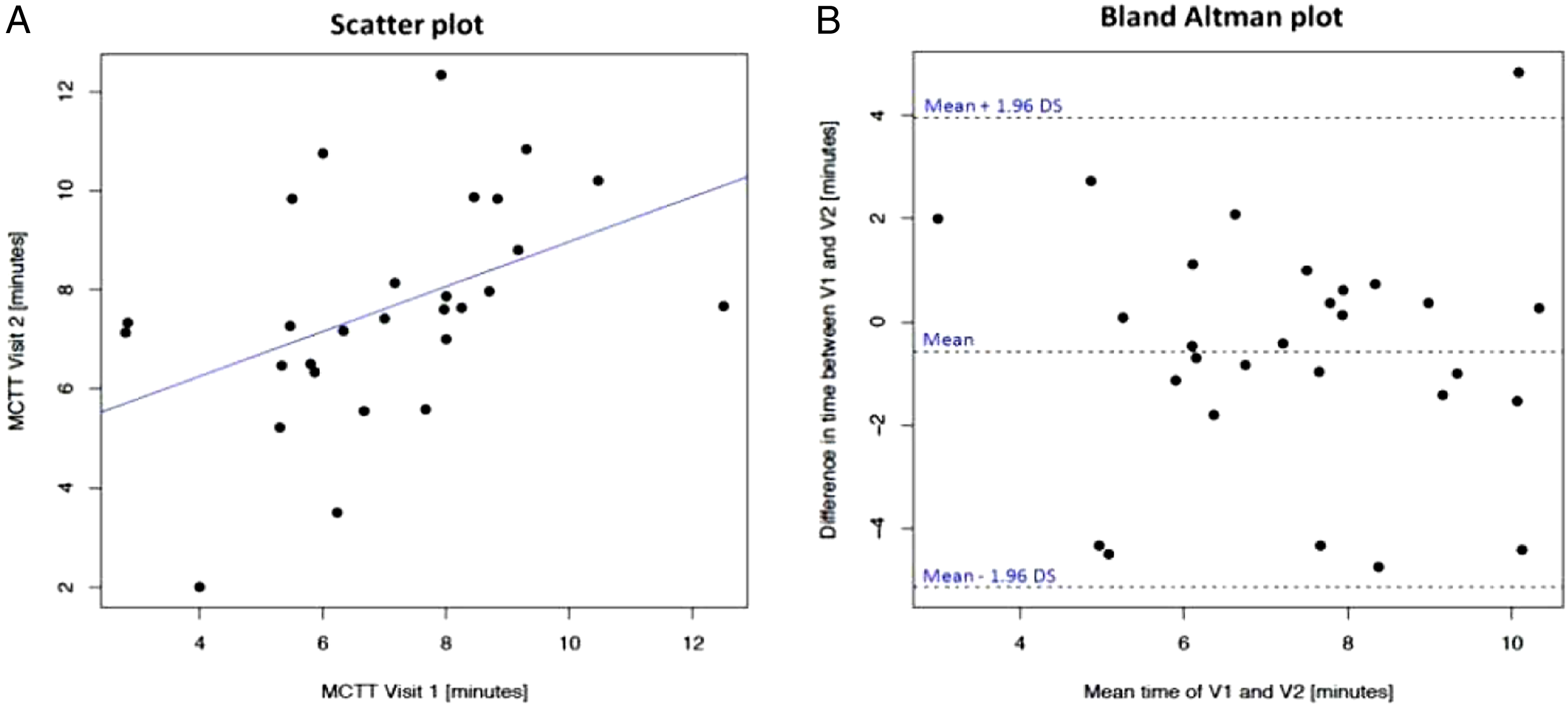
Saccharin test short-term repeatability. Panel A shows the scatter plot of regression analysis of MCCTT measurements between V2 and V1. Panel
Results of linear regression analyses performed to assess long-term repeatability of saccharin test.
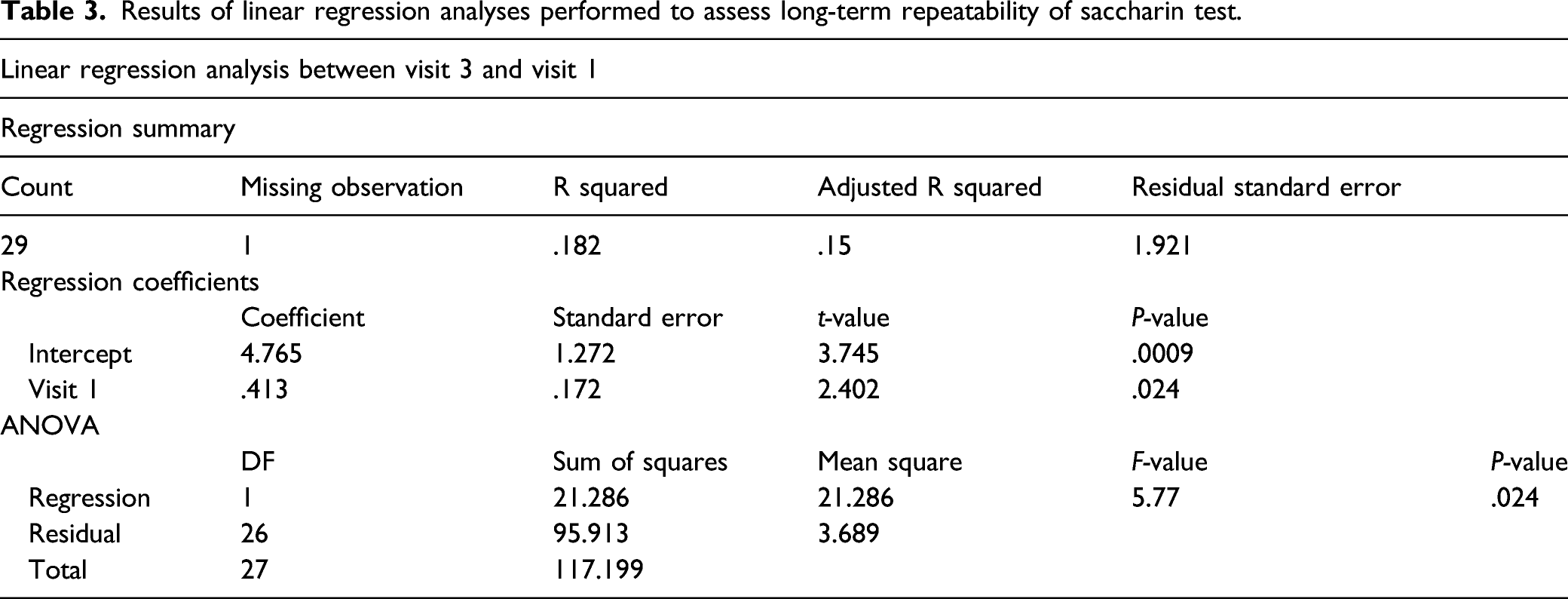
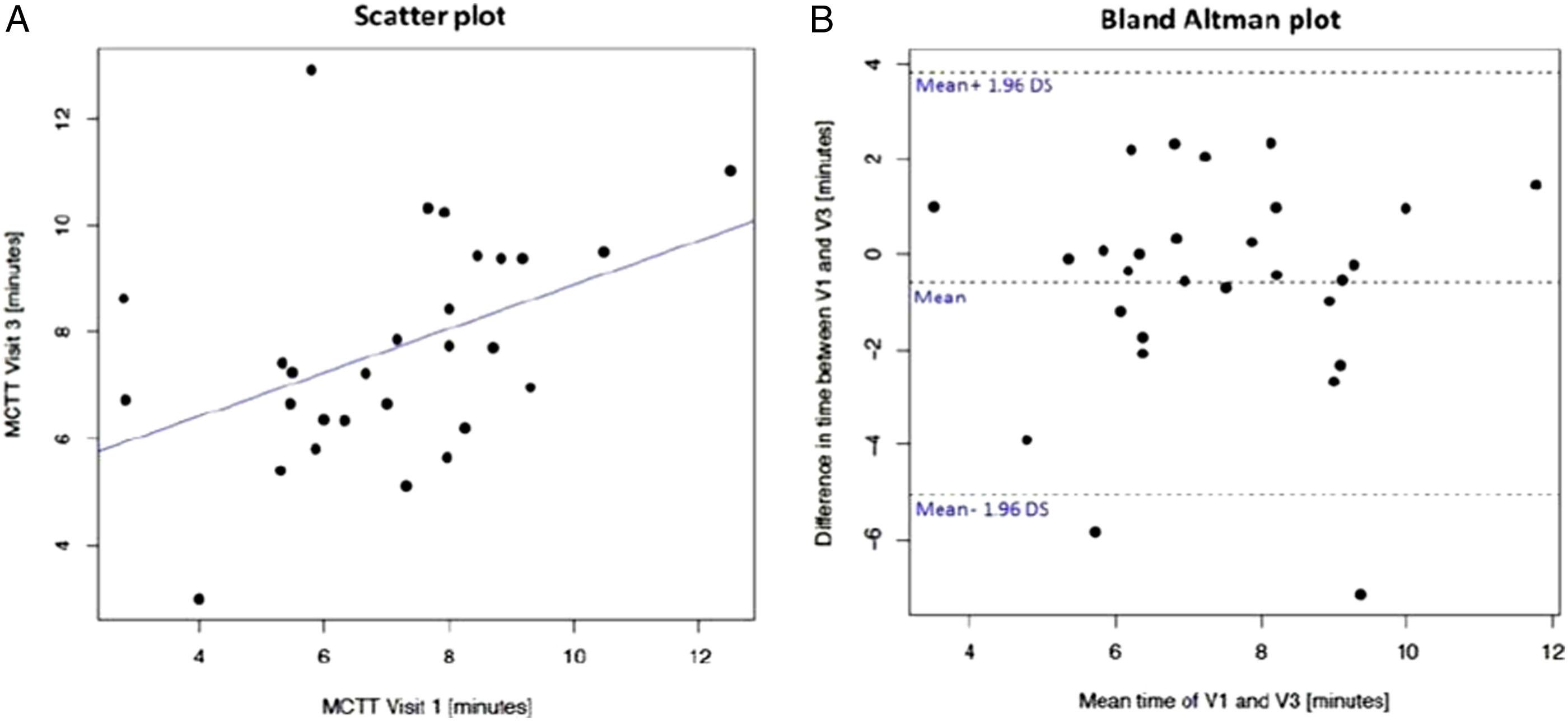
Saccharin test long-term repeatability. Panel A shows the scatter plot of regression analysis of MCCTT measurements between V3 and V1. Panel
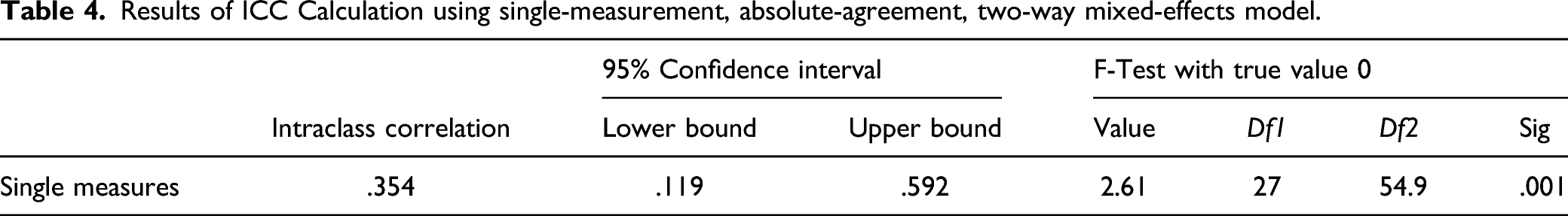
Results of ICC Calculation using single-measurement, absolute-agreement, two-way mixed-effects model.
Comparison with existing literature data on saccharine test
Retrieving research studies
The systematic review of the literature identified 301 potentially relevant studies (Figure 5). After removing the duplicates and applying the criteria listed above, an overall number of 298 records screened were potentially relevant to the topic. Through the records analysis and subsequent articles' full-text screening, we excluded all the studies that did not match inclusion criteria (n = 282). The remaining 18 papers were included in qualitative synthesis papers for the data extraction. Due to the meta-analysis established criteria, we did not perform a quantitative analysis. A graphical display of bias analysis outcomes is shown in Figure 6, summarizing the possible risk of bias. PRISMA flow-diagram. QUADAS 2 risk of bias.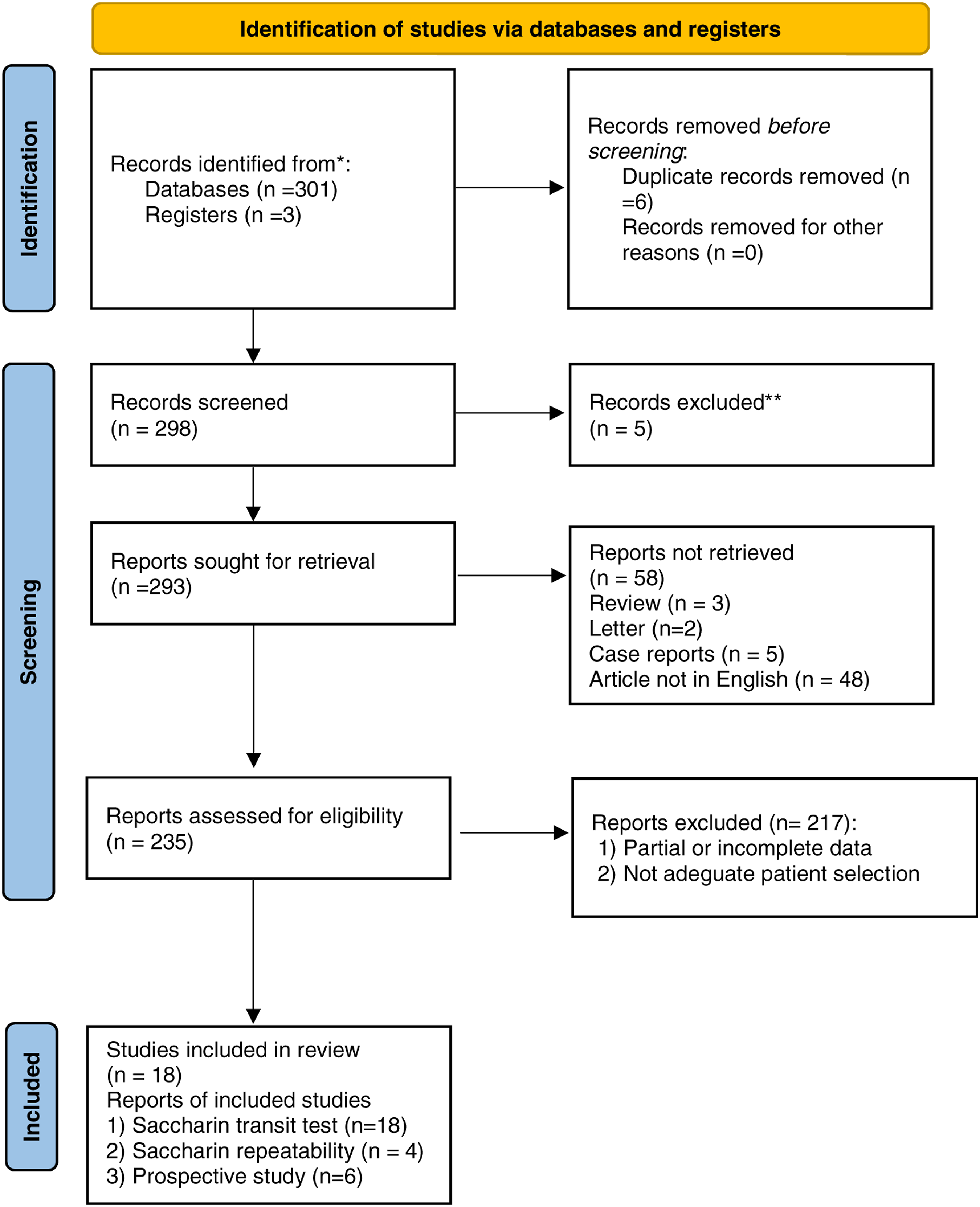
Study features
Eighteen studies were included.16,25–41 According to study design analysis, 11 were cross-sectional papers while 6 prospective controlled studies and one a cross-over study.
Main features of papers included in qualitative analysis.
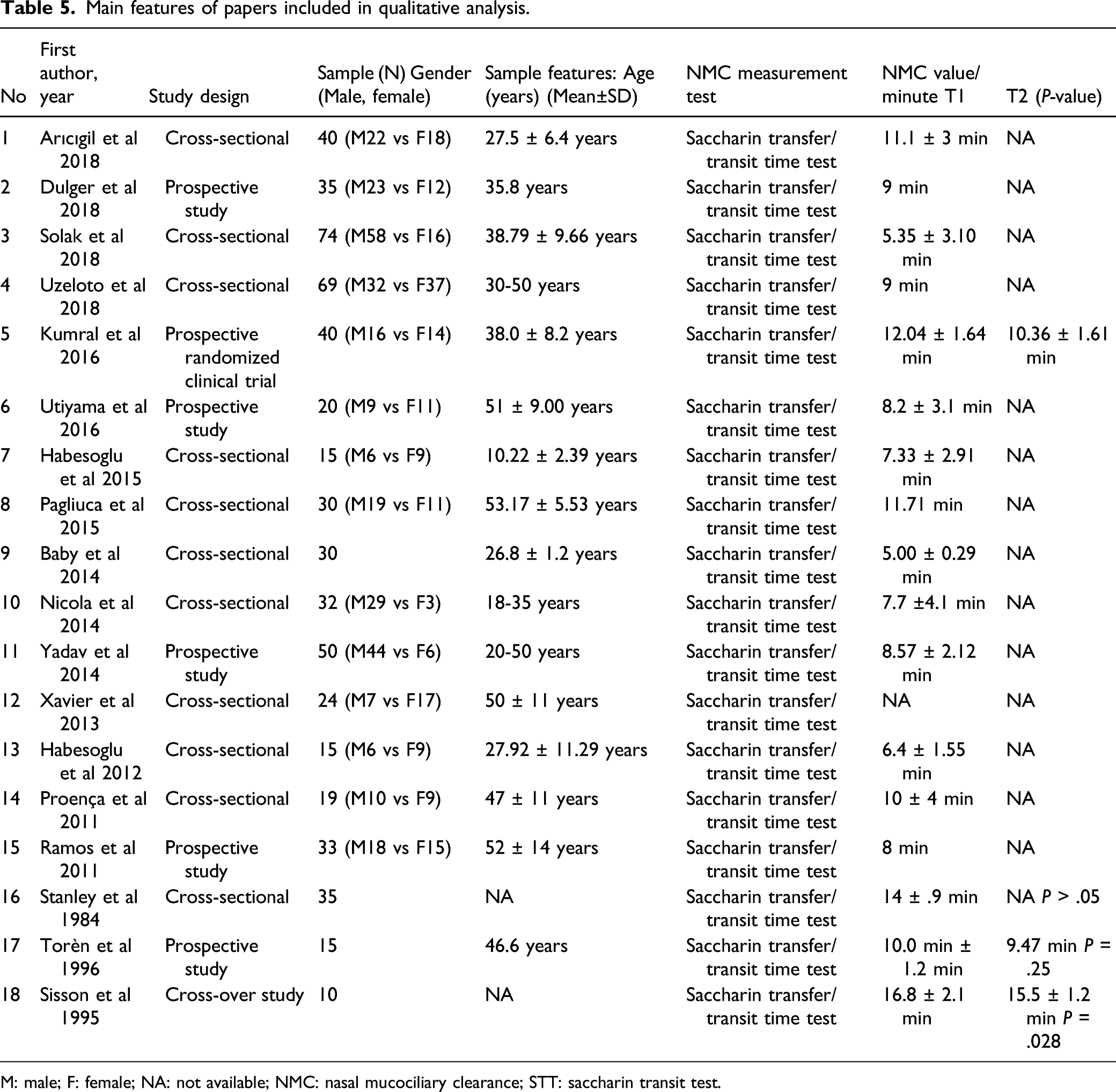
M: male; F: female; NA: not available; NMC: nasal mucociliary clearance; STT: saccharin transit test.
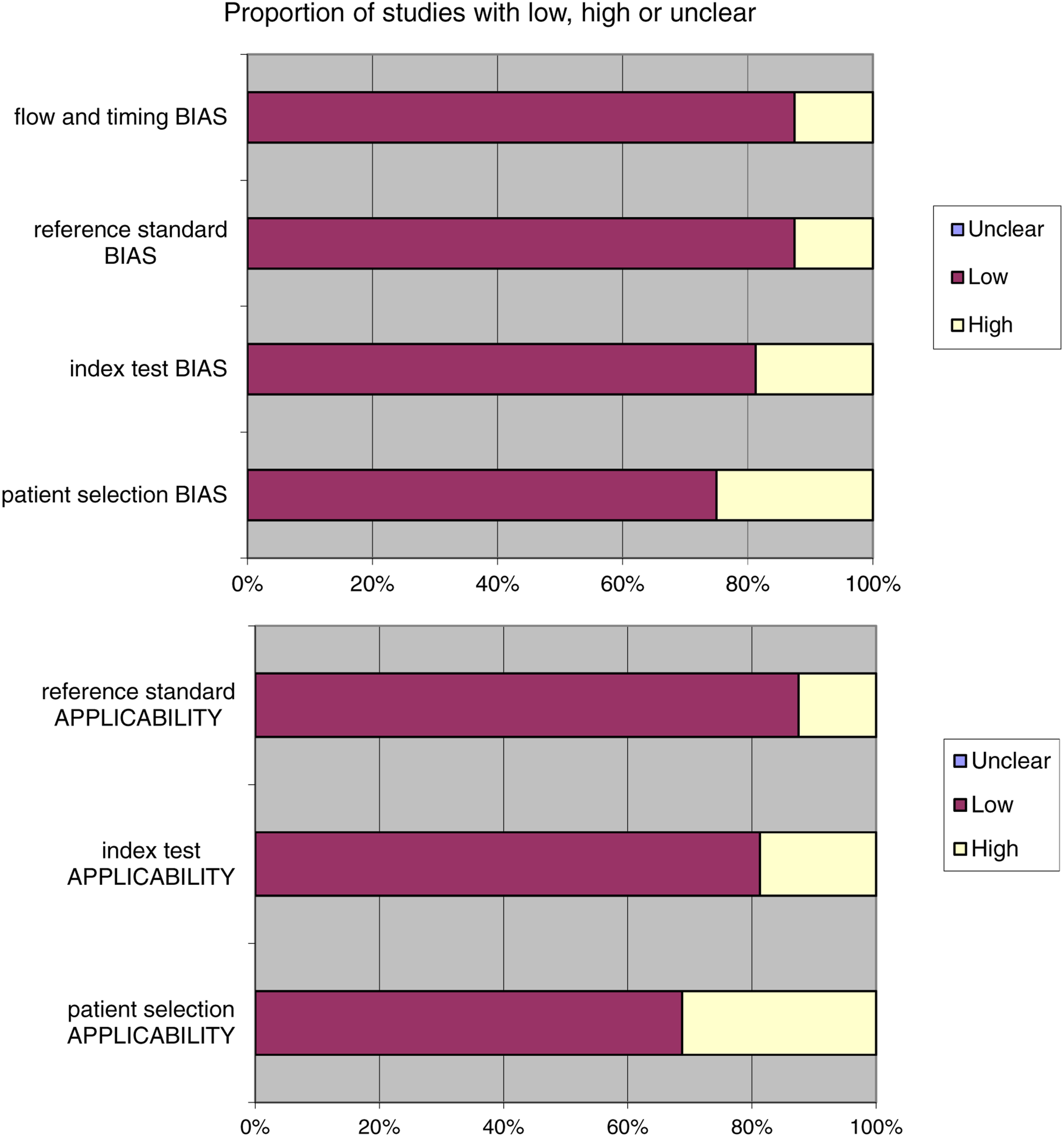
Heterogeneous methodology was performed mainly due to study design used (observational studies) and risk of bias in the included studies. The evidence appraisals are summarized in Figure 7. Risk of Bias summary author’s judgments for each included study, assessed by the Joanna Briggs Institute (JBI). Critical appraisal checklist for case–control studies.
Patients features, comorbidities, and treatment
The patients' average age was 42.8 ± 9.9 years, ranging from 10 to 53 years old. Among 586 subjects considered, 51% were male while 49% female.
Mucociliary transit time was performed in all papers included, with a mean pooled value of 9.42 ± 3.04 minutes. Moreover, 3 papers29,40,41 tested the repeatability of the test on the evaluation of mucociliary clearance (65 patients), reporting a mean value of 11.77 ± 3.25 with a non-significant difference (P = .34).
Quantitative analysis
Three papers were included according to the selection criteria established for quantitative analysis and consequent forest plot was generated.29,40,41 The analysis using random-effects modeling for the NMC Test–Retest value showed an SMD of .85 (95% CI 0.49, 1.22). The reported overall effect Z score was 4.64 (P < .0001), Q statistic P = .51 (no significant heterogeneity), and I = 0% (no inconsistency) as described in Figure 8. Forest plot of comparison: 1 repeatability, outcome: 1.1 repeatability test–retest evaluation.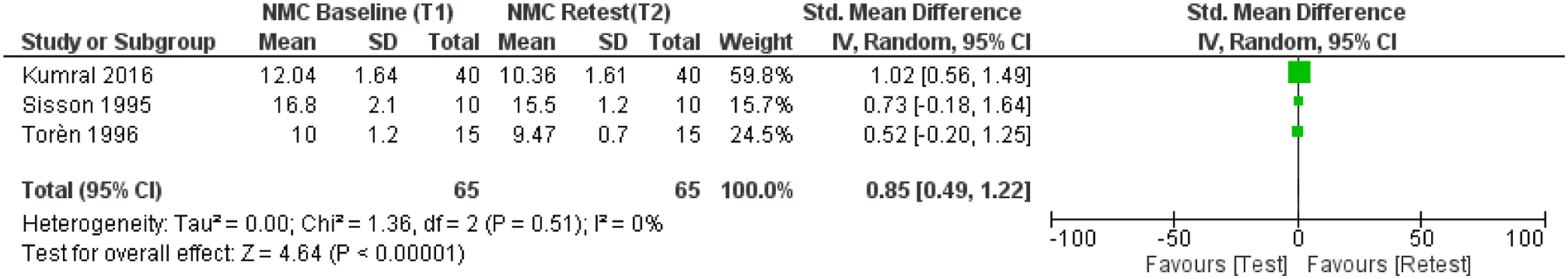
Discussion
Saccharin test is a practical, simple, and safe tool for assessing nasal mucociliary clearance in health and disease, particularly in primary or secondary ciliary disorders.42–44
However, large variability in STTT results has been reported in the literature due to differences in the protocols and subjects’ selection and very little is known about the reproducibility of the test.38–43 In the current study of well characterized healthy participants, between-subject variability was confirmed, but within-subject variability was moderate with a good level of test reproducibility not only in the short-term (within 3 days) but also in the long-term (within 30 days).
Notwithstanding the meticulous standardization used for the saccharin test in the current study, we confirm a substantial between-subjects variability for STTT. This is consistent with the results of the papers included in the systematic review. In keeping with the existing international reference values, this study carried out in healthy non-smokers from Sicily shows MCCTT values that are well within the 20 minutes duration cutoff point discriminating normal people from those with dysfunctional MCC.14,15,44
However, within-subjects variability is good. The linear regression analyses showed significant short-term repeatability (V2 vs V1) and long-term repeatability (V3 vs V1), despite the low value of R squared. Also, significant agreement in the repeatability of the intrasession measurements among the three visits (V1, V2, and V3) was observed in spite of the low ICC value. On the other hand, the mean values of differences (V1–V2 and V1–V3) were not significantly different from zero, which means that the first measurement is not affecting the second and the difference does not vary in any systematic way over the range of measurements. Such good level of repeatability is also confirmed by the results of the papers included in the systematic review of the literature (Table 5).29,40,41 Kumral et al 29 reported good STT reproducibility in patients after smoking cessation. In particular, the outcomes’ comparison between baseline vs three months follow-up did not show any significant difference (12.04 ± 1.64 min vs10.36 ± 1.61 min; P > .05) (Table 5).
The high level of STTT reproducibility reported in this study is likely due to meticulous standardization and competent conduct of the test as well as to careful subjects’ selection; in particular, great care was taken when reviewing clinical information and excluding all those conditions (and medications) that could impair cilia–mucus interaction or interfere with STTT measurements. For example, 10 out of 50 subjects (20%) were excluded due to allergic rhinitis, asthma, and recent flu and a further 3 subjects (6%) were excluded because they were probably lying about their smoking habit (eCO >7 ppm). Given that these conditions are not uncommon, it is important to consider that subjects’ recruitment may require larger pool of volunteers. Of interest, only one subject (2%) exhibited insufficient ability to perceive sweetness of the saccharin on its tongue (score <25 mm VAS mark for sweetness intensity rating). It is also likely that the high level of STTT reproducibility reported in this study was due to regulation of environmental conditions of the examination room (careful adjustment of ambient temperature and humidity). Last but not least, we believe that adequate subject’s preparation (nasal washing and ambient acclimatization) before conducting saccharin test and involvement of well-trained operators (correctly placing the saccharin tablet in the same position at each study visit) played a significant role.
The saccharin test was safe and very well tolerated. No changes in resting heart rate and systolic/diastolic blood pressure occurred during the study. Side effects were few and transient, lasting only a few minutes. In this study, the occurrence of symptoms that can invalidate the saccharin test (eg, sneezing and coughing) was rare. Only one subject who was very sensitive to test procedures experienced recurring sneezing and had to be withdrawn from the study.
Conclusion
The saccharin test is a fast, simple, accurate, and safe method for the evaluation of MCC. The measurement of STTT is not only relatively sensitive but also quite reproducible. These important features position this test as an emerging biomarker of physiological effect for the detection of early respiratory health changes. In addition to the obvious relevance to the investigation of potential health concerns, STTT could be important to provide contribution for a unique regulatory science endpoint for medical and regulatory research applied not only to pharmacological interventions for lung diseases characterized by defective mucus clearance (eg, anti-inflammatory or anti-allergic treatment for allergic and non-allergic diseases of the airways) but also to smoking cessation medications and combustion-free tobacco products (eg, e-cigarettes, heated tobacco products, and oral tobacco/nicotine products.).
Footnotes
Author Contributions
Conceptualization, P.C., R.P., A.M., and S.C.Methodology, S.F.Software, A.C., J.R.L., and S.C.Validation, M.C., F.B., and A.G.Formal analysis, JR. L.Investigation, A.G. and A.M.Data curation, R.P.Writing—original draft preparation, P.C.Writing—review and editing, A.G. and F.B.Visualization, R.E.Supervision, R.E. and S.F.Project administration, A.M.All authors have read and agreed to the published version of the manuscript.
Conflict of interest
Salvatore Ferlito and Salvatore Cocuzza are full-time employees of the University of Catania, Italy. Alessandro Gulino, Antonino Maniaci, and Rosalia Emma are fixed-term researcher at University of Catania, Italy. Francesca Benfatto is fixed-term researcher at Centro per la Prevenzione e Cura del Tabagismo, University of Catania. Jerome Lechien is an associate professor of Mons University; Angelo Ingrassia is an ENT specialist from Catania University. Riccardo Polosa was awarded an Investigator-Initiated Study award program established by Philip Morris International in 2017, but subsequently resigned from the role of Principal Investigator in 2018, before the trial began. Lecture fees from a number of European EC industry and trade associations (including Fédération Interprofessionnelle de la VAPE in France and Federazione Italiana Esercenti Svapo Elettronico in Italy) were directly donated to vaper advocacy no-profit organizations. RP is the Founder of the Center of Excellence for the acceleration of Harm Reduction at the University of Catania (CoEHAR), which has received a grant from Foundation for a Smoke Free World to develop and carry out eight research projects. RP is also currently involved in the following pro bono activities: scientific advisor for LIAF, Lega Italiana Anti Fumo (Italian acronym for Italian Anti-Smoking League), and Chair of the European Technical Committee for standardization on Requirements and test methods for emissions of electronic cigarettes (CEN/TC 437; WG4). Pasquale Caponnetto is paid by the University of Catania as an external part-time researcher and adjunct professor of clinical, addiction, and general psychology. He has been affiliated to the CoEHAR since December 2019 in a pro bono role. He is coauthor of a protocol paper supported by an Investigator-Initiated Study award program established by Philip Morris International in 2017. The other authors have no conflict of interests to declare
Funding
This research was supported by university grant no. 21040100 of “Ricerca Scientifica Finanziata dall’Ateneo di Catania”
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
