Abstract
Introduction
Bell’s palsy or idiopathic facial nerve palsy is the most common cause of facial nerve paralysis, accounting for at least half of all facial nerve palsies. 1 The annual incidence of Bell’s palsy is up to 32 per 1,00,000. 1 In majority of the cases, the palsy usually resolves within 3 weeks from the onset of the palsy. 1 Prednisolone has been shown to improve the outcome in Bell’s palsy, with a complete resolution of the symptoms in 83% of the patients, 2 and it has become an established treatment modality in the acute phase. Facial nerve palsy can also be caused, for example, by trauma, infections, or tumors.
Facial nerve palsy causes medical, functional, and esthetical harm to the affected individual. The deficit in eye closure and blinking is a major concern, as blinking is essential in lubricating and protecting the surface of the eye. Consequently, individuals with palsy suffer from the ocular surface problems associated with drying of the eye surface, such as discomfort, epiphora, and blurring of vision. Furthermore, individuals with palsy are also liable to suffer serious complications, such as corneal ulceration and scarring. At present, eye drops, gels, and mechanical eye closure, for example, with a tape during night-time, are the treatments of choice to prevent eye complications in the acute phase of Bell’s palsy. 3
An electrically elicited blink in facial nerve injury has been investigated in several animal studies. 4 -7 In human studies, it has been shown that a blink with natural kinematics can be achieved in healthy individuals using the appropriate stimulation parameters. 8 A 78-minute setting of stimulated blinks at 5 seconds intervals in healthy individuals was perceived to be somewhat uncomfortable but not painful. 9 In a study conducted on patients with acute facial nerve palsy, the electrical stimulation evoked a complete blink in 55% of participants. 10 In another study on individuals with unresolved facial nerve palsy, the electrically elicited blinking reduced subjective eye dryness in a two-hour setting. 11 The present aim is to study the functionality of a stimulus-induced blink by assessing visual acuity and ocular symptoms and the acceptability of this technique on individuals with acute facial nerve palsy in a 2-hour experimental setting that requires continuous visual attention.
Methods
Participants
Seventeen patients with facial nerve palsy were recruited after visits to the Department of Otorhinolaryngology, Tampere University Hospital, Tampere, Finland. Inclusion criteria were facial nerve palsy with less than 3 months from the onset of the symptoms and a deficit in eye closure during blinking, defined as a maximal blink covering less than half of the pupil, and age from 18 to 75 years. Exclusion criteria were history of diabetic neuropathy, unstable cardiovascular disease, neurologic disease causing severe cognitive or motor impairment, severe immunological deficiency, and malignant diseases that were not in remission. The participants were asked not to use eye drops or gels for 2 hours before the study visit. At the time of the study visit, 1 participant had developed other cranial nerve palsies and 1 showed full eye closure when blinking, and they were subsequently excluded. Thus, 15 participants, 8 females and 7 males, age range 32 to 72 years, mean (M) = 54, standard deviation (SD) = 12, were enrolled in the study. The etiology was Bell’s palsy in 12 participants, Ramsay Hunt syndrome in 2 participants, and sequelae of the temporo-mandibular joint replacement in 1 participant. At the time of the first study visit, the duration of the palsy ranged from 7 to 82 days (Median = 10, M = 21, SD = 21). The severity of the palsy was assessed with the Sunnybrook facial grading system (SB). The ocular symptoms of the participants were assessed with the Ocular Surface Disease Index© (OSDI©). The OSDI© scores ranged from 2.8 to 70.0. The study was approved by the Ethics Committee of Pirkanmaa Hospital District (R15067). All participants signed a consent form for the participation and for the possible use of video and photographic material.
Equipment
The stimulator used in the experiment was developed and manufactured at the Faculty of Medicine and Health Technology, Tampere University. 12 The safety of the stimulation hardware complies with the standard International Electrotechnical Commission (IEC) 60601-2-10 “Particular requirements for the basic safety and essential performance of nerve and muscle stimulators.” The stimulation waveform used in this study was a biphasic square wave with symmetric positive and negative phases of equal width using phase duration of .4 ms and 250 Hz pulse repetition frequency. Pulse train duration was 200 ms, and the pulse amplitude was linearly increased from zero to the target value within a period of 60 to 80 ms from the start of a pulse train. The parameters were selected based on earlier research. 8 -10 The participant’s face was recorded during the experiment with a digital video camera at 50 frames per second for offline video analysis.
Procedure
The experiment started with testing whether the eye-blink could be produced by electrical stimulation. For the stimulation, the skin was prepared with an alcohol swab and 2 commercial adhesive pregelled electrodes (Quirumed®, GMDASZ Manufacturing Co, Ltd, Shenzhen, China) were trimmed to a size of 1 cm2 and attached on the route of the zygomatic branch of the facial nerve. One electrode was attached just lateral to the orbital rim and the other at approximately .5 cm distance laterally. The stimulation was started at a current amplitude level of 1.0 mA. Thereafter, the current amplitude was raised in .5 mA steps until a complete eye closure was achieved as observed online by 2 investigators. The stimulation was stopped if the movement no longer seemed to increase in amplitude as evaluated during the online video analysis, the participant asked to discontinue, or the maximal current amplitude (48 mA) of the device was reached. If the first location of the electrodes did not yield sufficient movement, alternative locations for the lateral electrode were tested, where it was placed approximately .5 to 1 cm upward or downward from its initial position.
The blinks were then analyzed offline from the video. Those participants whose stimulated blink had a movement amplitude that was sufficient to cover the pupil, which approximately corresponds to the definition of a complete blink, 13 were eligible to continue in the study. If a sufficient blink could not be produced, the study visit was ended. The electric current amplitude used during the following experiment was determined offline from the videos as either the lowest current amplitude level that produced a complete eye closure or, if a full eye closure was not achieved, the level that produced the strongest blink.
The experiment continued on the same and the following day. During the experiment, the participant sat comfortably in an armchair and watched a freely chosen TV show on a 50-inch liquid-crystal display screen at a distance of 185 cm in controlled lighting conditions for approximately 2 hours. There were 2 conditions, 1 with the stimulation and another without the stimulation serving as a control condition. In the stimulation condition, the device produced a stimulation in order to elicit a blink at 5-second intervals. In the control condition, the participant watched TV without the stimulated blink. The 2 conditions were counterbalanced so that half of the participants had the stimulation condition on day 1 and the other half on day 2.
Assessment of the Effects of the Stimulation
The amplitude of both spontaneous and stimulated blinks was analyzed offline from the video recordings by 1 investigator. The amplitude of the spontaneous blinks was evaluated from the first eleven blinks of the control condition. The amplitude of the stimulated blinks was evaluated during the stimulation condition, the first time at 1 minute and then every 10 minutes, from 5 blinks where no simultaneous blink was observed on the unaffected side. The amplitude of the stimulated blinks was then compared with the amplitude of the participant’s spontaneous blinks. The classification for the blink amplitude was 5 = complete eye closure; 4 = nearly complete eye closure; 3= eyelid covers the pupil; 2 = eyelid partially covers the pupil; 1 = twitch; and 0 = no movement.
Visual acuity was evaluated from both eyes separately with a Logarithm of the Minimum Angle of Resolution (LogMAR) chart at a distance of 4 m with the participant’s own spectacles. The ocular symptoms were assessed with a questionnaire based on the Dry Eye Questionnaires (DEQ) 14,15 customized to also evaluate excessive eye-watering, which is a typical symptom in facial nerve palsy. The visual acuity and the ocular symptoms were assessed before and after the TV watching session. The pain and discomfort caused by the stimulation were evaluated with 2 numeric rating scales ranging from 0 (not at all painful or uncomfortable) to 10 (very painful or uncomfortable), naturalness of the stimulus-induced blink was assessed with a bipolar scale ranging from − 5 (unnatural) to + 5 (natural); these were assessed after the TV watching session with the stimulated blink. After the stimulation visit, the participant answered to a structured interview on the following experiences: Did they notice the stimulation and did the stimulation interfere with the TV watching? Did the stimulation affect the ocular symptoms? Was the frequency of the stimulated blink appropriate? And finally, would they be interested in using a portable device for this purpose if 1 was available?
Statistical Analysis
Bonferroni corrected pairwise t-test was used to test the difference between the amplitude of the stimulated blinks and the spontaneous blinks of both the unaffected and affected side. One-way repeated measures analysis of variance (ANOVA) with blink amplitude as a factor was used to test whether the blink amplitude changed during the TV watching task. 2-way 2 × 2 ANOVAs with condition (with or without stimulation) and time (before or after the TV watching) as factors were used to analyze the visual acuity test and the DEQ ratings. The subjective ratings on stimulation were analyzed with pairwise t-tests.
Statistical significance was set at a value of P < .05. Statistical analyses were performed using IBM SPSS Statistics for Windows software, version 25.0 (IBM Corp, Armonk, N.Y., USA).
Results
The electrical stimulation produced a blink in 8 out of 15 participants (53%). In these 8 participants, the spontaneous blink of the affected side was significantly more deficient when compared to the unaffected side, t (7) = −9.0, P < .001. At the beginning of the TV watching task, there was no significant difference between the movement amplitude of the spontaneous blink of the unaffected side and the stimulated blink of the affected side, t (7) = .8, P > .05 (Figure 1). The electric current amplitude that was needed to produce a blink ranged from 3.0 to 8.0 mA (M = 4.9 mA, SD = 1.8 mA). The mean blink amplitudes (± standard deviations) for the participants’ spontaneous unaffected side blink, spontaneous affected side blink and stimulated blink.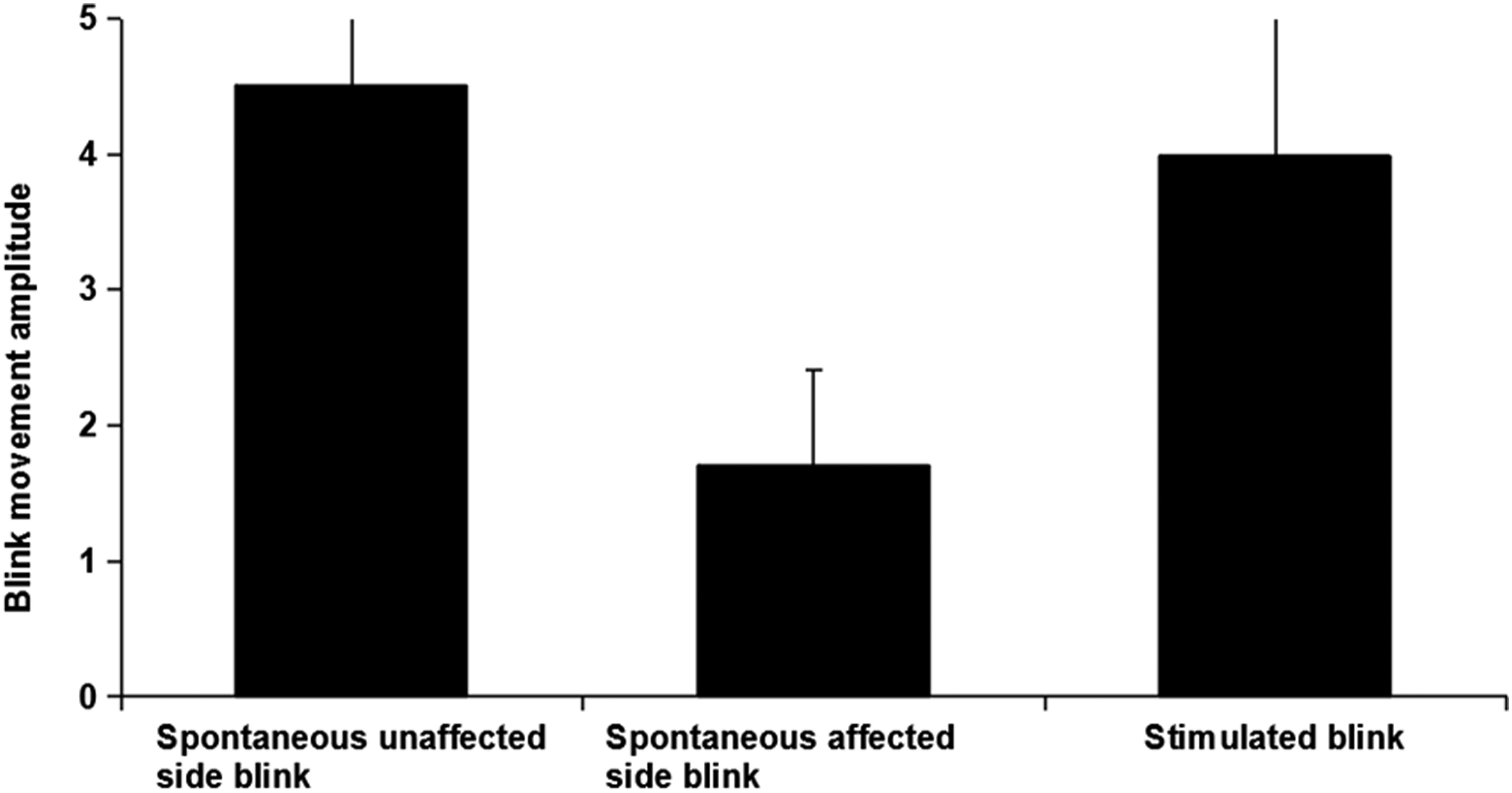
All 8 participants completed the two-hour TV watching session with the stimulation. The movement amplitude of the stimulated blink decreased gradually during the TV watching (Figure 2.) The decrement was statistically significant, F (11, 66) = 3.05, P < .01. Classified mean amplitudes (± standard deviations) of the stimulated blinks as a function of time.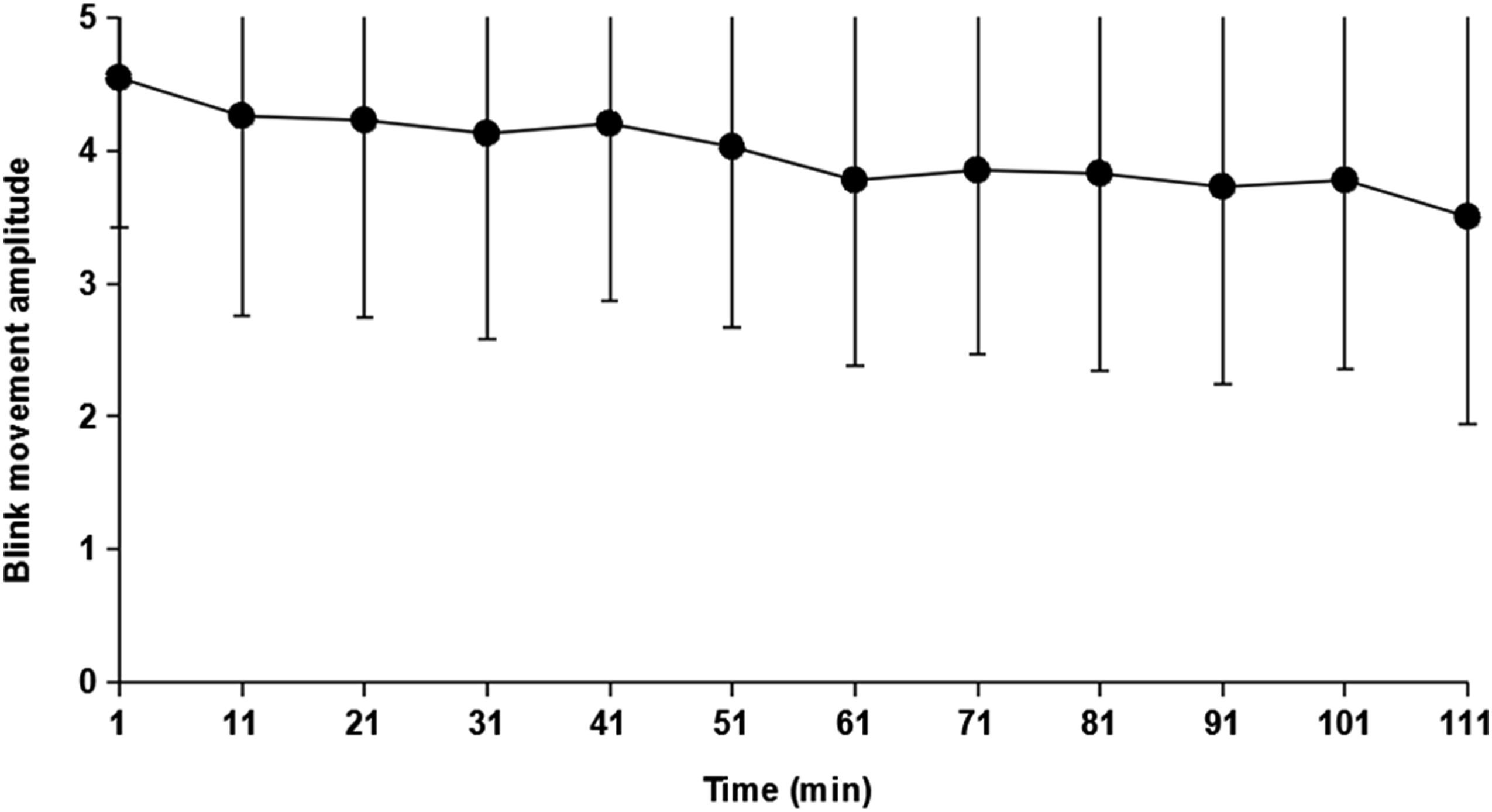
Visual Acuity and Ocular Symptoms
The mean visual acuity at different time points is presented in Figure 3. There was a significant interaction of the main effects of condition and time, F (1,7) = 5.7, P < .05. The visual acuity of the affected eye was significantly lower after the TV session in the control condition than before it, t (7) = −2.6, P < .05. In contrast, there was no significant change in the visual acuity of the affected eye before and after the TV session in the stimulation condition, t (7) = .4, P > .05. For the unaffected eye, there were no significant main effects or interactions for visual acuity. The mean visual acuity (± standard deviations) in Logarithm of the Minimum Angle of Resolution (LogMAR) scale at different time points for both eyes.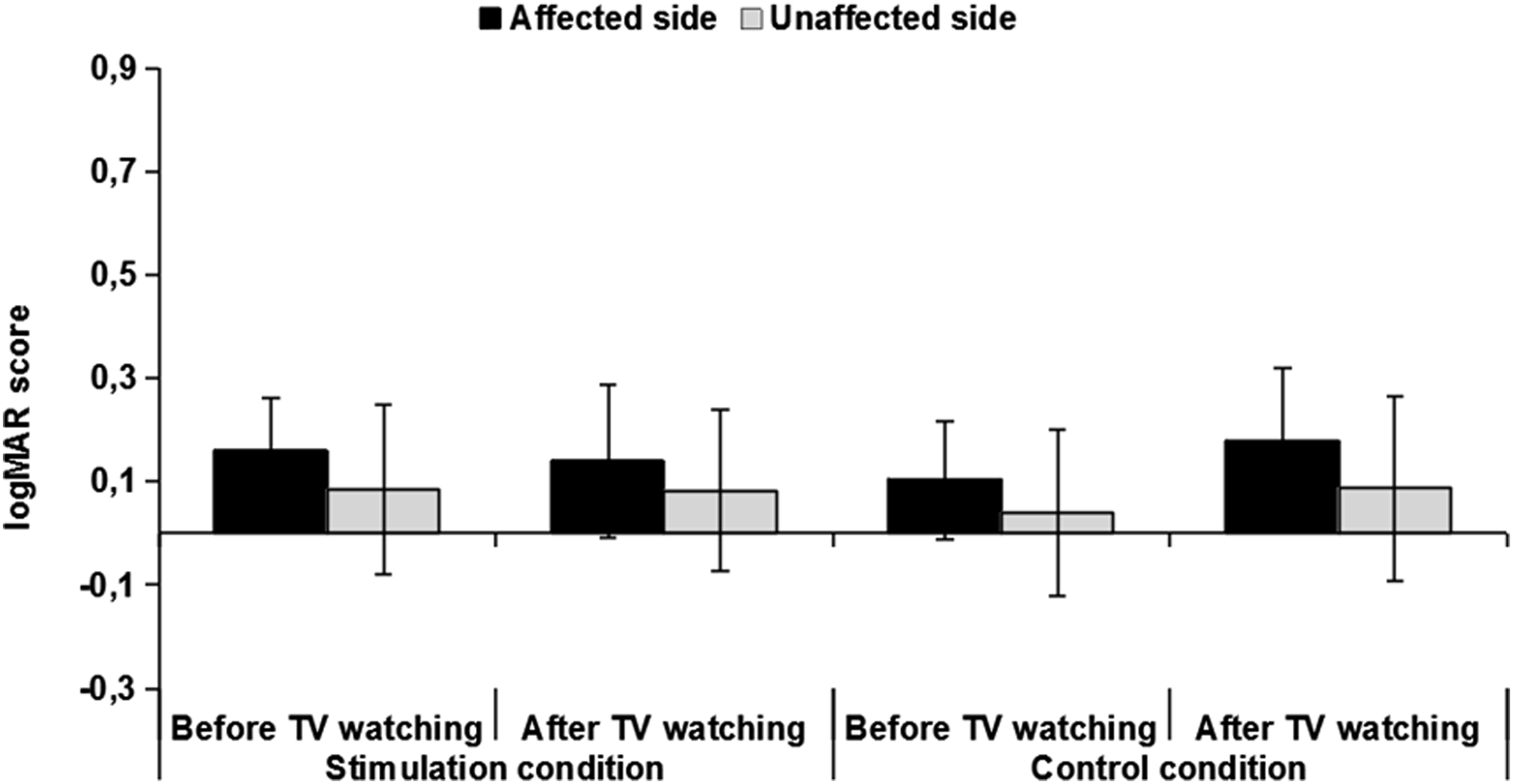
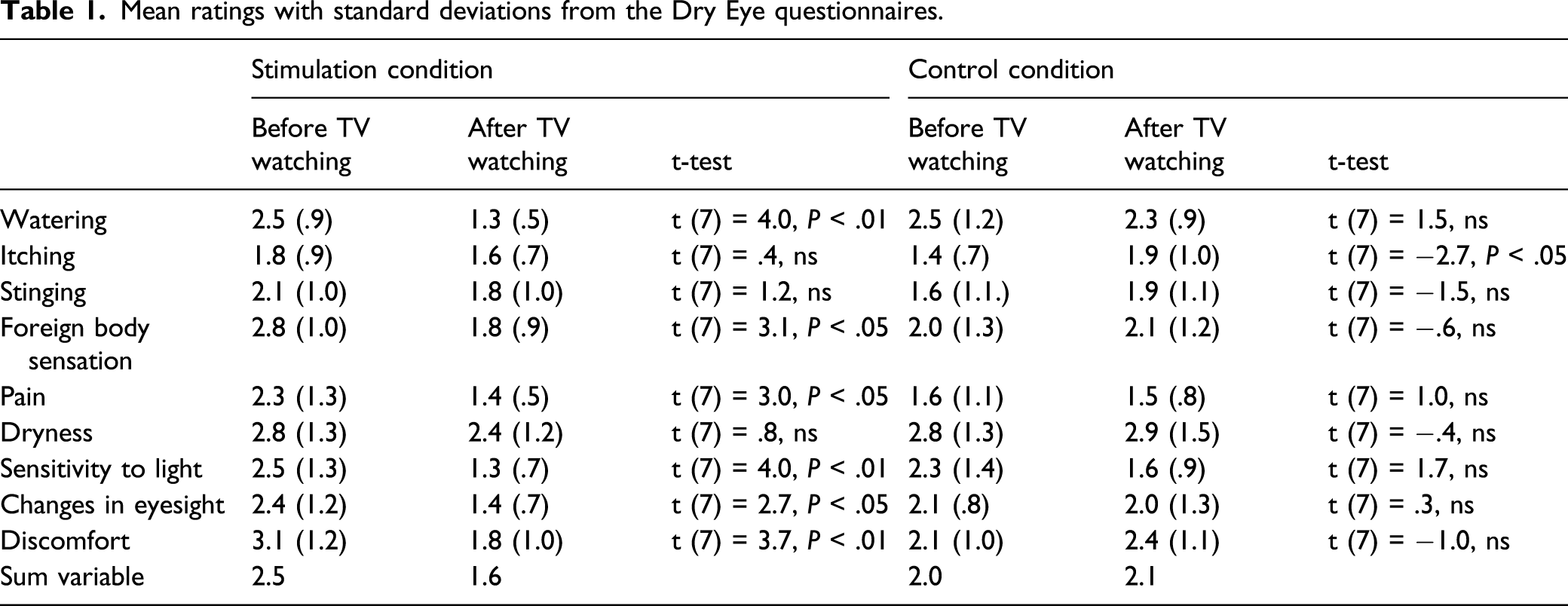
Mean ratings with standard deviations from the Dry Eye questionnaires.
Subjective Evaluations
The participants gave their ratings for stimulation painfulness, stimulation discomfort, and movement naturalness after the TV watching session with the stimulated blink. The mean rating was 1.5 (± 2.1) for stimulation painfulness and 2.1 (± 2.2 SD) for stimulation discomfort. The mean rating for movement naturalness was 0 (± 2.1 SD). In the interview after the experiment, 7 of the 8 participants reported that the stimulation passed unnoticed during the TV watching. Five said that the stimulation reduced their dry eye symptoms. All participants considered that the stimulated blink rate was appropriate. Moreover, 6 participants were interested in trying out a portable blink stimulator in everyday use if available, 1 was unsure and 1 answered with a definite no.
Outcome of the Palsy
The severity of the palsy was clinically evaluated during the experiment visit and at 3 months from the onset of the palsy. One participant whose stimulation had been successful was lost from the follow-up. The first SB scores ranged from 37 to 74 (M = 54, SD = 14) for the participants whose stimulation had been successful and from 5 to 42 (M = 13, SD = 13) for those whose stimulation had not been successful. At 3 months, the SB scores ranged from 63 to 100 (M = 89, SD = 12) for the former group and 4 to 91 (M = 31, SD = 32) for the latter. Mann–Whitney test showed that the scores were significantly higher at both assessments for those whose stimulation had been successful (U = 2.0, P < .01 and U = 2.5, P < .01, respectively). The scores were also significantly higher in those participants whose stimulation had been successful in the follow-up compared with the first evaluation (Wilcoxon signed-rank test, Z = −2.37, P < .05). For those whose stimulation had not succeeded, there was no significant difference between the 2 evaluations (Z = −1.53, P > .05).
Discussion
In this study, the success rate in producing a blink by electrical stimulation was 53%, which is in line with the results of a previous study. 10 In contrast, the average electric current amplitude that was necessary to produce a blink in this study was lower 10 (4.9 mA vs 7.2 mA). This may be explained by several factors, such as the different definition for a sufficient blink in our protocol (eyelid covering the pupil vs complete eye closure), or the different stimulation parameters used.
We are unaware of other studies that have evaluated the effect of electrically induced blinks on visual acuity and ocular symptoms in patients with acute facial nerve palsy. During a two-hour TV watching session, visual acuity declined in the compromised eye. This decline was halted by the electrically elicited blinking during the TV watching task. Participants also reported a reduction in their ocular symptoms after the 2-hour session with a stimulated blink. The electrical stimulation was well tolerated, as none of the participants wanted to interrupt the 2-hour stimulation session. Furthermore, for 7 out of 8 participants, the stimulation passed unnoticed. The reported pain and discomfort ratings were also relatively low.
The offline analysis of the blinks showed that the blink movement amplitude decreased over time. However, even at the end of the 2-hour stimulation session, the mean blink amplitude was still covering the pupil. In healthy subjects, less than half of the spontaneous blinks result in full eye closure 16 so even partial closure is probably sufficient to protect the ocular surface. However, a further decline in blink amplitude would inevitably affect the feasibility of the electrically induced blinking by limiting the duration for which the device could be used at a time. Whether the blink movement amplitude would continue to decline or reach a plateau, and whether this decline could be avoided by adjusting the stimulation parameters needs to be addressed in further investigations.
Studies have reported blink rates (i.e., blinks per minute) of 12.55 to 24.36 at rest. 17 -19 Individuals with acute facial nerve palsy are reported to have an increased blink rate, which is thought to be due to the compensatory hyperactivity of the contralateral facial muscles. 20 Blink rate is influenced by the activities involved, ranging from 4.5 when reading to 26 during conversation. 18 A mean blink rate of 14 has been reported for healthy subjects during movie watching, 21 which is quite close to the rate we used in this experiment (12). The participants found this rate to be appropriate. Further, they also reported that the stimulated blink did not disturb their concentration during TV watching.
It seems that the stimulation was more likely to be successful in participants with a less severe palsy. It is well known that during axonal degeneration, the nerve loses its excitability. It is thus probable that in these participants, the palsy was due to a predominantly demyelinating lesion of the nerve and the extent of the axonal loss was less important than in those with a more severe palsy. This would also explain the better recovery of the participants for whom the stimulation was successful. Hence, whereas electrically induced blinking is a promising method to be used temporarily to reduce ocular symptoms in milder palsies where the recovery is usually expected to occur in a matter of weeks, 1 the other treatment modalities for protecting the eye would still be necessary in more severe cases.
The small number of participants is a limitation of this study and the results would need to be confirmed in larger controlled trials. However, the results of this preliminary study suggest that electrically induced blinking is a well-tolerated and feasible method for maintaining visual acuity and reducing ocular symptoms in acute facial nerve palsies, and thus encouraging for further studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Academy of Finland (278529, 276567, 278312, and 292477) and Tampere University.
