Abstract
We report a case of laryngeal cancer with multiple lung metastases that maintained a complete response (CR) for 18 months after discontinuing nivolumab treatment, with colitis developing 5 months after drug discontinuation. A 65-year-old man was diagnosed with T3N2cM0 stage IVA right supraglottic squamous cell carcinoma that progressed after 1 course of TPF (cisplatin, docetaxel, and 5-fluorouracil) as induction chemotherapy. He underwent total laryngectomy, bilateral neck dissection, pharyngeal reconstruction with anterolateral thigh flap, and creation of a permanent tracheostoma; extranodal extension was detected in the right cervical lymph node metastasis, and the patient underwent adjuvant radiotherapy. Multiple lung metastases occurred during radiotherapy, and the patient was deemed platinum refractory; nivolumab treatment was thus initiated. The tumor proportion score for programmed death-ligand 1—evaluated via antibody testing of the laryngeal tumor—was <1. The patient received 240 mg/body nivolumab every 2 weeks; a computed tomography performed after course 16 of nivolumab treatment confirmed a CR. He exhibited grade 2 thyroid dysfunction, grade 1 interstitial pneumonia, and grade 2 colitis after 6, 7, and 14 months of receiving nivolumab, respectively; treatment was discontinued as despite maintaining a CR, interstitial pneumonia occurred twice. Colitis appeared 5 months after nivolumab discontinuation; nevertheless, a CR was maintained after 18 months.
Keywords
Introduction
Nivolumab is an immune checkpoint inhibitor (ICI) and anti-programmed death-1 monoclonal antibody. In the CheckMate 141 trial—a multinational phase III study—the overall survival (OS) of patients with platinum-refractory recurrent/metastatic head and neck cancer (R/M HNC) treated with nivolumab was significantly longer than that of patients administered the investigator’s choice of therapy (methotrexate, docetaxel, or cetuximab). 1 The National Comprehensive Cancer Network guidelines list nivolumab as a category-1 preferred regimen for the subsequent treatment of platinum-refractory R/M HNC. 2 Cases of R/M HNC that maintain long-term complete response (CR) even after discontinuation of nivolumab are rare. In addition, the appearance of immune-related adverse events (irAEs) after a long period of time had passed since the discontinuation of nivolumab is uncommon. Here, we report the case of a patient with laryngeal cancer and multiple lung metastases in whom a CR was achieved and maintained for 18 months after nivolumab discontinuation; additionally, colitis as an irAE developed 5 months after nivolumab discontinuation.
This case report is in line with the principles laid out by the World Medical Association’s Declaration of Helsinki; written informed consent for publication was obtained from the patient.
Case Report
A 65-year-old male was referred to our hospital due to dysphagia that had appeared 1 month earlier; his medical history included acute pancreatitis. The patient reported smoking 40 cigarettes per day between the ages of 20 and 65 years, and drinking 3 gō of sake per day. Flexible fiberoptic laryngoscopy findings revealed a raised, irregular tumor in the epiglottis; a biopsy was performed and pathological examination indicated a squamous cell carcinoma (negative for p16). The patient underwent cervical contrast-enhanced computed tomography (CT) and fluorodeoxyglucose positron emission tomography/CT that showed a T3N2cM0 stage IVA right supraglottic squamous cell carcinoma.
For initial treatment and chemoselection, the patient underwent 1 course of TPF (cisplatin, docetaxel, and 5-fluorouracil) 3 as induction chemotherapy. The treatment outcome was evaluated as progressive disease (PD); therefore, surgical treatment was selected. He underwent total laryngectomy, bilateral neck dissection, pharyngeal reconstruction with anterolateral thigh flap, and creation of a permanent tracheostoma. During surgery, extranodal extension of the right cervical lymph node metastasis was detected; part of the right sternocleidomastoid muscle, right internal jugular vein, right accessory nerve, and right cervical nerve were therefore resected. Postoperative pathological examinations confirmed the extranodal extension of the right cervical lymph node metastasis, and concurrent chemoradiotherapy (cisplatin + radiotherapy) was suggested as adjuvant therapy. However, the patient opposed long-term hospitalization; therefore, he received radiotherapy alone after surgery.
Total radiation doses were 40 and 66 Gy (both administered in fractions of 2 Gy) for the whole and right neck, respectively. Upon irradiation of 40 Gy, cervical CT was performed to narrow the irradiation field; however, metastases were identified in the right lung apex. Cervicothoracic CT, performed after radiotherapy, identified multiple lung metastases (Figure 1A-C). The therapeutic effect of TPF used as induction chemotherapy was PD; thus, the cancer was deemed platinum refractory, and nivolumab was initiated as first-line pharmacotherapy. The tumor proportion score (TPS) for programmed death-ligand 1 (PD-L1) on the laryngeal tumor, evaluated using Dako 28-8 antibodies (Dako, Carpinteria, CA, USA), was <1.
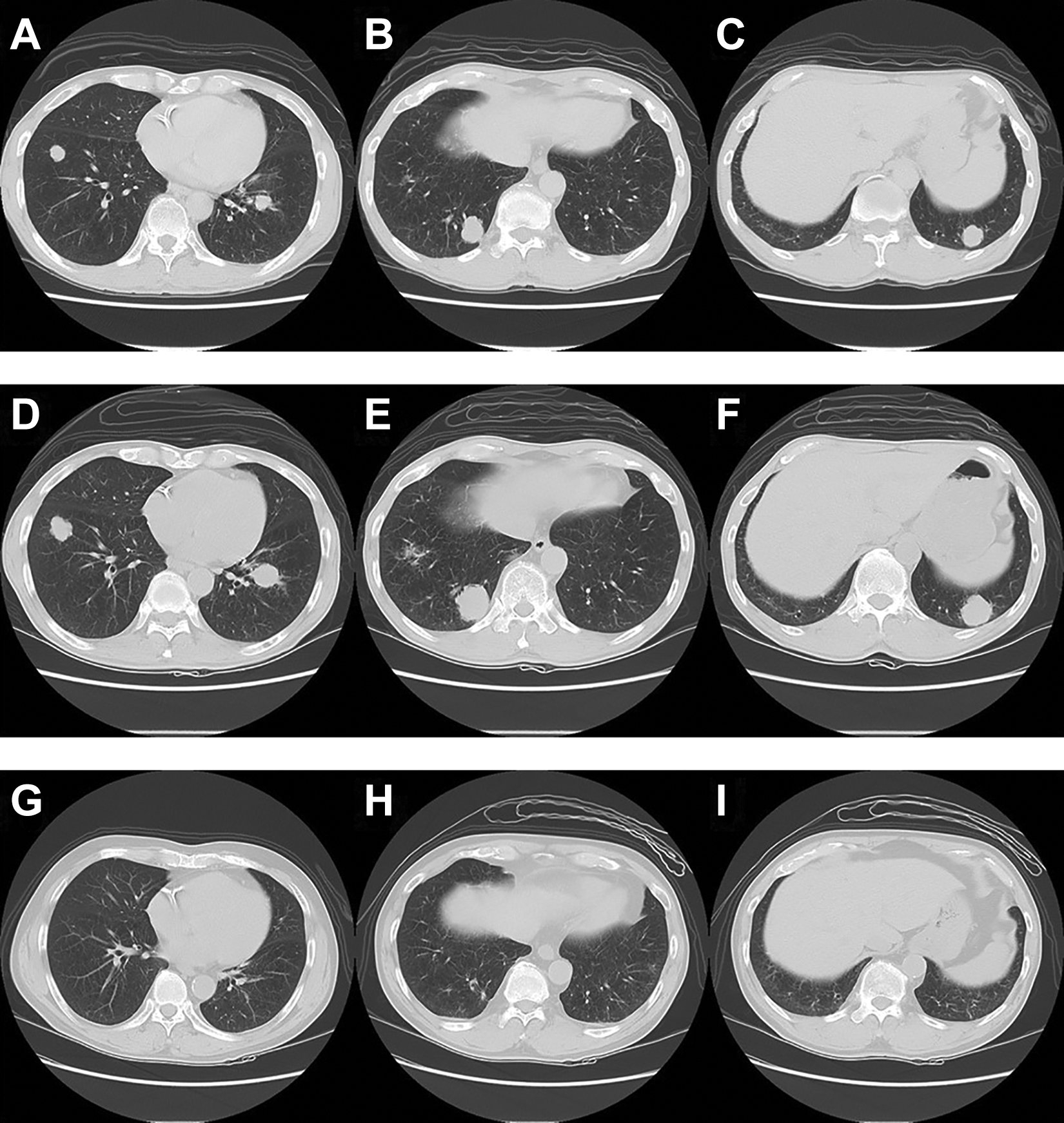
Computed tomography findings before and after nivolumab administration. A-C, Before nivolumab administration. Multiple small and large nodules are visible in both lungs, compatible with metastatic lung tumors. Approximately 20 nodules are seen, with a maximum nodule size of 25 mm. D-F, Day before the second course of nivolumab. All affected lung nodules are enlarged, with no increase in the number of nodules. G-I, After course 16 of nivolumab. All lung nodules have disappeared, and the shadow of interstitial pneumonia has improved.
Nivolumab (240 mg/body) was administered every 2 weeks from day 32 postradiotherapy. The patient did not exhibit any symptoms on the day before initiating the second course of treatment; however, he presented a white blood cell count of 9570/µL and an elevated C-reactive protein (CRP) levels of 8.5 mg/dL. Cervicothoracoabdominal CT scans evidenced no signs of infection; however, the metastatic lesions in the lungs had grown rapidly (Figure 1D-F). Despite this rapid lesion enlargement, we continued with nivolumab treatment as the patient exhibited a favorable general condition. Computed tomography scans repeated after courses 4, 8, and 12 of treatment showed a gradual reduction of the metastatic lesions. Since blood analyses performed during course 14 indicated grade 2 hypothyroidism, the patient was initiated on oral levothyroxine sodium hydrate (50 µg/d); subsequent normalization of thyroid function ensued.
A CT scan performed after course 16 of nivolumab suggested complete disappearance of the metastatic lesions in the lungs, and the patient was deemed to have achieved a CR. Grade 1 interstitial pneumonia was simultaneously identified; therefore, a descending dose regimen of prednisolone (10 mg/d) was initiated. A CT scan performed 14 days after initiating prednisolone treatment showed an improvement of the interstitial pneumonia (Figure 1G-I), and the patient resumed nivolumab treatment 14 days later. Courses 17 and 18 of nivolumab were administered, and CT performed after course 18 identified a recurrence of the interstitial pneumonia. As the patient had developed interstitial pneumonia twice while maintaining a CR for the lung metastases, we suspended nivolumab treatment (to be resumed upon lesion reappearance).
He also experienced frequent diarrhea (baseline + 6 times/) 5 months after discontinuing nivolumab treatment; therefore, he underwent abdominal CT and colonoscopy. An abdominal CT scan identified a circumferential wall thickening in the rectum (Figure 2A), while colonoscopy revealed ulcerative colitis-like findings in the rectum, with disappearance of visible vascular patterns and a coarse mucosa (Figure 2B and C). The biopsy tissue from the rectum primarily revealed infiltration of inflammatory cells including mononuclear cells, mucosal erosion, and apoptotic cells in the crypts (Figure 2 D). Immunohistochemical staining (Figure 2 E-I) revealed the inflammatory infiltrate to be composed of CD3-positive T cells and CD68-positive histiocytes. There was infiltration of both CD4+ and CD8+ T cells, and almost no infiltration of CD20+ B cells. Although histopathologically nonspecific, a diagnosis of exclusion was also performed for diseases such as cytomegalovirus enteritis. Finally, the patient was diagnosed with grade 2 colitis as an irAE; prednisolone treatment (60 mg/d) began after subsequent hospital admittance. The dosage was later reduced, and the patient was discharged on day 16 of hospitalization, receiving 30 mg/d of prednisolone. Although the patient was able to temporarily discontinue prednisolone, diarrhea recurred 61 days after the first event. Prednisolone treatment (40 mg/d) was therefore resumed and continued until 134 days after the initial diarrhea event; recurrence of colitis has not been observed since discontinuation of glucocorticoids. The patient continues to maintain a CR status without re-enlargement of the lung metastases 18 months after discontinuing nivolumab treatment.
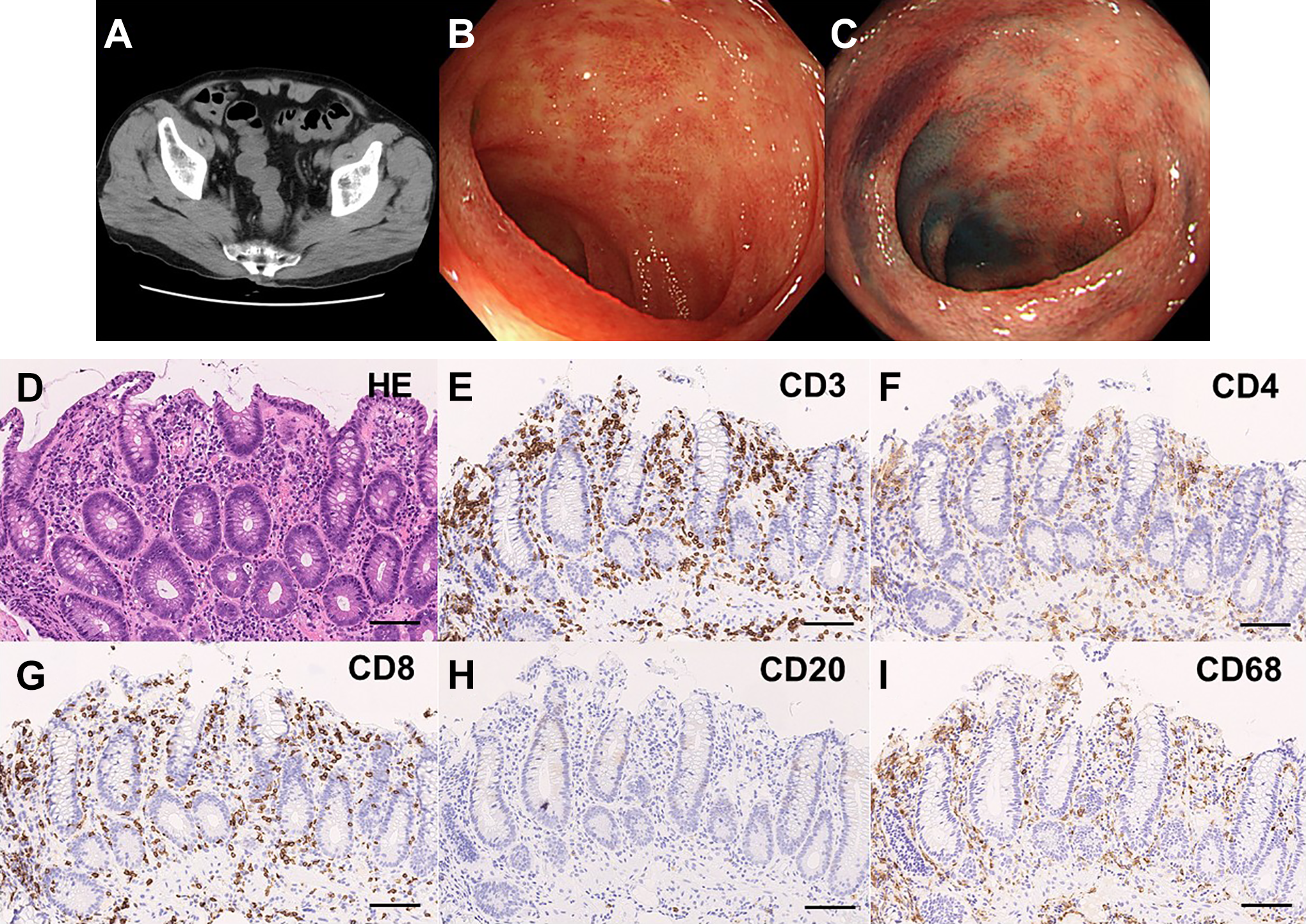
A-C, Abdominal computed tomography and colonoscopy findings. A, Abdominal plain computed tomography. Circumferential thickening of the colon wall was observed, with no mass suggesting a tumor in the colon. This is consistent with findings of colitis. B, Colonoscopy presentation (under normal observation). Normal observations indicated coarse mucosa and disappearance of visible vascular patterns, with fading areas in the rectal mucosa. C, Colonoscopy presentation (under dye spray). The dye spray finding of the same site revealed erythematous mucosa and erosions; all endoscopy findings were similar to those of ulcerative colitis. D-I, Histopathological findings of the rectal biopsy. D, Hematoxylin-eosin (HE) staining showing an inflammatory infiltrate (primarily composed of mononuclear cells), mucosal erosion, and apoptotic cells in the crypt. E-I, immunohistochemical staining using (E) anti-CD3, (F) anti-CD4, (G) anti-CD8, (H) anti-CD20, and (I) anti-CD68 antibodies. Almost no CD20+ B cell infiltration was observed; inflammatory infiltrate was composed of CD3+ T cells and CD68+ histiocytes. The extent of infiltration by CD4+ and CD8+ T cells was similar.
Discussion
The patient exhibited elevated CRP levels a day before receiving the second course of nivolumab, while CT revealed rapid enlargement of the lung metastases. We observed a reduction of the lesions via the CT scans taken after courses 4, 8, and 12 of treatment; and by the end of course 16, a CR status was confirmed. The rapid enlargement of multiple metastatic lung lesions was characterized by enlargement of each of the affected lung nodules, with no increase in the number of nodules, suggesting pseudoprogression. 4 We observed PD-L1—evaluated based on TPS using a surgical specimen—to be <1. In the CheckMate 141 study, 1 OS was longer in the nivolumab group than in the investigator’s choice treatment group in patients with a PD-L1 ≥1. In a previous report, examination of actual clinical experience showed a favorable OS, with a cutoff of ≥40 for PD-L1. 5 Our patient achieved a CR despite having a low PD-L1. Nivolumab proved effective despite PD-L1 being negative, suggesting that an abscopal effect 6 might have occurred. Considering that nivolumab was initiated early (on day 32 after radiotherapy), the multiple metastatic lesions in the lungs may have shrunk due to an abscopal effect of radiotherapy.
Our patient exhibited 3 irAEs, namely grade 2 thyroid dysfunction, grade 1 interstitial pneumonia, and grade 2 colitis. According to Okamoto et al, 7 the OS of patients with R/M HNC treated using nivolumab was better in those with irAEs. Matsuo et al 8 also reported that patients with R/M HNC exhibiting irAEs and receiving nivolumab had better OS, progression-free survival, objective response, and disease control rates. Additionally, despite experiencing 3 irAEs, our patient simultaneously achieved a CR status via nivolumab treatment. We therefore believe that controlling irAEs is key to ensure long-term nivolumab use in patients who respond to treatment.
According to a report by Weber et al, 9 85% of irAEs appear within 16 weeks of initiating treatment. Our patient exhibited grade 2 thyroid dysfunction, grade 1 interstitial pneumonia, and grade 2 colitis 6, 7, and 14 months after initiating nivolumab treatment, respectively, which is considerably later than what was reported by Weber et al. Colitis ensued 5 months after discontinuing nivolumab treatment; therefore, this case highlights the necessity of remaining attentive to irAEs, even after discontinuing treatment. In many cases, salvage chemotherapy with cytotoxic anticancer agents and molecular targeted drugs is administered, even in PD status patients who received nivolumab treatment. Fushimi et al 10 reported that salvage chemotherapy led to a significantly longer OS compared to best supportive care, even for R/M HNC. Regular medical interviews, blood tests, and diagnostic imaging are important to screen for irAEs, even if salvage chemotherapy is performed after discontinuing treatment. Additionally, Wang et al 11 reported higher lethal irAEs during the early stages of treatment; however, in our patient, irAEs appeared after a relatively long period after nivolumab induction. This timing may relate to the fact that we were able to control and improve the irAEs.
Histopathologically, colitis is reported to be characterized by the infiltration of lymphocytes and mononuclear cells (including histiocytes) in the lamina propria, cryptitis and crypt abscesses, and an increase in apoptotic cells in the crypt. 12 Although our patient did not present crypt abscesses, CD3+ T cell-dominant lymphocyte infiltration, CD68+ histiocyte infiltration, and crypt apoptosis were observed. The extent of CD4+ and CD8+ T cell infiltration was similar; codevelopment of other irAEs would thus suggest the colitis to be an irAE, rather than inflammatory bowel disease. 12 In our patient, interstitial pneumonia and thyroid dysfunction codeveloped, which was consistent with the presentation of colitis as an irAE. Wang et al 13 reported that the development of ICI-induced colitis was an independent prognostic factor in various malignant tumors (including melanoma) treated with ICI.
Yasumatsu et al 14 reported that patients who achieved a CR status via nivolumab treatment had a better prognosis than those who achieved a partial response or stable disease, even after treatment discontinuation. Additionally, Komiya et al 15 reported that in cases of non-small cell lung cancer treated with nivolumab or pembrolizumab alone, the response rate was significantly higher, and PFS significantly longer, in patients who discontinued treatment due to irAEs, rather than for other reasons. In our patient, the favorable prognosis may be related to the fact that a CR was achieved, and that the treatment was discontinued due to irAEs. There is currently no evidence to suggest when nivolumab treatment can be discontinued; therefore, treatment should continue even if a CR has been achieved. Still, as reported, some patients may continue to maintain CR status even after discontinuing nivolumab use; new evidence regarding when to discontinue ICI use upon achievement of a CR may therefore arise through accumulation of similar cases.
Conclusion
We reported a case of platinum-refractory recurrent/metastatic laryngeal squamous cell carcinoma that maintained a CR for 18 months after discontinuing nivolumab treatment. The patient exhibited irAEs such as grade 2 thyroid dysfunction, grade 1 interstitial pneumonia, and grade 2 colitis 6, 7, and 14 months after starting nivolumab treatment, respectively; colitis emerged 5 months after discontinuing treatment. It is thus necessary to consider the emergence of irAEs through regular testing, even after discontinuing nivolumab treatment.
Footnotes
Authors’ Note
Our institution does not require ethical approval for reporting individual cases. Written informed consent was obtained from the patient for his anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K. Tsukahara has received grant support from Ono Pharmaceutical Co., Ltd. The other authors have no potential conflicts of interest or financial relationships to disclose.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
