Abstract
Objectives:
To determine the efficacy of flurbiprofen 8.75 mg lozenges for patients with laboratory-confirmed streptococcal pharyngitis both before and concomitant with antibiotics.
Methods:
This post hoc analysis comprised adult participants from 2 earlier randomized, double-blind, placebo-controlled studies evaluating the analgesic efficacy of flurbiprofen 8.75 mg lozenges in acute pharyngitis. Throat swabs were obtained to diagnose streptococcal infection. Prior to and 2 hours after each dose of study medication (flurbiprofen or placebo lozenges), patients rated 3 symptoms of acute pharyngitis (sore throat pain, difficulty swallowing, and swollen throat) using visual analogue scales. Appropriate antibiotic treatment was initiated when culture results were reported. Mean changes in each pharyngeal symptom were compared over the immediate 24 hours before and during the initial 24 hours of antibiotic treatment.
Results:
Twenty-four patients provided both preantibiotic and concomitant antibiotic efficacy outcomes. Relief of throat pain was 93% greater in the flurbiprofen group than in the placebo group before antibiotic coadministration and 84% greater than placebo during antibiotic administration (both P < .05). Relief of difficulty swallowing was 71% greater in the flurbiprofen group than in the placebo before antibiotic administration (P = .16) and 107% greater during concomitant antibiotic administration (P = .04). Relief of the sensation of throat swelling was 295% greater with flurbiprofen than placebo before antibiotic administration (P = .008) and 70% greater during concomitant antibiotic administration (P = .06). For placebo-treated patients, relief from throat pain and difficulty swallowing were similar before and during antibiotic treatment (P > .05), indicating no benefit with antibiotic administration for these symptoms. No treatment-related discontinuations or serious adverse events were reported.
Conclusions:
Irrespective of antibiotic use, flurbiprofen 8.75 mg lozenges provide well-tolerated, effective relief of pharyngeal symptoms in patients with streptococcal infection. In the 24 hours after administration, antibiotics provide no relief of throat pain or difficulty swallowing beyond the topical demulcent effects of placebo lozenges.
Keywords
Introduction
Sore throat is a common symptom of pharyngitis due to upper respiratory tract infection (URTI), which affects adults 2 to 4 times a year on average. 1 Both viral and bacterial pathogens can trigger a sore throat by damaging oropharyngeal mucosa. It is the response to this tissue injury, in particular the production of proinflammatory mediators such as prostaglandins, which causes throat soreness and swelling. 2 In most individuals with sore throat, whether viral or bacterial in etiology, these symptoms are self-limiting. 3
Sore throat usually causes the most discomfort in the first few days of URTI, 4,5 when symptomatic relief can minimize throat pain, difficulty swallowing, and a swollen throat. It is for this therapeutic rationale that the use of analgesics and other nonantibiotic treatments are recommended by clinical practice guidelines and antimicrobial stewardship groups. 1,6,7 Symptomatic management tends to be short-term, as most sore throats are self-limiting. 3 However, because the use of analgesics could mask symptoms of disease progression, re-evaluation of any cases that are not improving or worsening after several days should be considered. As throat pain, throat swelling, and difficulty swallowing (dysphagia) are symptoms of localized pharyngeal inflammation, 2,8 a locally applied nonsteroidal anti-inflammatory drug (NSAID) such as flurbiprofen is a sensible therapeutic choice.
As up to 80% of pharyngitis in adults is caused by viruses 9 (against which antibiotics are ineffective 10 ), symptomatic management of nonbacterial pharyngitis avoids unnecessary and inappropriate use of antibiotics, 1,6,7 which contributes to the continuing medical challenge of global antibiotic resistance. 11,12 A number of bacterial species have been implicated in the etiology of sore throat, but group A, β-hemolytic streptococcus (GABHS) and group C β-hemolytic streptococcus (GCBHS) are the most common bacterial etiologies implicated and those for which antibiotic treatment may be indicated. 1,13,14 Assessment of the clinical presentation of sore throat using criteria such as the Centor scoring system can try to identify individuals with a higher likelihood of having a sore throat of bacterial origin. 1,15,16 However, because of the overlap in physical findings, this approach may not accurately predict etiology, leading to diagnostic inaccuracy and potential inappropriate use of antibiotics. 6,16 Rapid antigen tests for GABHS have a high specificity (∼95%) 17 ; however, they cannot distinguish between true GABHS infection and GABHS carriage. 17 The definitive test for bacterial “Strep throat” is a throat culture; however, a major limitation of this method is the inconvenience and 48-hour delay to obtain results.
In the absence of diagnostic confirmation or while awaiting culture results, there is a need to provide patients with symptomatic relief of pharyngitis. Furthermore, even in patients with confirmed streptococcal infection, antibiotics have only a modest effect on throat symptoms, shortening their duration by approximately 16 hours. 3 In this context, placebo-controlled studies have demonstrated that flurbiprofen 8.75 mg lozenges provide rapid, long-lasting and clinically meaningful relief on a single- and multidose basis 18 -22 regardless of microbiologic etiology. An earlier publication reported the efficacy of multiple doses of flurbiprofen 8.75 mg lozenges to relieve throat symptoms in patients with GABHS or GCBHS infection in the 24 hours before administration of antibiotics. 23 These findings prompted the research reported here. This article reports a post hoc analysis of data from 2 clinical trials designed to determine the efficacy, safety, onset, and duration of action of flurbiprofen 8.75 mg lozenges compared with placebo lozenges in patients with acute sore throat. 21,24 To the best of our knowledge, no placebo-controlled study has been published that demonstrates the efficacy of a topical and/or NSAID treatment of streptococcal pharyngitis both before and during administration of antibiotics, showing consistent efficacy and no drug–drug interaction. In so doing, we were also able to examine the efficacy of antibiotics for relief of throat symptoms in patients with laboratory-confirmed streptococcal pharyngitis receiving placebo lozenges.
Patients and Methods
Study Design
In order to examine a sufficient sample of patients with culture-proven Streptococcal pharyngitis, data were combined from 2 almost identical randomized, double-blind, placebo-controlled studies of similar design (NCT01049334 and NCT01048866) which evaluated the efficacy of flurbiprofen 8.75 mg lozenge in adult patients with acute sore throat. 21,24 The studies followed the International Conference on Harmonisation Good Clinical Practice and the ethical principles contained within the Declaration of Helsinki (South Africa, 1996) and complied with the Code of Federal Regulations of the US Food and Drug Administration. Ethical approval was obtained from institutional review boards, and patients provided written consent before enrollment. 21,24
Study Population
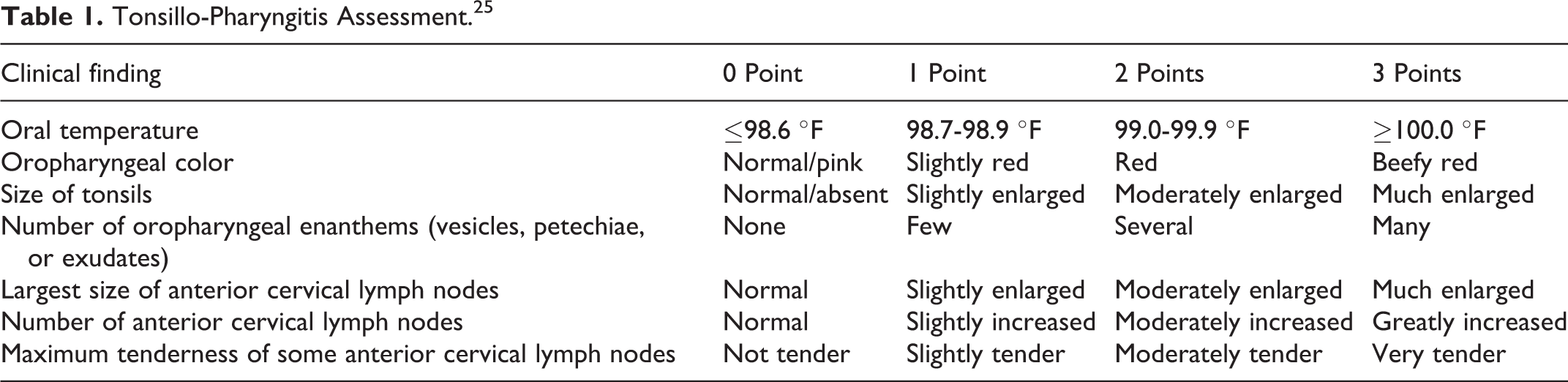
The studies enrolled patients >18 years of age with recent onset (≤3 days) sore throat. All patients were required to present with a complaint of moderate or severe sore throat pain (as assessed on a categorical Throat Pain Scale). Objective evidence of pharyngeal inflammation was confirmed by a score ≥5 on the Tonsillo-Pharyngitis Assessment (TPA; index comprised of the sum of 0-3 ratings for seven clinical features; Table 1). 25 All patients reported at least 1 URTI symptom on the URTI Questionnaire 26 and 3 symptoms commonly associated with tonsillopharyngitis: sore throat pain (≥66 mm on the 100-mm Sore Throat Pain Intensity Scale [STPIS]), difficulty swallowing (≥50 mm on the 100-mm Difficulty Swallowing Scale [DSS]), and the sensation of a swollen throat (≥33 mm on the 100-mm Swollen Throat Scale [SwoTS]). 21,27
Tonsillo-Pharyngitis Assessment. 25
Patients were excluded if they had: any known allergy and/or hypersensitivity to NSAIDs (including aspirin) or paracetamol; use of any throat/cough medication 1 hour prior to the initial dose of study medication; use of any cold/influenza medications or immediate-release analgesic/antipyretic within 4 hours or sustained-release analgesic/antipyretic within 12 hours preceding the initial dose of study medication; use of an antibiotic within 24 hours before the initial dose of study medication; or any evidence of mouth-breathing (causing throat dryness), coughing (that caused throat discomfort), or any active disease (eg, bronchopneumonia) which could compromise respiratory function.
Study Medications
Patients were randomly assigned to treatment with sugar-based, flavored flurbiprofen 8.75 mg lozenges or sugar-based, identically flavored, shaped and sized vehicle lozenges (placebo) by computer-generated randomization. Patients sucked 1 lozenge and after the full dose was dissolved in the mouth, the patient was not permitted to have anything by mouth for the next 2 hours in the clinic. Patients were then discharged and supplied with the same assigned study lozenges for use every 3 to 6 hours as needed, up to a total of 5 lozenges in 24 hours for the following 6 days. Rescue medication (paracetamol 650 mg) was permitted every 4 to 6 hours, if needed, up to 5 times in 24 hours.
Antibiotics (primarily oral penicillin, amoxicillin or azithromycin, as prescribed by the attending physician) were administered at approximately 48 hours after the first dose of study medication to patients with laboratory-confirmed streptococcal A infection and, at the practitioner’s discretion, to patients with still-symptomatic laboratory-confirmed streptococcal C throat infection.
Study Assessments
Pretreatment evaluations comprised the practitioner’s physical examination, including the TPA and clinical assessment of the extent of pharyngeal inflammation on the Practitioner’s Assessment of Inflammation (a categorical scale indicating the overall severity of pharyngeal inflammation, from mild to severe).
Throat swabs were taken (over the sides and back of the oropharynx) in all patients to conduct rapid streptococcal A tests (Directigen™ EZ Group A Strep Test; Becton Dickinson and Company) and to prepare throat cultures for streptococcal infection. Both throat tests were reported at approximately 48 hours (ie, 2 days after the patients’ and practitioners’ pretreatment assessments) when antibiotic therapy was initiated, if indicated.
At baseline, patients rated sore throat pain on the STPIS, difficulty swallowing on the DSS, and the sensation of a swollen throat on the SwoTS. Following the first dose of study medication, qualifying patients continued to record these ratings at hourly intervals for 24 hours (while awake). After 24 hours, patients rated their symptoms on the STPIS, DSS, and SwoTS immediately before and 2 hours after each dose of study medication. Patients reported any adverse event (AE) they experienced; all treatment-emergent AEs were recorded using Medical Dictionary for Regulatory Activities version 14.0 terminology.
Statistical Analysis
Because the patient populations and study designs of each study 21,24 were almost identical, STPIS, DSS, and SwoTS data were pooled from the 2 studies to provide a larger sample of patients with culture-proved streptococcal pharyngitis. Comparisons between treatment groups were conducted on the mean change from baseline in the 3 end points at 2 hours after each dose for 2 time periods: over the 24 hours immediately before antibiotic treatment and over the initial 24 hours during antibiotic treatment. Rescue medication use after a dose of study medication was considered treatment failure for that dose and all subsequent doses of study medication. Efficacy was calculated using least square means from an analysis of variance mixed model. Treatment before/during antibiotic administration and the corresponding interaction term were fitted as fixed effects. Each lozenge taken was treated as a repeated measure with a compound symmetry covariance structure with the patient fitted as a random effect. Two-sided statistical tests were performed with significance determined by reference to the 5% significance level. The AE records were analyzed for the same 2 time periods and statistical significance determined via chi-squared test.
Results
Patient Population
All patients presented with acute sore throat (mean onset: 2.2 days ± 0.97 standard deviation [SD]) and throat pain which they assessed as moderate or severe in intensity (mean: 2.45 ± 0.5 SD at onset).
Of the 402 enrolled patients, 1 patient did not have a throat culture test. Among the remaining 401 patients, on culture 40 (10.0%) were positive for streptococcal A and 56 (14.0%) were positive for streptococcal C (consistent with GABHS and GCBHS rates in the general adult population). 6,9
All 40 patients who tested positive for streptococcal A were treated with antibiotics when throat culture results were reported 2 days later. Additionally, 11 of the 56 streptococcal C-positive patients received antibiotics because they were clinically unimproved when culture results were reported. Of these 51 patients who received antibiotics, 26 were in the flurbiprofen 8.75 mg treatment group and 25 were in the placebo group, including, in particular, 11 patients who used flurbiprofen both before and during antibiotic administration and 13 patients who used placebo both before and during antibiotic administration. These 24 patients comprised the sample of antibiotic-treated patients in which the efficacy of flurbiprofen 8.75 mg compared with placebo was examined in the same patients during the 24-hour periods before and during antibiotic treatment. At baseline, patients in the treatment groups had relatively severe throat symptoms (as would be expected for patients seeking medical intervention), with no differences (all P > .05) between treatment groups in pretreatment levels of throat pain and inflammation or TPA score (Table 2).
Baseline Features of Patients Who Received Flurbiprofen 8.75 mg or Placebo Lozenges Before and During Antibiotic Administration.
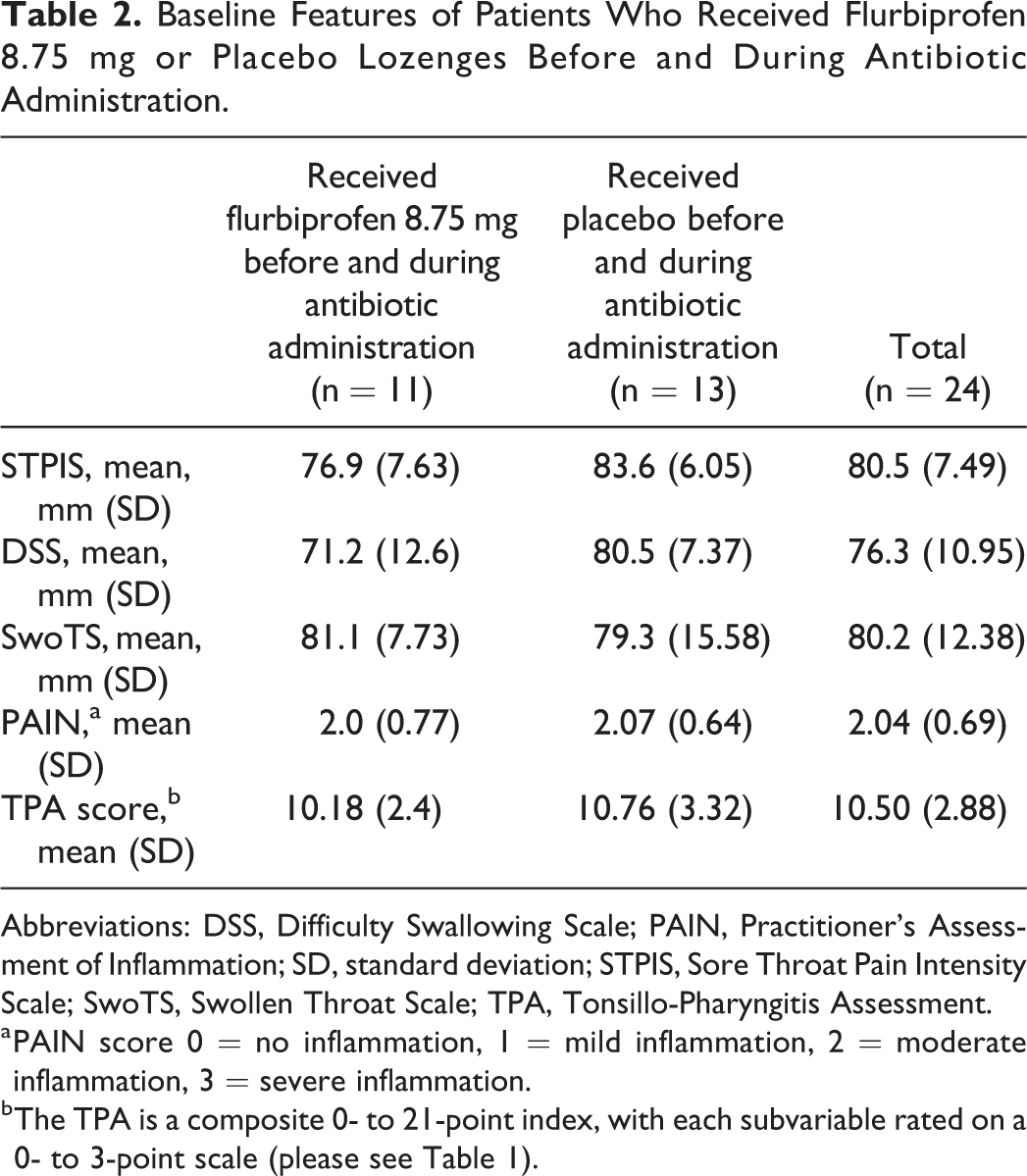
Abbreviations: DSS, Difficulty Swallowing Scale; PAIN, Practitioner’s Assessment of Inflammation; SD, standard deviation; STPIS, Sore Throat Pain Intensity Scale; SwoTS, Swollen Throat Scale; TPA, Tonsillo-Pharyngitis Assessment.
a PAIN score 0 = no inflammation, 1 = mild inflammation, 2 = moderate inflammation, 3 = severe inflammation.
b The TPA is a composite 0- to 21-point index, with each subvariable rated on a 0- to 3-point scale (please see Table 1).
In the 24-hour period before antibiotic administration, relief of sore throat pain was 93% greater in patients treated with flurbiprofen 8.75 mg lozenges than with those given placebo lozenges (P = .05; Figure 1A). During antibiotic coadministration, patients who used flurbiprofen 8.75 mg lozenges had 84% greater relief of sore throat pain than those who used placebo (P = .04; Figure 1B).
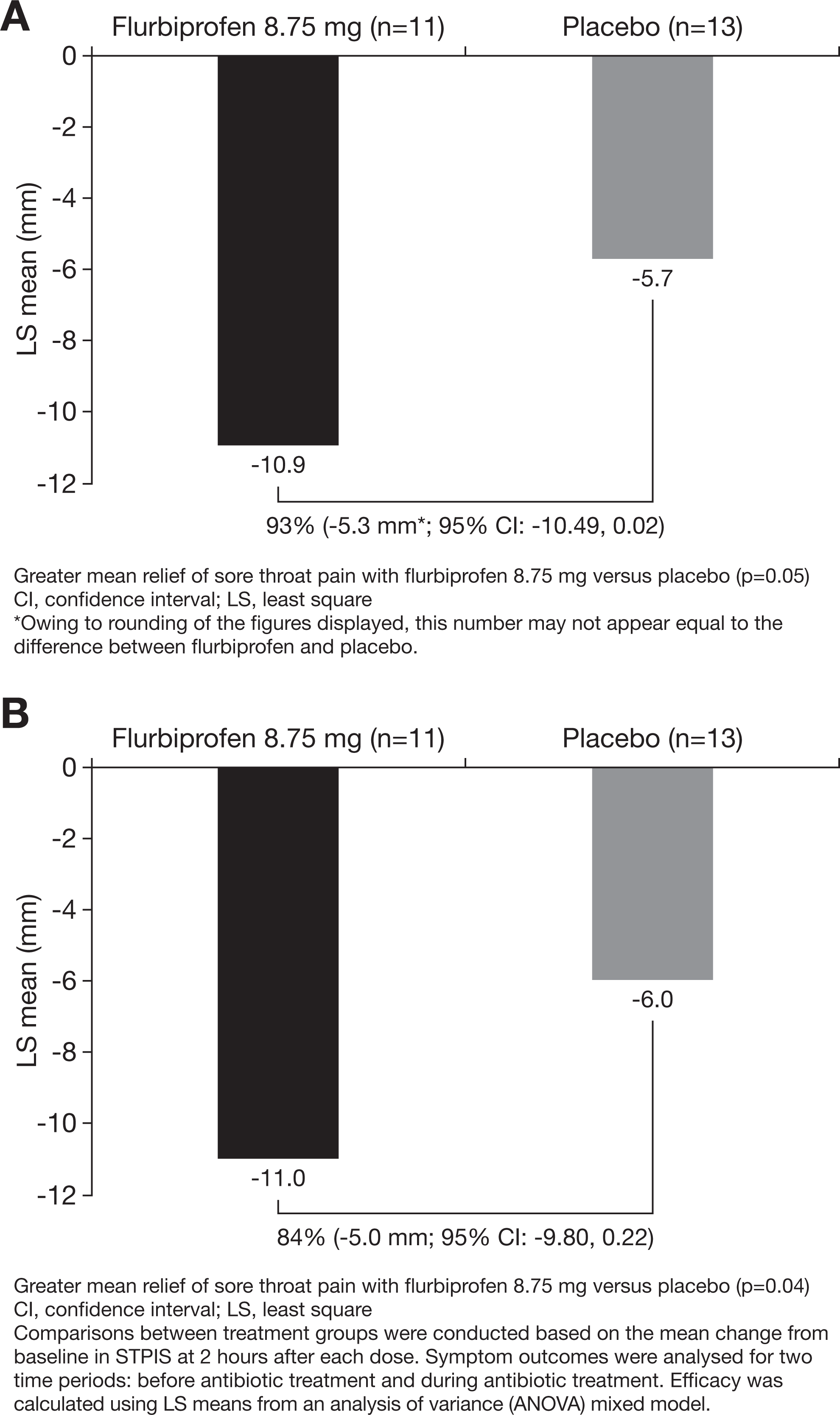
Differences between patients receiving flurbiprofen 8.75 mg and placebo lozenges measured on the Sore Throat Pain Intensity Scale (STPIS): A, During the 24-hour period before antibiotic administration and (B) during antibiotic administration.
Reduction in difficulty swallowing was 71% greater in patients who used flurbiprofen 8.75 mg lozenges than those who used placebo lozenges in the 24 hours before antibiotic administration (P = .16; Figure 2A). During antibiotic coadministration, the reduction in difficulty swallowing was 107% greater for the patients who used flurbiprofen 8.75 mg lozenges than for those who used placebo (P = .04; Figure 2B).
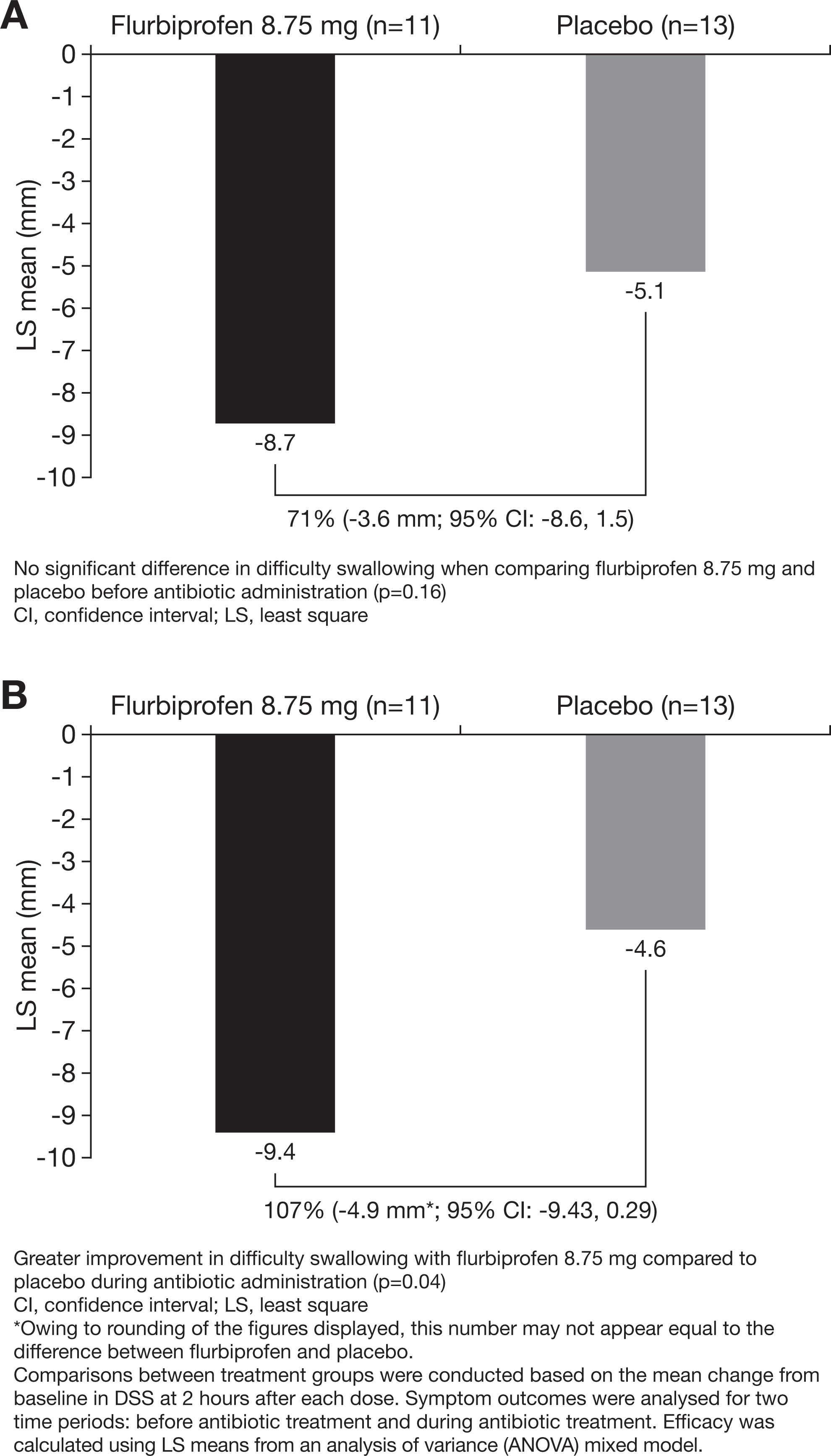
Differences between flurbiprofen 8.75 mg and placebo lozenges measured on the Difficulty Swallowing Scale (DSS): A, During the 24-hour period before antibiotic administration and (B) during antibiotic administration.
Before antibiotic administration, the reduction in the sensation of swollen throat was 295% greater for patients treated with flurbiprofen 8.75 mg lozenges than placebo (P = .008; Figure 3A). During antibiotic coadministration, relief of swollen throat was 70% greater for patients using flurbiprofen 8.75 mg lozenges compared with placebo (P = .06; Figure 3B).
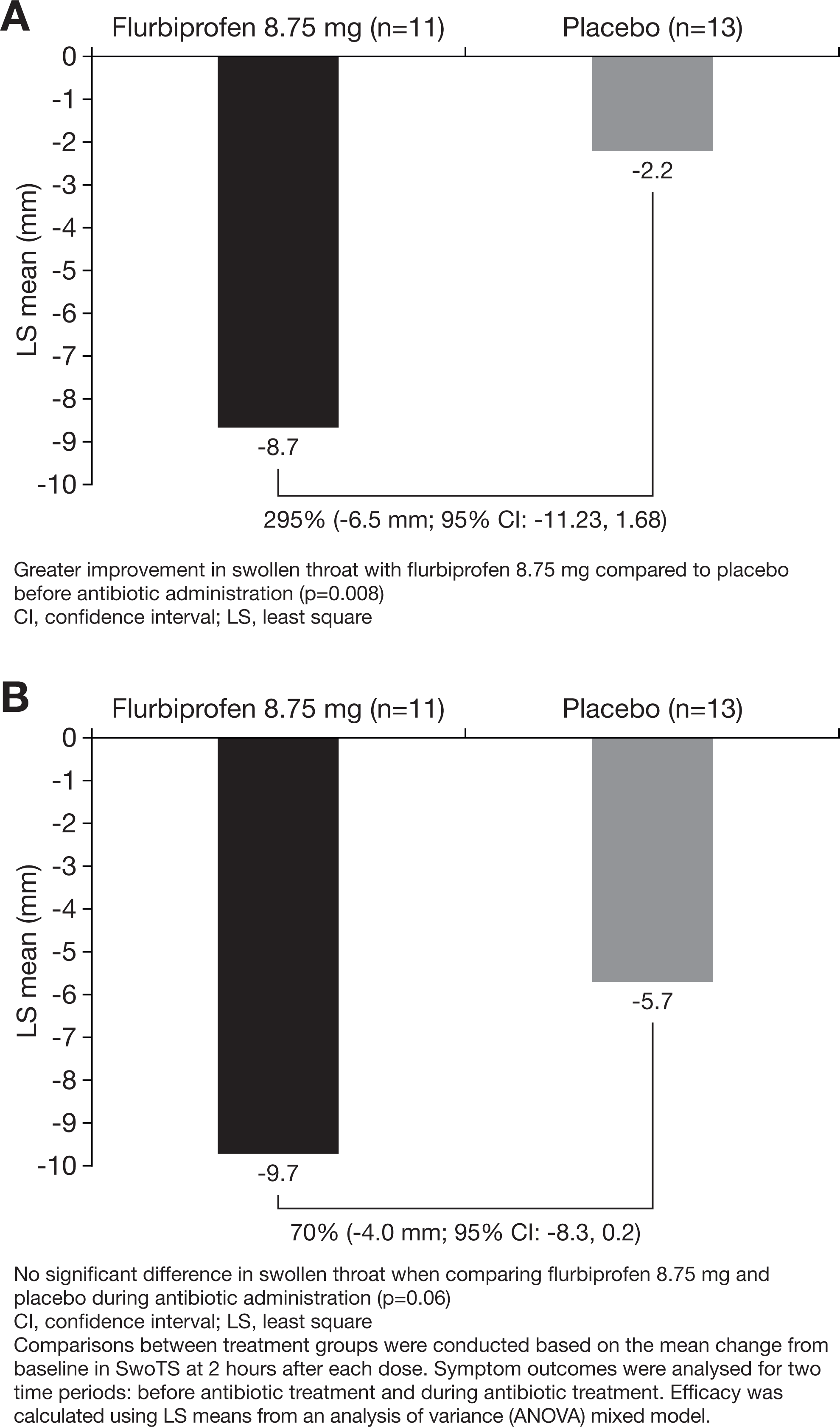
Differences between flurbiprofen 8.75 mg and placebo lozenges measured on the Swollen Throat Scale (SwoTS): A, During the 24-hour period before antibiotic administration and (B) during antibiotic administration.
In flurbiprofen-treated patients, comparisons of the 24-hour changes in STPIS, DSS, and SwoTS scores before and during antibiotic treatment show that these outcomes were not influenced by the administration of antibiotics (P = .96, P = .66, P = .54, respectively; Figure 4A-C).
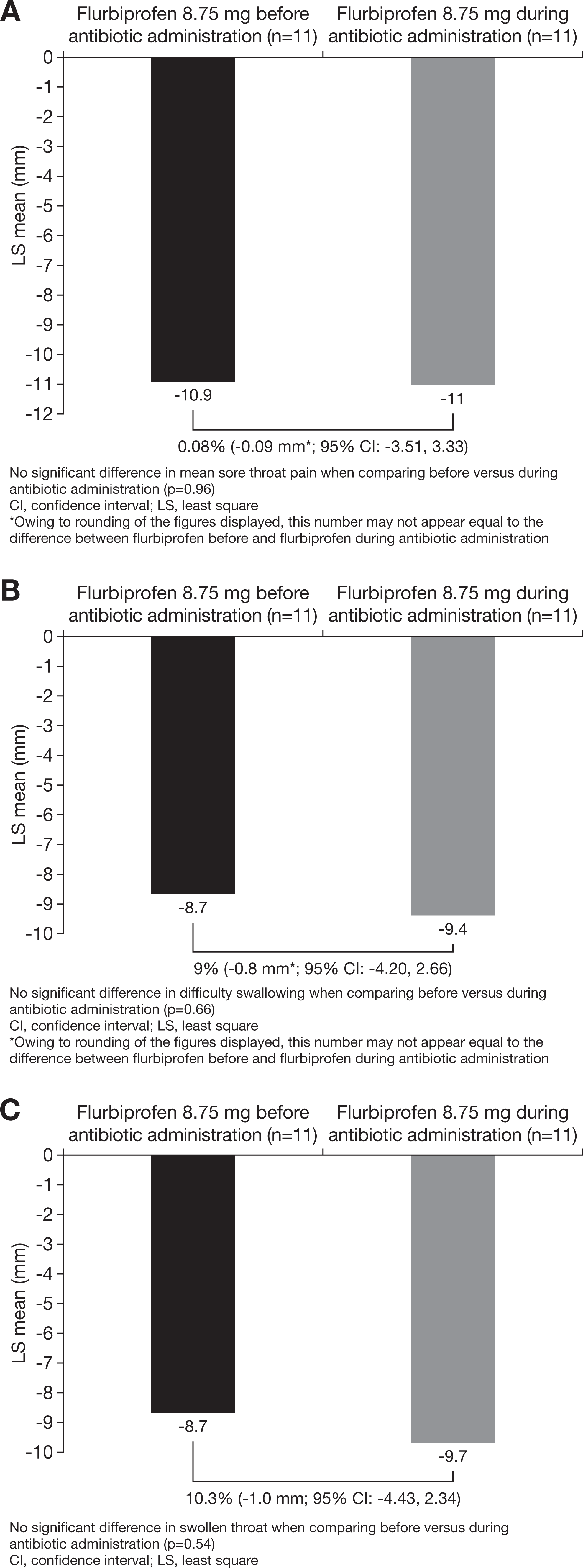
Pharyngeal symptoms in patients taking flurbiprofen 8.75 mg lozenges before and during antibiotic administration measured on the (A) Sore Throat Pain Intensity Scale (STPIS); (B) Difficulty Swallowing Scale (DSS); (C) Swollen Throat Scale (SwoTS).
For placebo-treated patients, the 24-hour changes in STPIS scores before and during antibiotic treatment were similar (P = .8), indicating no significant effect of antibiotics on relief of sore throat pain. Changes in DSS scores for the placebo group were also not significantly different before and during antibiotic administration (P = .7). Antibiotics provided no relief for these 2 pharyngeal symptoms, but SwoTS scores for placebo-treated patients indicated a 61% greater reduction in the sensation of a swollen throat during antibiotic administration (P = .02).
Adverse Events
Among the 51 patients who received antibiotics, the percentage of patients who reported any AE in the 24 hours before antibiotic administration was higher in the placebo group than in the flurbiprofen 8.75 mg group (12.0% vs 3.8%, respectively, P = .57; Table 3). During the 24-hour period with concomitant antibiotic administration, the percentage of patients reporting any AE was 34.6% in the flurbiprofen 8.75 mg group and 24.0% in the placebo group (P = .6). No serious AEs were reported and none of the patients discontinued use of flurbiprofen because of an AE before or during use of an antibiotic.
Comparison of the percentage of patients who reported AEs before and during antibiotic administration (regardless of treatment group assignment) revealed a higher occurrence rate in the period of antibiotic administration (7.8% vs 29.4%; P = .01). The percentage of patients with gastrointestinal AEs was 11.8% during antibiotic administration compared with 3.9% before antibiotics (P = .28). Placebo-treated patients reported twice as many AEs during antibiotic use compared with the period before antibiotic administration (24% and 12%, respectively), although this did not achieve statistical significance (P = .46).
Incidence of AEs in the Population Who Received Antibiotics.
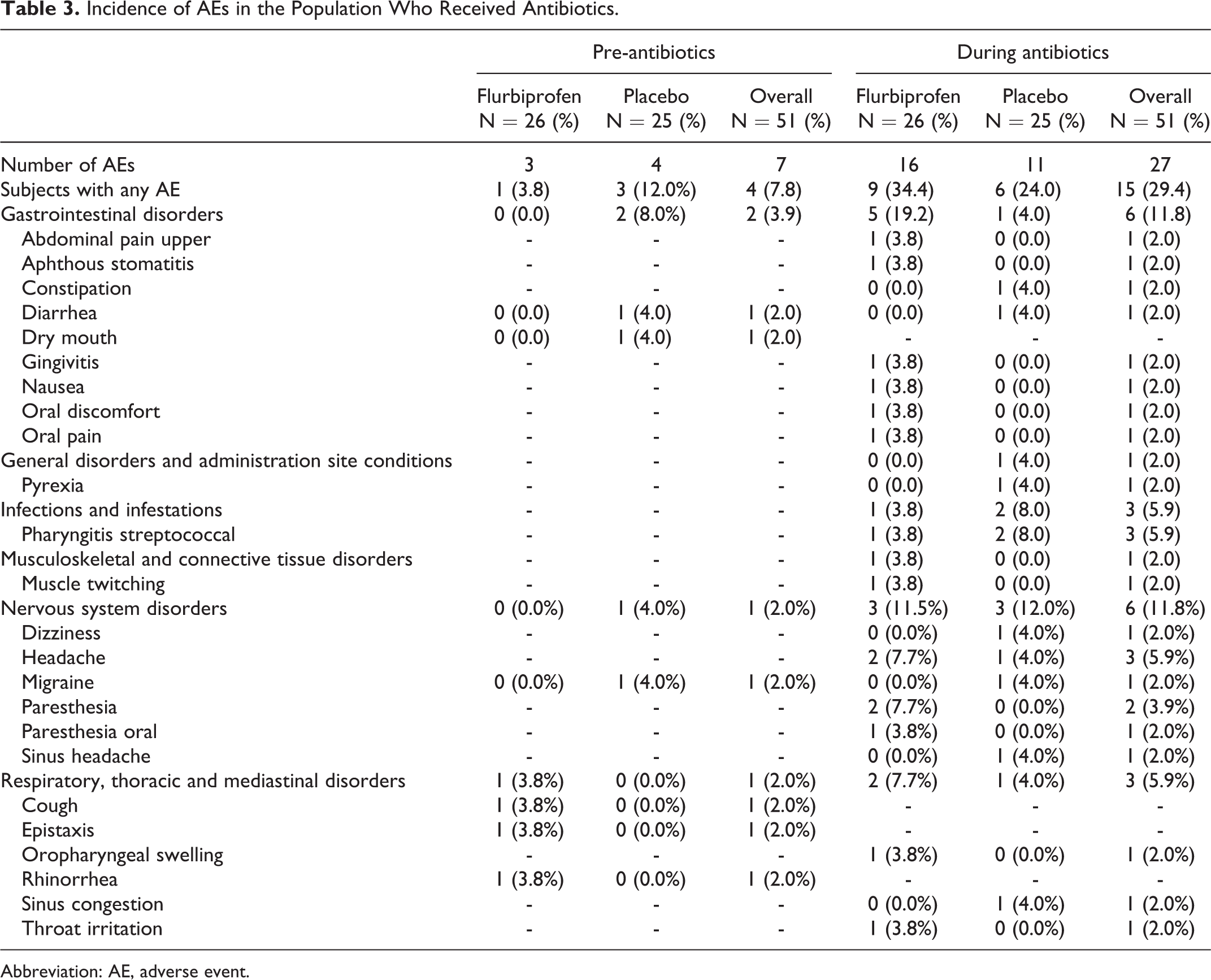
Abbreviation: AE, adverse event.
Discussion
These results demonstrate that flurbiprofen 8.75 mg lozenges provided effective sore throat pain relief in patients who were prescribed antibiotics for laboratory-confirmed streptococcal pharyngeal infection and that relief of sore throat pain was not affected by subsequent coadministration with antibiotics. Despite the small sample size (due to ca 10% incidence of proven streptococcal A pharyngitis), analgesic efficacy of flurbiprofen 8.75 mg lozenges compared with placebo lozenges was evident both before administration of antibiotics and while patients were also receiving antibiotics, with 93% and 84% differentiation from placebo for each treatment period (P = .05 and P = .04, respectively).
Flurbiprofen 8.75 mg lozenges also provided significantly (107%) greater relief of difficulty swallowing compared with placebo during antibiotic administration (P = .04). Prior to antibiotic administration, flurbiprofen provided 71% greater reduction in difficulty swallowing than placebo lozenges, although not statistically significant (P = .16). Relief from swollen throat was significantly (295%) greater with flurbiprofen than placebo prior to the administration of antibiotics (P = .008), with 70% greater reduction in swollen throat compared with placebo lozenges during antibiotic administration (P = .06). The limited number of patients with laboratory-confirmed streptococcal pharyngitis reduced the power to detect statistically significant differences in all treatment comparisons. The demulcent effect 28 of the dissolving placebo lozenges also provided relief of swollen throat and difficulty swallowing, somewhat masking the differences between the placebo and flurbiprofen lozenge treatments. Despite the small sample size and the effect of the “active” placebo lozenges, the efficacy of flurbiprofen 8.75 mg lozenges in these “streptococcal-positive” patients was maintained during preantibiotic and concomitant antibiotic administration for sore throat pain, as well as for the other patient-reported outcomes, swollen throat and difficulty swallowing.
As observed in previous studies of flurbiprofen 8.75 mg lozenges, there were no serious AEs or clinical sequelae. 19,21,24,29 -31 However, AEs were reported more frequently when antibiotics were taken (P = .01), providing another reason to avoid antibiotics unless indicated on a proven microbiologic basis.
This study also provides further insight about the analgesic role of antibiotics in culture-proven streptococcal infection. In placebo-treated patients, antibiotic treatment did not provide relief of throat pain or difficulty swallowing beyond the demulcent effects of the vehicle ingredients of the lozenge itself.
Streptococcal pharyngitis occurs in only approximately 10% of adults. 6 The research reported here was designed to compare the outcomes in the same culture-positive patients before and during antibiotic treatment; consequently, the study sample was limited by the natural incidence of this infection. Nevertheless, clinically impressive and statistically significant differences were observed between flurbiprofen and placebo regardless of concomitant antibiotic therapy, with no evidence of analgesic effect associated with antibiotics. There was no difference in the efficacy of flurbiprofen 8.75 mg between pre- and concomitant-antibiotic administration and no additional relief of pharyngeal pain attributable to coadministration of antibiotics.
Sore throat is a common reason for patients to utilize primary care, however the majority of patients are unlikely to require antibiotics due to the predominant viral etiology of pharyngitis. 9,28 Inappropriate antibiotic usage for minor self-limiting conditions is a major contributor to antibiotic resistance. 11,12 Thus, avoiding unnecessary antibiotic use in this population is important. In patients with confirmed streptococcus infection, of course, treatment with antibiotics may be appropriate. However, this study showed that over the initial 24 hours of treatment, antibiotic treatment confers no additional relief of sore throat beyond the topical demulcent effects of dissolving placebo lozenges. 32 As sore throat pain and difficulty swallowing are symptoms of pharyngeal inflammation, treatment with a locally delivered low-dose NSAID such as flurbiprofen 8.75 mg could complement antibiotic treatment when needed. In addition, in regions where throat swabs are rarely taken (such as the United Kingdom), flurbiprofen 8.75 mg lozenges could support a delayed antibiotic prescription strategy in suitable patients, where antibiotics are only taken if symptoms deteriorate or fail to improve. 33 Flurbiprofen 8.75 mg lozenges are indicated for 3 days’ use and re-evaluation of any cases that do not improve within this time frame is recommended. The safety of a delayed antibiotic prescription strategy in sore throat has been shown in a large observational study. 34
Previous studies have shown that flurbiprofen 8.75 mg lozenges offer effective relief of pain, difficulty swallowing, and swollen throat irrespective of the cause of sore throat. 18 -23 The research reported here demonstrates that coadministration of flurbiprofen lozenge and antibiotics does not affect the efficacy or AEs profile of flurbiprofen in patients with culture-proven “Strep throat”. Flurbiprofen lozenges represent an accessible and effective first-line choice for symptomatic relief of pharyngitis allowing antibiotics to be reserved for patients with culture-proven streptococcal pharyngitis.
Footnotes
Authors’ Note
Medical writing assistance was provided by Louisa Reed at Elements Communications Ltd, Westerham, United Kingdom. Bernard Schachtel, Adrian Shephard, Emily Schachtel, and Tim Shea were involved in the design and conduct of the studies from which data has been pooled for this manuscript. Adam Smith and Evi Tselenti conducted the statistical analyses reported in the article. All authors were involved in the conception and development of this manuscript and have critically reviewed the drafts, approved the final content, and are wholly accountable for its content.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tim Shea, Adam Smith, and Adrian Shephard are employees of Reckitt Benckiser. Evi Tselenti was an employee of Reckitt Benckiser when this manuscript was being prepared. Bernard Schachtel and Emily Schachtel (at Schachtel Research Company) received a research grant from Reckitt Benckiser to design and conduct the studies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Reckitt Benckiser Healthcare International Ltd.
