Abstract
Objectives:
To evaluate the feasibility, morphological, and functional outcomes of endoscopic retrograde transcanal mastoidectomy.
Study Design:
Prospective study.
Settings:
Tertiary Referral Hospital.
Materials and Methods:
We analyzed 31 patients with a diagnosis of chronic otitis media with cholesteatoma that extended to the mastoid cavity who underwent endoscopic transcanal retrograde canal wall down mastoidectomy under general anesthesia. The tympanic membrane and posterior canal wall reconstruction were done using the tragal cartilage palisade technique reinforced with perichondrium. Morphological and functional results were reported in the follow-up of a minimum of 1 year. The graft uptake was also compared with the presence of granulations and discharge status.
Results:
Graft uptake and disease-free conditions were achieved in 21 of 24 patients, that is, 87.5%, and 3 patients required revision surgery. The graft uptake rate was not affected by the presence of granulation tissues and discharge. The hearing was improved after the surgery, which was statistically significant.
Conclusion:
Endoscopic retrograde mastoidectomy is a new technique to deal with cholesteatoma with excellent results. It is more functional, ideal for sclerotic mastoid, allows faster wound healing, has excellent outcomes, and is a minimally invasive procedure that avoids postauricular incision and tissue dissection. Because the set up for the surgery costs less, it holds a promising future for the developing countries.
Introduction
Chronic otitis media is one of the common ear problems encountered by the otologist. 1 It can be divided as cholesteatomatous or noncholesteatomatous. 2 The pathogenesis of the cholesteatoma is still unknown. 3 The risk factors include low socioeconomic status, Eustachian tube dysfunction, congenital birth defects as cleft palate, undernutrition, and low birth weight. 4 The condition is highly prevalent in developing countries. The management of cholesteatoma is still surgical to date. 5
The microscope has been the gold standard tool for mastoid surgery. 6 Over 5 decades have passed, the posterior canal wall management is still controversial. Canal wall down or up techniques for mastoidectomy are still debated widely. 7 The preference depends on different factors such as the surgeon’s experience, belief, and confidence. 8 These techniques described were widely based on microscopic ear surgery.
Modern microscopes provide excellent views of the surgical field, 3-dimensional binocular vision, and 2-handed surgery, but the visualization of deeper recesses in the middle ear is limited. The microscope provides straight-line vision, and its optical properties necessitate an adequate amount of light to reach the surgical plane. As a result, microscope-based surgical approaches frequently require postauricular or endaural incisions, soft tissue retraction, or bony drilling for adequate visualization of the targeted pathology. 9
The concept of otological surgeries is gradually evolving after the introduction of the endoscope. 10 Endoscopic ear surgery (EES) is a recently introduced field in otology, and many otologists are now accepting it. 11 The endoscope was introduced in the world by Harold Hopkins as Hopkins rod endoscope in 1966. 12 However, the first study on transcanal EES (TEES) was only reported in 1992 by El Guindy. 13 Before that, the endoscope was utilized as an additional tool to assist microscopic ear surgery.
Endoscopes allow the surgeon to address the target pathology transcanal while reducing the need for postauricular/endaural incisions, canalplasty, and avoiding the associated morbidity leading to the evolution of minimally invasive ear surgery. 10 Good transcanal endoscopic access into the attic and antrum could be provided by removing a relatively limited amount of bone, which is ideal for sclerotic mastoid with contracted antrum. 8 - 10
There are numerous articles on attic cholesteatoma and limited articles on the endoscopic management of the mastoid cholesteatoma. This study extends the indications for EES by reporting the feasibility, outcomes, advantages, and disadvantages of EES by doing transcanal retrograde mastoidectomy.
Materials and Methods
We prospectively analyzed 31 patients who underwent endoscopic transcanal retrograde mastoidectomy in our hospital from December 2016 to April 2018. Of 31 patients, 4 patients lost to follow-up, and 3 patients were converted to microscopic ear surgery; thus, only 24 patients were included.
The study’s inclusion criteria were patients presenting with attic cholesteatoma extending into the mastoid cavity with or without the involvement of mesotympanum along with sclerotic mastoid in HRCT temporal bone. Revision cases, chronic otitis media with simple perforation without cholesteatoma, cholesteatoma involving only attic, atelectasis, and retraction pocket without cholesteatoma or debris, and the patients who needed microscopic assistance were excluded from the study. Preoperative scan revealing extensive cholesteatoma involving mastoid tip, sinodural angle, dehiscence of tegmen, or with complications as facial nerve palsy, intracranial abscess was excluded. The study received approval from the institutional review board of the hospital. Informed consent was obtained from all the participants.
Surgical Procedure
All surgeries were done under hypotensive general anesthesia with patient head elevation at 15° to 30° to decrease the venous congestion and intraoperative bleeding. A 3-chip high-definition video camera system, rigid high-resolution of 4 mm diameter, 18 cm length, 0°-, and 30° wide-angle otoendoscopes were used along with angled elevators, picks, and forceps. The light intensity of the endoscope was kept at less than 50% (Figures 1 and 2).
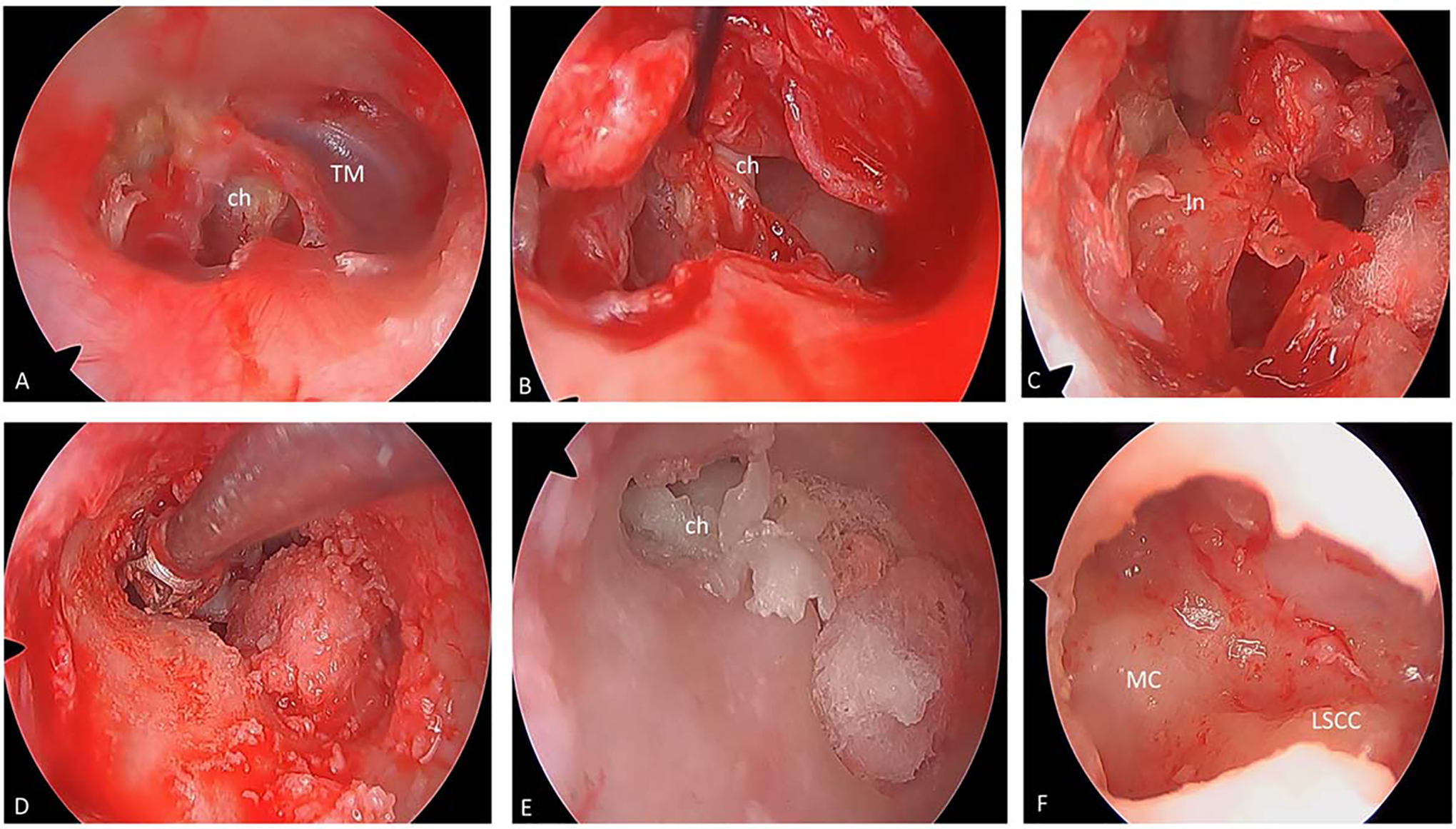
A, Preoperative evaluation, destroyed attic with cholesteatoma and posterosuperior tympanic membrane (TM) perforation. B, Elevation of tympanomeatal flap with cholesteatoma sac at posterosuperior quadrant. C, Necrosed incus removed. D, Drilling was done at the attic and posteriosuperior canal wall. E, Cholesteatoma removed. F, Mastoid cavity after drilling posterior canal wall and removal of cholesteatoma.
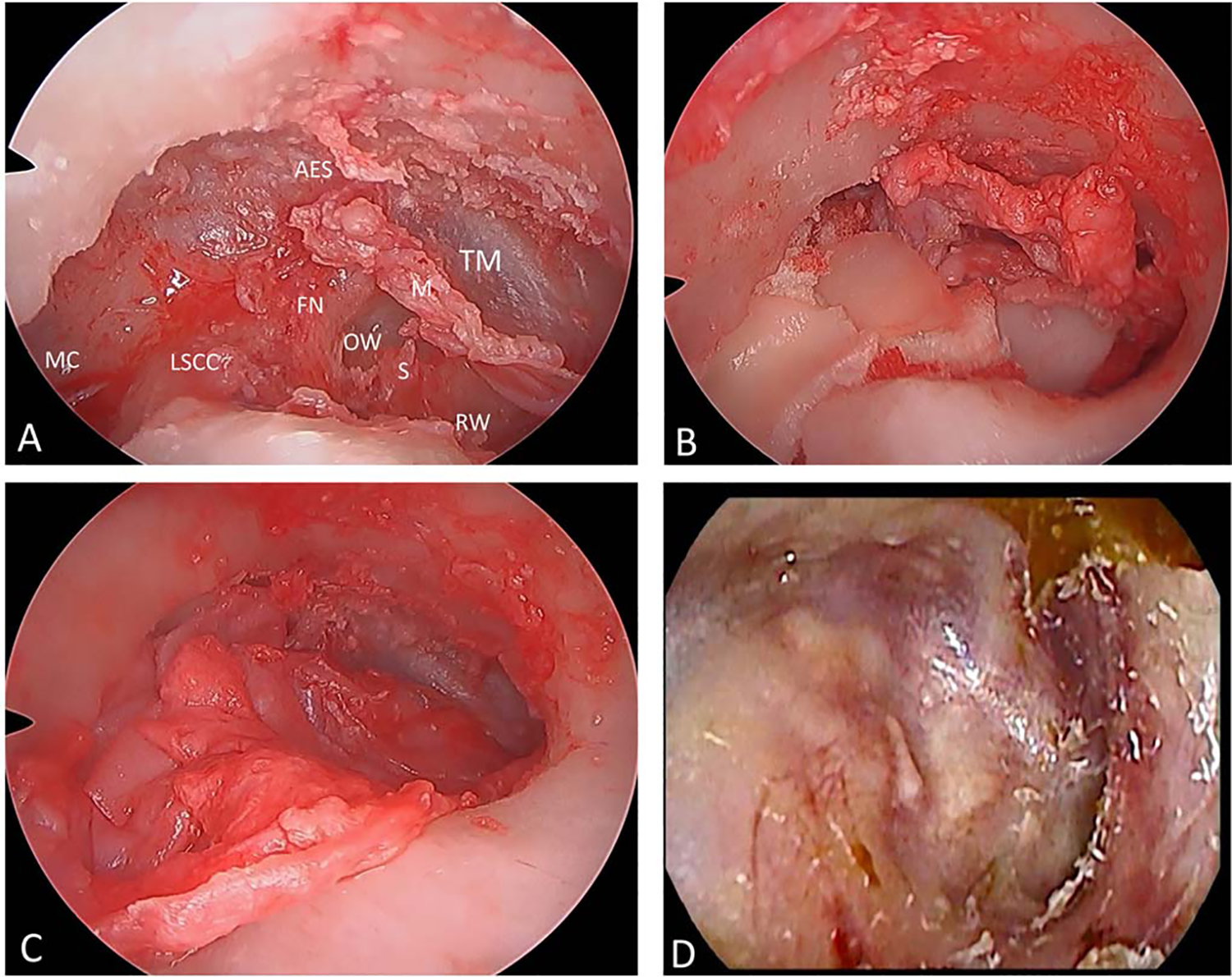
A, Evaluation of the middle ear and mastoid cavity to check for clearance of disease. B, Tympanic membrane (TM) and canal wall reconstruction done with tragal cartilage palisades. C, Final cavity and TM reconstruction after reinforced with perichondrium. D, Graft uptake after 1-year follow-up. AES indicates anterior epitympanic space; Ch, cholesteatoma; In, incus; FN, horizontal segment of the facial nerve; LSCC, lateral semicircular canal; M, malleus; MC, mastoid cavity; OW, oval window; RW, round window; S, stapes.
Two percent lidocaine with 1:200,000 adrenaline was injected around the ear canal and at the tragus. A wide posterior tympanomeatal flap was elevated by incising the canal from 2 to 5’o clock for the right ear and 10 to 7’o clock for the left ear. The flap was elevated from the posterior annulus and placed inferiorly if the cholesteatoma was found in the middle ear under a closed or perforated tympanic membrane (TM). The middle ear status, ossicular chain, and tympanic isthmus were evaluated. The cholesteatoma involving the middle ear was dissected gradually.
The outer attic wall, if intact, was removed using a curette and/or drill, and atticotomy was performed. Incus was removed if the lenticular and long process was necrosed. The head of the malleus was removed as well to clear the disease involving anterior epitympanic space. The cholesteatoma was followed transcanally starting from the middle ear. The drilling was carried out in the posterosuperior canal wall. Complete exposure of the lateral semicircular canal and the horizontal facial nerve was done. The speed of the burr was kept at 30 000 rpm, less than the usual speed. Cutting and diamond burr of size 1 to 3 mm were used as needed. The drilling was done intermittently for flushing and suctioning. Once the bone was paper thinned, a curette was used to remove the bones. These steps were continued until the end of the cholesteatoma sac was reached. Drilling was done carefully under vision using 0°- and a 30°-endoscope, avoiding damage to the critical structures such as tympanomeatal flap, facial nerve, lateral semicircular canal, chorda, and stapes. As much as possible, a minimal amount of the posterior canal wall was removed to clear the disease completely.
The cholesteatoma was then removed under vision. Common hidden sites of cholesteatoma were thoroughly checked, such as sinus tympani, anterior epitympanum, mastoid cavity, posterior sinus, hypotympanum, and protympanum. The final check of the mastoid cavity and the clearance of the cholesteatoma was inspected.
Ossicular reconstruction was done with the sculptured autologous incus if it was not grossly necrosed or cartilage.14,15 The TM defect and the posterior canal wall were reconstructed with tragal cartilage reinforced with perichondrium. The ear canal was packed with the ear pack. The tragus incision site was not sutured, and the bandage was not applied. The patients were discharged the next day following the surgery.
Follow-Up
After surgery, an antibiotic (amoxicillin 500 mg plus clavulanic acid 125 mg) was given per oral 3 times a day for 7 days. Analgesics (paracetamol 500 mg plus ibuprofen 400 mg) were given 3 times a day for 3 days and then as needed. The patient was followed up after 7 days, and the ear pack was removed. Patients were followed up after 1 month, 3 months, 6 months, and yearly or as needed after that. The average on 4 frequencies (0.5, 1, 2, and 3 kHz) of hearing thresholds in air and bone conduction and the air–bone gap (ABG) were evaluated 7 days before surgery, 12 weeks after, and yearly basis on follow-up.
Statistical Tools
Data were analyzed for graft uptake rate, change in pre- and postoperative hearing status using the Fisher exact test, paired t test in IBM SPSS Statistics for Windows, version 23.0 (IBM Corp). The level of statistical significance was set at P < .05.
Results
During the study period, 24 patients underwent endoscopic retrograde mastoidectomy for cholesteatomatous chronic otitis media. The demographic profile of the patients is shown in Table 1. The mean age of patients was 24.25 ± 6.46; age ranged from 16 to 39 years. There were 13 (54.2%) males and 11 (45.8%) females. As for the laterality of the disease, 10 (41.7%) patients had disease on the right side, and 14 (58.3%) had on the left side.
Distribution of Enrolled Patients.a

Abbreviations: M, Malleus; I, incus; S, stapes; (−), absent; (+), present.
a Fisher exact test.
The TM was perforated in 13 (54.2%) patients, intact in 6 (25%) patients, and retracted (Sade’s Grade III) in 5 (20.8%) patients. The outer attic wall, that is, scutum, was destroyed in 17 (70.8%) and intact in 7 (29.2%) patients. The epitympanic cholesteatoma extending to the mastoid cavity only was observed in 9 (37.5%) patients, and in 15 patients (62.5%), the cholesteatoma involved both the mesotympanum and mastoid cavity. The anterior and posterior isthmus of the ventilatory pathway was obstructed due to cholesteatoma in all the patients. The most common ossicular defect was necrosed long and lenticular process of incus in 17 (70.8%) patients, followed by absent ossicles, but intact stapes footplate in 5 (20.8%) patients, and 2 (8.3%) patients had absent malleus and incus, but intact and mobile stapes.
Eighteen (75%) patients had active discharge during the surgery, along with cholesteatoma, and 6 (25%) patients had no discharge. We evaluated the granulation tissues and cholesteatoma, 16 (66.7%) patients had granulations and the cholesteatoma, and 8 (33.3%) patients had no granulations. Chorda tympani nerve could not be preserved in 13 (54.2%) patients, and it was kept intact in 11 (45.8%) patients. All the patients were followed up for a minimum of 1 year, with a mean follow-up of 18.92 ± 7.9 months. The graft uptake and disease-free status were seen in 21 (87.5%) patients and failure that required revision surgery in 3 (12.5%) patients. We compared the graft uptake and disease-free condition with granulations and found it statistically not significant (P = .216). Furthermore, we compared graft uptake with the middle ear discharge, which was also not significant (P = .285).
Hearing evaluations were also done pre- and postoperatively (Tables 2 and 3). The mean pre- and postoperative air conduction thresholds were 48.33 ± 14.29 and 36.66 ± 14.12, respectively. The mean pre- and postoperative ABG was 30.41 ± 10.99 and 21.27 ± 11.20, respectively. The difference was statistically significant (P = .000). The maximum preoperative ABG was 21 to 30 dB in 37.5%, followed by more than 40 dB in 25%. Postoperatively, the maximum ABG was 11 to 20 dB in 37.5%, followed by 21 to 30 dB in 20.8%. Thus, there was improved hearing after the surgery.
Pre- and Postoperative Hearing Outcomes.
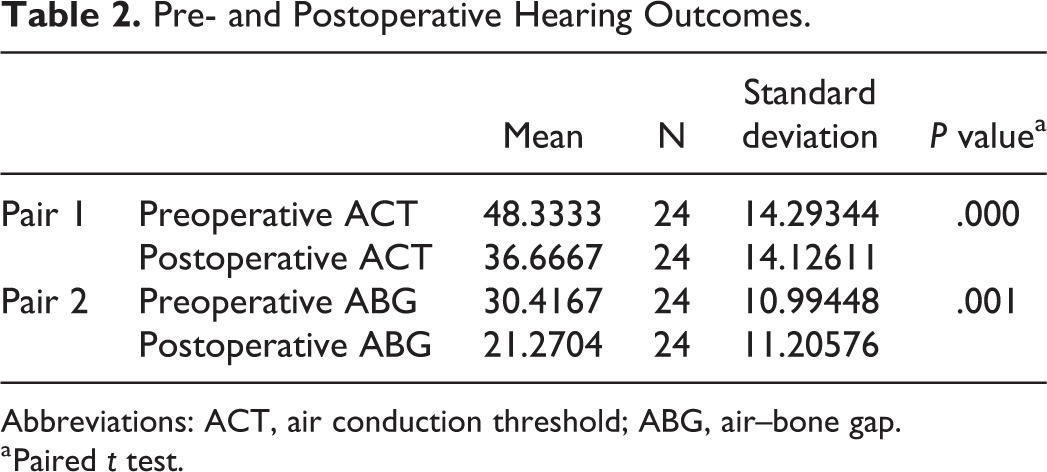
Abbreviations: ACT, air conduction threshold; ABG, air–bone gap.
a Paired t test.
Air–Bone Gap Range.
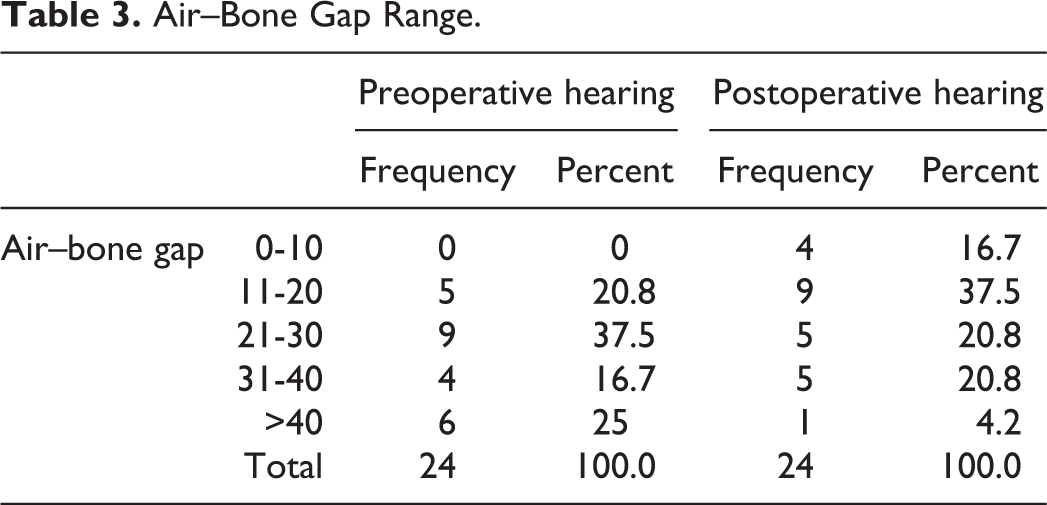
There were no significant postoperative complications noted after the surgery, such as vertigo, facial nerve palsy, cerebrospinal fluid leakage, and sensorineural hearing loss. There were no complaints regarding postsurgical alteration in taste sensation in patients with loss of chorda tympani, which may be due to the disease’s chronicity. 16 However, 4 patients had a laceration in the outer ear canal due to the friction created by the drilling. All the frictional skin lacerations healed by 1 week. None of the patients experienced canal stenosis.
Discussion
History has witnessed many changes in the mastoidectomy procedure. It has improved mainly due to the advances in surgical equipment during the last century. 17 The procedure is further evolving with the introduction of the endoscope in ear surgery. In this study, endoscopic retrograde mastoidectomy was feasible for moderate cholesteatoma. Controversies on doing canal wall down or up, mastoidectomy still linger even after 5 decades. 18 The canal wall up technique preserves the posterior canal wall, which prevents the cavity problem. 19 The hearing aid insertion after the surgery is straightforward. The patients have no restrictions on water activity. However, it has the drawbacks of having a high recurrence rate and recidivism with studies reporting from 15% to 70%. 7 The use of an endoscope at the end of the surgery has decreased the residual cholesteatoma rate from 0% to 15%. 10
Although it may not be universal in all cases, drilling from the outer mastoid cortex to the sclerotic mastoid antrum can be difficult and tedious. The preferred surgical approach in tight sclerotic mastoid cavities with attic cholesteatoma has been to proceed with a canal wall down mastoidectomy.7,8 The residual and recurrent cholesteatoma is decreased with this technique. However, the cavity requires regular cleaning and long-term or lifelong follow-up. 20 Based on the literature, the canal wall down technique remains one of the most effective procedures in preventing recurrent disease after a single-stage surgery and is considered a gold standard technique.8,21,22
On the other hand, retrograde mastoidectomy combines the virtues of both canal wall down and up techniques by the partial removal of the bony canal wall depending on the extent of the disease.7,23 Most importantly, it creates the smallest cavity needed to remove all the cholesteatoma to preserve more external auditory canal bone to facilitate reconstruction. 24 The inside-out mastoidectomy is not a new technique and was reported by McQuiston in 1950 and by Moore in 1957 using endaural incision.25,26 Similar technique of small cavity retrograde mastoidectomy was reported by Smyth, which resulted in small cavity surgery. 27 The disease was followed from the bony canal to the mastoid cavity in the retrograde fashion. The technique was then modified by Professor Jan Helms, who did posterior canaloplasty and widening the canal until the air cells were seen through the bone. 7 The disease was followed up, and if it extended only to the antrum, the reconstruction of the canal wall defect was done via conchal cartilage. However, if the disease was extensive involving mastoid tip, “turning around the corner,” the canal was taken down as described by Smyth.
The technique was further modified by Dornhoffer, 7 where the author was able to preserve the lower two-thirds of the posterior canal wall, even if the disease was extensive, “turning around the corner” to the mastoid tip. The preservation of the posterior canal wall helped the author to reconstruct the posterior canal wall with the cartilage. However, the above procedures were all done via a microscope using a postauricular approach.
Roth et al studied 604 ears with a mean follow-up of 10 years and concluded that the inside-out technique allowed the safe removal of cholesteatoma and constituted a minimally invasive concept. 28 Retrograde mastoidectomy for the cholesteatoma involving sclerotic mastoid can safely be performed with good results. For small cholesteatoma limited to epitympanum, the microscopic retrograde mastoidectomy can be performed transcanally with little damage to the ossicular chain. 28 The moderately large cholesteatoma eroding epitympanic space, antrum, and ossicles require limited retroauricular or endaural epitympanectomy with ossiculoplasty. The extensive cholesteatoma growing far into the mastoid requires classical retroauricular radical cavity operation. 28
We performed the retrograde endoscopic mastoidectomy for moderate cholesteatoma via an exclusive transcanal approach. The disease was followed from the middle ear to the mastoid cavity. Only diseased bone and an on-demand minimal amount of healthy bone were removed to have access to target the pathology. We found endoscopic mastoidectomy can be ideal for small or moderate cholesteatoma. However, combined microscopic and endoscopic postauricular mastoidectomy is needed for extensive cholesteatoma for optimum results.
The endoscope’s main advantage is the wide-angle view and availability of the angled scope that allows visualization of the nooks and corners including the mastoid cavity. As Tarabichi 29 explained, most of the cholesteatoma originates from the middle ear and then spreads to the mastoid. The excision of the cholesteatoma through the ear canal is the functional approach. Most of the time, mastoid is just an innocent culprit of the disease, and often unnecessary mastoidectomy is performed. 10
Tarabichi 30 studied endoscopic mastoidectomy for cholesteatoma in 31 patients, followed for a mean of 41 months, and reported only 1 case that required revision surgery. He concluded that the endoscopic techniques allowed transcanal, minimally invasive management, and cholesteatoma surveillance with long-term results compared to those of postauricular methods. Good transcanal endoscopic access, without the need for a postauricular incision, into the attic and antrum could be provided by removing a relatively limited amount of bone mostly with a curette or microdrill.
Most surgeons claim that a postauricular approach is not particularly an invasive approach. Tarabichi responded to this argument stating avoiding postauricular incision would be the logical choice for patients and surgeons alike if it does not compromise the outcome of the procedure. 29 The patient would choose the transcanal approach to postauricular for the surgery. 6 As the pathology was accessed directly through the canal, avoiding postauricular large cavity drill, Tarabichi claimed that the average surgical duration became shorter. However, he did not perform a comparative study regarding surgical duration between the microscopic and endoscopic mastoidectomy. 30
Migirov et al 31 published a retrospective study on exclusive EES for 31 patients and concluded it to be a minimally invasive surgery that allowed eradication of cholesteatoma with good functional results. Their arguments were similar to Tarabichi. Their results were comparable with the traditional approaches.
Sajjadi 32 reported a study on the endoscopic transcanal modified canal wall down mastoidectomy for cholesteatoma showing a good outcome. The posterior canal was also reconstructed using cartilage. However, the limitation of this study was sample size of only 4 patients. The study reported canal stenosis secondary to drilling, which was prevented by retraction technique. We also had canal skin laceration in 4 patients due to the friction created by drilling. However, they all healed without any stenosis or complications. An ultrasonic bone curette can be useful to avoid such damage. Nevertheless, these instruments are costly for low- and middle-income countries to afford, and the handpiece is also bulky to use.
A similar retrograde mastoidectomy was also done by Kakehata et al, 33 where the authors used an ultrasonic bone curette device to remove the bone. Of 39 ears that underwent TEES, 13 ears had cholesteatoma involving the mastoid cavity, and the rest involving only the attic. All the patients had a good outcome after the surgery. They concluded the endoscopic approach to the antrum as being less invasive and more functional.
Another study was done by Mehta et al, 34 who reported on “inside-out” mastoidectomy using an ultrasonic bone curette. However, the sample size was only 5 patients, and follow-up was for 12 months. They also reported endoscopic inside-out mastoidectomy to be safe and effective for sclerotic mastoids. The disadvantages of an ultrasonic bone curette are its cost and a bulky handpiece.
We performed the surgery using the routine otological surgical setup. We also created a malleable copper elevator and needle to elevate the flap and remove the disease from the mastoid cavity. The endoscopic system is the same system used for sinus surgery. One of the essential advantages of EES is the easy setup of endoscopic tools, especially when microscope access is difficult. This advantage is very crucial to all otologists who are working in a limited economic circumstance. Common otological surgeries like myringoplasty and ventilation tube insertions can be easily performed with less expenditure. Overall, the endoscopic setup costs less, easy to arrange, and affordable for developing countries.
The disadvantages of EES include one hand surgery that requires surgical skills.6,10,11 Bleeding is always the issue, but once the flap is raised, it is not much of the problem even if granulation tissues are present. We had 3 patients who were converted into microscopic ear surgery due to bleeding. All 3 patients had polyp at the attic that bled on touch. Thus, bleeding is still one of the drawbacks of the TEES. Case selections play a vital role in EES. The permeatal drilling can be difficult initially as one might feel instruments getting crowded. Nevertheless, this can be overcome once the technical skill is learned.
One of the significant limitations of this study is the small sample size. The attic-only cholesteatoma and the extensive cholesteatoma were excluded, the reason for our less sample size. The extensive cholesteatoma was managed via combined microscopic and EES. We included only moderate-sized cholesteatoma. Short follow-up is another limitation of this study. The present study reported on early 1-year results only. Long-term follow-up would have been better. Several authors have claimed endoscopic surgery to be faster than microscopic surgery especially for myringoplasty. 35 A comparative study between the microscopic and endoscopic mastoidectomy would have yielded the real advantages between the two.
Conclusion
Endoscopic transcanal retrograde mastoidectomy was feasible for moderate cholesteatoma with good results. It is more functional, ideal for sclerotic mastoid, avoids postauricular incision, allows minimal tissue/bone removal, and faster wound healing.
Footnotes
Authors’ Note
The research was done at Civil Service Hospital, Department of Otorhinolaryngology, Kathmandu, Nepal.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
