Abstract
Objective:
To develop a French Short Version of the Questionnaire of Olfactory Disorders-Negative Statements (Fr-sQOD) to assess the quality of life impairments of patients with olfactory dysfunction (OD).
Methods:
Patients with OD and controls were enrolled from 2 academic centers. Individuals completed the Fr-sQOD, an OD visual analog scale severity, and the French version of the sinonasal outcome tool-22 (SNOT-22). Cronbach α was used to measure the internal consistency of Fr-sQOD. The reliability and the external validity of Fr-sQOD were assessed through a test–retest approach and by correlating Fr-sQOD with SNOT-22 scores, respectively. The external validity was assessed by correlation analysis between Fr-sQOD and the result of an assessment of the severity of OD on a visual analog scale.
Results:
Eighty patients completed the evaluations. The internal consistency was adequate (Cronbach α .96), and the test–retest reliability was high in the entire cohort (rs = 0.877, P < .001). The correlation between Fr-sQOD total scores and the severity of OD was moderate but significant (rs = −0.431; P = .001) supporting an acceptable external validity. Patients with OD had a significantly higher score of Fr-sQOD than healthy individuals (P < .001), indicating a high internal validity.
Conclusion:
The Fr-sQOD is a reliable and valid self-administered tool in the evaluation of the impact of OD on quality of life of French-speaking patients.
Introduction
Olfactory dysfunction (OD) affects one-fifth of the general population. 1 Recently, anosmia has been identified as a key symptom of the coronavirus disease 2019 (COVID-19), 2,3 indicating the current need for standardized olfactory-specific quality of life (QOL) patient-reported outcome questionnaires. In 2012, Simopoulos et al developed the Questionnaire of Olfactory Disorders-Negative Statements (QOD-NS) composed by 17 items measuring olfactory-specific QOL. 4 This questionnaire is well used in clinical practice around the world and presents high validity and reliability properties. 4 Questionnaire of Olfactory Disorders-Negative Statement evaluates the OD impact on QOL, through social, anxiety, annoyance, and eating-related problem questions. Recently, Mattos et al developed a short version of QOD-NS (sQOD-NS) composed of 7 items, with excellent validity and reliability properties. 5 To date, sQOD-NS is only validated in English and Spanish. 6 There is no validated sQOD-NS version for French-speaking countries that include more than 400 million inhabitants. The aim of this study is to develop a French Short Version of the Questionnaire of Olfactory Disorders-Negative Statements (Fr-sQOD).
Methods
The study protocol was approved by the Institutional Ethics Committee (n° CHUSP20032020). Patient electronic informed consent was obtained.
Translation of Fr-sQOD
A multidisciplinary team composed of 2 otolaryngologists, a psychologist, a statistician, and a linguist worked on the French adaptation of sQOD-NS (Fr-sQOD-NS). The Fr-sQOD-NS was translated from the US version by the linguist. Experts of the team were native French speakers. Before the validation, the Fr-sQOD-NS was sent to 10 patients to detect potential misunderstanding(s). The final version of Fr-sQOD is available in Table 1.
Short Questionnaire of Olfactory Disorders-Negative Statements.a
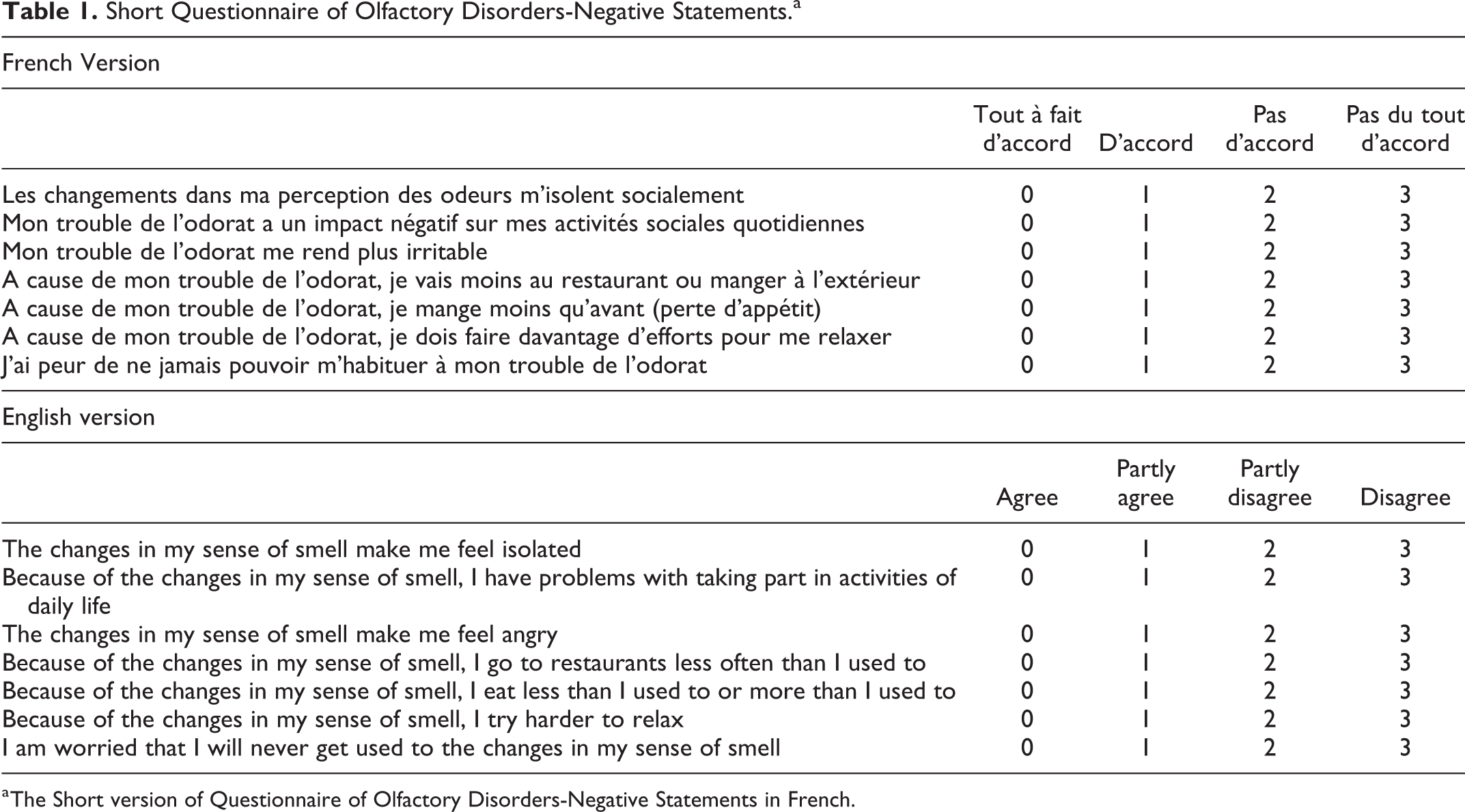
a The Short version of Questionnaire of Olfactory Disorders-Negative Statements in French.
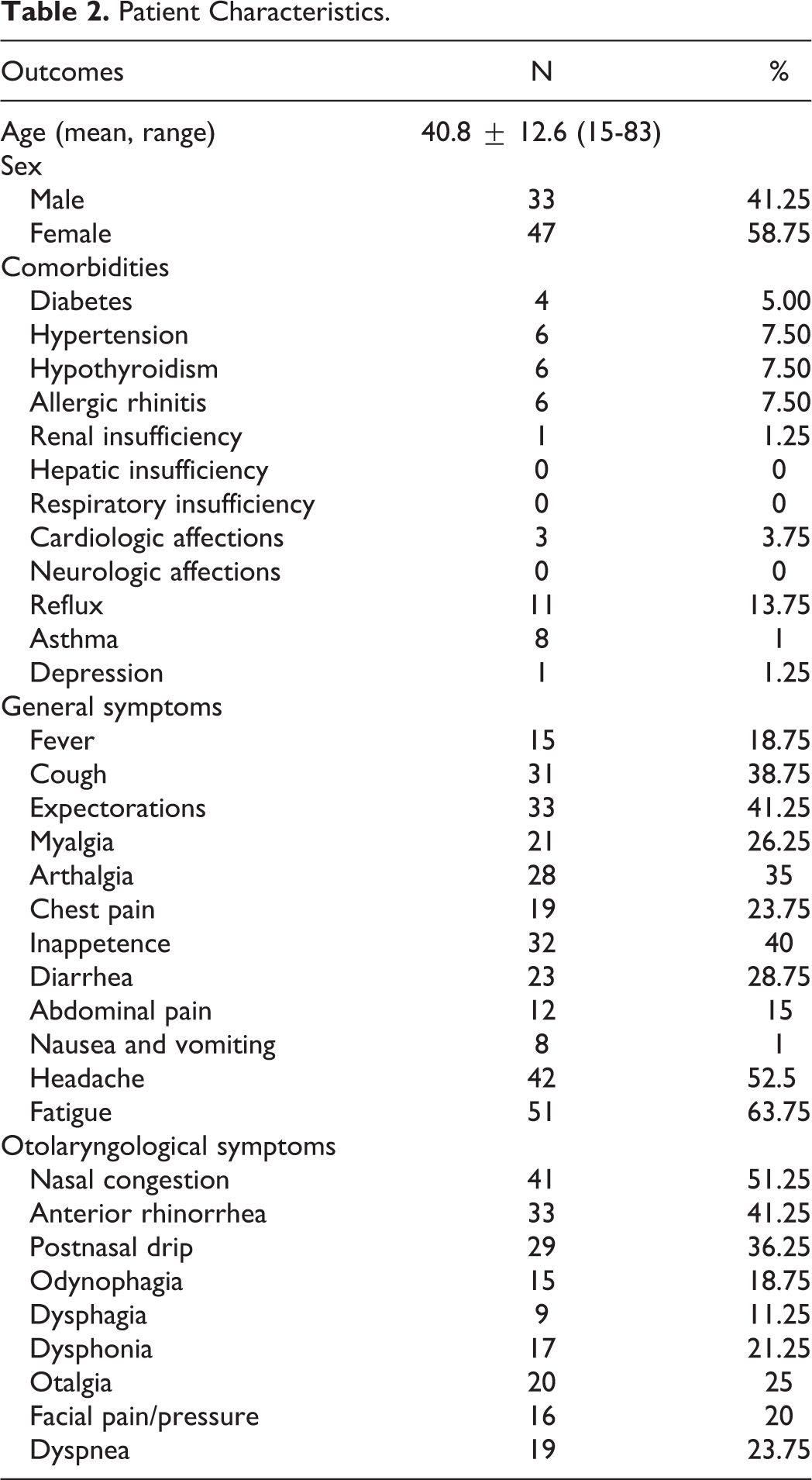
Patients with OD were enrolled from March 2020 to June 2020 from the Departments of Otolaryngology–Head & Neck Surgery of Foch Hospital (Paris, France), CHU Saint-Pierre (Brussels, Belgium), and EpiCURA Hospital (Baudour, Belgium). Patients developed OD during the pandemic of COVID-19. There were 47 males and 33 females with a mean age of 40.8 ± 12.6 (ranged from 15 to 83 years old). A control group of healthy individuals was composed with a mean age of 37.7 ± 15.7 (range 18 to 70 years old). Individuals with severe neurological diseases limiting the understanding of the study protocol, history of chronic OD, or those who were not native French speaker were excluded. Eighty patients with demonstrated anosmia or hyposmia and 100 healthy individuals (62 males) completed the study. The characteristics of patients are reported in Table 1. The diagnosis of COVID-19 was performed through nasal swabs and reverse transcriptase-polymerase chain reaction.
Olfactory and Nasal Evaluations
Nasal complaint evaluation was performed using the French version of the sinonasal outcome tool-22 (SNOT-22). 7 Patients and controls fulfilled subjective evaluations of olfaction based on olfactory and gustatory questions of the smell and taste component of the National Health and Nutrition Examination Survey. 8 Psychophysical olfactory evaluations were performed with Sniffin-Sticks tests (Medisense), which is a standardized and validated evaluation using 16 smell pens. The patient had to choose the adequate term describing the smell between 4 given options. 9 The total score ranges from 0 (complete anosmia) to 16 (no olfactory disorder). Regarding results, 3 categories were defined: normosmia (score between 12 and 16), hyposmia (score between 9 and 11), and anosmia (score <9). 9
Validity, Reliability, and Responsiveness to Change
The Fr-sQOD-NS was completed twice over 2-day period (test–retest reliability) to assess the internal consistency (Spearman correlation coefficient). Internal consistency was evaluated with Cronbach α for the 7 items of the tool. External validity was assessed by correlation analysis between Fr-sQOD-NS and the result of an assessment of the QOL impact of OD through a visual analog scale, ranging from 0 (=no QOL impact) to 4 (=severe QOL impact) using Spearman correlation coefficient. The sQOD-NS scores were compared between patients and healthy patients to measure internal validity (Mann-Whitney U test.). Because Fr-sQOD-NS is not a clinical tool aiming to detect pathology but a QOL questionnaire, we did not assess the normative value of Fr-sQOD-NS.
Statistical Analyses
The statistical analyses were performed with Statistical Package for the Social Sciences for Windows (SPSS version 22.0; IBM Corp). A level of significance of P < .05 was used. The difference in Fr-sQOD-NS scores between patients and healthy individuals was evaluated with the Mann-Whitney U test. Spearman correlation coefficient was used to perform the different correlation analyses (test–retest reliability, external validity).
Results
A total of 80 patients with hyposmia or anosmia completed the evaluations. The epidemiological and clinical characteristics of patients are available in Table 2. There were respectively 72, 6, and 2 patients with mild, moderate, and severe forms of the COVID-19 according to the World Health Organization classification. 10 The most prevalent general symptoms in COVID-19 patients were fatigue, headache, and myalgia. Prevalent otolaryngological symptoms were nose congestion, anterior rhinorrhea, and postnasal drip. Among the cohort of patients with OD, there were 49 anosmic and 31 hyposmic patients according to Sniffin-Sticks tests (Table 3). The mean Fr-SNOT-22 score of patients was 36.6 (Table 3).
The Cronbach α value was .955 for the 7 items of Fr-sQOD-NS, indicating high internal consistency. The test–retest reliability was high for total scores (rs = 0.877, P < .001) and moderate-to-high for all item scores (Table 4). The correlation between Fr-sQOD-NS total score and the visual analog scale OD assessment was moderate but significant (rs = −0.431; P = .001), suggesting high external validity. Concerning internal validity, the mean score of Fr-sQOD-NS was significantly higher in patients compared with controls (Table 5).
Patient Characteristics.
Nasal, Smell, and Taste Features of Patients.
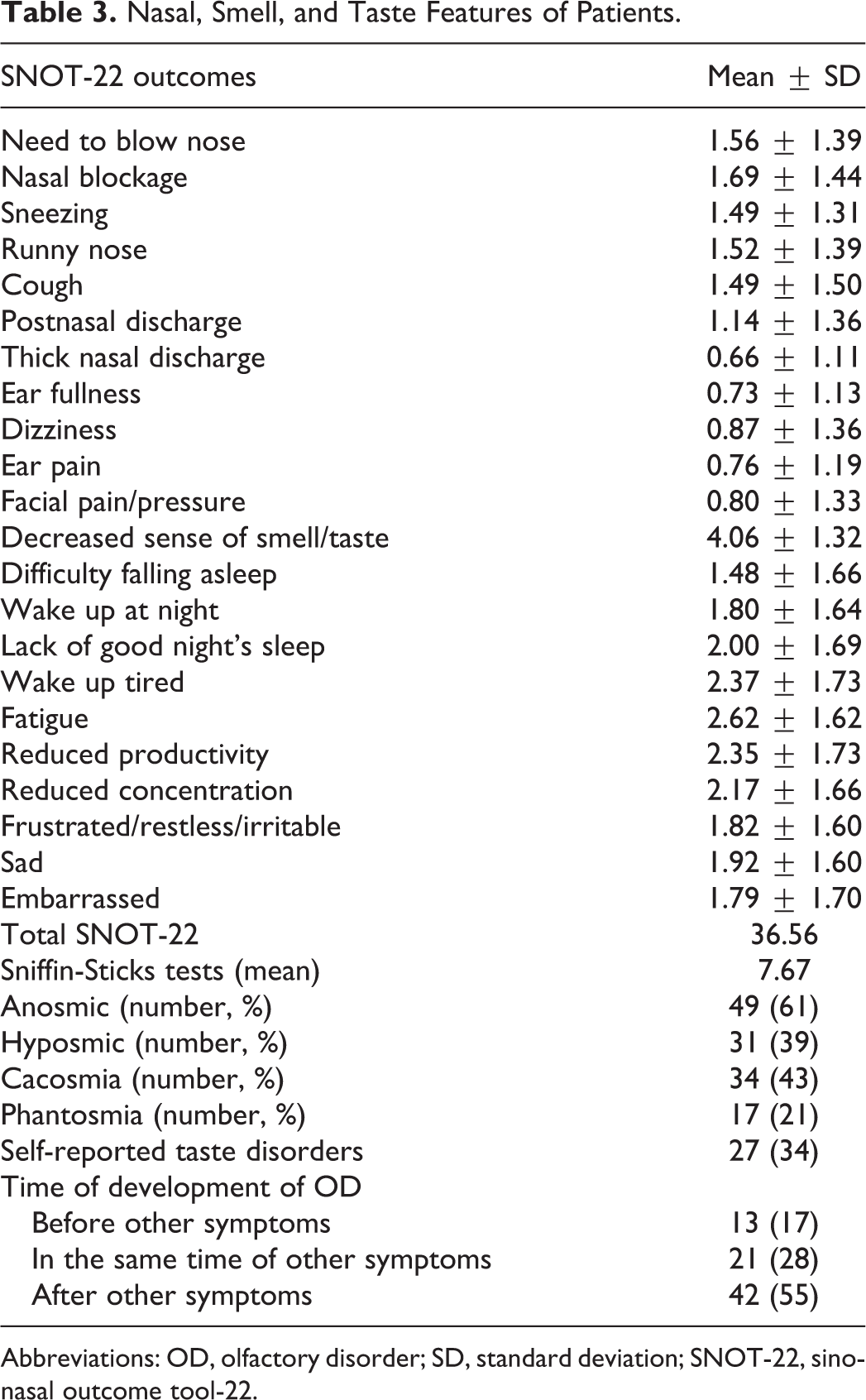
Abbreviations: OD, olfactory disorder; SD, standard deviation; SNOT-22, sinonasal outcome tool-22.
Test–Retest Reliability Analysis.a
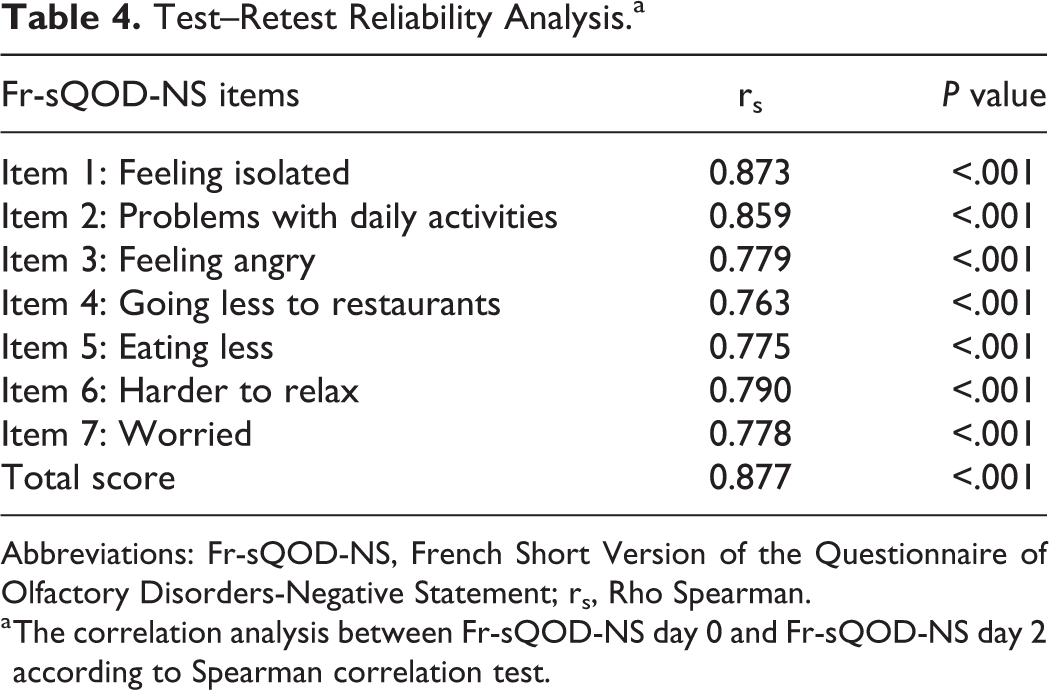
Abbreviations: Fr-sQOD-NS, French Short Version of the Questionnaire of Olfactory Disorders-Negative Statement; rs, Rho Spearman.
a The correlation analysis between Fr-sQOD-NS day 0 and Fr-sQOD-NS day 2 according to Spearman correlation test.
Comparison of sQOD-NS Between OD Patients and Healthy Individuals.
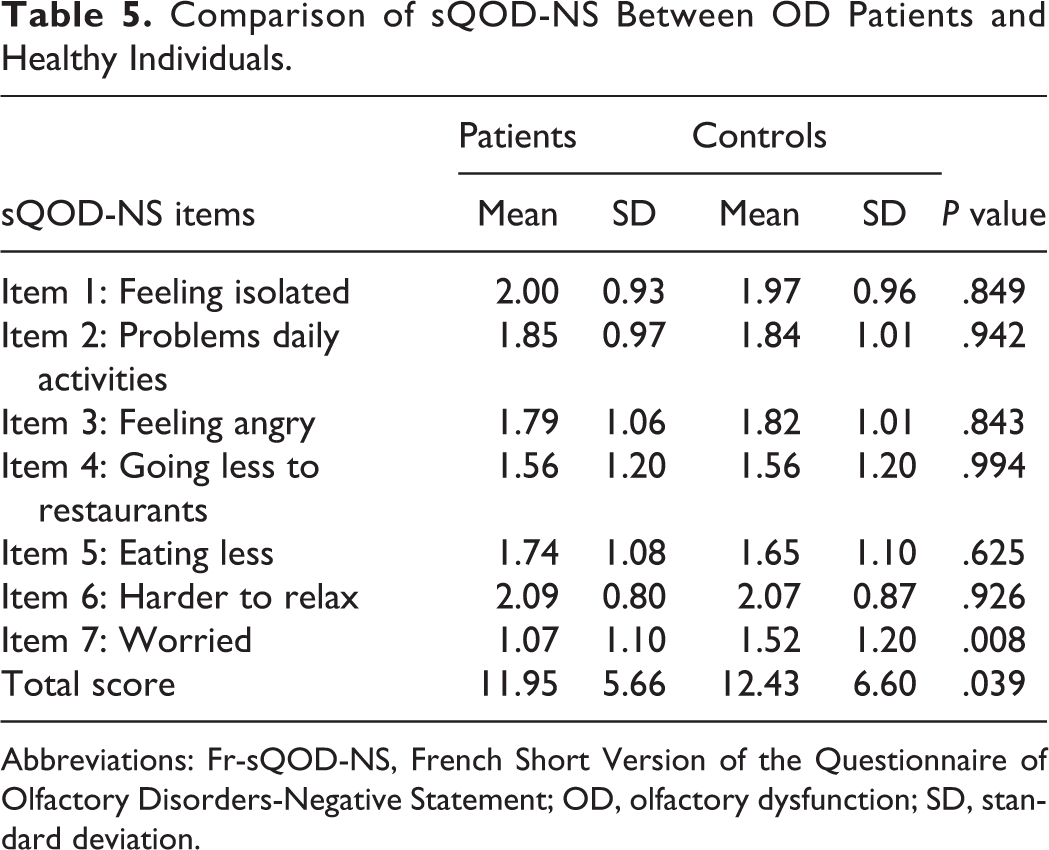
Abbreviations: Fr-sQOD-NS, French Short Version of the Questionnaire of Olfactory Disorders-Negative Statement; OD, olfactory dysfunction; SD, standard deviation.
Discussion
The high prevalence of anosmia and hyposmia in COVID-19 11,12 and the risk of reinfection and second anosmia episodes 13 make the development of QOL patient-reported outcome questionnaires important. The initial version of QOD-NS was time-consuming with these 17 items, and consequently, the development of a reliable and valid short version makes sense. In this study, we developed a French version of sQOD-NS that reports high internal consistency regarding the Cronbach α value (.955). The internal consistency value of the Fr-sQOD-NS is comparable to the value of the original paper of Simopoulos et al who reported a Cronbach α of .88. 4 In the Spanish version of sQOD-NS, Chiesa-Estomba et al 14 found a Cronbach α of .861, which was also comparable to ours. 6
In the same vein, the test–retest reliability of Fr-sQOD-NS, also named external reliability, was high for total scores (rs = 0.877, P < .001) and moderate-to-high for item scores. In the original version of QOD-NS, Simopoulos et al reported adequate external reliability of QOD-NS since they did not find significant differences between the values of the initial (test) and the second (retest) fulfill of QOD-NS (P = .567). 4 However, the result comparison between our studies is still limited because they used a different statistical approach to assess the external reliability. The comparison with the Spanish study is not possible because the authors did not provide external reliability analysis.
The Fr-sQOD-NS total score of OD patients was significantly lower than the score of healthy individuals (P = .039), indicating good internal validity. Concerning the item scores, only the item 7 (“I am worried that I will never get used to the changes in my sense of smell.”) was significantly lower in OD patients compared with controls. According to our data, this item is probably the most relevant to report the fear of patients about the risk to not recover olfaction and the related significant negative impact on QOL. Our internal validity findings are comparable to those of Simopoulos et al who compared the QOD-NS values of anosmic, hyposmic, and normosmic in order to evaluate the internal validity. The authors found that the value of QOD-NS of normosmic individuals was significantly higher than those of hyposmic and anosmic. 4
Currently, there is a lack of patient-reported outcome questionnaires focusing on olfactory QOL impairment. For this reason, we assessed the external validity through a correlation analysis between sQOD-NS and a visual analog scale describing OD QOL impairment. Then, the correlation analysis reported moderate but significant association (rs = −0.431; P = .001), suggesting high external validity. We did not evaluate external validity through a correlation analysis with SNOT-22 because many COVID-19 patients are known to be free of nasal complaints. 15,16 The comparison of our data of external validity with the other studies is still limited because Simopoulos et al only assessed the external validity of QOD-positive statements and not QOD-NS, while Chiesa-Estomba et al did not report such analysis. 6
The main limitation of this study is the low number of patients and the focus on individuals with COVID-19. Indeed, OD in COVID-19 is probably related to neurological mechanisms, leading to total loss of smell (anosmia) in the high majority of cases. The features of patients with neurological post-viral anosmia may be different from the OD of patients with chronic rhinosinusitis with nasal polyps or other rhinological common diseases. In that way, the properties of sQOD-NS would be different for these types of patients. Moreover, some items of sQOD-NS involved social habits of individuals. During the pandemic, many European governments have imposed lockdown in some regions, modifying the social habits of populations, which may impact the patient responses. The strength of this study is the realization of psychophysical olfactory evaluations, allowing the confirmation of OD.
Conclusion
The Fr-sQOD-NS is a short, reliable, and valid self-administered tool for the evaluation of the impact of OD on the QOL of French-speaking patients.
Footnotes
Authors’ Note
Jerome Lechien and Sven Saussez have equally contributed to this work and should be regarded as joint senior authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Electronic informed consent was obtained from all individual participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
