Abstract
The coronavirus disease 2019 (COVID-19) is a global pandemic caused by severe acute respiratory syndrome coronavirus 2. Rapid spread with rampant growth of cases and deaths brought forth an urgent need for novel therapies including vaccinations. The mRNA vaccines for COVID-19 disease have been implemented at an unprecedented scale in an effort to combat the unrelenting pandemic. Such a massive scale vaccination program is bound to coincide with adverse events related to treatment. We present a case of massive cervical lymphadenopathy in a 58-year-old male patient post-Moderna COVID-19 vaccination. Additional investigations did not identify malignancy and he was diagnosed with vaccine-related lymphadenopathy. Patient significantly improved with corticosteroid treatment within 2 days of admission. Lymphadenopathy is reported as the second most common local reaction to the Moderna vaccine. Promoting knowledge of this side effect, particularly in the setting widespread vaccination efforts, would allow for better management of cases, especially in relation to oncologic patients.
Introduction
The United States Food and Drug Administration has allowed emergency use authorization for the Moderna coronavirus disease 2019 (COVID-19) vaccine to minimize the effects of the pandemic in December of 2020. The vaccine has a 94.1% effectiveness and is given through two separate doses 28 days apart. The Centers for Disease Control and Prevention (CDC) data show that as of September 6, 2021, 62.3% of Americans have received one vaccine dose and 53% are fully vaccinated, with 66.1 million people receiving 2 doses of Moderna COVID-19 vaccine. 1
The Moderna COVID-19 vaccine contains mRNA that instructs the body’s immune cells to produce a spike protein. This protein is recognized as an antigen that elicits an immune response to provide defense against said antigen. Because this spike protein contains characteristics of the COVID-19 virus, the resulting immune response solicits production of B-lymphocytes and T-lymphocytes thereby allowing for future protection against exposure to SARS-CoV-2.
Though considered safe, COVID-19 vaccines may have side effects, and with increased efforts to vaccinate the nation, it is imperative that clinicians are aware of potential adverse effects. Axillary swelling and tenderness are the second most frequently reported local reaction to the Moderna COVID-19 vaccine, occurring in 11.6% of patients receiving the first dose and 16.0% of recipients receiving the second dose. Such responses are seen as less common with those who received Pfizer–BioNTech vaccine. 2 We have observed one such case of massive lymphadenopathy in a recently vaccinated patient.
Case Presentation
A 58-year-old man was admitted to the hospital with low-grade fever, fatigue, myalgia, left neck swelling, and dysphagia for 2 weeks prior to admission. His past medical history was significant for hypertension. He reports no history of this type of swelling in the past, no history of intravenous drug use, and no history of cancer. He had a recent Moderna COVID-19 vaccination just 2 days prior to onset of his symptoms. On physical examination, the patient had an approximately 5.0 cm × 3.0 cm large, firm, and tender areas of edema along the left mandible and extending over the left neck. There was no pharyngeal erythema, oral ulceration, exudate, or rash. No laryngeal lesions were observed with flexible fiberoptic laryngeal endoscopy.
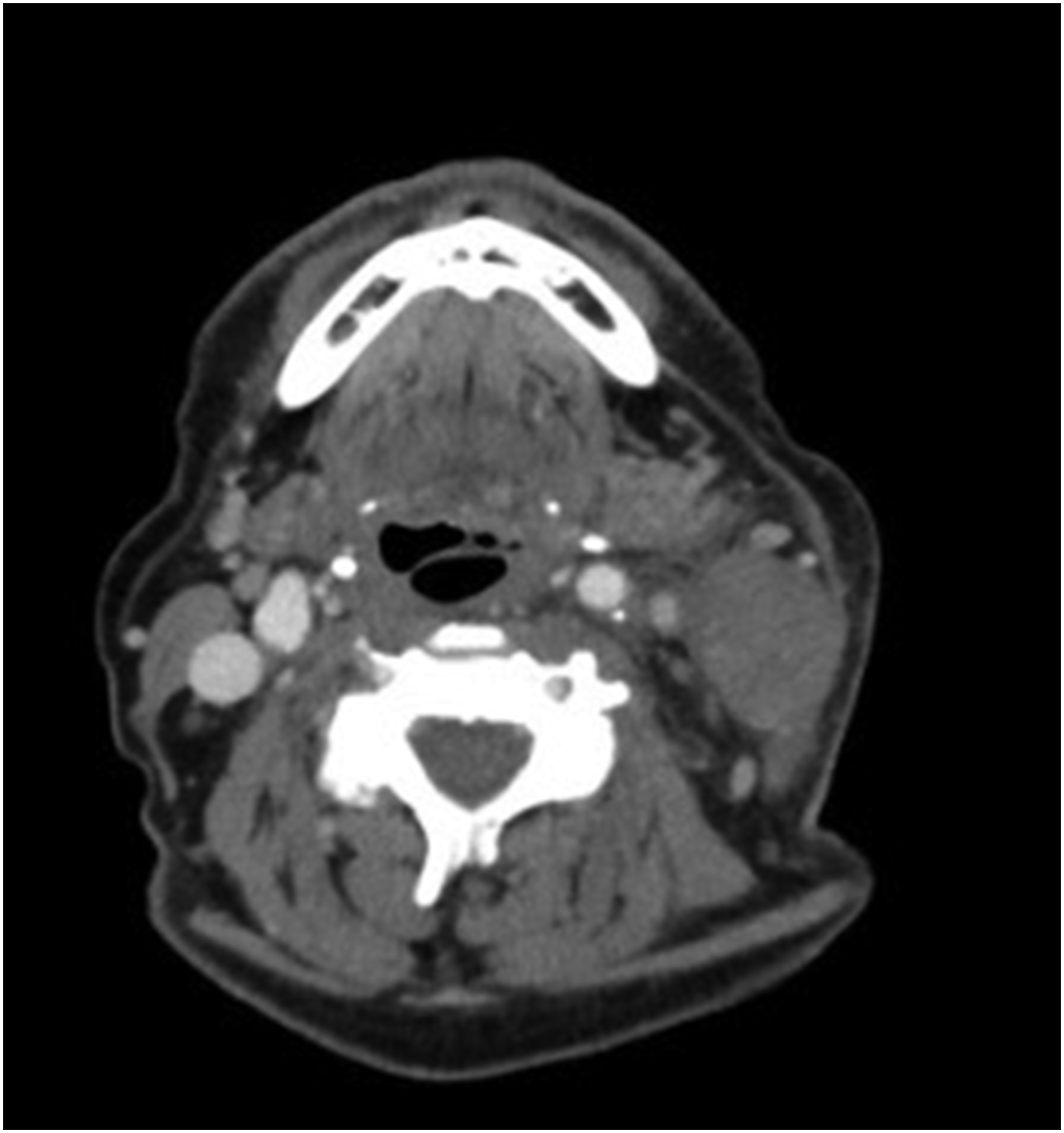
During the hospitalization, he had intermittent low-grade fever (99.9°F). Laboratory findings revealed an erythrocyte sedimentation rate (ESR) of 84 mm/hr, C-reactive protein (CRP) of 5.6 mg/dL, and complete blood count (CBC) and complete metabolic panel within normal limits. HIV test, COVID-19 test, respiratory viral panel, blood cultures, and strep screen were all negative, along with normal serum procalcitonin levels. Computed tomography (CT) scan of the neck showed marked irregular thickening and inflammation of the sternocleidomastoid area with adjacent swelling on the left side of the neck, measuring 5.9 cm × 3.7 cm × 1.8 cm (Figures 1 and 2). Ultrasound of the neck was positive for large mass, causing mass effect upon the left internal jugular vein. Ultrasound-guided biopsy of the left neck lymph node was done; pathology was negative for malignancy and positive for inflammatory cells. Computed tomography scan of the neck with contrast pre-treatment showing significant swelling on the left side of the neck and inflammation of the sternocleidomastoid area. Computed tomography scan of the neck with contrast pre-treatment showing significant swelling on the left side of the neck and inflammation of the sternocleidomastoid area.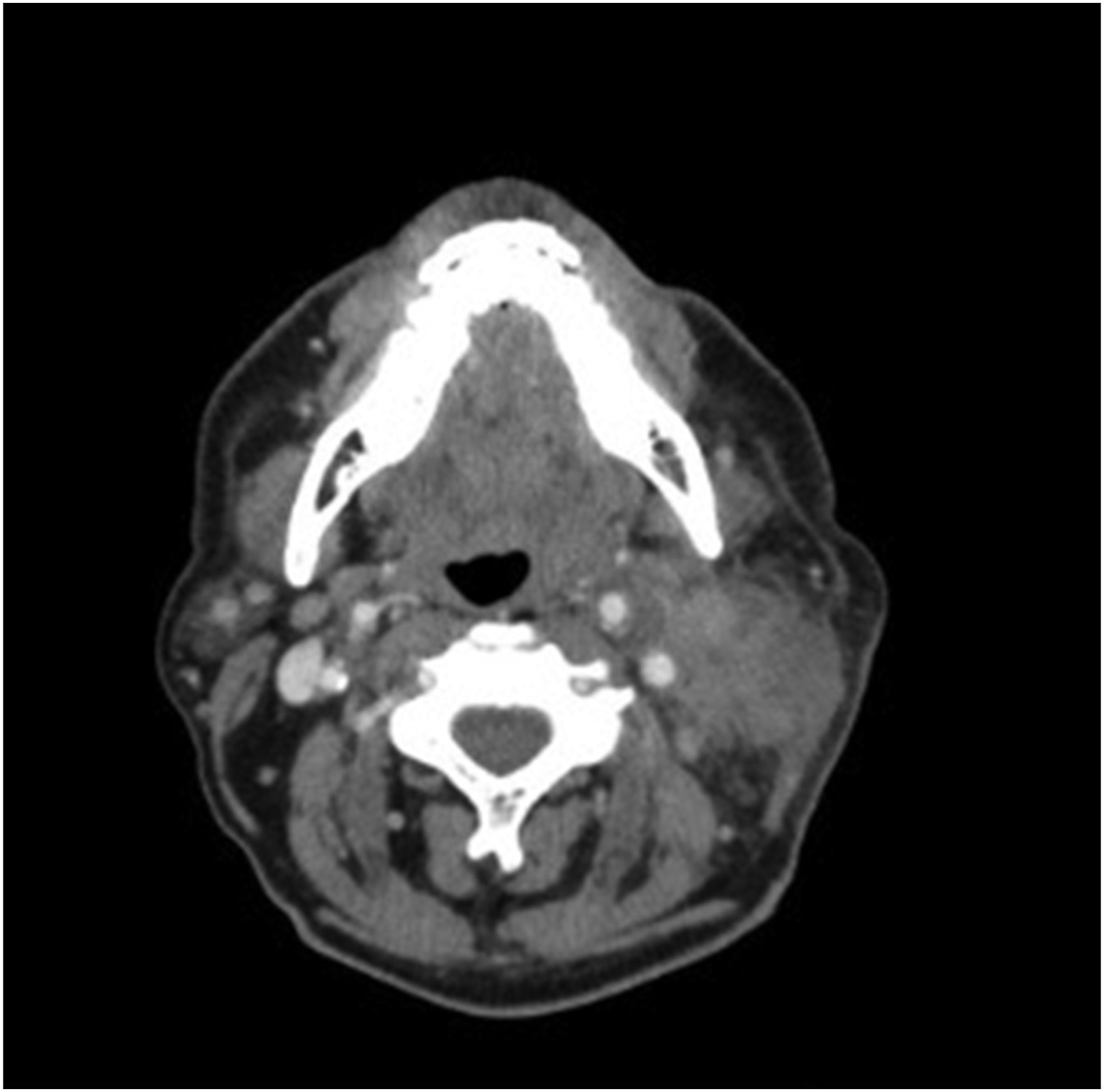
This case was evaluated by otolaryngology and infectious disease services. Hospitalization treatment course included IV piperacillin/tazobactam as empiric antibiotic pending blood culture and an IV dexamethasone 6 mg once a day for 2 days. Symptoms improved with resolution of fevers, diminished left neck swelling, and resolution of dysphagia. Repeat CT scan of the neck on the second day of admission showed significantly decreased left neck mass swelling (Figure 3). The patient was then discharged home in stable condition. During a follow-up telephone interview 2 weeks later, the patient reported no residual symptoms. Computed tomography scan of the neck with contrast post-treatment showing deceased left neck mass swelling.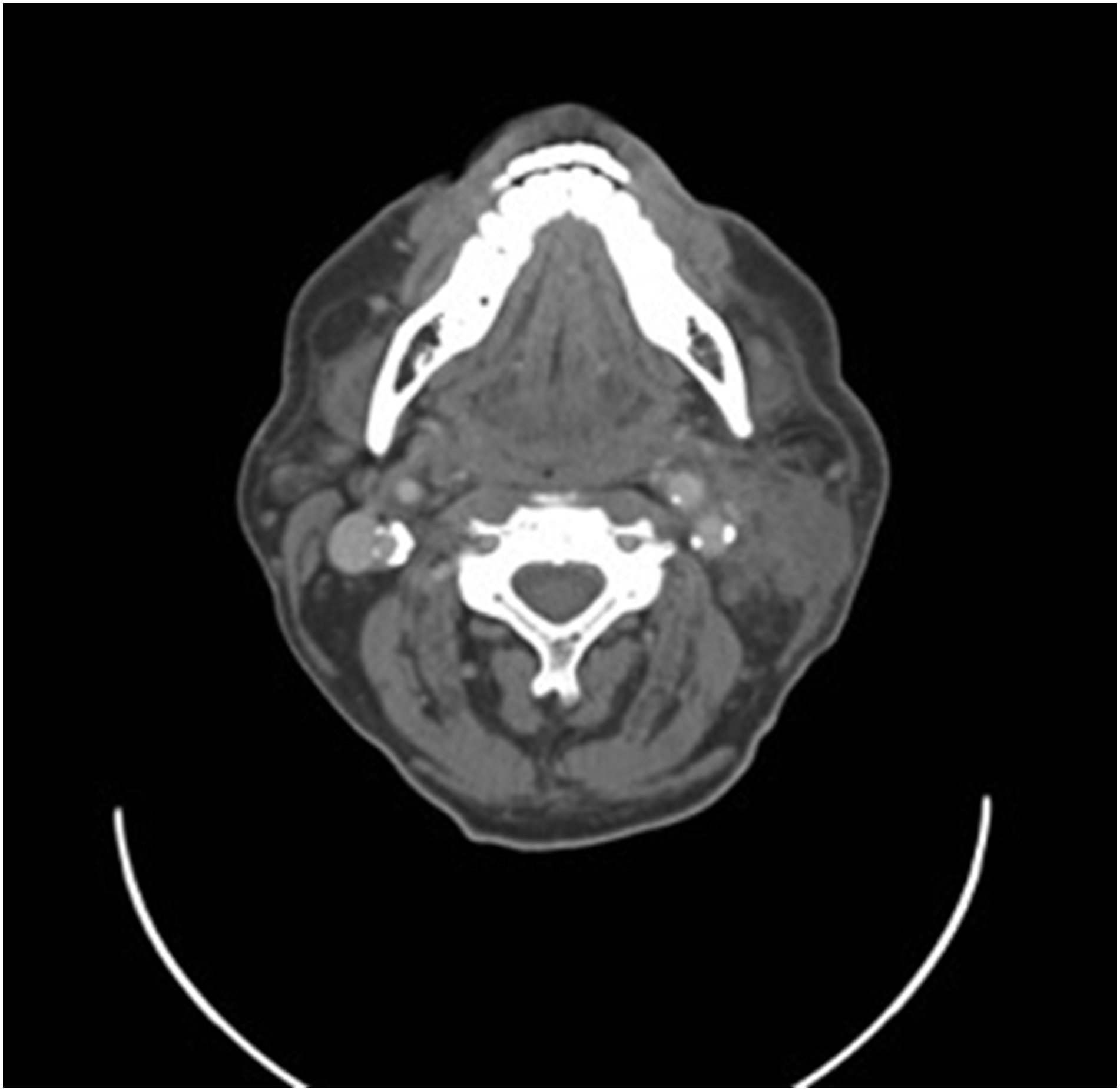
Discussion
In this case, exuberant lymphadenopathy was observed in the setting of COVID-19 vaccination. Initial differential diagnosis was concerning for infectious versus neoplastic etiology. However, further questioning revealed that this patient had received COVID-19 vaccination 2 days prior to onset of symptoms. For those who received the Moderna vaccine, reports of lymphadenopathy were more common (1.1%) when compared with the placebo group (0.6%), 2 typically occurring within 2 to 4 days after vaccination. Given our patient’s history and negative work up, it was concluded that lymphadenopathy has most likely developed because of recent COVID-19 vaccination.
Lymphadenopathy on the same side of vaccination has been previously reported as a side effect of other vaccinations. 3,4 The median duration of lymphadenopathy across reported vaccinated COVID-19 cases was 1 to 2 days. 2 Yet, many cases, including this one, report complete resolution of swelling weeks after treatment. 5 -7 The pathophysiology of vaccine-related lymphadenopathy remains unclear. One study proposes that vaccination performed too high on the arm, as opposed to injecting into the deltoid muscle, may be associated with such a response, though this hypothesis has not been validated. 8 There are cases of transient fluorodeoxyglucose (FDG) uptake in the cervical lymph nodes of patients receiving positron emission tomography (PET) or CT scans after COVID-19 vaccination. Since interpretation of PET/CT scans of post-vaccination lymphadenopathy cases may be confounded with metastasis, delaying routine cancer surveillance imaging after vaccination in oncology patients may be beneficial to avoid confusion. 9
Though this was an unusual case of exuberant lymphadenopathy in the setting of COVID-19 vaccination, it is important for clinicians in internal medicine, infectious disease, emergency room, and otolaryngology to be familiar with this potential side effect that may involve cervical and axillary lymph nodes. Obtaining accurate vaccination history when encountering patients with new lymphadenopathy will aid in management and appropriate diagnostic tool utilization.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
