Abstract
Most reported cases of middle ear adenoma (MEA) have focused on histopathology because MEA is usually diagnosed postoperatively, which is considered as a major setback. We focused on the surgical aspect of the disease to facilitate a preoperative diagnosis, resulting in prompt and proper treatment, without requiring a second stage of surgical treatment. In this report, we present the differential diagnoses in a 40-year-old man with MEA requiring surgical treatment. Preoperatively, the patient was suspected to have an MEA. An analysis of the surgical procedures in similar misdiagnosed tumors has enabled us to assess surgical procedures in cases wherein the preoperative diagnosis does not coincide with the postoperative histopathological results.
Introduction
Approximately 120 cases of middle ear adenoma (MEA) have been reported in the Medline database to date. However, the actual rate of diagnosis in clinical practice may be higher. Middle ear adenoma is estimated to represent less than 2% of all middle ear tumors. 1 The mean age at the time of diagnosis of this low-grade malignant tumor is 44 years, with no sex preponderance. The tumor presents as a yellowish, grayish, or pale reddish mass behind the tympanic membrane and can be diagnosed by fine-needle aspiration, tympanotomy, and biopsy; however, diagnosis is usually confirmed by histopathological examination after surgery. The histopathological frozen tumor section obtained during surgery is often misdiagnosed as adenocarcinoma or paraganglioma. 2 Establishing the correct diagnosis is necessary for the provision of proper surgical treatment and prevention of potential recurrence. Therefore, ear, nose, and throat surgeons should have sufficient knowledge about this pathology and its differential diagnosis.
A 40-year-old man was admitted to our clinic with complaints of gradual hearing loss in the right ear, ear fullness, and tonal tinnitus. The tympanic membrane was found to be intact on otoscopic examination; however, a reddish mass was observed in the posterior quadrants (Figure 1). Tonal audiometry revealed conductive hearing loss with a hearing level of 20 to 30 dB (Figure 2). A type-b tympanogram was obtained using tympanometry. High-resolution computed tomography (HRCT) of the right ear showed a restrictive mass with soft-tissue density in the posterior epi- and mesotympanum, without involving the Eustachian tube. The lesion had no connection with the promontory (Figure 3), involved an ossicle that was not yet destroyed, and had not infiltrated the mastoid bone. Additionally, dense fluid was noted in the mastoid cells. Magnetic resonance imaging (MRI) was performed using the non-echo planar diffusion-weighted technique. The mass was limited to the middle ear with no connection to the internal ear structures and showed low to intermediate intensity on T1-weighted imaging and high intensity on T2-weighted imaging. There was no diffusion surrounding the pathology in the non-echo planar imaging (EPI)-diffusion-weighted imaging (DWI) protocol (Figure 3). The patient was thus qualified for ear surgery. Mastoidectomy, posterior tympanotomy, and resection of the tympanic cavity tumor with the incus and head of the malleus were performed. Finally, partial ossicular replacement prosthesis tympanoplasty was performed. Postoperative histological and immunohistological examination of the tumor revealed a carcinoid tumor (Figure 4).
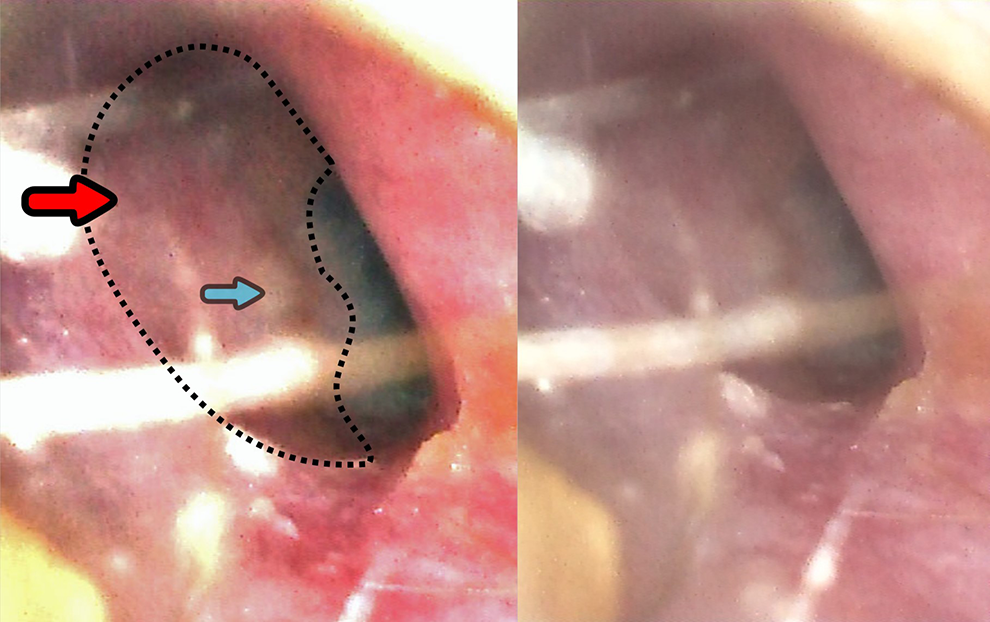
Otoscopy tumor—red arrow, umbo—blue arrow.
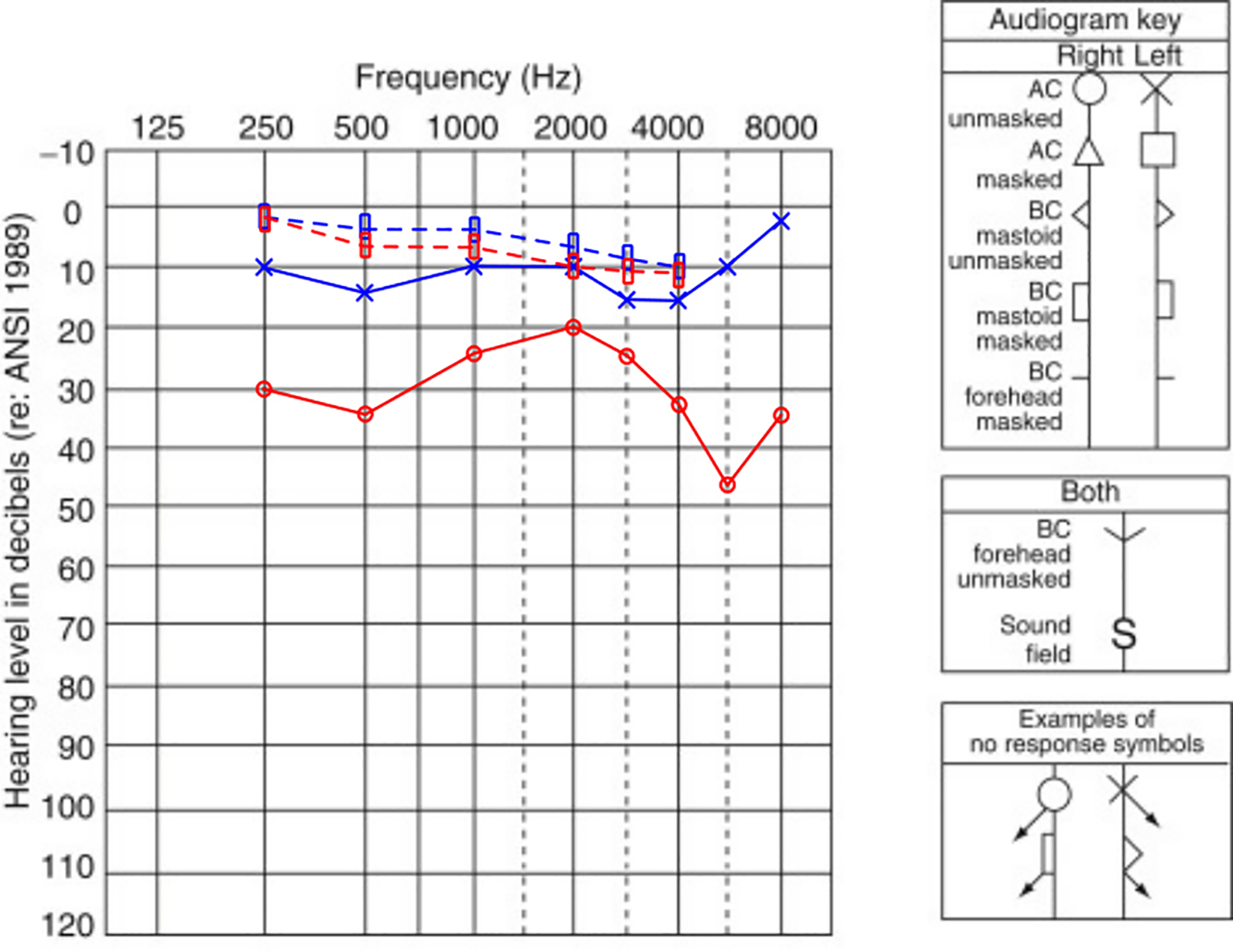
Tonal audiometry—right-side conductive hearing loss.
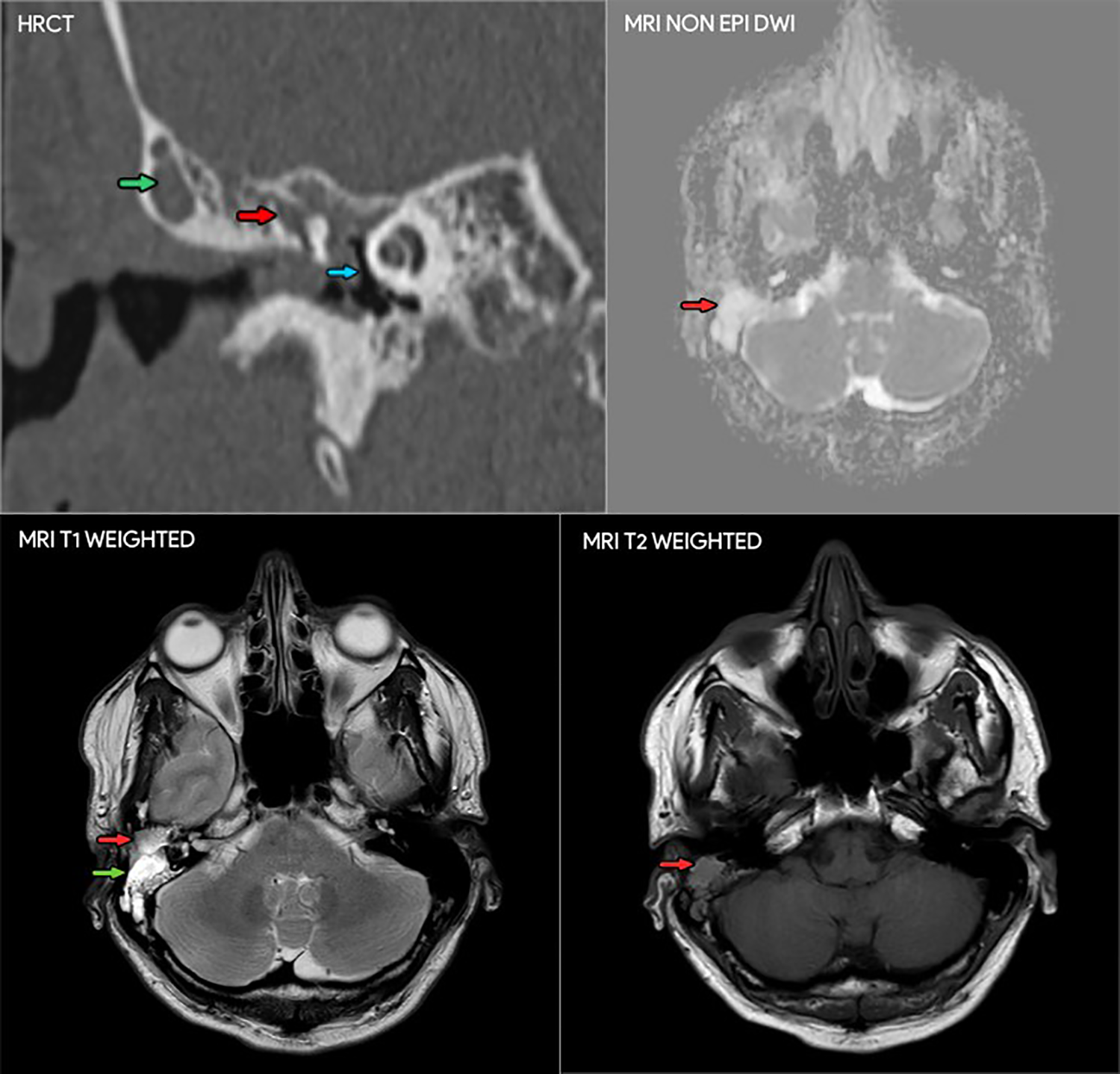
Radiological immaging: tumor—red arrow, and fluid in the mastoid—green arrow, and promontory—blue arrow.
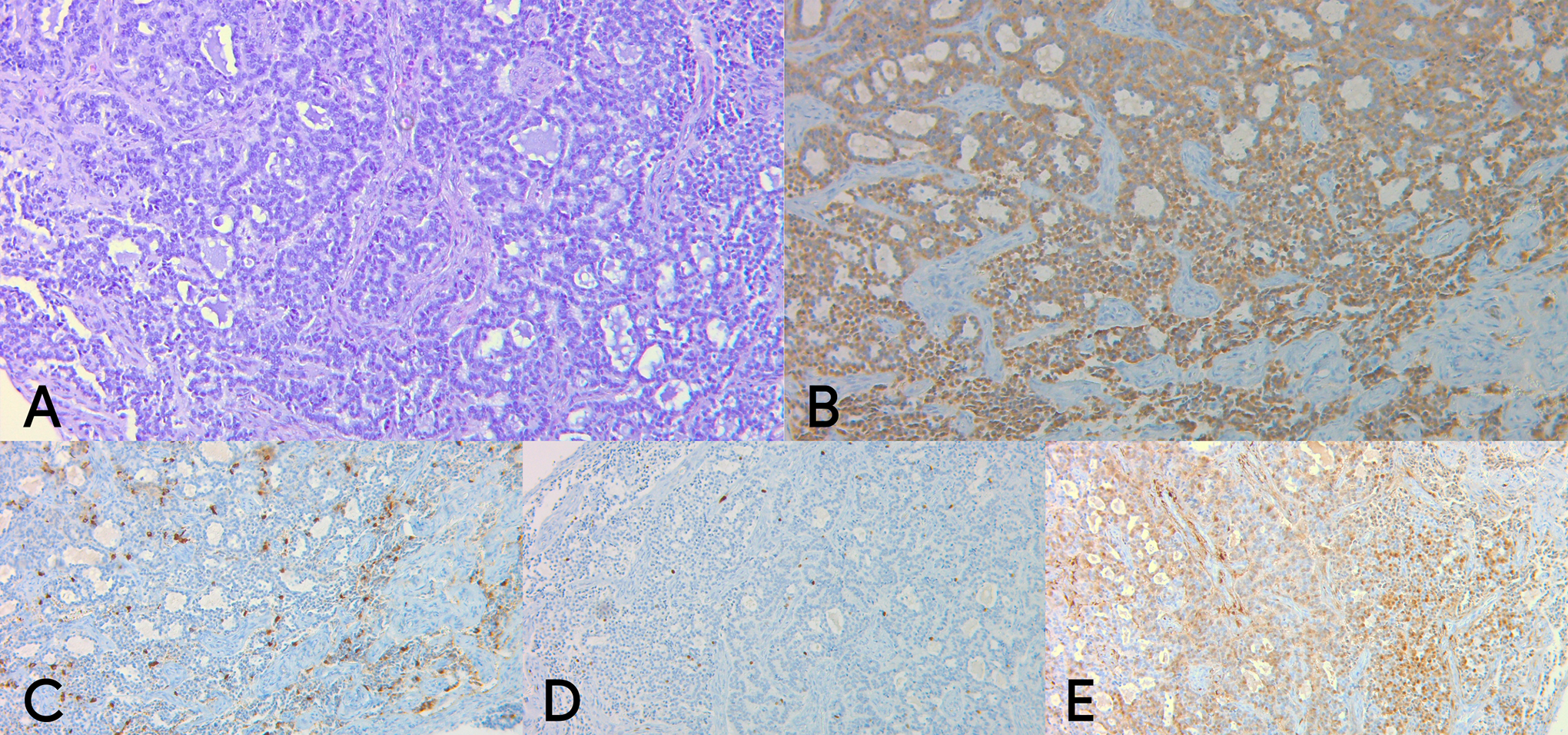
Histopathological examination: A, Hematoxylin-eosin (HE) stain. Cuboidal and uniform cells with acidophilic cytoplasm, (B) Synaptophysin-positive immunohistochemical staining, (C) Chromogranin focally positive immunohistochemical staining. D, Ki-67 cell proliferation index <5%, E. S100 protein-negative stain.
Discussion
Imaging modalities such as HRCT and MRI should be performed preoperatively in order to exclude the diagnosis of paragangliomas, schwannomas, ceruminous gland adenomas, Schneiderian-type mucosal papillomas, lipomas, and cholesteatomas. Paragangliomas almost always arise from the Jacobson nerve and its branches at the level of the promontory. 3 In this case, the location of the pathology ruled out the possibility of a paraganglioma; however, digital subtraction angiography was performed to confirm this. The location of the tumor also indicated the possibility of MEA, which is seldom seen in the Eustachian tube but usually surrounds the ossicular chain. 1 In contrast to MEAs, gland adenomas, meningiomas, cranial nerve VII and VIII neuromas, retrotympanic vascular masses, and parotid carcinomas or metastases arise outside of the middle ear, involving it in later stages. 4 Thus, the location of the discussed pathology involves only the middle ear, as seen on MRI and HRCT, which ruled out this disease group. Moreover, MRI with a T1-weighted sequence may allow differentiation between lipomas and MEAs. The absence of diffusion on non-EPI-DWI eliminated the possibility of a cholesteatoma. Radiographic findings of Schneiderian-type mucosal papillomas mimic those of MEAs, except for a more common localization within the Eustachian tube region (45%). Schneiderian-type mucosal papillomas and MEAs can occur at the same age, but the diagnosis of Schneiderian-type mucosal papillomas precludes a long-standing history of otitis media with otorrhea (usually treated with antibiotics) and concomitant sinonasal disease. 5,6
The described tumor may be classified as T2A according to the staging system proposed by Marinelli et al. 7 Owing to a recurrence rate of 9% to 22%, 1,8,9 the tumor should be radically resected along with ossicle removal, followed by ossicular chain reconstruction. In our case, the most probable misdiagnoses were a lipoma or Schneiderian-type mucosal papilloma. In the literature, more than 30 cases of Schneiderian-type mucosal papilloma of the ear have been described. 5 Owing to its significant association with malignancy (approximately 41%), the tumor required radical resection with an ossicular chain if encased. In addition, since only 8 cases have been reported in the literature, there is no standard treatment protocol for ear lipomas. However, Buen et al suggested aggressive treatment, including the possibility of ossicle removal. 10 In conclusion, for all cases of well-restricted middle ear tumors surrounding the ossicle, radical resection should be performed with the removal of ossicles.
Radiological diagnosis, including HRCT of the ear and MRI with a non-EPI-DWI protocol in case of a suspected middle ear tumor, is sufficient to establish a differential diagnosis of MEA. For surgery of a tumor that involves well-preserved ossicles, we suggest removing the tumor along with the ossicles to facilitate easier access to the tumor mass thus decreasing the risk of recurrence.
Footnotes
Authors’ Note
This article describes the stage of the case at the time of treatment in accordance with current standards. Consent from the Ethics Committee was not required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
