Abstract
Purpose:
To evaluate the impact of parathyroid gland vasculature preservation in-situ technique (PGVPIST) on postoperative parathyroid hormone (PTH) and calcium plasma levels in thyroid patients undergoing total thyroidectomy for papillary thyroid carcinoma (PTC).
Study Design:
Retrospective cohort study.
Methods:
Patients with PTC who underwent total thyroidectomy by either the conventional technique (group 1, January 2019 to January 2020) or PGVPIST (group 2, January 2020 to January 2021) were compared. Postoperative blood calcium levels and PTH levels were assessed in these groups.
Results:
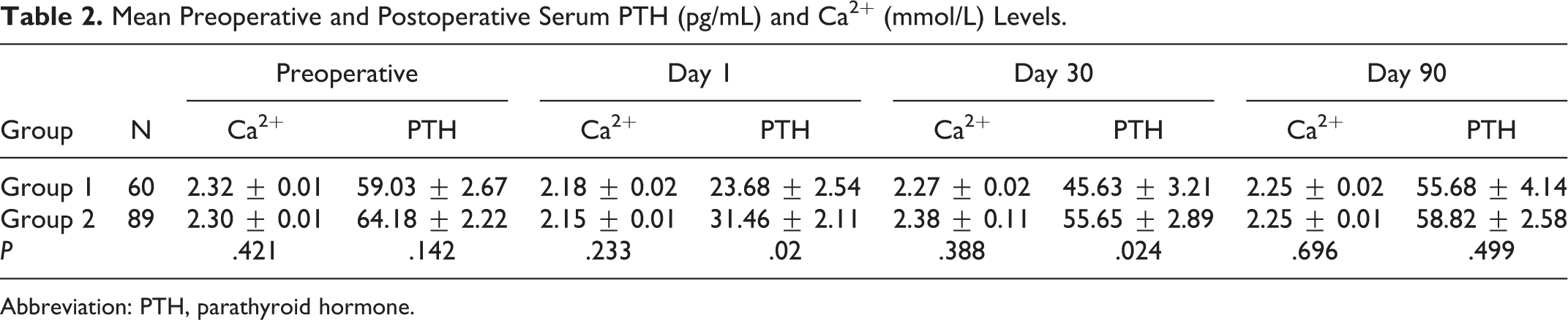
Totally 149 patients with consecutive PTC underwent total thyroidectomy, including 60 patients in group 1 and 89 patients in group 2. Postoperative serum calcium levels in group 1 were insignificantly lower than in group 2 at day 1 (2.18 ± 0.02 vs 2.15 ± 0.01 mmol/L) and day 30 (2.27 ± 0.02 vs 2.38 ± 0.11) after surgery. But postoperative serum PTH levels in group 1 were significantly lower than that in group 2 at day 1 (23.68 ± 2.54 vs 31.46 ± 2.11 pg/mL) and day 30 (45.63 ± 3.21 vs 55.65 ± 2.89 pg/mL) after surgery.
Conclusion:
Parathyroid gland vasculature preservation in-situ technique for PTC is associated with higher PTH level after total thyroidectomy. The parathyroid gland vasculature mostly strongly adheres with adjacent thyroid parenchyma. Therefore, deferred processing of tiny thyroid parenchyma of parathyroid gland vessels is essential to prevent devascularization.
Keywords
Introduction
Thyroid surgery is one of the most frequently performed procedures in the general surgery setting, so its related complications (recurrent laryngeal nerve injury and hypocalcemia) affect a large number of people worldwide, though the development of new techniques and technologies permits better surgical results. The incidence rates of transient and permanent hypocalcemia are about 10% to 50% and 0.5% to 2%, respectively, in patients after total thyroidectomy. 1 The rates of transient and permanent hypoparathyroidism are about 19% to 38% and 0% to 3%, respectively, in patients after thyroidectomy. 2 The clinical symptoms of hypocalcemia include Trousseau’s sign, Chvostek’s sign, and paresthesia and numbness of fingertips which cause discomfort. 3 Hypocalcemia occurs due to vascular injury or inadvertent removal of parathyroid glands in thyroidectomy. Nevertheless, parathyroid gland damage or unintentional excision may occur in up to 19% of cases. Hypoparathyroidism is a principal cause of hypocalcemia, but continued administration of vitamin D and calcium supplementation lengthens hospital stay. 4 Until now, the widely accepted way is to preserve parathyroid glands in situ during total thyroidectomy, besides decreasing short-term hypoparathyroidism. 5 Many methods of parathyroid vascular preservation in situ have been described, and each shows both advantages and drawbacks. In particular, many of preservation sites do not permit to check preserved gland function and blood supply vitality, leading to a higher risk of procedure-related complications. In a previous preliminary report, we investigated the preservation of relative thyroid artery/vein or parenchyma as a supply for nonpathologic parathyroid glands, demonstrating excellent results from a short-term follow-up.
The aim of this study is to verify, in a series of unselected patients, the results of parathyroid glands with vasculature preservation in-situ technique (PGVPIST) after short-term follow-up and consider patients’ expectations and satisfaction in clinical practice.
Materials and Methods
A retrospective cohort study of patients with papillary thyroid carcinoma (PTC) was performed by a team of surgeon experts in the Department of Thyroid Surgery at the First Affiliated Hospital of Yangtze University between January 2019 and January 2021. Patient characteristics including gender, age, tumor size, lymph node metastasis, extent of lymph node dissection, and hypoparathyroidism were analyzed (Table 1). Preoperative checks included clinical examination, thyroid and cervical lymph node ultrasound scans, and assay of serum calcium, parathyroid hormone (PTH), and thyroid hormone. Each patient received laryngoscopy before surgery. All patients with PTC underwent total thyroidectomy. Inclusion criteria were diagnosis of PTC, normal preoperative serum calcium, and PTH levels. Patients with parathyroid diseases, non-PTC, parathyroid gland autotransplantation, parathyroid tissues in pathological specimens, or reoperation were excluded. The research protocol was approved by the Ethics Review Board of the Hospital.
Patients Characteristics and Data.
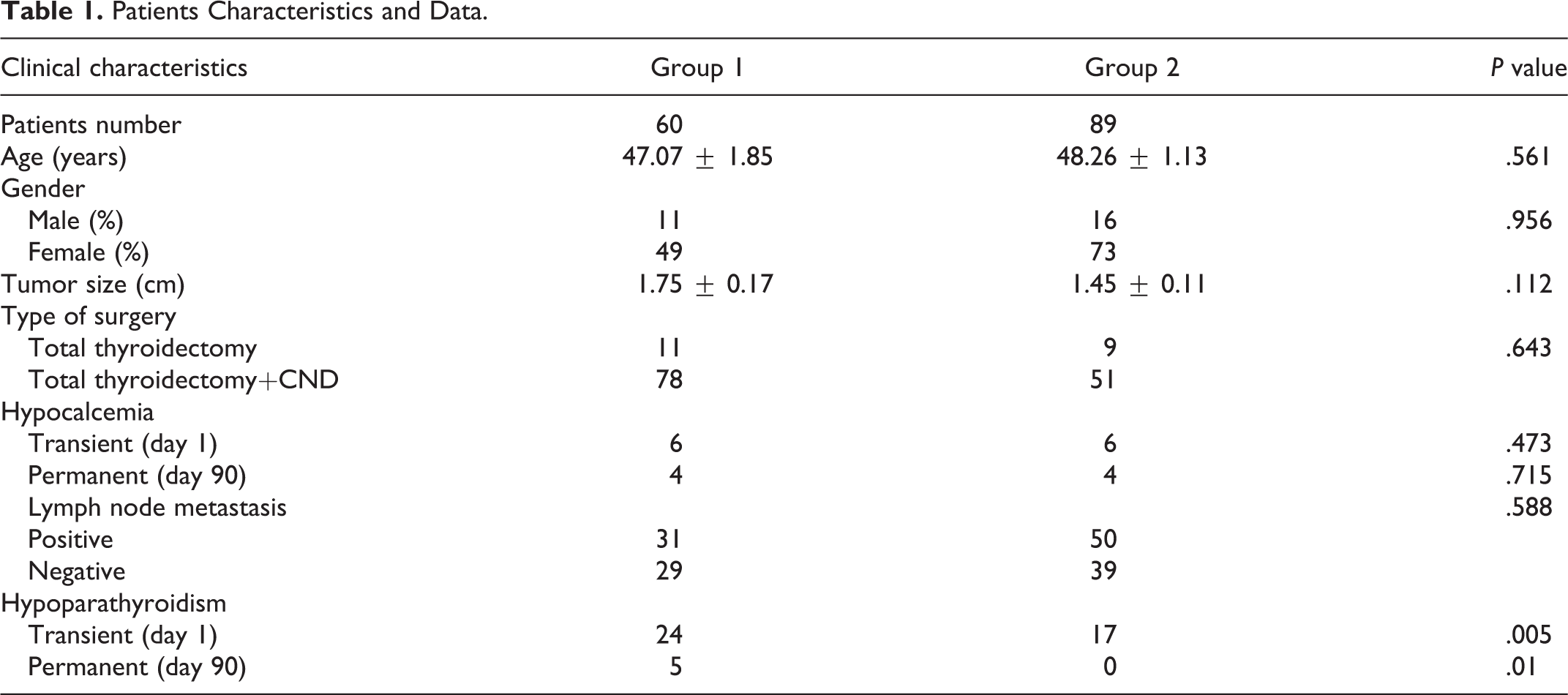
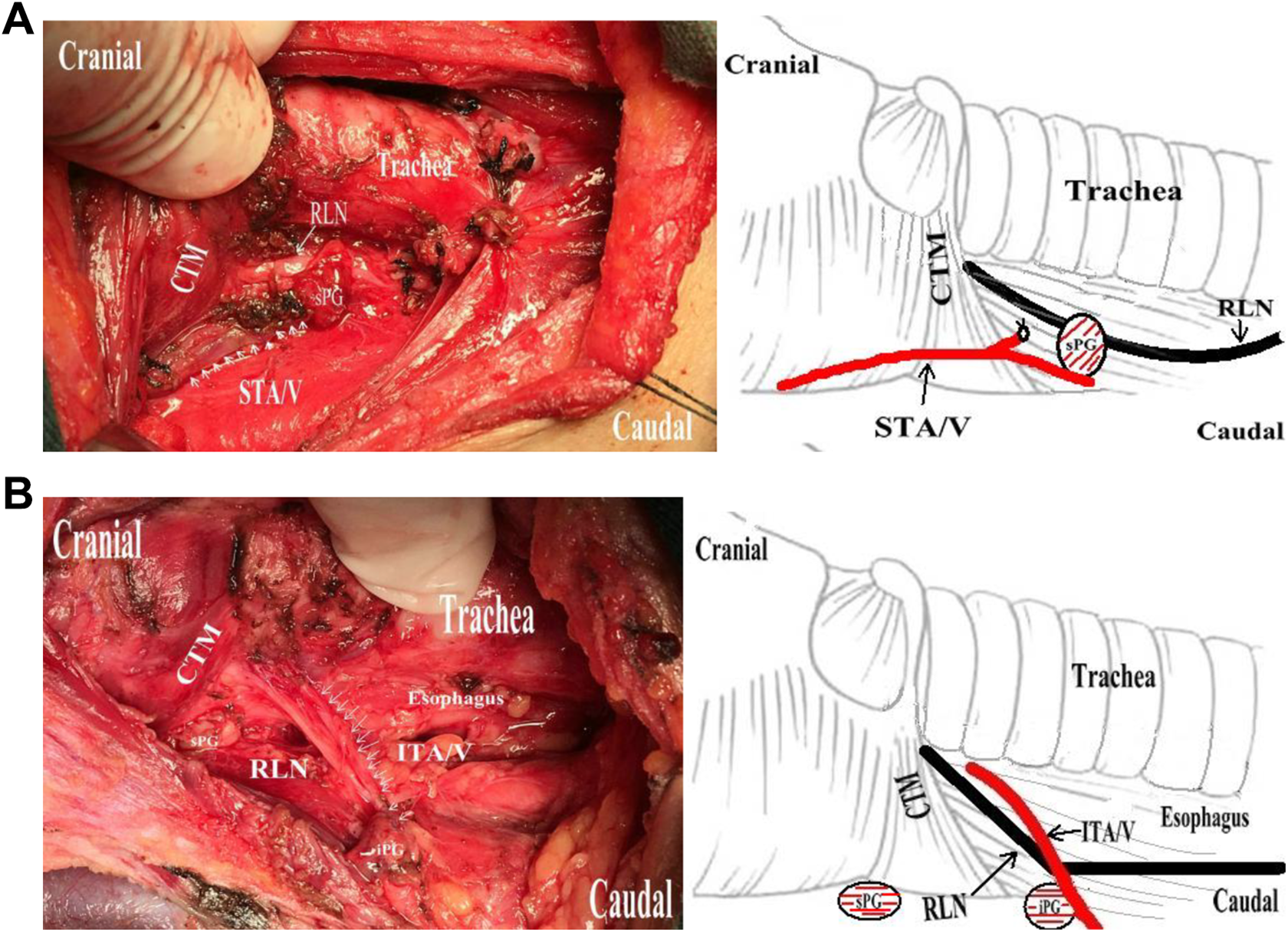
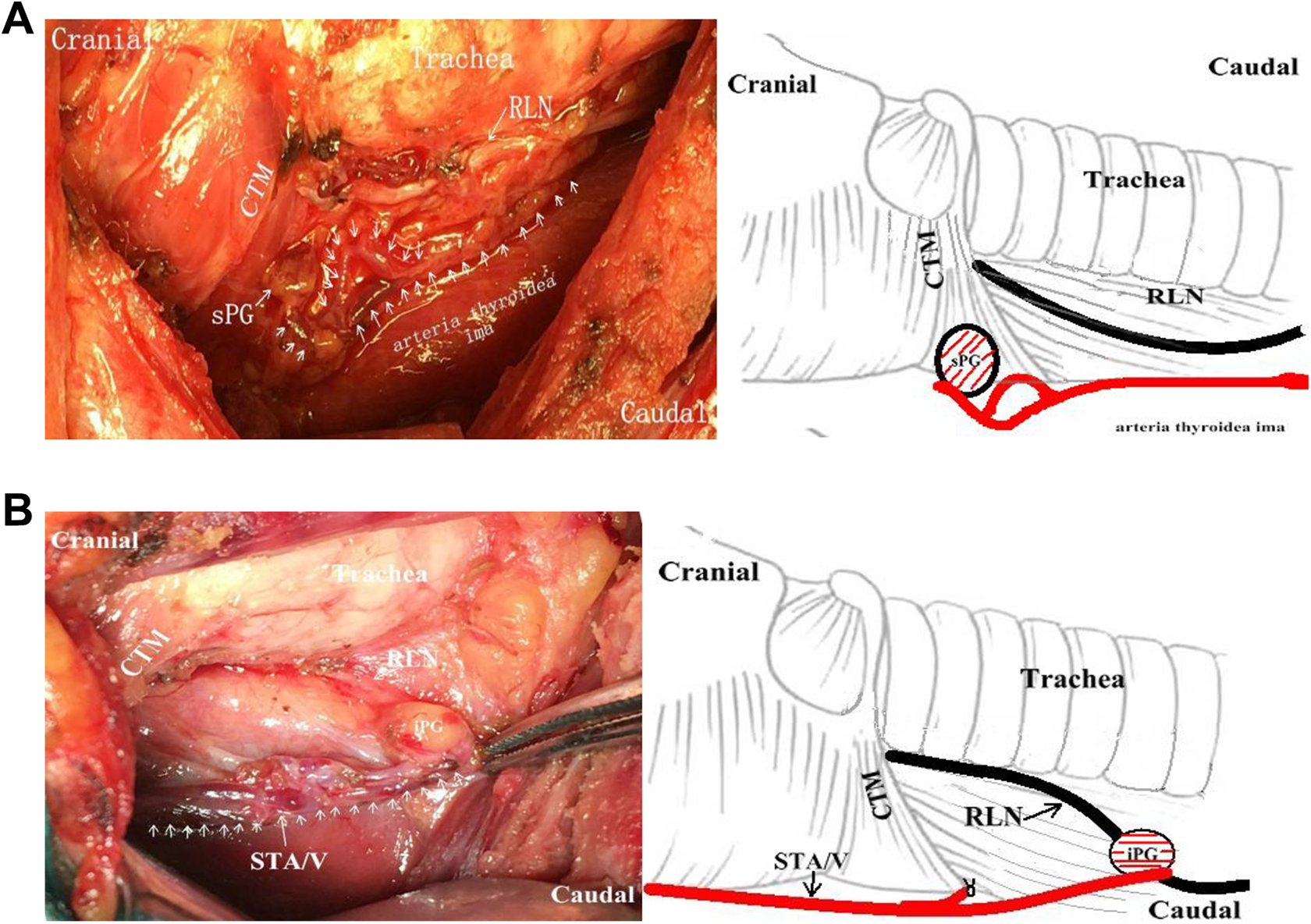
For analysis of operation outcomes with hypoparathyroidism and hypocalcemia before and after the application of PGVPIST, the thyroid patients were divided into group 1 with conventional technique (between January 2019 and January 2020) and group 2 with PGVPIST (between January 2020 and January 2021). The protocol was done in accordance with the guidelines of the Ethical Committee of the Hospital and the Declaration of Helsinki. All procedures were performed under the same general anesthesia. In group 1, patients underwent standard thyroidectomy by the conventional technique. The parathyroid glands in group 1 were simply preserved in situ without distinct blood vessels around these parathyroid glands. In group 1 patients, the artery and vein of inferior thyroid (ITA/V) and superior thyroid (STA/V) trunk ligature were performed no closely to the thyroid capsule on both sides. In group 2, the suspected parathyroid glands with surrounding blood supply or tiny thyroid parenchyma were preserved in situ. The dorsal trunk and terminal branches of STA/V with superior parathyroids were preserved in situ (Figures 1A and 2B). Terminal branches of ITA/V were ligated close to the thyroid parenchyma (Figures 1B and 5A). Inferior parathyroid with blood vasculature was well preserved in situ and close to the thyroid glands. In some special cases, the dorsal branch of thyroide ima artery (Figures 2A, 4B, and 8B) or tiny thyroid parenchyma (adjacent thyroidal tissue 1 × 1 mm3) supplying parathyroid glands were preserved in situ (Figure 3A and B). Each parathyroid gland with vascularity was assessed at the end of the operation. Preservation of the parathyroid gland with its blood supply is a standard surgical goal in group 2.
In all patients, parathyroid glands were identified according to visual macroscopic features, and a frozen-section biopsy was performed in some unidentified cases. Intraoperative confirmation of the frozen-section diagnosis of PTC was followed by total thyroidectomy with central or lateral lymph node clearance. The remaining adjacent thyroidal tissues should be removed by ophthalmic scissors without affecting the parathyroid gland with blood supply. All patients received oral calcium carbonate (1000 mg/d) or intravenous calcium gluconate (2 mg/kg/h). In all patients, serum calcium and PTH levels were measured preoperatively, at day 1, day 30, and day 90 after surgery. Transient hypocalcemia was defined as the serum calcium level below the normal range (Ca2+ <2 mmol/L, 2-2.8 mmol/L) combined with the paresthesias and numbness of the perioral area, peripheral paresthesia, laryngospasm for no more than 6 months. Hypoparathyroidism (PTH < 15 pg/mL, 15-68.3 pg/mL) was defined as permanent when the supplementation period of calcium or vitamin D exceeded 3 months. There were 8 types of blood supply of the superior and inferior parathyroid glands with preservation measures (Figures 1A-8A and 1B-8B). Seven patients with PTC were lost to follow-up. Parathyroid hormone and blood calcium were routinely tested from day 1 after surgery to day 30, day 90 following hospital discharge to confirm the effectiveness of PGVPIST for protecting parathyroid function.
A, Type I sPG was characterized by a single dorsal terminal branch of superior thyroid artery and vein (STA/V; right side). B, Type I iPG was characterized by a single blood supply of inferior thyroid artery and vein (ITA/V; right side).
A, Type II sPG was characterized by a single blood supply of thyroid ima artery (right side). B, Type II iPG was characterized by a single blood supply of superior thyroid artery and vein (STA/V; right side).
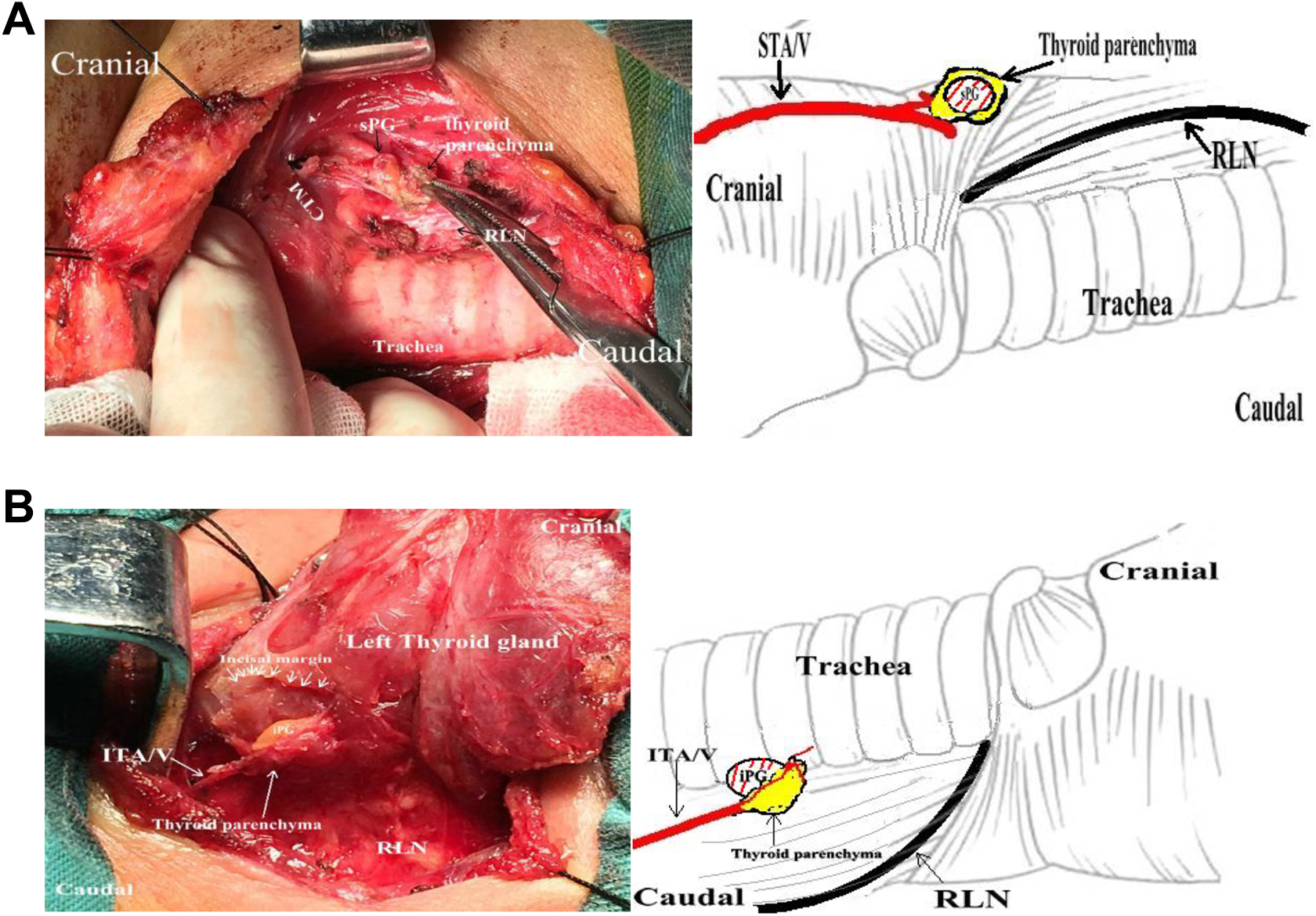
A, Type III sPG was characterized by a single blood supply of thyroid parenchyma or within the thyroidcapsule (left side). A small portion of adjacent thyroid tissue can be left in place to preserve the vasculature. B, Type III iPG was characterized by the blood supply of thyroid parenchyma (left side).
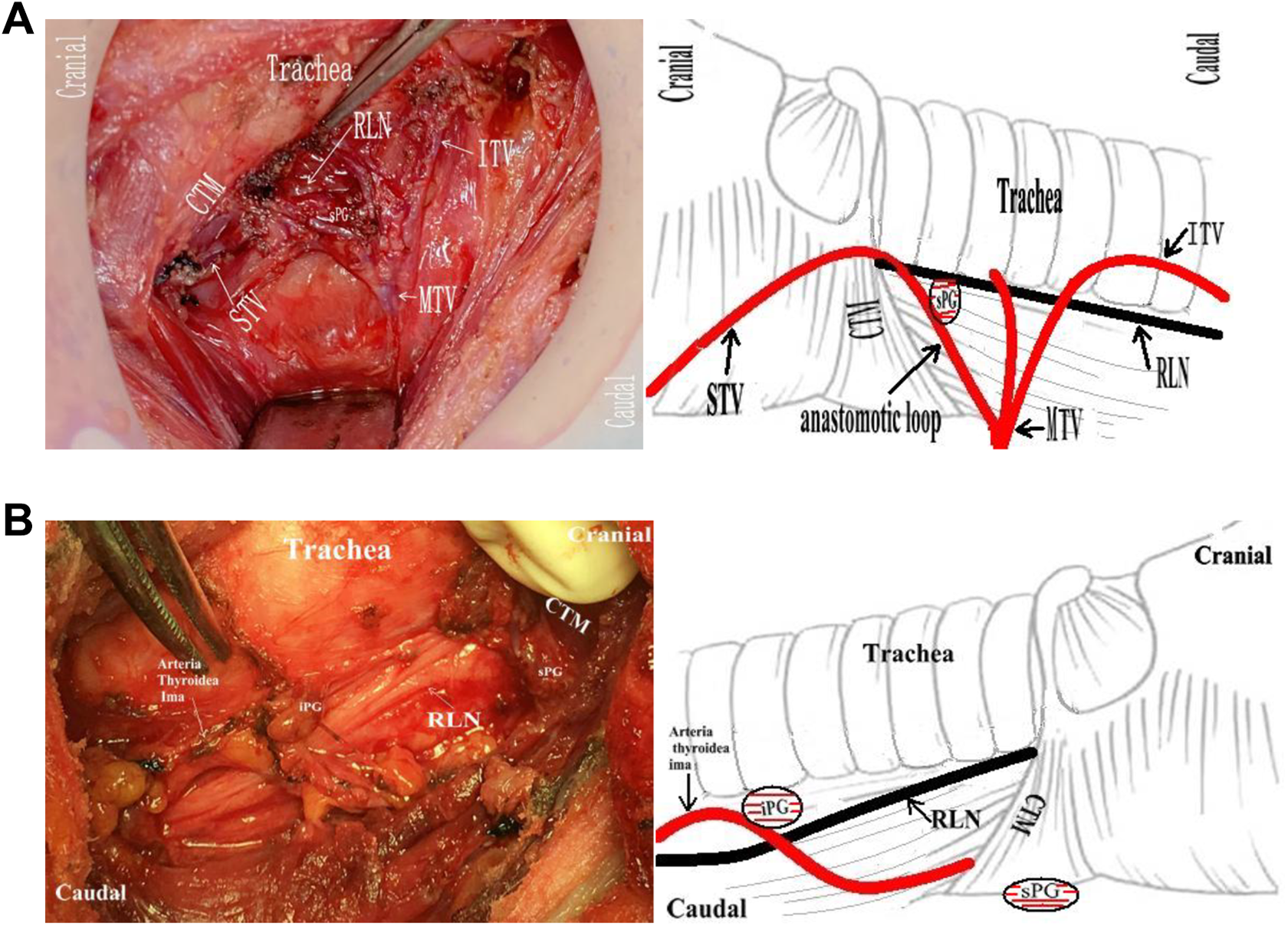
A, Type IV sPG was characterized by a single blood supply of anastomotic loop of superior thyroid artery and vein (STA/V) and inferior thyroid artery and vein (ITA/V; right side). B, Type IV iPG was characterized by a single blood supply of thyroid ima artery (left side).
A, Type V sPG was characterized by a single blood supply of inferior thyroid artery and vein (ITA/V; right side). B, Type V iPG was characterized supply by an anastomotic loop of superior thyroid artery and vein (STA/V) and ITA/V (right side).
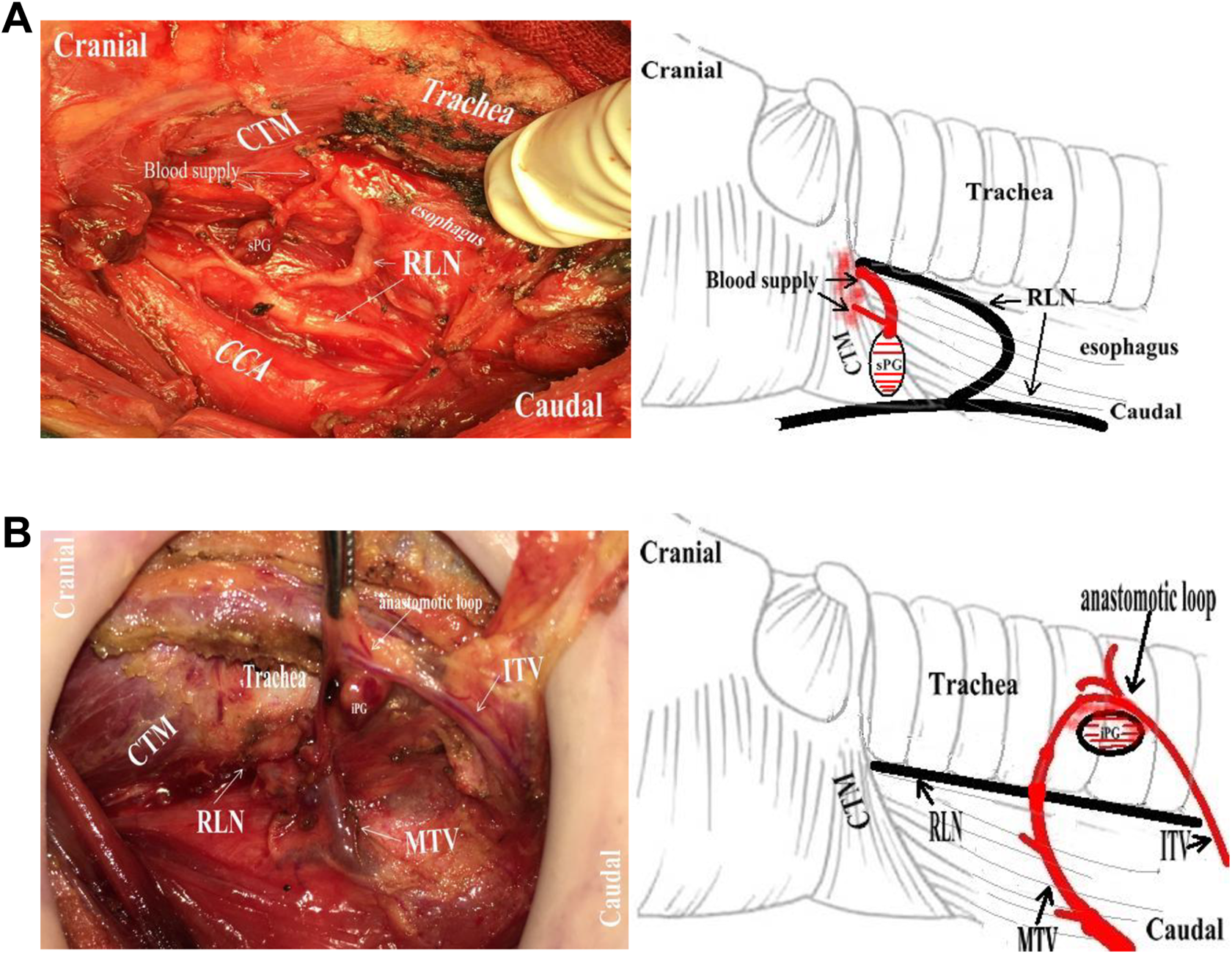
A, Type VI sPG was characterized by 2 branches of esophagus blood supply (right side). B, Type VI iPG was characterized by a single blood supply of anastomotic loop between MTV and inferior thyroid artery and vein (ITA/V; right side).
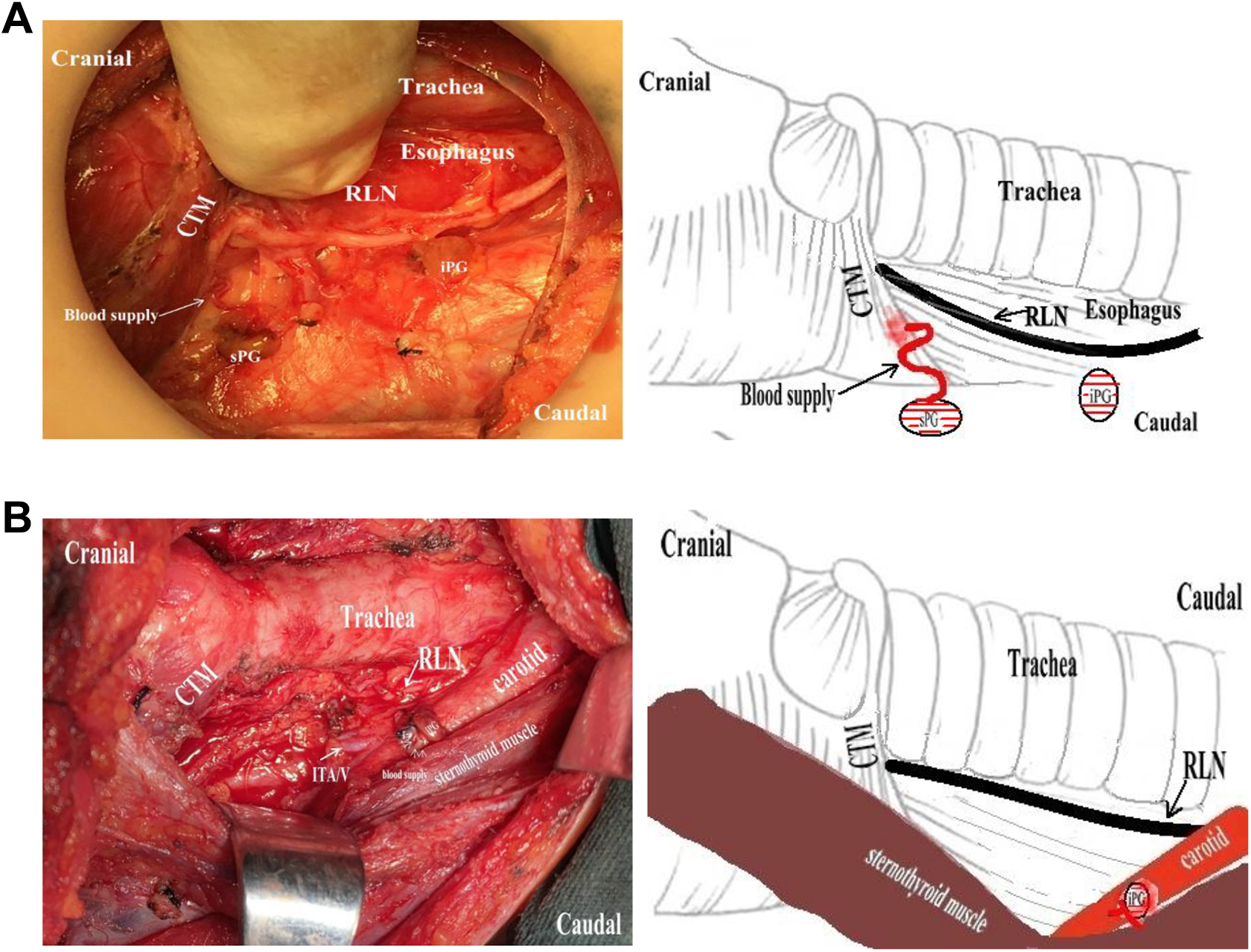
A, Type VII sPG was characterized by a single blood supply of esophagus (right side). B, Type VII iPG was characterized by a single blood supply of sternothyroid muscle (right side).
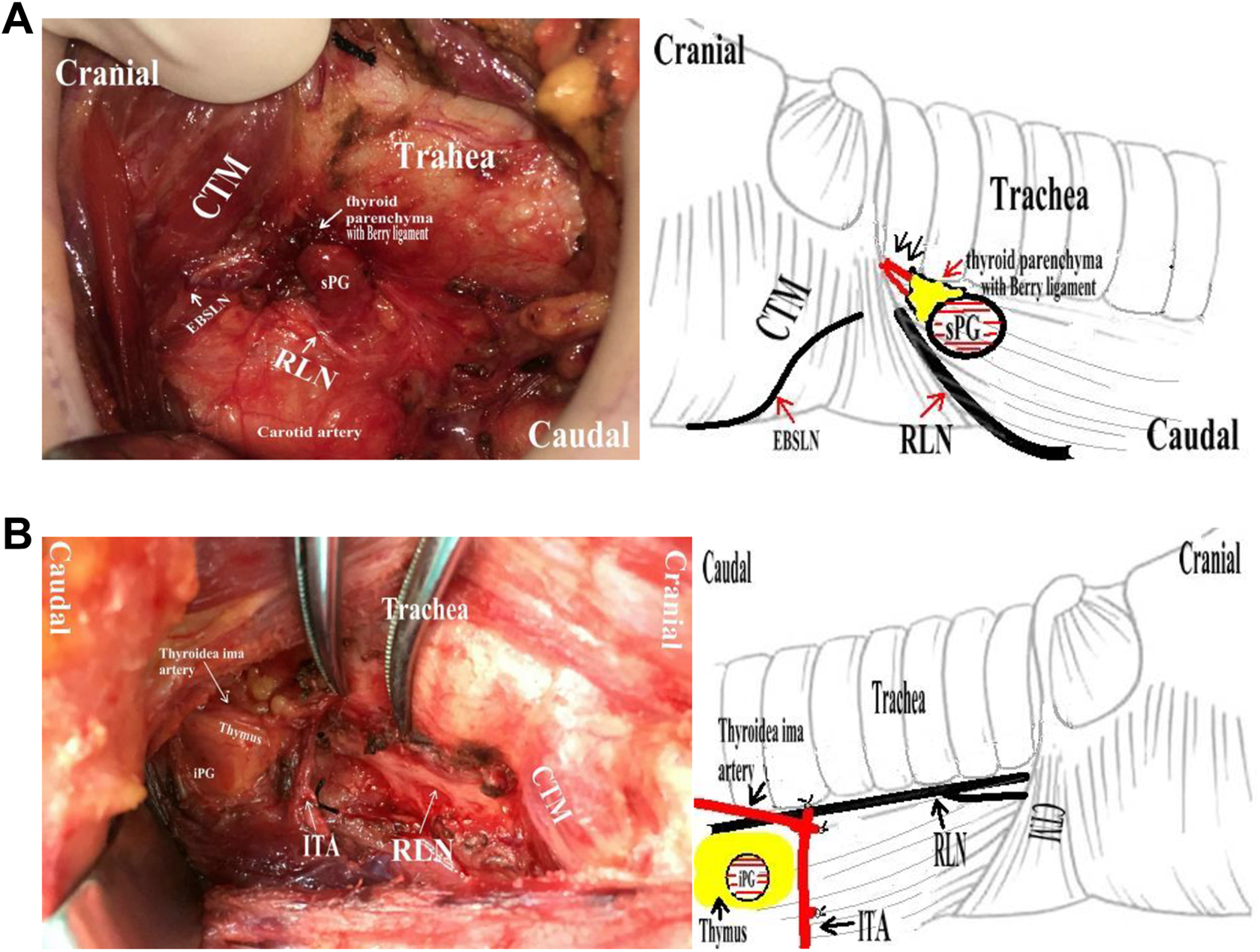
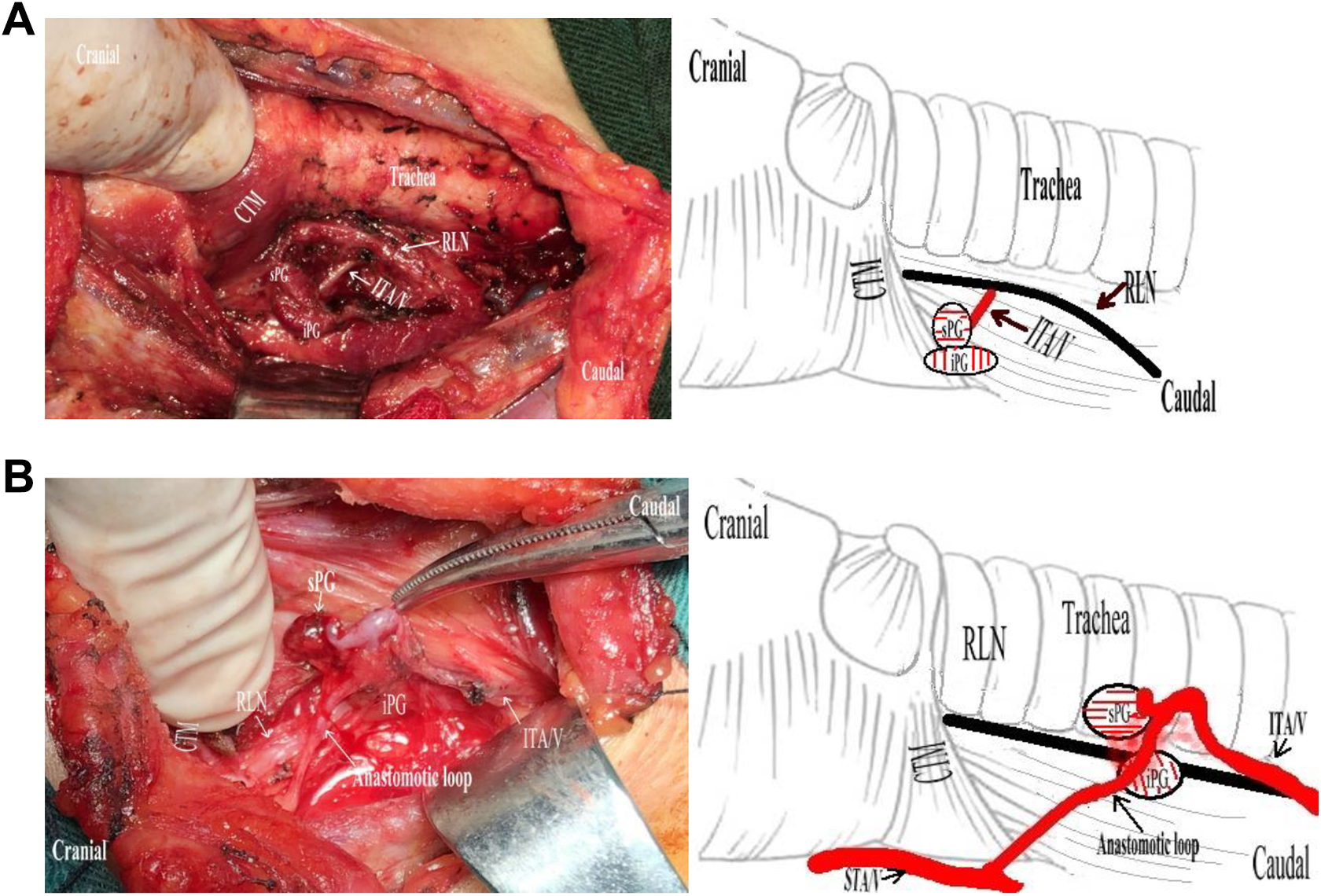
A, Type VIII sPG was characterized by the blood supply of the Berry ligament and thyroid parenchyma (right side). The 8 types of blood supply for the inferior parathyroid gland with specific preservation measures. B, Type VIII iPG was characterized by its blood supply of the thymus (left side). CCA indicates common carotid artery; CTM, cricothyroid muscle; EBSLN, external branch of superior laryngeal nerve; iPG, inferior parathyroid gland; ITA/V, inferior thyroid artery and vein; MTV, middle thyroid vein; RLN, recurrent larynx nerve; sPG, superior parathyroid gland; STA/V, superior thyroid artery and vein.
Statistical Analysis
Serum calcium and PTH levels (preoperatively and postoperatively at day 1, day 30, and day 90 after surgery) were compared between groups using Student t test on SPSS 23.0 (IBM Corp). Chi-square test was used for data of transient hypocalcemia and hypoparathyroidism between groups. The significant level was P < .05.
Results
Some 170 patients fulfilled our inclusion criteria; 21 patients with papillary cancer were excluded because of either parathyroid tissue found in a pathological specimen or insufficient data. Finally, a retrospective cohort research of 149 patients who underwent total thyroidectomy for malignant disease (PTC) was reviewed. Group 1 involved 60 patients (11 men and 49 women) undergoing standard thyroidectomy, who were aged 47.07 ± 1.85 years. Group 2 involved 89 patients (16 men and 73 women) receiving PGVPIST, who were aged 48.26 ± 1.13 years (Table 1). The average values of preoperative PTH and serum calcium levels were similar between groups (Table 2). No RLN injury was observed after surgery. Mean PTH levels were significantly lower in group1 than in group 2 at day 1 (23.68 ± 2.54 vs 31.46 ± 2.11 pg/mL, P < .05) and day 30 (45.63 ± 3.21 vs 55.65 ± 2.89 pg/mL, P < .05) after surgery. However, no difference in PTH levels of the 2 groups remained significant at day 90 after surgery (Table 2). In addition, serum calcium levels were insignificantly higher in PGVPIST group at day 1 (2.18 ± 0.02 vs 2.15 ± 0.01 mmol/L, P > .05) and day 30 (2.27 ± 0.02 vs 2.38 ± 0.11 mmol/L, P > .05) after surgery (Table 2).
Mean Preoperative and Postoperative Serum PTH (pg/mL) and Ca2+ (mmol/L) Levels.
Abbreviation: PTH, parathyroid hormone.
Postoperative hypocalcemia was found in 12 (8.1%) patients at day 1 after surgery in patients of both groups, but occurred no more frequently in group 1 than in group 2 (10% vs 6.7%, P > .05; Table 1). Postoperative hypoparathyroidism was found in 41 (27.5%) patients at day 1 after surgery in patients of both groups and occurred more frequently in group 1 than in group 2 (40% vs 19.1%, P < .05) (Table 1). Significant difference in hypoparathyroidism was found between these 2 groups (8.3% vs 0%, P < .05) at day 90 after surgery (Table 1). No patient reported vocal cord paralysis after thyroidectomy.
Discussion
Postoperative hypocalcemia and hypoparathyroidism caused by devascularization or accidental removal of parathyroid glands 6 are frequently observed after total thyroidectomy. Hence, normalization of parathyroid function after surgery is one of the important goals unremittingly pursued by thyroid surgeons. The incidence of these consequences depends on thyroid disorders, experience of surgeons, and specific type of thyroidectomy. 5 Studies evaluating postoperative hypocalcemia and hypoparathyroidism in relation to the blood supply of parathyroid gland mostly involve a part of patients undergoing total thyroidectomy. 7 In our study, about 8.1% of patients had transient hypocalcemia and 27.5% of patients had transient hypoparathyroidism (Table 1). The reported incidence of transient hypocalcemia is lower than 10% to 50% in patients after total thyroidectomy. 1 This was because each patient received calcium supplementation after surgery. In concordance with our results, Yong et al reported a 10% to 46% rate of transient hypoparathyroidism after total thyroidectomy. 8 It is recommended that vascularized parathyroid glands should be preserved regardless of intraoperative intact PTH levels, 9 but devascularized parathyroid glands should be routinely transplanted in thyroid operation. 10 In our study, patients with PTC undergoing total thyroidectomy with PGVPIST in group 2 versus conventional technique in group 1 have higher mean postoperative PTH levels at day 1 (31.46 ± 2.11 vs 23.68 ± 2.54, P < .05) and day 30 (55.65 ± 2.89 vs 45.63 ± 3.21, P < .05) after surgery (Table 2). The reason for these results may be that PGVPIST protects the vasculature circuits between blood supply and parathyroid glands after total thyroidectomy. The parathyroid gland blood supply and preserving method are based on some observations and research.6,11-14 The number of parathyroid glands in our patients is 2 to 6. The reported number of arteries supplying parathyroid glands per patient is 1 to 4.11,15 The superior parathyroid glands are frequently located at the posterior margin of the upper two-thirds of the thyroid gland or are embedded in the parenchyma. 16 The location of the inferior parathyroid gland is variable, as it is embedded in the fatty tissue surrounding the lower pole of either the thyroid gland or the ectopic intrathymic parathyroid. The inferior parathyroid glands are generally distributed between the isthmus and the lower pole of the thyroid gland. 17 A parathyroid gland is supplied by several arteries arising from branches of thyroid arteries or their anastomotic branches. 18 Permanent hypoparathyroidism easily occurs after less than 3 parathyroid glands are identified intraoperatively. 19 We conclude that true capsular vessels of the thyroid can nourish parathyroids in 7 ways. (1) Superior parathyroid glands derive blood supply from the dorsal terminal branch of the STA/V trunk (grade 2-3, Figure 1A). (2) Most of the blood supply of parathyroid glands comes from the terminal branches of the ITA/V away from the thyroid (grade 2-3; Figures 1B, 4A, 5A, 5B, and 6B). The parathyroid glands are supplied with blood from (3) the thyroid ima artery (Figures 2A, 4B, and 5B), (4) the thyroid parenchyma (Figure 3A and B), or (5) the surrounding dorsolateral “arc” vein structure of the thyroid (Figures 4A, 6B, and 8B). Blood supply originates (6) from anastomotic loop between superior and inferior thyroid arteries or veins (Figures 4A, 5B, and 6B), or (7) from the Berry ligament (Figure 8A), esophagus (Figures 6A and 7A), trachea, and surrounding muscle tissue (Figures 7B and 8B).
Preservation of parathyroid glands and blood supply in situ is essential for postoperative parathyroid function recovery. 20 Due to restriction on medical costs among patients in China, we designed the PGVPIST for preserving parathyroid glands with blood supply without complementary measures. The parathyroid glands were preserved without distinct blood vessels by the conventional technique in group 1 but were well preserved with nearby blood vessels by the PGVPIST in group 2. We propose a new concept of using the peripheral thyroid vein as a marker to conduct anatomy and protect the parathyroid gland and blood supply. Because the thyroid artery is thinner and often located on the dorsal thyroid, it can be hardly distinguished with naked eyes. As reported more recently, post-thyroidectomy hypocalcemia can be reduced by the preservation of the inferior thyroidal vein. 21 Recognizing the size, color, possible vascular supply, and location of parathyroid glands is essential. According to our observations, the supplying vessels of uninjured parathyroid tissues cannot be easily separated via simple dissection of the thyroid capsule, because the blood vessels enter the thyroid parenchyma and come out to nourish the parathyroid gland. The blood may be supplied by small branches (4-5 mm long) of the ITA. 22 We adopted protective anatomy and preferentially preserved the blood supply sources related to the parathyroid glands. PGVPIST is generally conducted by 5 key steps. (1)The posterior branch of the STA, as well as the vein trunk with tertiary branches, is preserved (Figures 1A, 1B, 2B, and 5A). (2) The ITA and the vein trunk with tertiary branches away from the gland side, micro-ligation, and resection of thyroid side vessels, are preserved (Figures 1B and 5A). (3) The dorsal branch of the thyroid ima artery trunk with tertiary branches away from the gland side is preserved (Figures 2A, 4B, and 8B). (4) Anastomotic loop between these vessels is conducted, and the ultrafine ligature of blood supply is placed in contact with thyroid parenchyma (Figures 4A, 5B, 6B, and 8B). (5) Parathyroid gland and a tiny adjacent thyroidal parenchyma (1 × 1 mm3; Figures 3A and B, and 8A) with thyroid adherent vasculature should be preserved, when the source of blood supply to the parathyroid gland cannot be identified. All vascular supply should be ligated close to the thyroid parenchyma. Kocher’s vein trunk should be preserved with ligation of thyroid side branches (Figure 6B). Parathyroidal branches can be preserved by peripheral ligation of ITA. 23 Finally, each preserved parathyroid gland with vasculature should be evaluated, and the devascularized parathyroid glands should be autologously transplanted. This is because the devascularized parathyroid glands proved by biopsy can be salvaged with autotransplantation. 24
We precisely dissected parathyroid gland vessels and micro-ligatures of thyroid side vessels through surgery, aiming to reduce the incidence of iatrogenic hypoparathyroidism. When a parathyroid gland was identified as avascular, and it is a tiny adjacent thyroidal tissue (1 × 1 mm3) with thyroid adjacent vasculature should be preserved in order not to harm its vasculature. The parathyroid blood supply is usually buried in the adjacent thyroid tissue. If the malignant thyroid tumor requires a total thyroidectomy, the remaining dense thyroid tissue should be removed by ophthalmic scissors to protect the parathyroid gland and its blood supply. A normal parathyroid gland turns brownish-red or yellow with an attached pedicle at the end of operation. 25 The position of parathyroid gland vasculature in relation to the thyroid gland parenchyma is the most important risk of parathyroid gland devascularization during thyroidectomy. 13 A dramatic change in normal color from a buff tan parathyroid gland to a cyanotic, dark or pale parathyroid gland can easily be observed. The parathyroid score may be helpful for predicting the function of parathyroid glands after thyroidectomy. 1 Kuriloff DB et al salvaged devascularized parathyroid glands by using lidocaine spasmolysis for avoiding unnecessary autotransplanatation, 26 which was proved in our surgery. Some researchers believe that autotransplantation of parathyroid glands may prevent permanent hypoparathyroidism and increase the incidence of transient hypocalcemia, but this belief remains controversial.27,28 Parathyroid function failure was found in many parathyroid autotransplantation studies.23,29 Intrathyroidal parathyroid gland was not found in our operation. Despite the preservation of the parathyroid glands with vessels in group 2, we still find 17 of 89 patients with hypoparathyroidism at day 1 after surgery. This result probably means visual assessment of vascularization may be more reliable with an indocyanine green fluorescence technique.30,31 The intrathyroidal parathyroid glands will be preserved and studied in future studies. About 3% to 6% of patients with PTC had permanent hypoparathyroidism when they underwent thyroidectomy with cervical lymph node dissection. 28 In comparison, we found no hypoparathyroidism in group 2 at day 90 after operation (Table 1), which means PGVPIST is helpful for recovering parathyroid function after thyroidectomy.
In conclusion, PGVPIST during total thyroidectomy for PTC is associated with higher PTH level after the operation. However, the incidence rates of permanent hypoparathyroidism are significantly different between patients undergoing PGVPIST and standard thyroidectomy. Parathyroid gland vasculature preservation in-situ technique has high clinical values in recovering parathyroid function and preventing hypoparathyroidism after thyroidectomy for patients with thyroid cancer. We recommend that PGVPIST be used and verified in every thyroidectomy case to preserve parathyroid glands with the vasculature. We are waiting for your verification of our technique.
Footnotes
Authors’ Note
Chuanchang Yin and Bin Song contributed to study conception and design. Xiaoyan Wang, Chuanchang Yin, and Huichuan Zhao contributed to acquisition of data. Xiaoyan Wang and Weihong Zheng contributed to analysis and interpretation of data. Chuanchang Yin and XingruiLi contributed to drafting of manuscript. Xiaoyan Wang contributed to critical revision of manuscript. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Acknowledgments
The author would like to thank Prof Bin Song and Xingrui Li sharing knowledge on PGVPIST dissection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
