Abstract
Objective
The purpose of this paper is to review the literature and compile promising and clinically relevant biomarkers in otolaryngology—head & neck surgery not related to autoimmune disorders.
Study Design
Narrative review.
Methods
PubMed and Google Scholar were queried using combined key words such as “biomarkers” and “otolaryngology.” Additional queries were made with combined key words such as “biomarkers” and a particular subspecialty such as “rhinology” or “otology” to maximize yield of relevant titles. Subsequently, specific biomarkers identified, such as “beta-2 transferrin,” were used as key words. Relevant titles were reviewed and selected for abstract review. Applicable abstracts were then selected for review of the full text.
Results
Biomarkers currently in clinical use within the field of otolaryngology were included in this review. The compiled biomarkers were then detailed individually regarding their molecular characteristics, function, and clinical significance.
Conclusions
The number of biomarkers in use in otolaryngology is rapidly expanding representing a new diagnostic modality for our field. This review defines the key biomarkers that are currently or likely to be soon translated into clinical use within the field of otolaryngology. The majority of these biomarkers are in the form of proteins such as beta-2 transferrin, thyroglobulin, and P16. Given their growing impact on diagnosis, management and surveillance of otolaryngologic disorders periodic surveys are needed for education and to guide further advances and applications of otolaryngologic biomarkers.
Introduction
Biomarkers such as proteins, miRNA, DNA sequences, cytokines, and cell receptors serve as surrogate indicators of disease and aid in diagnosis, disease monitoring, and treatment outcomes. The quantity of biomarkers currently under investigation is innumerable; however, the number of biomarkers that are accessible, appropriately specific, cost-effective, and validated for clinical practice pales in comparison. In our previous paper, we presented a review of the literature and characterized notable biomarkers in use for autoimmune disorders affecting the head and neck. 1
The purpose of this article is to serve as the second part of our two-part comprehensive resource detailing key clinically relevant biomarkers currently in use or potentially promising for near future application, within otolaryngology and its subspecialties. In particular, we examined biomarkers in use within rhinology and skull base surgery, otology, laryngology, and head and neck cancer. Instead of cataloging each biomarker for each organ, we have broadly grouped them into 3 categories: biomarkers of injury to the inner ear and skull base; inflammation or infections of the upper respiratory tract; and malignant neoplasms of the head and neck.
Methods
The databases PubMed and Google Scholar were queried using combined key words such as “biomarkers” and “otolaryngology.” Additional searches were made with combined key words such as “biomarkers” and a particular subspecialty such as “rhinology” or “otology” to maximize yield of relevant titles. Subsequently, specific biomarkers identified, such as “beta‐2 transferrin,” were used as key words. Relevant titles were reviewed and selected for abstract review. References of these articles were also searched for original articles and systematic reviews. Applicable abstracts were then selected for review of the full text. Articles that specifically discussed biomarkers in active clinical use within the field of otolaryngology and related subspecialties were selected for complete review.
Injury to the Inner Ear and Skull Base
Identification and molecular characterization of inner ear proteins creates a foundation by which targeted diagnostic and therapeutic strategies can be implemented in hearing loss. 2 -5 A recent literature review by Mulry et al characterized 8 inner ear specific proteins identified within perilymph: otolin-1, otoconin-90/95, prestin, otoancorin, otogelin, α-tectorin, β-tectorin, and cochlin. 2 While each of these proteins has been implicated in inner ear related hearing loss, only 2, prestin and otolin-1, have been described to exist in the peripheral circulation. 2,4
Of these candidates, the molecule that has come closest to entering clinical use as a biomarker for hearing loss is prestin. 6 -13 A paper by Parham in 2015 posed the hypothesis that prestin could be used as a biomarker for the early detection of hearing loss. 6 An 80 kDa transmembrane anion transporter protein, unique to outer hair cells (OHCs), prestin functions in electromotility and cochlear amplification. 6,7 Proof of concept was provided in experimental studies of noise trauma and ototoxicity. 8 -12 Application to clinical populations has been demonstrated more recently in sudden sensorineural hearing loss, noise exposure, lead and cisplatin ototoxicity. 13 -15 As discussed by Parham in the 2015 article, and subsequent papers, the specificity for OHCs makes prestin a promising candidate for monitoring OHC-specific related inner ear insults, such as cisplatin toxicity or noise trauma. Its extracellular domain, which binds a specific ligand, makes it a potential therapeutic target for drug delivery research. 6 Furthermore, its size and ability to cross the blood-labyrinthine barrier allow it to enter circulation and therefore make it susceptible to collection and analysis via sensitive assays such as ELISA. 10 -12 Thus, prestin encompasses qualities that make it an attractive candidate for further development as a clinical biomarker to quantify and monitor acquired hearing loss.
Another promising candidate in inner ear biomarker research is otolin-1, a 70 kDa scaffolding protein specifically expressed in the inner ear, which forms the multimeric complex of the otoconia. 16 Prior studies have investigated the association between otolin-1 and vestibular pathology, including benign paroxysmal positional vertigo (BPPV). 4,16,17 Most notably, Parham et al demonstrated that patients with BPPV had elevated levels of otolin-1 in peripheral circulation. 17 This finding has now been replicated by others and extended to another inner ear protein, otoconin-90. 18 -21 Due to its specificity for the inner ear, its ability to be detectable and measurable in peripheral circulation, and its correlation to BPPV, otolin-1 is another attractive candidate for use as a clinical biomarker.
In addition to noise trauma and medication toxicity, injury to the skull base can occur from other etiologies as well. Direct trauma to the skull base can result in CSF (cerebrospinal fluid) leaks, which occur when there is a connection between the dura and the skull base resulting in otorrhea or rhinorrhea. 22 Non-traumatic CSF leaks can occur from disorders leading to disruption of the meninges and the bony skull base. 23 Irjala et al 24 first demonstrated an extra band of transferrin located in the beta-2 fraction on protein electrophoresis, pathognomonic for CSF, and not found in serum, tears, saliva, nasal secretions, or endolymph. 24 This molecule, a de-sialated isoform of the protein transferrin, found almost exclusively in CSF, denoted, beta‐2 transferrin, can be used as a specific marker for CSF leaks. 24 Subsequently, beta-2 transferrin has been validated as a key clinically relevant diagnostic biomarker to identify CSF. 25,26 It is safe and easy to collect, only a small amount is needed, and moderate contamination does not invalidate the assay. 24 -26 Historically, CSF was said to leave a “ring sign” or “halo” when combined with blood on a white fibrillar surface; however, reliability of this sign is poor due to the same sign occurring if blood is mixed with any clear fluid such as water, nasal fluid, or saline. 24,27 Comparatively, beta-2 transferrin is more specific, accurate, and reliable. 24 -27 Using gel electrophoresis methods, beta-2 transferrin detection assays can take as quickly as 3-6 hours to result; however, labs may take longer to report the results due to delays in processing times. 26,27
Oakley et al 27 compared benefits, costs, and risks of the various techniques for CSF detection, including ring sign, glucose testing, radionuclide cisternography, or CT cisternography. They found beta‐2 transferrin to be the best confirmation test for identifying CSF. 27 Collection of the sample usually involves taking liquid from a patient’s rhinorrhea or otorrhea, making the process low-cost, low-risk and non-invasive. 24 -27 Regardless of the assays used, Oakley et al 27 identified an 87%-100% sensitivity and a 71%-100% specificity. 27 Of note, they also evaluated another biomarker, Beta-trace protein, a protein present in high concentrations in CSF, which they found to be superior to beta-2 transferrin in terms of specificity and sensitivity; however its application is limited due to its unavailability in the United States. 27 Detection methods for beta-trace protein can take anywhere from 15 minutes to 4 hours depending on the commercial assay used. 28
Inflammation or Infections of the Upper Respiratory Tract
Biomarkers can aid in monitoring and assessing bacterial and viral illnesses of the upper respiratory tract as well as characterize the immunologic milieu that predisposes certain patients to disease. For example, the molecular and biochemical framework of the allergic molecular pathway has been extensively described and can serve to distinguish chronic rhinosinusitis with and without nasal polyposis (CRSwNP and CRSsNP, respectively). 29 The distinctions between these 2 disease processes have been defined by their molecular signature. 29 Defining the molecular basis of disease in each of these processes allows targeted treatment and prognostic predictions.
A review by Workman et al investigated the inflammatory pathways of CRSwP and CRSsP, with particular attention to the biomarker composition that defines each of the various endotypes of disease. 29 Their review characterizes IgE as a useful biomarker in defining disease severity, risk of recurrence, and glucocorticoid responsiveness; however, it is not a predictive biomarker for CRSwNP due to its non-specific nature. 29,30 A separate review by Gurrola et al similarly concludes that IgE is an important clinical indicator of disease course and response to macrolide treatment. 30 Additionally, it can serve as a key target for biologic therapy, namely, the monoclonal antibody drug Omalizumab. 30
The role of biologics is becoming more prominent in CRSwNP. Both Workman et al and Gurrola et al describe the key biomarkers, including IL-4 and IL-13, that can serve as targets for directed biologic therapy, specifically dupilumab. 30,31 In a separate review of the contemporary pharmacotherapy for CRS, Ghadersohi et al discuss the important role of dupilumab, a human monoclonal antibody that targets the receptors of interleukin-4 (IL-4) and interleukin-13 (IL-13) by binding to the IL-4 alpha subunit which is a component of both receptors. 32 This attenuates the TH2 response. Ghaderoshi et al describe multiple randomized controlled trials that demonstrate significant improvements in biomarkers of type-2 inflammation following dupilumab treatment. 32 The cost for dupilumab in CRSwNP, however, cannot be ignored, as it can exceed the cost of surgical management. 32,33 Therefore, appropriate patient selection is key, further highlighting the importance of implementing biomarkers in diagnosis and management. Identifying which patients have the optimal biomarker composition for successful treatment response can help guide practitioners on when to use dupilumab.
Biomarkers downstream to IL-4 and IL-13 have also been identified in the pathway of CRSwNP, including the extracellular protein periostin. 34 Izuhara et al 34 define the importance of periostin for the onset of allergic inflammation as a matricellular protein that binds to integrin receptors in response to IL-4 and IL-13 resulting in fibrosis deposition and eosinophil recruitment. 34 One advantage of using periostin as a biomarker includes convenience of collection from blood, urine, tears, or sputum. 34 Additionally, periostin levels appear to decrease following treatment, and it can therefore be used to monitor disease course. 34 Although many kits designed to detect periostin as a biomarker are available, many of them requiring only a few hours to run, there is a lack of standardization among them, limiting current clinical use. 34
Another important biomarker studied as a potential target for management of CRS is P-glycoprotein (P-gp), an active ATP-dependent membrane efflux transporter. 35,36 Feldman et al conducted a study in which 39 steroid-naïve patients with CRS underwent sinus tissue collection and fluorescent immunostaining. They found that patients who had higher P-gp expression also had higher eosinophil counts and higher Lund-Mackay scores. 35 A separate study from the same group had previously demonstrated that P-gp was secreted in sinonasal mucous and that patients with CRSwP secreted higher levels of P-gp than CRSsP. 36 Additionally, they found that higher secreters had significantly higher SNOT-22 scores than low secreters. 36 Both these studies demonstrate that upregulation of P-gp in sinonasal mucosa is associated with CRSsNP and CRSwNP and is correlated to worse radiographic inflammation scores, including worse Lund-Mackay and SNOT-22 scores. 35,36 The existence of a secretory form of P-gp identified in nasal mucous makes it a readily collectable potential diagnostic biomarker. 35,36
Last, an important biomarker in bacterial-driven illnesses, procalcitonin, a 13 kDa protein, has also been studied as a serum biomarker for acute rhinosinusitis (ARS). 37 When utilized, procalcitonin has been linked to reduction in antibiotic therapy. 37 Dilger et al highlight the utility of procalcitonin in ARS and its promising role as a biomarker for monitoring disease course and outcomes. Procalcitonin is released in response to certain cytokines and can indicate a bacterial infection. 37 It has been validated for use in respiratory tract infections, such as pneumonia and bronchitis as well as in sepsis, and is currently being used in those clinical settings, with commercial kits available that can run in as quick as 20 minutes–5 hours. Although it is indicative of a bacterial process, it is not specific for sinusitis and does not replace culture for definitive antibiotic treatment. 37
In addition to sinonasal infections, viral infections of the larynx can result in significant disease, as seen in the extensive recurrent papillomas of recurrent respiratory papillomatosis (RRP). 38 -40 A review by Derkay et al described the causative etiology of RRP as infection with Human Papilloma Virus (HPV), particularly types 6 and 11. 33 They also report that HPV-11 has been associated with higher disease severity and tracheal involvement as well as higher likelihood of tracheostomy requirement. 38
Changes in the immunologic and biochemical milieu of RRP patients have been studied as an indirect marker of disease severity. 38,39 Rosenthal et al 40 sought to investigate TH2-like chemokine patterns in the serum of patients with RRP and correlate these patterns to disease severity. 40 They found a significant correlation between sustained clinical remission in patients with RRP and reduced levels of chemokines CCL17, CCL22, and CCL18. 40 They concluded that changes in expression of these plasma TH2-like chemokines may serve as useful biomarkers for predicting disease prognosis. 40
Malignant Neoplasms of the Head and Neck
Squamous cell carcinoma (SCC) encompasses 95% of head and neck cancer diagnoses. 40 An important risk factor in the pathophysiology of HNSCC is HPV positivity, particularly HPV-16. 41,42 For oropharyngeal squamous cell carcinoma (OPSCC), HPV positivity has become a mainstay in characterizing and defining trajectory of disease. 42 -44 Tumorigenesis appears to be induced by expression of E6 and E7 viral proteins that target and inactivate p53 and Rb tumor suppressor genes. 43,44 Patients with HPV-positive OPSCC have significantly improved overall and disease-free survival and radio-sensitivity compared with HPV-negative patients. 43,44 Pathogenesis involves upregulation of tumor suppressor gene CDKN2A, which produces the protein P16INK4a, (P16). 42 -44 P16 inhibits cell cycle progression by preventing complex formation of cyclin D1 and cyclin-dependent kinase. 43 Use of RT-PCR amplification to identify viral E6/E7 expression in patient serum is the gold standard of detection because clinically relevant infection necessitates demonstration of transcriptionally active HPV. 43,44 Circulating serum E6/E7 mRNA or protein elements can be detected via many different types of assays, each with its own sensitivity and specificity. 43,44 Alternatively, immunohistochemistry staining for P16 can be used as a surrogate marker for HPV detection and is 100% and 79% specific. 43 -45 Depending on the assay used, testing for HPV may take anywhere from a few hours to a few days. 43 -45
Other biomarkers are under investigation for surveillance and management of HNSCC; however, few have made it to clinical use as extensively as HPV testing. Chemokines such as CXCR2 and CXCR4 have been associated with rates of metastasis and poor survival outcomes in HNSCC. 40,41 These molecules, however, are identified in tissue samples from tumor biopsies rather than easily collectible secretion samples and lack specificity for HNSCC. 40,41
Among the other types of head and neck cancer, nasopharyngeal carcinoma (NPC) is a distinct entity which has been repeatedly shown to have an association with Epstein Barr virus (EBV). 46 In fact, EBV infection is present in most undifferentiated NPCs and is not detected in other head and neck cancers, except salivary gland tumors. 46 Additionally, EBV DNA has been shown to be detectable in plasma using PCR assays, which can yield results in a few hours to days. 47 Levels of EBV DNA prior to treatment with radiotherapy were shown to correlate to multiple clinical variables including tumor stage, nodal stage, and overall disease stage. 47 EBV levels measured from patient plasma after treatment with radiation therapy were shown to be an important and independent predictor of treatment outcomes, including freedom from relapse. 46,47 Thus, EBV DNA is detectable in circulation, specific for NPC vs other types of head and neck cancer, and correlates to NPC disease trajectory for prognostication and treatment, making it an important and reliable biomarker in the clinical management of NPC. 46,47
Several studies have determined correlations of certain serum markers in patients with either benign or malignant salivary gland tumors, but to date, these have not been validated to diagnose or track disease course. IL-33, a cytokine that is normally involved in type-2 immune responses, has also been implicated in tumor evasion from the immune system and was shown to be significantly elevated in patients with both malignant and benign salivary tumors when compared to healthy, matched control subjects. 48 Specifically, elevations in IL-33 were seen in patients with adenoid cystic carcinoma, mucoepidermoid carcinoma, and mixed malignant tumors more so than other malignant tumor groups, and serum IL-33 was correlated with tumor stage and size. 48 Furthermore, these groups had significantly higher IL-33 levels compared to the benign salivary gland tumors. 48 Galectin-3 (Gal-3) has also been demonstrated to be significantly elevated in salivary gland tumors when compared to healthy control patients. 49 However, Gal-3, a lectin family protein implicated in malignancy of several other human tumors, was not indicative of tumor size, metastasis, or prognosis. 49,50
In the treatment for differentiated thyroid carcinoma, thyroglobulin is a widely used biomarker in treatment follow-up. Thyroglobulin is a 660 kDa glycoprotein specific to normal and neoplastic thyroid follicular cells and should not be detectable in a patient who has undergone total thyroidectomy or total thyroid ablation. 51 Serum concentration of thyroglobulin can serve as an indicator of tumor burden and prognosis. Bachelot et al determined in their study that serum thyroglobulin correlated significantly with tumor volume and number of metastatic lymph nodes. 51 Absence of thyroglobulin after thyroid hormone withdrawal is an excellent predictor of prolonged disease-free survival. 51,52
While thyroglobulin detection assays can be run within a few hours, and the tissue specificity and correlation with prognosis make it a great biomarker, its accuracy can be affected by several factors, such as anti-thyroglobulin antibodies and thyroid replacement therapy. 52,53 In patients actively receiving thyroxine treatment, the sensitivity of serum thyroglobulin in picking up isolated lymph node metastases is only 80% compared to 90% after withdrawal of treatment. 52,53 This is because serum thyroglobulin is dependent on thyroid stimulating hormone (TSH). Without TSH stimulation, tumor production of thyroglobulin remains low and reduces sensitivity of the assay. 52,53 If unaccounted for, these factors can interfere with the accuracy of serum thyroglobulin measurements and its use as a biomarker.
Although also a thyroid-related malignancy, medullary thyroid cancer (MTC) represents a separate entity from other thyroid cancer types. MTC arises from the parafollicular cells (C-cells) of the thyroid and account for 3-4% of thyroid cancers. 54 C-cells are neuroendocrine cells involved in calcium homeostasis through production and secretion of the peptide hormone calcitonin. 54 This 3.45 kDa protein can be useful in screening for MTC treatment monitoring. 54 In a comparison of basal calcitonin levels in patients with MTC to normal controls, Pacini et al 54 found that normal subjects have a basal calcitonin typically 10 pg/mL or less, and subjects with MTC had significantly elevated levels, greater than 50 pg/mL. 54 In another study of 226 MTC patients, Cohen et al found that a calcitonin level greater than 1000pg/mL significantly correlated with a larger median tumor size (25.0 mm) than calcitonin levels below 100 pg/mL (3.0 mm). 55 This study also found that basal calcitonin levels significantly predicted the normalization of calcitonin postoperatively. 55 Serum calcitonin is also used postoperatively in MTC patients to evaluate for disease recurrence. 56 The half-life of calcitonin is approximately 30 hours and available assays can detect calcitonin levels within a few hours. 56,57 Serum calcitonin testing is recommended at 2-3 months after surgery. 56 While undetectable, calcitonin is a strong predictor of remission and is best confirmed with provocative testing, usually with pentagastrin. 55,57 Normalization of calcitonin is highly sensitive for disease remission and a good prognostic indicator. 56,57
Another useful key biomarker within the realm of head and neck cancers includes parathyroid hormone (PTH), a key hormone in calcium homeostasis. In 80-85% of the time, hyperparathyroidism is due to parathyroid adenoma. 58 Severe elevations in serum PTH levels and subsequent elevations or declines in serum calcium can result in severe systemic effects and even be fatal. 58,59 In the absence of chronic renal disease, very high serum PTH concentrations (5-10 × the upper limit of normal) as well as absolute PTH levels >500 mg/dL raise may be indicative of parathyroid cancer. 60 As such, monitoring of PTH is critical to the otolaryngologist. PTH is an 84-amino acid peptide hormone, secreted by the chief cells of the parathyroid gland with a half-life of less than 5 minutes. 61,62 First formed as a 115-amino acid prepro-PTH molecule, it is then cleaved at the N-terminus into the final, functional 84-amino acid product. 61,62
The molecular fragments of PTH become important when discussing the assays in use for measuring PTH levels. 63 -66 First generation methods have been largely replaced by second generation methods. 65,67 These newer tests, called immunometric assays, employ a “sandwich” model utilizing 2 different antibodies, one targeting the C-terminus and the other targeting the N-terminus. 65 -67 This assay therefore isolates the full-length bioactive PTH rather than fragments. 65 -67 This is the assay most commonly used currently and is the “intact” PTH test ordered at most facilities and can be run quickly, on the order of 10-30 minutes. 64 -67 Intraoperative PTH testing has been used to confirm successful management of hyperparathyroidism due to adenoma or cancer, to demonstrate existence of multi-glandular disease, or identify existence of ectopic glands. 64,68,69
Conclusion
Clinical Biomarkers in Otolaryngology.
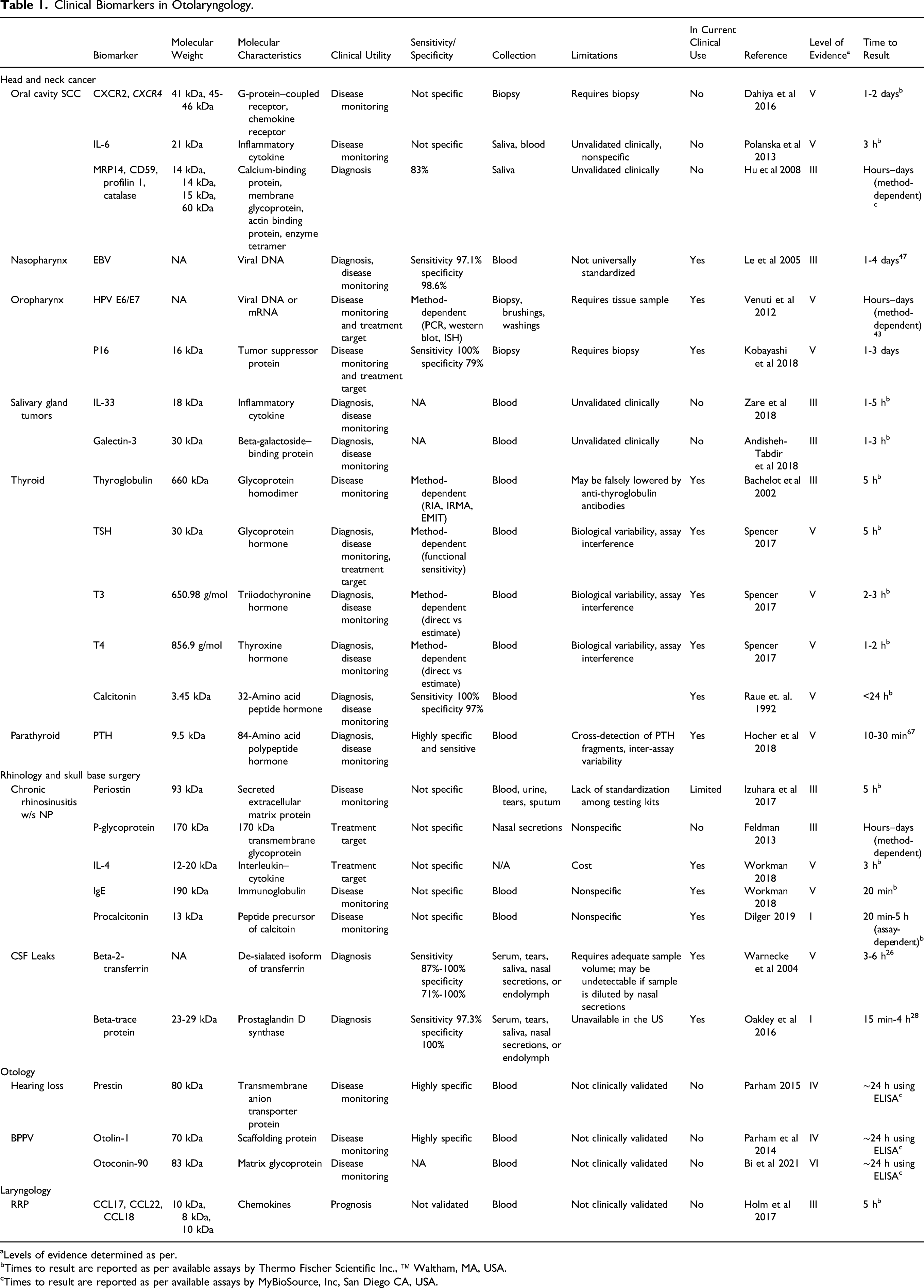
aLevels of evidence determined as per.
bTimes to result are reported as per available assays by Thermo Fischer Scientific Inc., ™ Waltham, MA, USA.
cTimes to result are reported as per available assays by MyBioSource, Inc, San Diego CA, USA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
