Abstract
We hereby present the first-reported pediatric case of a hard palate neurovascular hamartoma in a male newborn, in which the diagnosis was established following an initial nondiagnostic biopsy, extensive radiological investigations, and eventual wide local excision. These benign lesions can easily be mistaken for malignant diagnoses, leading to increased parental and child anxiety as well as avoidable diagnostic and therapeutic interventions.
The term hamartoma, first coined by Albrecht in 1904, describes a tumor-like developmental malformation composed of hazardous tissue elements endogenous to its anatomic location. 1,2 Within the oral cavity, the most commonly implicated subsites are the oral tongue, buccal mucosa, and lower lip. 2,3 Tissues that may proliferate include smooth and skeletal muscle, fat, salivary tissues, epithelium, as well as nervous, vascular, and lymphatic structures. Neurovascular hamartomas (NVH) of the oral cavity are exceedingly rare, although previous authors have hypothesized this may be due to unrecognition and underreporting. 3
A 20-day-old male was referred to our clinic for an ulcerated lesion of the anterior hard palate. The latter was first noticed a few days following an uncomplicated delivery at term and had since rapidly increased in size. Examination of the hard palate revealed an 8-mm, midline, ulcerated lesion that was soft to palpation (Figure 1). The child had no syndromic features and no palpable lymphadenopathy. An incisional biopsy revealed nonspecific ulcerated granulation tissue with prominent thin-walled vasculature and acute and chronic inflammatory infiltrates. Additionally, foci of reactive stromal cells were identified, which were negative for all immunohistochemical markers for rhabdoid, histiocytic, epithelial, and neuroectodermal lineages. No definitive evidence of malignancy was noted by histopathology and immunohistochemistry.
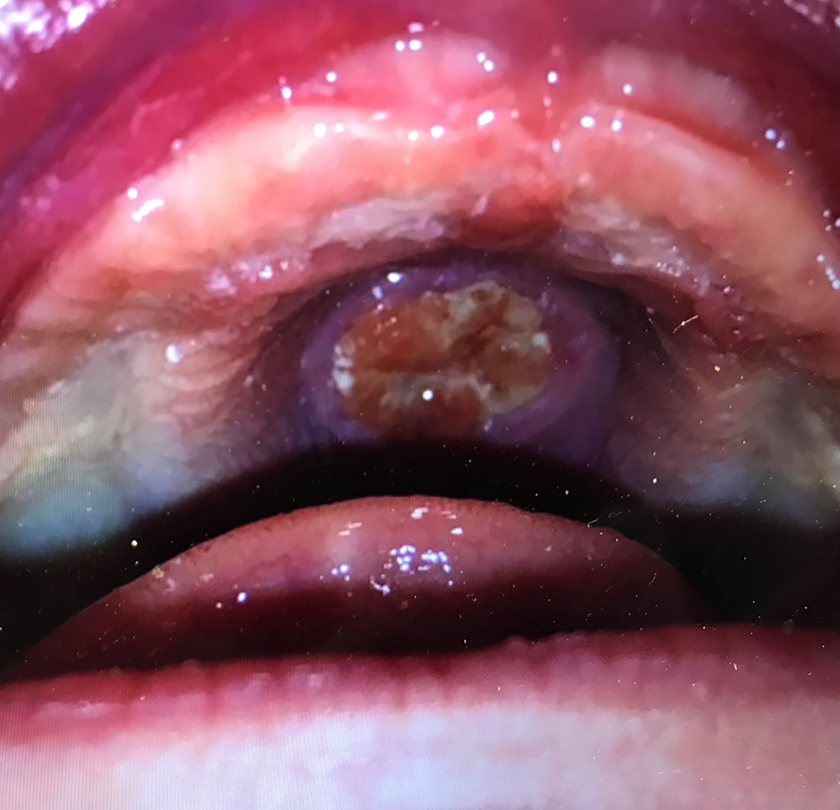
Post-biopsy ulcerated lesion of the anterior hard palate.
A post-biopsy magnetic resonance imaging study was obtained, demonstrating a residual 7 × 8 × 4 mm anterior hard palate mass, hyperintense on T1 and T2 with enhancement, and suspected osteolytic changes of the floor of the nasal fossa (Figure 2). On computed tomography, the lesion appeared to be causing focal erosion, almost transfixing the bone toward the nasal fossa.
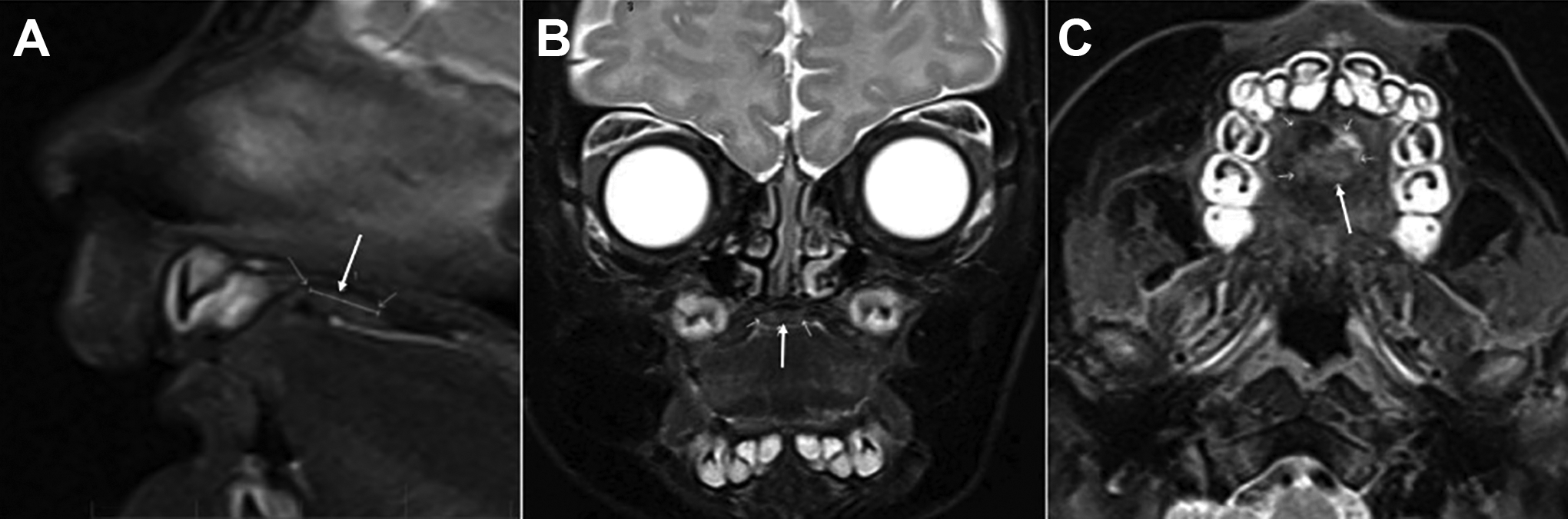
Sagittal (A), coronal (B), and axial (C) views of magnetic resonance imaging displaying a small solid lytic lesion of the anterior palatal mass (à) (T2 sequence).
In light of the aforementioned equivocal biopsy results and radiographic findings, the lesion was excised with wide circumferential margins and drilling of the palatine bone. Histologic examination of the resected specimen revealed organoid neurovascular bundles surrounded by adipose tissue wih thin-walled arteries, consistent with an oral NVH (Figure 3). Surgical margins were negative on pathology. At 6-month follow-up, the surgical site was well healed with no evidence of locoregional recurrence.
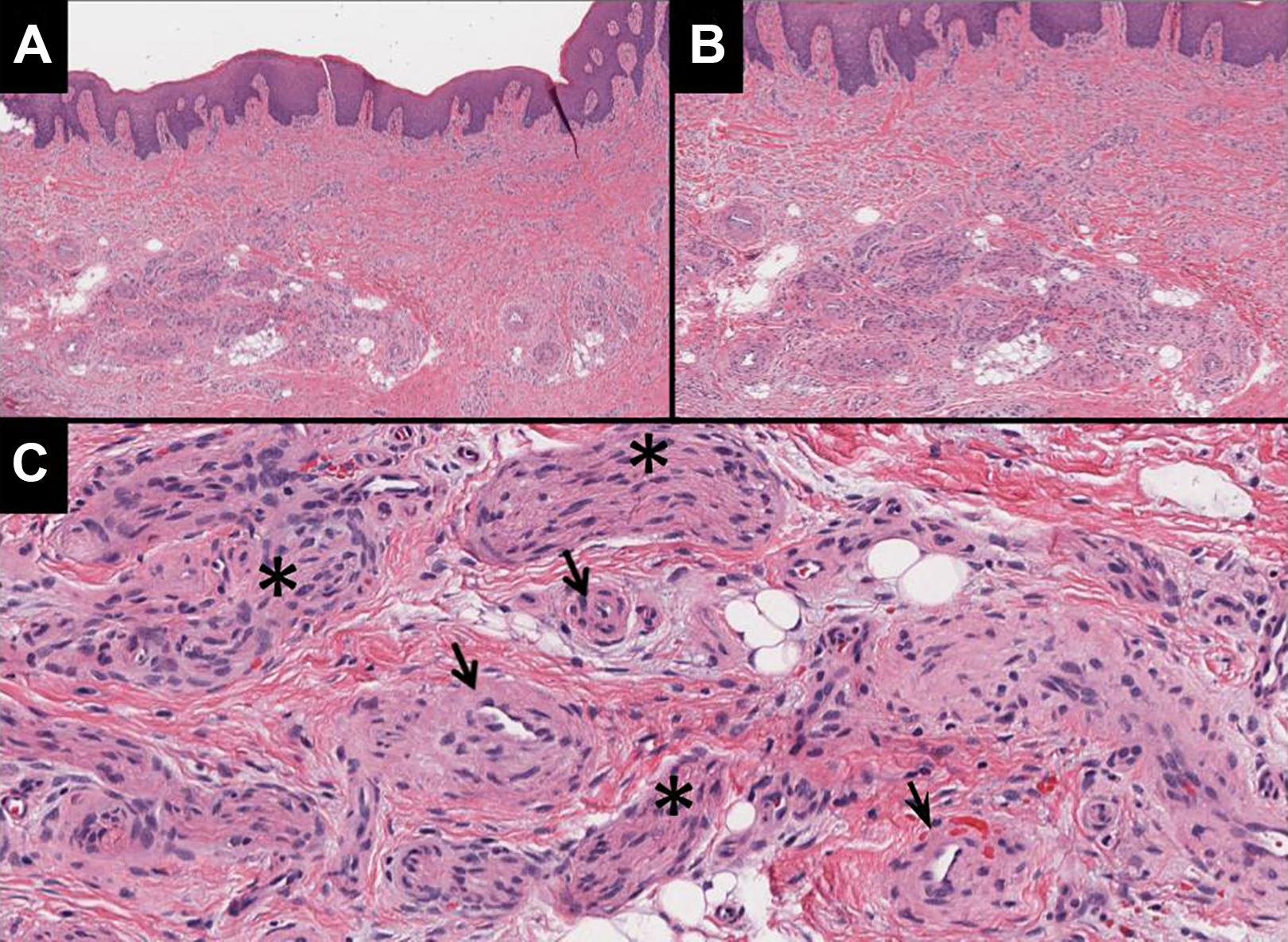
Histopathological H&E stain of the palatal neurovascular hamartoma lesion (A) low power 4× view of lesion’s architecture (B) higher 10× power view showing irregular organization of neurovascular structures within deep submucosa of the palate (C) high power 20× view with indicated spindle shaped nerve cell bundles (*), numerous small blood vessels (à), and surrounding adipose cells.
Classically, harmatomas present as asymptomatic exophytic tumoral growths at birth or in early childhood formed by disorganized, yet normal endogenous tissue. 4 To our knowledge, our case represents the first reported pediatric NVH of the hard palate. Indeed, even in the largest series of 25 cases of oral cavity NVHs, the most commonly involved subsites were the oral tongue, buccal mucosa, or lower lip, and only 1 NVH of the palate was listed in an elderly patient. 3
Other types of hamartomas have been reported in the hard palate; however, none were predominantly composed of neural and vascular tissues. 5 -7 Zaitoun et al reported on a case of a smooth muscle hamartoma of the hard palate, postulating that the latter anatomical subsite may be prone to hamartomas in virtue of its complex embryological events. 6
Palatal tumors pose a unique radiological challenge, and dual modality imaging may improve diagnostic performance in differentiating between benign and malignant tumors. 8 Bony erosion of the thin palatal bone (as was seen in our patient) or perineural invasion on imaging may increase clinical suspicion of malignancy and warrant further investigations.
Given the nonspecific clinical and radiological features of NVHs, histopathology is ultimately required to establish the diagnosis. Allon et al first described the microscopic characteristics of oral NVHs, describing them as nonencapsulated, poorly circumscribed masses comprising a high propensity of nerve bundles closely entwined with small- to medium-sized blood vessels within a loose connective tissue matrix. 3 Although these histopathological features may bear similarity with traumatic neuromas, in the latter, the neuronal and vascular components are separate and will additionally show inflammation and fibrosis. 9 A further diagnostic consideration is a benign mesenchymoma, defined as an unencapsulated tumor containing greater than 2 benign mesenchymal tissues, excluding fibroblastic elements, with no single predominant tissue. 9 Although this entity theoretically encompasses NVHs, this may prove solely a nomenclative quandary. 3,9
A recent review of head and neck hamartomas has shown that these benign entities may expand and recur, and chronic inflammation may heighten the risk of malignant transformation. 4 Hence, complete surgical resection is indicated, although partial resection may be considered for large hamartomas with anticipated poor functional or cosmetic results. 4 In conclusion, this rare case of oral NVH presenting as a palatal mass with apparent bony invasion on radiologic studies serves as a humbling reminder that hamartomas, although benign, may at times mimic aggressive malignancies.
Footnotes
Authors’ Note
Ethics approval was waived by the McGill University Health Centre Research Ethics Board. Consent was obtained from the child’s parents for publication of this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
