Abstract
Objective:
To determine taste and smell impairment rates in patients with coronavirus disease 2019 (COVID-19) who were hospitalized in the intensive care unit (ICU).
Methods:
Between March 2020 and May 2020, patients with COVID-19 hospitalized in the ICU were enrolled in this study. Upon discharge, patients were telephoned and asked to complete a survey related to taste and smell impairment. Characteristics were compared between patients with and without taste and/or smell impairment.
Results:
Fifty-two patients were enrolled (mean age, 61.32 ± 12.53 years; mean ICU stay, 10.19 ± 10.24 days). Age, sex, type/number of comorbid diseases, most ICU support modalities, and ICU stay duration did not significantly differ between groups. Patients in group B required more high-flow nasal oxygen therapy than patients in group A (P = .010). In total, 22 (42.3%) of 52 patients experienced taste and/or smell impairment. Three patients experienced isolated smell impairment and one patient experienced isolated taste impairment. Among the 21 patients who experienced smell impairment, 18 (85.78%) experienced hyposmia (mean visual analog scale [VAS] score: 6.33 ± 0.97), while 3 (14.28%) experienced anosmia. Among the 19 patients who experienced taste impairment, 16 (84.22%) experienced hypogeusia (mean VAS score: 6.43 ± 1.03) and 3 (15.78%) experienced ageuisa. Among 22 patients who experienced taste and/or smell impairment, 15 (68.18%) patients (n = 15/22) experienced smell and/or taste impairment before the ICU stay, while 7 (31.82%) patients (n = 7/22) experienced impairment during the ICU stay. Overall, 28.84% (n = 15/52) of the patients experienced taste and/or smell impairment before the ICU stay.
Conclusions:
Patients who were hospitalized in the ICU experienced lower rates of taste and/or smell impairment. Some patients experienced taste and/or smell impairment during the ICU stay.
Introduction
Taste and smell impairment was initially considered a common finding in patients with coronavirus disease 2019 (COVID-19). 1 A few months after the pandemic began, there were reports that taste and smell impairment was a standard manifestation during the clinical course of COVID-19. Accordingly, both the World Health Organization and the Centers for Disease Control listed smell and taste impairment as a symptom of interest when screening for COVID-19. 2 Previous reports indicated that patients with taste and/or smell impairment were 7- to 10-fold more likely to have COVID-19 infection. 1,3
The COVID-19 clinical course in infected patients is highly variable, ranging from asymptomatic disease to severe critical illness. 1,2 Some reports have indicated that taste and smell impairment is less frequent in severe to critically ill patients. 3 Thus, it has been hypothesized that 2 different forms of COVID-19 exist: a “nasal” form and a “pulmonary’’ form. 4 These forms of the disease have been reported to differ in terms of nasal obstruction, as well as taste and smell impairment. Accordingly, patients hospitalized in the intensive care unit (ICU) may experience less taste and smell impairment than patients with mild/moderate disease. Comparison of taste and smell impairment rates among patient subgroups may enable researchers to better understand the clinical course and pathophysiology of COVID-19.
To the best of our knowledge, there have been few reports of taste and smell impairment in critically ill patients. Existing reports mostly comprise case series of patients with COVID-19, and the results have been heterogeneous. 5 Therefore, the occurrence of taste and/or smell impairment in patients with varying COVID-19 severity has not been fully characterized. The present study evaluated the taste and smell impairment rates in critically ill patients hospitalized in the ICU. This study also evaluated whether other characteristics differed between among COVID-19 patient subgroups, on the basis of taste and smell impairment status.
Patients and Methods
This study was performed in the Anesthesiology and Reanimation Department of Sisli Hamidiye Etfal Training and Research Hospital between March 2020 and May 2020. Patients who were hospitalized in the ICU were enrolled in the study. Ethical approval was obtained from the hospital’s ethics committee. Patients with polymerase chain reaction (PCR)-confirmed COVID-19 were included. Patients who died during the ICU stay, those who could not be reached via telephone, those who had communication disorders, those who underwent tracheostomy, and those who declined to participate were excluded from the study. Upon discharge, patients completed a telephone survey related to the loss of taste and/or smell.
Patients’ demographic parameters (age and sex), comorbidities, onset of taste and smell impairment (before or during ICU stay), and types of taste and smell impairment (ie, anosmia/hyposmia or ageusia/hypogeusia) were recorded. When patients reported hyposmia and/or hypogeusia, they were asked to indicate smell and/or taste impairment on a visual analog scale (VAS; 0-10, where 10 indicates normal function). Noninvasive ventilation, mechanical ventilation, high-flow nasal oxygen (HFNO) therapy, and mask oxygenation data were recorded. Patients were divided into 2 groups according to the presence or absence of taste and/or smell impairment. Group A included patients who did not experience any taste or smell impairment, while group B included patients who experienced taste and/or smell impairment. Clinical characteristics were compared between the 2 groups.
Statistical Analysis
IBM SPSS Statistics (version 22.0; IBM Corp) was used for the statistical analysis. Descriptive statistics (eg, mean, standard deviation, median, frequency, ratio, minimum, and maximum) were generated. The Shapiro-Wilk test was used to assess whether quantitative data exhibited a normal distribution. Student t test was used to compare normally distributed variables between the 2 groups. The Pearson χ2 test was used to compare qualitative data between the 2 groups. A P value <.05 was considered to indicate statistical significance.
Results
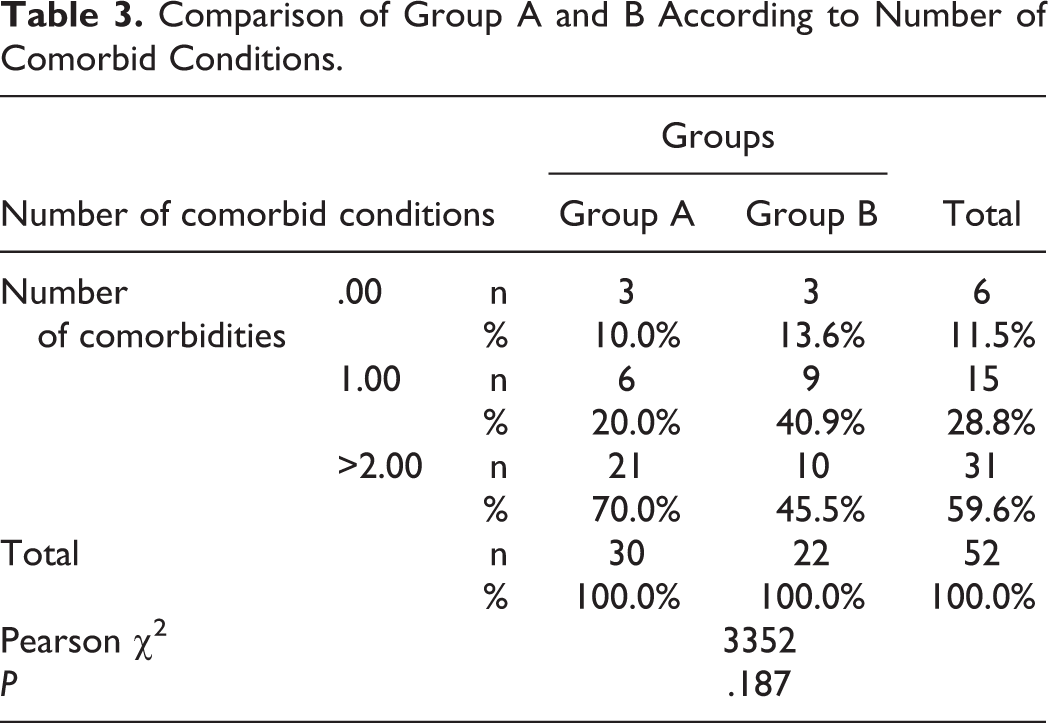
Fifty-two patients were included in this study. Flow chart of the study was presented in Figure 1. The mean patient age was 61.32 ± 12.53 years (Table 1) and 16 (30.8%) patients were women. The mean ICU stay of the overall cohort was 10.19 ± 10.24 days. Table 2 shows the distribution of comorbid diseases. Six patients had no comorbid diseases, while 15 patients had 1 comorbid disease and 31 patients had 2 or more comorbid diseases. The age, sex, type and number of comorbid diseases, and length of ICU stay did not significantly differ between groups A and B (Tables 1 -3).
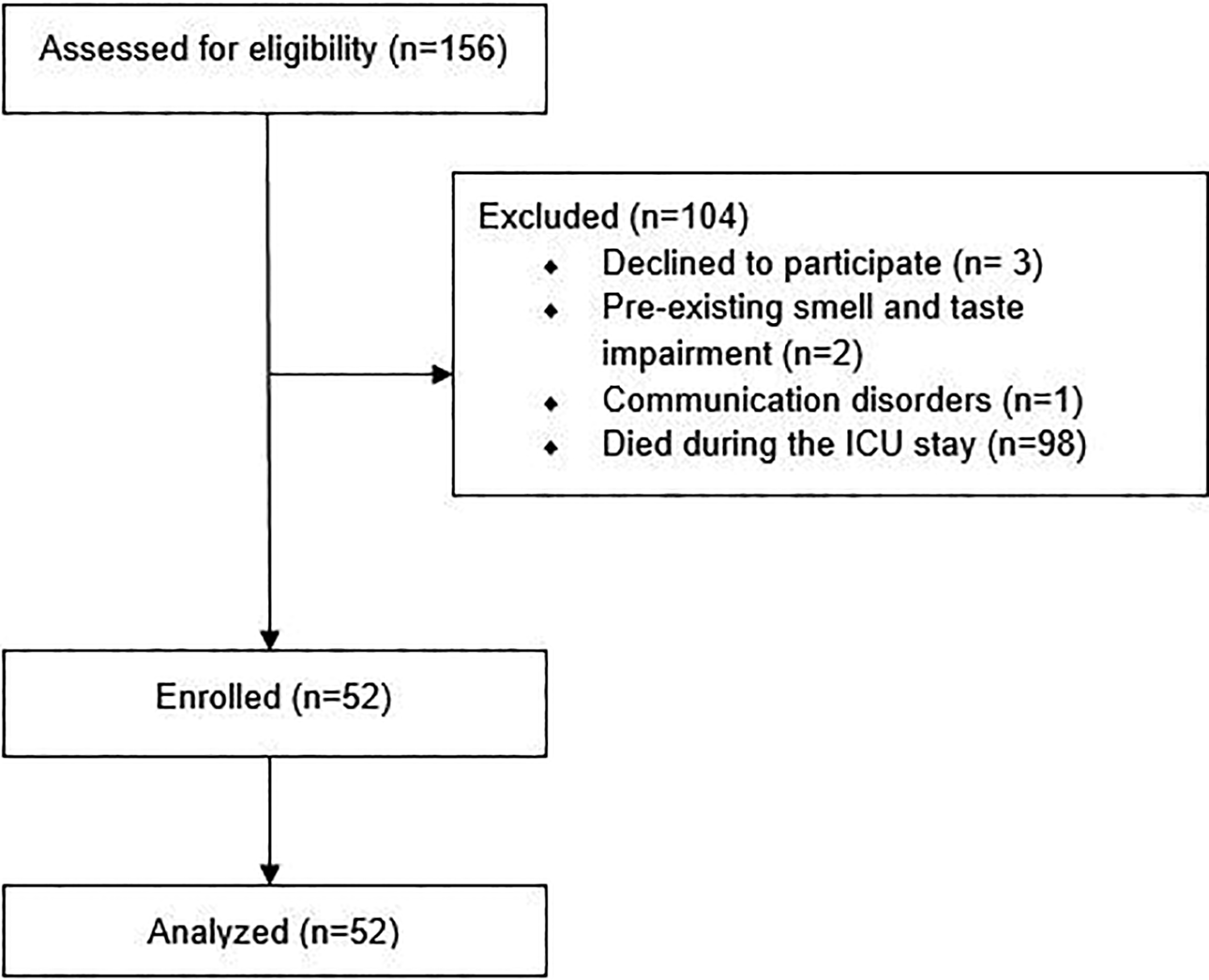
Flowchart of the study.
Demographic Characteristics, Taste and Smell Impairment Data of the Study Group.
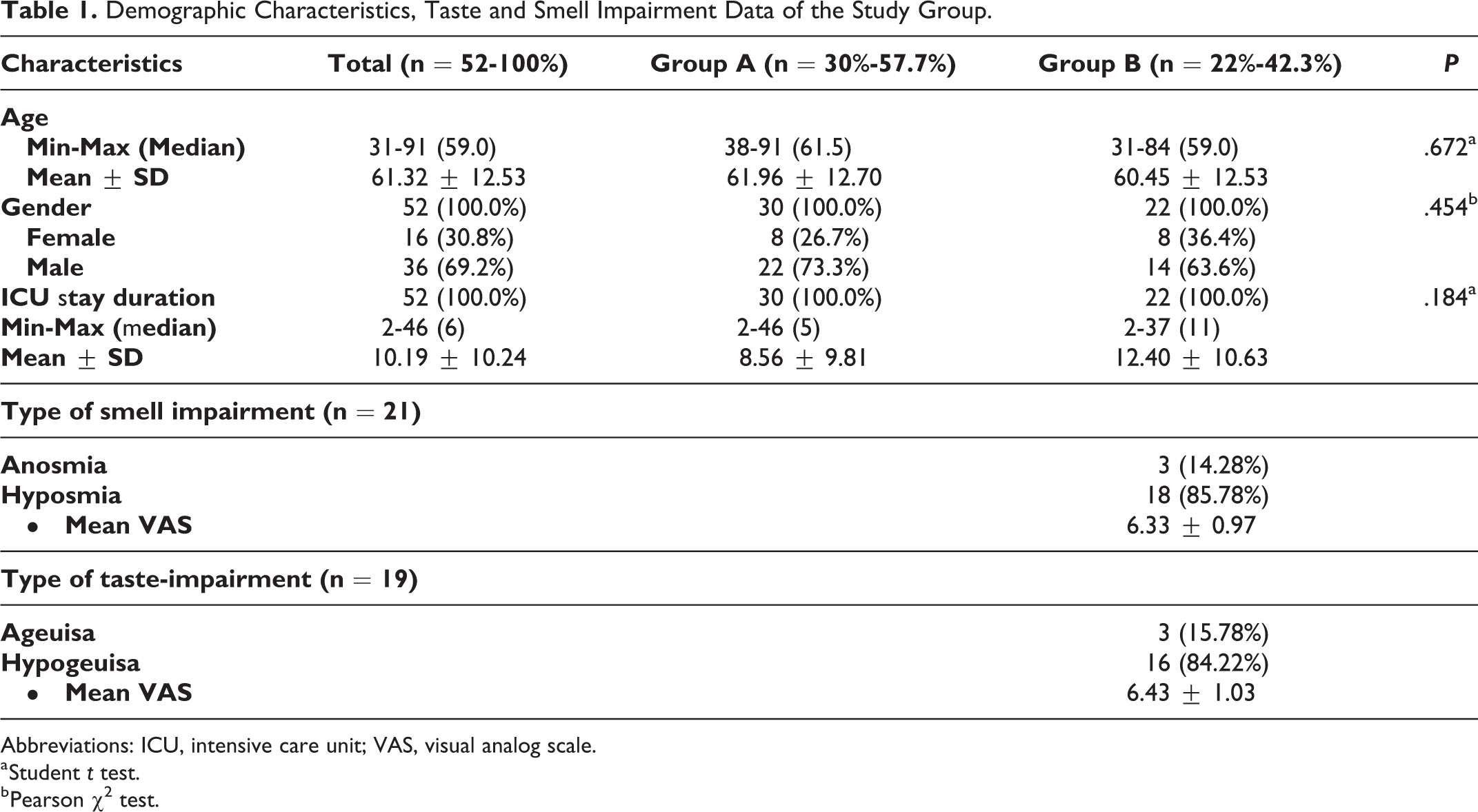
Abbreviations: ICU, intensive care unit; VAS, visual analog scale.
a Student t test.
b Pearson χ2 test.
Comorbidities of the Study Group.
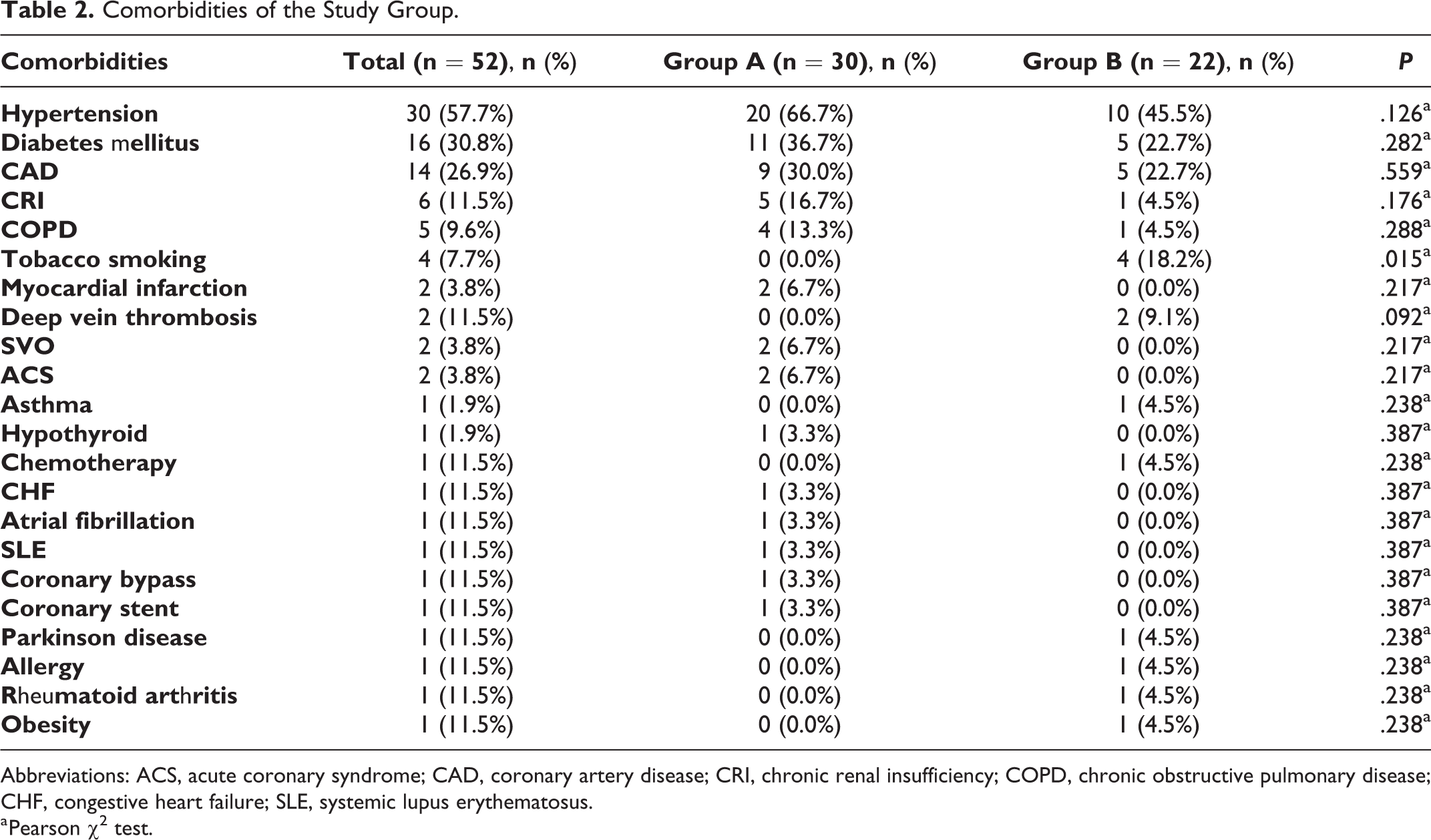
Abbreviations: ACS, acute coronary syndrome; CAD, coronary artery disease; CRI, chronic renal insufficiency; COPD, chronic obstructive pulmonary disease; CHF, congestive heart failure; SLE, systemic lupus erythematosus.
a Pearson χ2 test.
Comparison of Group A and B According to Number of Comorbid Conditions.
Twenty-two (42.3%) patients (n = 22/52) experienced taste and/or smell impairment (Table 1). Among these 22 patients, 3 experienced smell impairment alone, 1 experienced taste impairment alone, and 18 experienced both smell and taste impairment. Among the 21 patients who experienced smell impairment, 18 (85.78%) experienced hyposmia and 3 (14.28%) experienced anosmia. Among the 19 patients who experienced taste impairment, 16 (84.22%) experienced hypogeusia and 3 (15.78%) experienced ageusia (Table 1). Patients who experienced hyposmia and hypogeusia reported mean VAS scores of 6.33 ± 0.97 and 6.43 ± 1.03, respectively. Among 22 patients who experienced taste and/or smell impairment, 15 (68.18%) patients (n = 15/22) experienced smell and/or taste impairment before the ICU stay, while 7 (31.82%) patients (n = 7/22) experienced impairment during the ICU stay. Overall, 28.84% (n = 15/52) of the patients experienced taste and/or smell impairment before the ICU stay.
Concerning intensive care support modalities, the use of mechanical ventilation, noninvasive ventilation, and masks did not significantly differ between groups A and B. Patients in group B required significantly more HFNO therapy than patients in group A (n = 11, 36.7% in group A, and n = 16, 72.7% in group B; P = .010, Table 4).
Comparison of Ventilator Support and Oxygenation Modalities Between Group A and Group B.
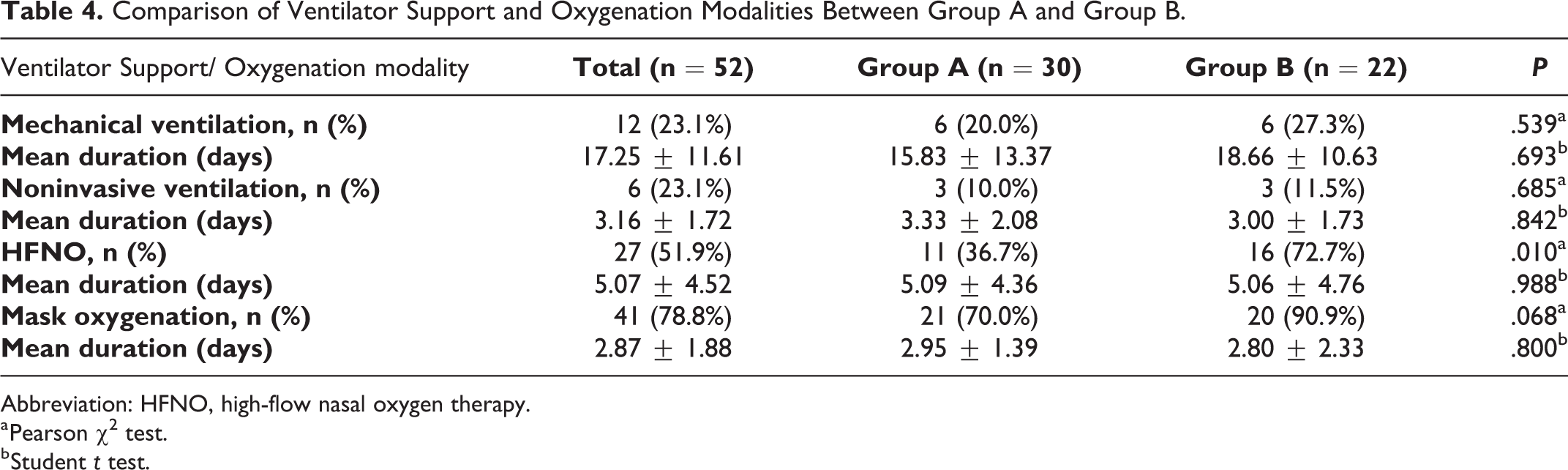
Abbreviation: HFNO, high-flow nasal oxygen therapy.
a Pearson χ2 test.
b Student t test.
Among the patients who required mechanical ventilation, noninvasive ventilation, mask application, and HFNO therapy, the durations of these support modalities did not significantly differ between groups A and B. Intensive care unit stay duration and ventilation/oxygenation parameters were not significantly associated with the onset of taste and/or smell impairment (before/during ICU stay; Table 5).
Comparison of Ventilator Support and Oxygenation Modalities on Onset of Taste and Smell Impairment.
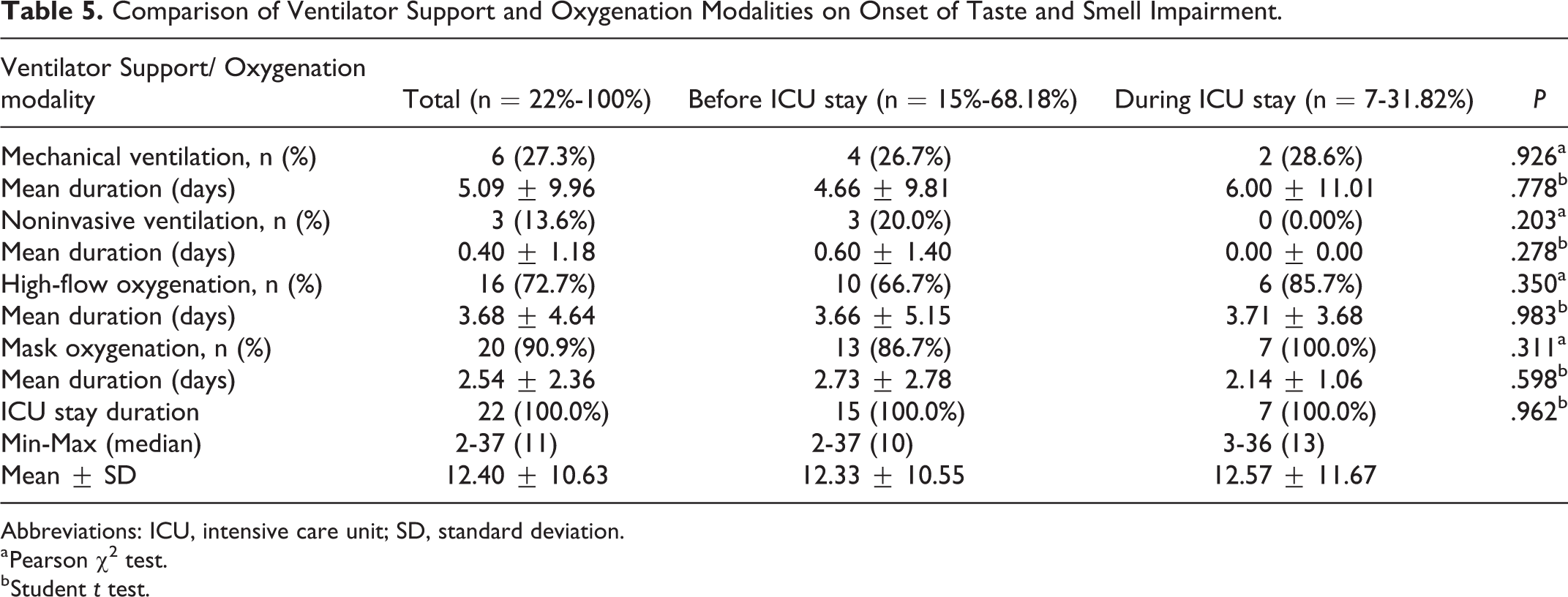
Abbreviations: ICU, intensive care unit; SD, standard deviation.
a Pearson χ2 test.
b Student t test.
Discussion
In this study, 42.3% of patients who required hospitalization in the ICU reported taste and/or smell impairment. Among the intensive care support modalities, only HFNO therapy differed between the groups; patients with taste and/or smell impairment required significantly more HFNO therapy. Among 22 patients who experienced taste and/or smell impairment, 68.18% experienced loss of taste and/or smell before the ICU stay, while 31.82% experienced loss of taste and/or smell during the ICU stay.
Smell and taste impairment is a well-known sequelae of upper respiratory tract infections. 6 The reported incidence of olfactory dysfunction following upper respiratory tract infection is between 20% and 40%. 1 For typical upper respiratory tract infections, the associated smell and taste impairment mostly occurs as a result of nasal obstruction, which differs from the impairment in patients with COVID-19. 7 Notably, patients with COVID-19 have high rates of smell and taste impairment in the absence of nasal obstruction. The virus that causes COVID-19, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), exerts its effects by binding to the ACE2 and TMPRSS2 receptors. Because neural structures do not contain ACE2 receptors, non-neural involvement has been suggested as a cause of COVID-19-related smell and taste impairment. 8 Most patients with COVID-19 exhibit partial taste and/or smell impairment, which tends to resolve early and spontaneously. 9 However, some patients do not exhibit early resolution of these symptoms, and baseline severity is reportedly an important predictor of early recovery. 5 Radiological studies also support this hypothesis. Eliezer et al evaluated 20 patients with magnetic resonance imaging and found that 19 (95%) of them had edema in the olfactory cleft. At the 1-month follow-up visit, olfactory edema was resolved in 63% of those patients. 10
Taste and/or smell impairment is now considered an important symptom when screening for COVID-19. This biomarker is robust in young and mildly symptomatic patients with COVID-19. 11 However, the cases reported thus far have been heterogeneous, and there are limited data concerning critically ill patients.
Previous reports indicated that taste and/or smell impairment may occur frequently in patients with mild to moderate COVID-19. Nasal and pulmonary forms of disease have also been suggested, which may be useful in prognostic assessment. Yan et al reported that anosmia is strongly predictive of outpatient care. 4 In a study of 59 hospitalized patients with COVID-19, Giacomelli et al reported that 33.9% exhibited smell and/or taste impairment. 12 In a separate study of 99 hospitalized patients with COVID-19, Barry et al reported that only 9.1% of severely ill patients experienced loss of smell. 13 Finally, in 47 hospitalized patients with severe COVID-19, Leichen et al observed a smell impairment rate of 38.3%. 14 In contrast, Neto et al reported that taste and smell impairment were not associated with disease severity in 655 patients with COVID-19. 15 Von Bartheld et al performed a meta-analysis of 104 studies including 38 198 patients. They found that the rates of olfactory and taste dysfunction were 43.0% and 44.6%, respectively. 16 The reported rates differ among meta-analyses. For example, Hajikhani et al analyzed 3739 patients from 15 studies with PCR-confirmed COVID-19. 17 The taste and smell impairment rates were 49% and 61%, respectively. Among patients hospitalized in the ICU, we found that 42.3% (n = 22/52) experienced taste and/or smell impairment, consistent with the findings of previous studies. However, approximately one-third of the patients reported that smell and/or taste impairment occurred during the ICU stay. When these patients were excluded, the taste and/or smell impairment rate decreased to 28.84% (n = 15/52). These results support the existing hypothesis that smell impairment is less frequent with increasing disease severity.
Another important finding of our study was taste and/or smell impairment during the ICU stay. To the best of our knowledge, no study has evaluated how ICU stay affects taste and smell in hospitalized patients. Although the exact causes of ICU-related taste and smell impairments are unclear, increasing disease severity, use of additional medications, and/or electrolyte imbalances may all be factors. However, additional studies are required for confirmation of this. Furthermore, the presence of these impairments may not reflect pre-ICU onset, and critically ill patients should be evaluated separately.
No definitive treatment exists for COVID-19-related smell impairment. However, British Rhinological Society consensus guidelines recommend the use of olfactory training after 2 weeks, as well as nasal/systemic steroids and omega-3 supplements on an individual basis. The use of alpha-lipoic acid and vitamin A is not recommended for patients with COVID-19-related smell impairment. 18
With the exception of HFNO therapy, we found no significant differences in demographic characteristics or ventilation/oxygenation parameters between patients who experienced taste and/or smell impairment and those who did not. Only HFNO therapy was required more by patients who experienced taste and/or smell impairment. In our study, 7 (31.82%) of 22 patients developed taste and/or smell impairment during the ICU stay. Furthermore, ICU stay duration was not associated with the onset of taste and/or smell impairment.
This study had some limitations. First, objective testing methods were not used. Although self-reported smell impairment may be regarded as a subjective parameter, previous studies showed that self-reported smell impairment was strongly correlated with objective testing methods. 2 Second, the exact number of critically ill patients with smell and taste impairment could not be determined because the survey was administered after patients had been discharged.
Conclusion
Patients who were hospitalized in the ICU experienced lower rates of taste and/or smell impairment. Some patients experienced taste and/or smell impairment during the ICU stay.
Footnotes
Authors’ Note
P.S. helps the conception of the work, acquisition, analysis of the data, drafted the work. M.A. helps the design of the work, interpreted the data for the work, drafted the work. A.S.C. helps the design of the work, analyzed the data, interpreted the data for the work, revised the manuscript for important intellectual content. H.M.O. helps the design of the work, analyzed the data, interpreted the data for the work, revised the manuscript for important intellectual content. All authors approved the final version of the manuscript. NCT04532632 (![]() ). Sisli Etfal Training and Research Hospital Local Ethics Committee-2855.
). Sisli Etfal Training and Research Hospital Local Ethics Committee-2855.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
