Abstract
Introduction
Locally advanced oral cavity cancer accounts for 25–40% of all stages. The treatment must strike a balance between oncological, functional, and aesthetic goals. Apart from that, operating on a large tumor is difficult, and achieving a negative margin is extremely difficult. 1 Preoperative chemotherapy, like many other studies, did not increase the likelihood of radical surgery, function preservation, or overall survival (OS), indicating that concurrent chemoradiotherapy (CCRT) is an appropriate alternative treatment method for locally advanced stage, and CCRT has been widely used in Vietnam.
Tumor hemorrhage and bleeding are a common complication in the advanced stage, occurring in up to 6% to 14% of patients. It could be the direct cause of death, the result of a treatment interruption, or both. Additionally, it may impair a patient’s quality of life. Medical intervention is almost completely ineffective. Numerous studies have demonstrated the importance of vascular intervention in managing bleeding complications. However, the studies examined only palliative care without evaluating its effect on the patient’s chemoradiotherapy outcomes. 2,3 At the Department of Head and Neck Radiation Oncology, Vietnam National Cancer Hospital, we documented 16 clinical cases of advanced oral cavity cancer with tumor bleeding during CCRT. Evaluate the efficacy of hemostasis and its effect on treatment outcomes.
Materials and methods
A retrospective descriptive study was conducted on 16 patients with locally advanced oral cavity cancer who received definitive CCRT and experienced tumor bleeding complications. From September 2018 to June 2021, 90 patients with advanced oral cavity cancer received definitive CCRT at our center. There were 16 patients with bleeding complications, and they all received selective embolization with diagnostic angiography at Vietnam National Cancer Hospital.
Persistent or recurrent bleeding that cannot be controlled locally (via compression, dressing, packing, or administration of antifibrinolytic agents) or via blood transfusion is indication for embolization.
Age, gender, ability to control bleeding, clinical stage of TNM (as defined by UICC 8th), response to CCRT, overall survival, and progression-free survival were all analyzed. After one month of treatment, the response to CCRT was evaluated using rigid ENT endoscopic, head and neck MRI, and PET/CT scans, according to RECIST version 1.1 criteria.
Procedure
The patient is positioned supine, with a monitor monitoring vital signs, particularly the airways. A thorough medical history of allergies is obtained. Local anesthesia is used, and the catheter is inserted through the femoral arteries. Using digital subtraction angiography (DSA) to identify the tumor’s bleeding and feeding arteries, examining the common carotid artery, internal carotid artery, external carotid artery, and its branches to ascertain the source of bleeding and guide the intervention. Tumor bleeder (a site of active bleeding included vessels with active contrast extravasations during the artery phase), tumor staining (a contrast-enhanced mass caused by tumor vascular proliferation), and pseudoaneurysm formation were all abnormal imaging findings (an enlargement or sac-like structure in the arterial branches). Numerous embolization materials may be used, including temporary embolization materials (Gelfoam), Gelatin sponge gel, and permanent embolization materials such as PVA (polyvinyl alcohol, Embosphere), Bio glue (Histoacryl, Onyx), and NBCA glue (N-butyl 2-cyanoacrylate), as well as metal helices of various sizes (steel or platinum coils, micro coils), or balloons. We may or may not intervene based on the tumor staining morphology, after consultation with the radiologist, oncologist, and patient’s family. In severe bleeding cases, a balloon and coils are preferable.
Evaluation of vascular interventional side effects/complications: prolonged bleeding into the approach site (> 10 hours), hematoma, allergies, or even anaphylactic reactions to contrast agents, renal failure, technical complications (vessel injury or dissection), and reverse flow of embolization materials (headache, facial edema, transient paralysis, hemiplegia, etc.). Evaluation of hemostasis effectiveness: Follow-up of patients following CCRT to assess treatment response, overall survival, and disease-free survival with re-examination every 3 months using head and neck MRI, CT scans, neck ultrasound, and ENT endoscopic examinations, as well as maintaining contact with patients.
Results
Patient characteristics (Table 1)
Patient and digital subtraction angiography characteristics.

The results of angiography
The femoral artery was approached in 16/16 patients during digital subtraction angiography. No patients had a history of contrast allergy or reaction. The results of angiography revealed that in all 16 cases where a bleeding site was discovered, the initial source of bleeding was from the lateral branches of the external carotid artery, with 13/16 (81.25 %) cases coming from the lingual artery, 2/16 (12.5 %) cases coming from the facial artery (including 1 patient with a tongue tumor and 1 patient with a floor of the mouth tumor), and 1/16 cases coming from the maxillary artery. Bleeding from the lingual artery occurred in ten out of eleven (91 %) of tongue tumor patients (see Figure 1). A: DSA showed the source of tumor bleeding: lingual artery, (a). branch of external carotid artery. (b). Contrast extravasation prior to embolization. (c). After trans-arterial embolization lingual artery by Embosphere 300–500 µm, no contrast extravasation.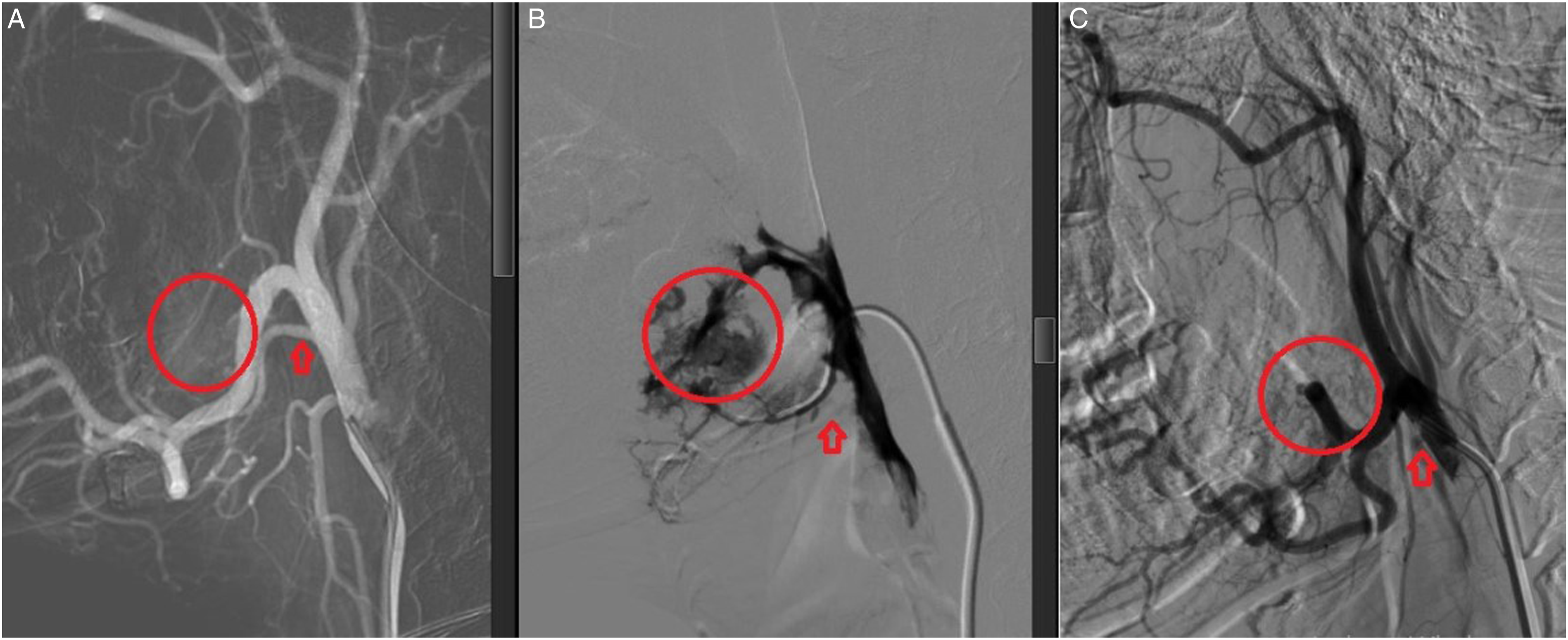
All cases of hemostatic embolization were successful, with two patients receiving Gelfoam, two receiving coils, nine receiving PVA or Embosphere, and four receiving a combination of coils and sponge gel. 16/16 patients’ experienced immediate clinical hemostasis. One patient with tongue cancer experienced re-bleeding 3 weeks after initial trans-arterial embolization; the patient underwent re-intervention and the bleeding was controlled.
There have been no procedure-related complications, such as anaphylactic shock to the contrast agents, emergent neurological difficulties during or following the angiographic procedure due to vessel spasm or infarction, contrast-related nephrotoxicity, or puncture site hematoma. 4/16 patients experienced local pain, but these instances were well controlled by analgesics.
Survival
The average duration of chemoradiotherapy interruption due to intervention was 6.7 days. This time period begins when the patient is forced to discontinue treatment due to bleeding complications and ends when radiation therapy is resumed following the intervention and the patient’s condition has improved.
Following definitive CCRT, 9/16 (56.25%) patients had a complete response, 6/16 (37.5%) patients had a partial response, and 1 patient had stable disease. For the first two years, all patients were followed every 3 months; the OS was 14 months [3–26], and the PFS was 10.5 months [3–20]. Three patients died in the end stage of the disease due to progressive/recurrent disease-related multi-organ failure. 7/16 (43.75%) patients currently have no detectable disease recurrence, while 6/16 patients are receiving symptomatic or palliative care for recurrent disease. (see Figure 2). (a). cT3N2M0 tongue cancer. (b). Completed response after definitive CCRT (70Gy/35fractions, IMRT technique, Cisplatin 100mg/m2 every 3 weeks).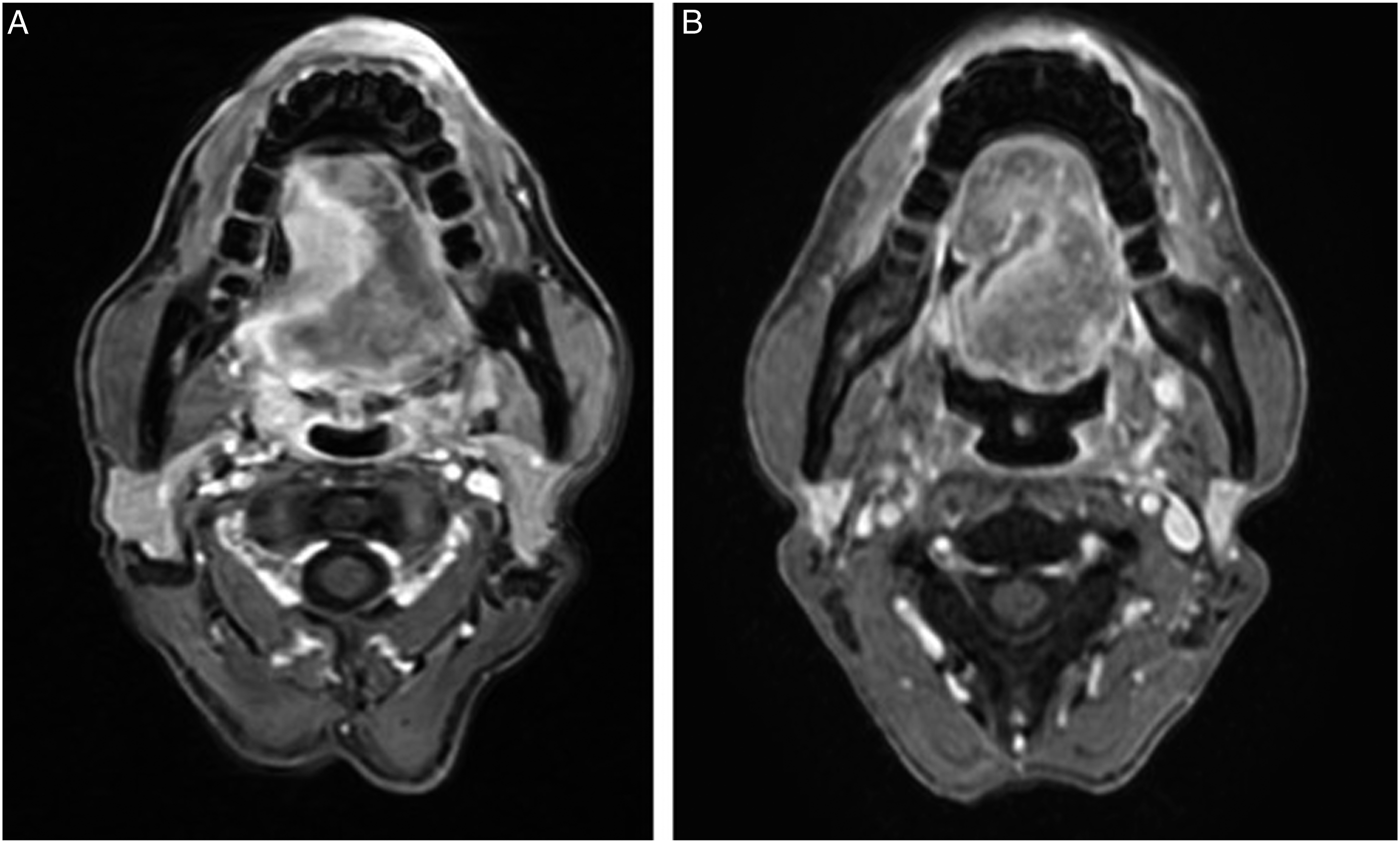
Discussion
Locally advanced head and neck cancer, particularly oral cavity cancer with extensive local invasion, frequently presents with a variety of complications, including intractable pain, tumor bleeding, poor nutrition, infection, and fistula. In this instance, bleeding is a significant complication that is both acute and dangerous, necessitating prompt intervention. The most severe form, carotid blowout syndrome (CBS), occurs in 4.3 percent of head and neck cancers. The mortality rate is approximately 50%. CBS is frequently seen in patients who have previously received radiation therapy or surgery. The primary treatment is trans-artery embolization because the vessel wall becomes weak, fragile, radio-induced necrosis, which makes ligation operate more difficult. 4
The mobile tongue, the mouth floor, the hard palate, the posterior triangle, gingiva, and buccal mucosa are all included in the oral cavity. When large tumors invade widely, they cause infiltration of the jaw, oropharynx, pharynx, and larynx, as well as local necrosis infection, resulting in the formation of a fistula with a folder mucosal structure and a dense vascular system derived from branches of the external carotid artery. Tumor bleeding can be caused by invasive tumors that invade adjacent vessels, ulcerative tumors, and almost all oral cavity tumors are proliferating blood vessels. Our patients received definitive CCRT with a favorable outcome, resulting in tumor necrosis, radiation-induced necrosis of tumor feeding vessels, radiation-induced fibrosis, and breakage of elastic fibers in vessel walls. As a result, blood vessels were vulnerable to injury and bleeding. Additionally, cancer-related abnormalities, anticoagulant use, and NSAID anti-inflammatory drug use all contribute to an increased risk of bleeding. By and large, patients with tongue, gingiva, and palate cancer receive blood supply primarily from the lingual and maxillary arteries, whereas patients with buccal cancer receive blood supply primarily from the facial arteries, which originate primarily from a branch of the external carotid artery. However, many cases exhibit significant blood vessel proliferation or extensive anastomosis from both the external and internal carotid arteries. 5
Additionally, this study revealed that all patients had a bleeding source from a branch of the external carotid artery, with 81.25% from the lingual artery and 12.5% from the facial artery, which is consistent with previously obtained anatomical data. Endovascular embolization is indicated in head and neck cancer for preoperative treatment of highly angiogenic tumors such as nasopharyngeal fibroma (Juvenile nasopharyngeal angiofibroma) to reduce tumor size and intraoperative blood loss, or for emergency hemostatic intervention with large tumors causing bleeding or invasive tumors, lymph nodes, or fistula causing vascular erosion, or for treatment-impairment.
Hemorrhage from oral cavity tumors has traditionally been treated with open surgical exploration and ligation of the involved vessels, as well as the usual ligation of the unilateral or bilateral external carotid arteries. This operation has a high rate of hemostatic efficiency, ranging from 9 to 100%. 6 Surgical treatment, on the other hand, is frequently difficult due to fistulas, infections, radiation-induced necrosis, or prior surgical exploration. Radiation therapy, in particular, causes fibrosis and enlargement of invasive lymph nodes in patients who have undergone extensive neck lymph node dissection or flap surgery, resulting in anatomical changes that make it difficult to approach the carotid artery. Ligation surgery can cause significant postoperative pain, difficulty healing the wound, an extended hospital stay, and even neurologic complications and death. Additionally, surgery can put a patient at risk of re-bleeding from the collateral circulation, and endovascular treatment becomes more challenging as a result of the altered vascular anatomy following ligation. Embolization is the preferred method of emergency hemostasis in patients with head and neck cancer, particularly those who have previously undergone lymph node dissection surgery or received cervical radiation. Although the risk of recurrent bleeding is between 0% and 33%, re-embolization is still a viable option.
Yung-Fang Chen et al. (2015) conducted an analysis of 25 patients who underwent vascular intervention, including nine patients with tongue cancer (36 %). The majority of patients had received prior treatment. Following that, 33% (3/9) of patients experienced recurrent bleeding. Additionally, tongue cancer required a longer hospital stay than other cancers, approximately 21 days. 7 According to Kakizawa et al., 8 the recurrence rate of oral bleeding due to head and neck cancer was 22%, and some patients with facial pain were well controlled with conventional pain relief. However, these two studies found no significant complications such as hemiplegia or vision loss.
Rzewnicki et al 9 (2012) studied 66 patients who underwent selective embolization, 15 of whom had oral cancer and nine of whom had palate cancer. The majority of them were successfully treated with Co-60 radiation therapy (in conjunction with chemotherapy or surgery), and 85 percent of bleeding tumors were controlled. However, in four (5%) of the cases, re-bleeding occurred, necessitating additional external carotid ligation operations, and other patients required a second embolization after 3 to 7 days. However, 21 (27.6 percent) of the cases had adverse effects, including 14 patients who developed headaches after 3 days, six patients who developed facial edema, and one patient who developed hemiplegia. Chou et al. analyzed 63 patients in 2007, 14 of whom had oral cavity cancer and had received chemotherapy or radiotherapy (84 percent in the T4 stage). They performed 93 angiographies and 56 trans-arterial embolizations, with 19 patients undergoing two scans, four patients undergoing 3 scans, and one patient undergoing four scans. After bleeding, the average survival time was 16 days (0–644 days). Patients who underwent embolization had a significantly longer survival time than those who underwent angiography alone (26 vs 8 days, P = .008). Gender, tumor stage, pathology, prior therapies (chemotherapy and radiotherapy), and embolization materials had no predictive value for survival time. 10
Embolization materials were chosen based on DSA results, vascular diameter, vessel location, and intervention doctors' experience. Several complications occurred during the procedure: the source of bleeding could not be identified, and the extensive collateral system failed to identify the primary tumor blood source. Microcatheters, in our experience, can be used to optimize approach and selective embolization. Typically, the initial particles used in these procedures were 150–250 microns in diameter to block small branches, followed by larger particles or Gelfoam. In larger vessels, coils can be combined with other materials. The timing of the intervention is largely determined by the clinical oncologist, whereas the outcome is highly dependent on the radiologist’s experience and skill. At our hospital, we used both temporary and permanent hemostatic materials, as well as combinations of the two. Similar hemostatic effects were observed.
The majority of retrospective studies have demonstrated a low rate of complications, demonstrating the efficacy and safety of trans-arterial embolization. There were no significant adverse events associated with contrast agents or neurological complications in our study. Certain patients experienced local pain but responded well to conventional analgesia. Embolization improves hemostasis. However, there is concern that it will reduce the tumor’s sensitivity to radiation, resulting in poorer treatment outcomes. According to theory, radiation therapy is more sensitive to tumors that are vascular and oxygen-rich. When vascular intervention is performed, the amount of oxygen delivered to the tumor is reduced, resulting in a halt to the treatment process. Both of these critical factors will have an effect on treatment outcomes. Additionally, it is for these reasons that, when acute bleeding occurs, hemostasis is preferred over continued treatment in palliative care.
In comparison to the previous studies, our OS and DFS were significantly longer (14 and 10.5 months, respectively), possibly because our patients were at an earlier stage and had received definitive CCRT, whereas the other studies focused on palliative treatment. Additionally, there is an explanation for why patients receiving definitive CCRT have a better response and a higher risk of tumor necrosis resulting in bleeding. Our study had a higher rate of treatment response, with an overall response rate of 93.75 percent, including a 56.25 percent complete response. To the best of our knowledge, this is the first study to report on the outcomes of definitive concurrent chemoradiotherapy in patients with oral cancer who have undergone trans-arterial embolization. The study established that embolization did not impair the patient’s response to chemoradiotherapy.
Conclusions
Tumor bleeding is a frequent and serious complication during definitive CCRT treatment for advanced oral cavity cancer. The external carotid artery and its branches are the source of bleeding. Trans-arterial embolization is an effective and safe method of managing acute bleeding, whereas embolization during definitive CCRT has no adverse effect on the overall treatment outcome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for this article because it did not contain human or animal trials.
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
